Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Abanico veterinario
On-line version ISSN 2448-6132Print version ISSN 2007-428X
Abanico vet vol.11 Tepic Jan./Dec. 2021 Epub Apr 04, 2022
https://doi.org/10.21929/abavet2021.33
Original Article
Habitat characteristics of the white-toothed woodrat (Neotoma leucodon) in the Potosino-Zacatecan plateau
1Posgrado de Innovación en Manejo de Recursos Naturales, Colegio de Postgraduados, Campus San Luis Potosí, Iturbide 73, Colonia Centro, CP. 78620. Salinas de Hidalgo, San Luis Potosí. S.L.P. México.
2Centro de Investigación Aplicada en Ambiente y Salud de la Facultad de Medicina-CIACYT, Universidad Autónoma de San Luis Potosí. Av. Sierra Leona No. 550, Col. Lomas 2a, CP. 78210 San Luis Potosí, S.L.P. México.
3Departamento de suelos. Universidad Autónoma Chapingo. Km 38.5 Carretera México-Texcoco, CP. 56230 Chapingo, Estado de México.
The white-toothed woodrat (Neotoma leucodon) inhabits the arid zones of north-central Mexico and it is important for rural communities for food and economic income. The objective was to characterize the habitat of the white-toothed woodrat in the Potosino-Zacatecano Plateau. In 44 sites with burrows and 44 random sites, the altitude, slope, slope exposure, ground cover: shrubs, grasses, rock, woody material and bare soil, as well as the density of prickly pear cactus, agave, shrubs and palms were recorded. In addition, 21 hunter-gatherers (H-G) were surveyed about woodrat habitat conditions. The woodrat builds its burrows in agaves (40.4 %) and 47.6 % of the H-Gs stated that the woodrat was more frequent in prickly pear cactus. Ninety % of the burrows were located on terrain with low slopes (≤ 4 %), where shrub density and cover were dominant (2085.6 ± 1825 ha -1, 36.1 ± 17.5%, respectively). Rat burrows are associated with shrubs and cactus, as they provide them with food and protection from predators. The H-G consider that in order to conserve the woodrat it is necessary to allow them to reproduce, establish hunting seasons and maintain the vegetation.
Keywords: Soil cover; local knowledge; density; burrow
La rata nopalera (Neotoma leucodon) habita las zonas áridas del centro-norte de México y es importante para las comunidades rurales como alimento e ingreso económico. El objetivo fue caracterizar el hábitat de la rata nopalera en el Altiplano Potosino-Zacatecano. En 44 sitios con madrigueras y 44 aleatorios, se registró la altitud, la pendiente, exposición de la pendiente, la cobertura del suelo: arbustivas, gramíneas, roca, material leñoso y suelo desnudo, así como la densidad de nopal, maguey, arbustos y palmas. Asimismo, se encuestaron 21 cazadores-recolectores (C-R) acerca de las condiciones del hábitat de la rata. Ésta construyó sus madrigueras en maguey (40.4 %) y el 47.6 % de los C-R aseguran que la rata es más frecuente en los nopales. El 90 % de las madrigueras se ubicaron en terrenos con pendientes bajas (≤ 4 %) donde la densidad y la cobertura de arbustivas fueron dominantes (2085.6 ± 1825 ha -1, 36.1 ± 17.5%, respectivamente). Las madrigueras de rata se asocian con arbustos y nopales, ya que les brindan alimento y protección contra depredadores. Los C-R consideran que para conservar la rata es necesario dejar que se reproduzcan, establecer temporadas de caza y mantener la vegetación.
Palabras clave: Cobertura de suelo; conocimiento local; densidad; madrigueras
INTRODUCTION
Rodents are the largest group of mammals in the world; in Mexico there are about 245 known species of rodents. The genus Neotoma is composed of four subgenera (Teonopus, Hodomys, Teonoma and Neotoma), the latter consisting of four species (albigula, floridana, lepida and mexicana) (Hall, 1982) and more recently N. leucodon, which, through cytochrome-b sequencing was separated from the N. albigula group (Edwards et al., 2001).
The species distributed in the Potosino-Zacatecan high plateau are N. leucodon and N. mexicana. The distribution of N. leucodon ranges from southeastern Colorado, eastern New Mexico, western Oklahoma and Texas (USA). Southward through Chihuahua, Coahuila, Guanajuato, Querétaro and into central Mexico, (Ceballos, 2010a), while the distribution of N. mexicana starts from southeastern Utah and central Colorado, U.S.A., south to western and interior Mexico (Ceballos, 2010b).
White-toothed woodrats, in addition to playing an important role in community dynamics, are prey for some birds and mammals, including humans. In the Potosino-Zacatecan high plateau, this species is used by the rural population as a source of food and for local trade (Márquez-Olivas, 2002). Inhabitants of this area attribute various nutritional properties to this species and consider it to be a safe food, since their diet is based on plant consumption. However, its habitat preferences are unknown.
White-toothed woodrat is territorial, and like other rodents is ecologically important, as it disperses seeds (Schupp et al., 2010) and enhances the regeneration of plant communities (Nathan & Muller-Landau, 2000); likewise, plant communities allow the establishment of diverse rodent populations (Riojas-López, 2012). In addition, rat burrows enhance water infiltration, allow for nitrogen mineralization, and they are refugia for arthropods (Whitford & Steinberger, 2010). N. albigula is a generalist and feeds on Opuntia spp, Yucca spp, Prosopis spp and Agave spp. (92%), cactus fruits and insects (Sorensen et al., 2005); however, little is known about the vegetation influence on the settlement of its populations (Edwards & Bradley, 2002).
N. leucodon is associated with desert scrub (Edwards et al., 2001); however, another study showed that N. leucodon and N. mexicana do not prefer any specific vegetation type (Villanueva-Hernández et al., 2017). Unlike other species of the same genus such as, N. lepida, which requires rocky habitats for thermoregulation (Murray & Smith, 2012). Given such differences, it is required to complement the knowledge related to the vegetation selection by wildlife species. The objectives of this work were: 1) to characterize the white-toothed woodrat habitat (Neotoma leucodon) in sites with burrows and random sites; 2) to determine the habitat variables with which the presence of the white-toothed woodrat is mostly associated and 3) to complement the information on habitat characteristics with local knowledge. This study contributes to the knowledge of the genus Neotoma, and its results can be considered to improve habitat conditions and establish management plans for this species.
MATERIAL AND METHODS
Study area. This study was conducted during the period from September to December 2017 in the Potosino-Zacatecan high plateau in San Luis Potosí, Zacatecas and Guanajuato states (Figure 1). The Potosino-Zacatecan high plateau is located in the central plain physiographic region of the Mexican high plateau, within the high plateau of central and southern San Luis Potosí and southeastern Zacatecas and includes Aguascalientes and adjacent regions of Guanajuato and Jalisco. The study area is located at coordinates 21°30' to 23°30' NL and 100°45'to 102°45' WL. This zone is characterized by the presence of mountain ranges, hills and plains, with altitudes ranging from 1,000 to 2,600 m. The climate is predominantly dry and temperate. The climate is predominantly dry temperate (BS0 kw) with rainfall in the summer and winter precipitation between 5 and 10.2% of the annual total, with average temperatures in the warmest month between 12 and 18 °C and the coldest month between -3 and 18 °C (INEGI, 2021). In the plant communities it is common to find succulent, rosetophyllous and microphyllous scrub.
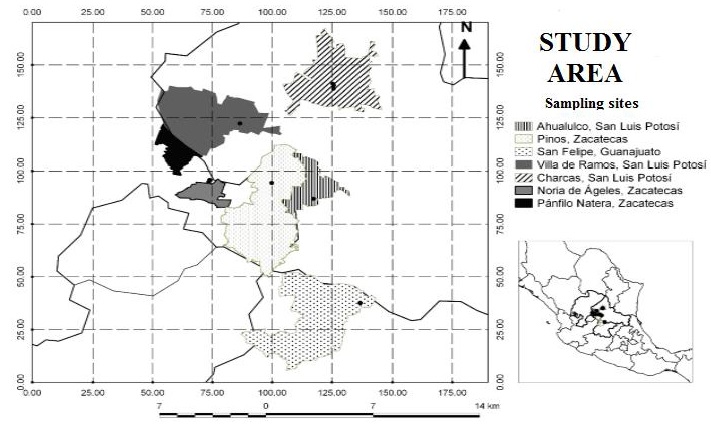
Figure 1 States and municipalities in which the habitat of the white-toothed woodrat (Neotoma leucodon) was characterized in the Potosino-Zacatecan high plateau
The predominant plant species in the succulent scrub are of the Opuntia genus, including:O. leucotricha, O. streptacantha, and shrub species such as: Dalea tuberculata, Jatropha dioica, Mimosa aculeaticarpa. In the microphyllous desert scrub, Larrea tridentata, Parthenium incanum, Prosopis laevigata, Zinnia acerosa. The rosetophyllous scrub is dominated by Agave lechuguilla, Dasylirion acotriche, Yucca carnerosana, Y. filifera, and Salvia ballotaeflora (Giménez & González, 2011).
Habitat characterization. The white-toothed woodrat habitat was characterized in circular plots of 20 m diameter (Solís & Gutiérrez, 1990), considering active burrows as the center and in random sites. Burrows were located with the support of local hunters by means of field trips and active burrows were considered to be those where the construction material was found arranged, with corridors free of vegetation, presence of cactus and gnawed maguey. The sampling of active burrows consisted of selecting those that maintained a distance of at least 50 m between one burrow and another. A random plot was evaluated for each plot established in the active burrows, these were established at 50, 100, 150 or 200 m and in a north, south, east and west direction, considering the home range, whose greatest distance was 50 m for N. magister (Hornsby, 2005) and the activity area of N. fuscipes, which according to Innes et al. (2009) is restricted to the central area of its burrow. Likewise, Cranford (1977) reported that the home range of males and females of N. fuscipes was 2 289 m2 and 1 924 m2, respectively.
Burrowed and random sites were georeferenced with a Global Positioning System (GPS Garmin Etrex 10) and the following variables were evaluated: a) elevation, b) slope (%) (Suunto PM-5/360 PC clinometer), c) slope exposure (Brunton 5007 compass), d) substrate (plant where the burrow is built), e) number of prickly pear cactus (Opuntia spp.), cardenches (Cylindropuntia spp.), magueys (Agave spp.), palms (Yucca spp.) and shrubs, as well as ground cover. To quantify ground cover in the categories of shrubs, grasses, herbaceous, bare soil, woody material and rock, two 20 m Canfield lines were used, each divided into 40 intersecting sections (every 50 cm).
Local knowledge about the white-toothed woodrat. In order to know the perception of hunter-gatherers (H-G) about the use of the habitat by the white-toothed woodrat, its food and some actions that they consider taking to conserve their populations, surveys were applied using the snowball methodology, which consists of asking each person who has been surveyed to recommend other people to be interviewed (Ledesma et al., 2002).
Statistical analysis. Descriptive statistics (mean and standard deviations) were obtained for the white-toothed woodrat habitat variables (ground cover and vegetation density). Also, to compare variables´ information of burrowed and random sites, a Kruskal-Wallis nonparametric test was performed in JMP v.13.10 software (2016). To decrease the variance in the set of independent variables (Ground cover categories, plant density, elevation, slope, slope exposure) a Principal Component Analysis (PCA) was performed in R software v. 3.4.3 (R Core Team, 2013). The graphical association of the presence of burrows with the variables that resulted from the PCA was obtained with a Simple Correspondence Analysis (SCA) (Ledesma, 2008) in Statistica v. 13.3 software (2017); for this, ordinal variables were converted to nominal variables and categorized. In all cases, a 95 % confidence interval and α = 0.05 were considered. Finally, frequencies and percentages of the variables included in surveys were obtained in Microsoft Excel (2016).
RESULTS
The white-toothed woodrat burrows were located at an average altitude of 2105 ± 87.38 m on low slopes (4 %) and only one was located on a site with a slope of 11 %. Slope exposure did not represent a key habitat component for burrow establishment by N. leucodon, as there was no preference for any exposure; however, 12 and 10 burrows were found on northeast (NE) and northwest (NW) exposures, respectively.
In the field it was identified that most of the burrows were built at maguey base (40.9 %) and prickly pear cactus (36.4 %), while 47.6 % of the H-G mentioned that white-toothed woodrats build their burrows in the prickly pear cactus and 33.3 % of them mentioned that in the maguey. However, 42.8% of the H-G said that the rats are bigger when they are in the maguey. The white-toothed woodrat burrows were found where there was a higher density of shrubs and maguey (Table 1).
Table 1 Means and standard deviations of plant density (individual ha-1) in burrowed and random sites in the white-toothed woodrat (Neotoma leucodon) habitat of the Potosino-Zacatecan high plateau
| Site | ||
| Burrow (n = 44) | Ramdom (n = 44) | |
| Maguey | 740.1 ± 899 | 263.3 ± 362.3 |
| Prickly pear cactus | 590.3 ± 473.1 | 413 ± 563.1 |
| Cardenche | 102 ± 133.9 | 122.2 ± 154.7 |
| Shrubs | 2085.6 ± 1825 | 2156.6 ± 2004.9 |
| Palms | 28.9 ± 67.9 | 20.3 ± 47.1 |
In the burrowed and random sites, the plant density and ground cover variables did not show significant differences (α = 0.05), even though in the burrowed sites a higher density of maguey and prickly pear cactus was found, as well as higher percentages of cover by shrubs and woody material, while random sites were more likely to contain rock, herbaceous and grasses (Table 2). The PCA suggests that, of the 14 variables evaluated, six explain in greater proportion the phenomenon under study (Table 3).
Table 2 Means and standard deviations of ground cover percentages of burrowed and random sites in the white-toothed woodrat (Neotoma leucodon) habitat of the Potosino-Zacatecan high plateau
| Site | ||
| Ground cover (%) | Burrow (n = 44) | Ramdom (n = 44) |
| Shrubs | 36.1 ± 17.5 | 23.1 ± 16 |
| Bare soil | 18.6 ± 16.1 | 19.3 ± 19 |
| Rock | 13.6 ± 13.2 | 23.2 ± 22.4 |
| Woody material | 8.4 ± 5.6 | 1.5 ± 2.6 |
| Grasses | 13.4 ± 13.7 | 18.6 ± 18 |
| Herbaceous | 9.9 ± 15 | 14.2 ± 19.3 |
Table 3 PCA results for habitat variables of burrowing (44 sites) and random (44 sites) sites in the habitat of the white-toothed woodrat (Neotoma leucodon) in the Potosino-Zacatecan high plateau
| Variable | Comp. 1 | Comp. 2 | Comp. 3 |
|---|---|---|---|
| ALTITUDE | 0.3044 | 0.2360 | - 0.1649 |
| SHR COVER | 0.0639 | - 0.5350 | 0.2142 |
| GR COVER | - 0.0031 | 0.2899 | 0.4895 |
| WOODMC | 0.0610 | -0.3566 | - 0.294 |
| FOR COV | - 0.0846 | 0.2016 | - 0.5663 |
| ROCK COV. | - 0.4076 | 0.2375 | 0.0086 |
| BS COV | 0.4437 | - 0.0717 | - 0.0969 |
| SHR DENS | 0.4095 | - 0.0596 | - 0.1404 |
| CARDE DENS | 0.3586 | 0.1626 | 0.1045 |
| MAGUEY DENS | - 0.2506 | - 0.3536 | - 0.0717 |
| PPEAR DENS | - 0.0257 | - 0.3696 | 0.3121 |
| PALMS DENS. | - 0.1699 | - 0.2079 | - 0.3700 |
| SLOPE | - 0.3746 | 0.0977 | - 0.0047 |
| Importance of components | |||
| Comp. 1 | Comp. 2 | Comp. 3 | |
| Standard deviation | 1.7429231 | 1.5177777 | 1.2400455 |
| Proportion of variance | 0.2336755 | 0.1772038 | 0.1182856 |
| Cumulative proportion | 0.2336755 | 0.4108792 | 0.5291649 |
SH COVER= Shrub cover (%), GRA COVER = Grass cover (%), WOODMC = Woody material cover (%), FOR COVER. = forage cover (%), ROCK COV. = Rock cover (%), BS COV = Bare soil cover (%), SHR DENS = Density of shrubs ha -1, CARDE DENS = Density of cardenches ha -1, MAGUEY DENS = Density of maguey ha -1, PPEAR DENS = Density of prickly pear cactus ha -1, PALMS DENS = Density of palms ha-1.
The simple correspondence analysis showed a graphic association of variables that resulted from the PCA with the presence of white-toothed woodrat burrows in the study area, which identified the conformation of three groups that are related with an inertia that explains 37.5% (Figure 2). Groups one and three show that the white-toothed woodrat a preference for prickly pear cactus density, shrub cover and density, rock cover, grass cover and bare ground.
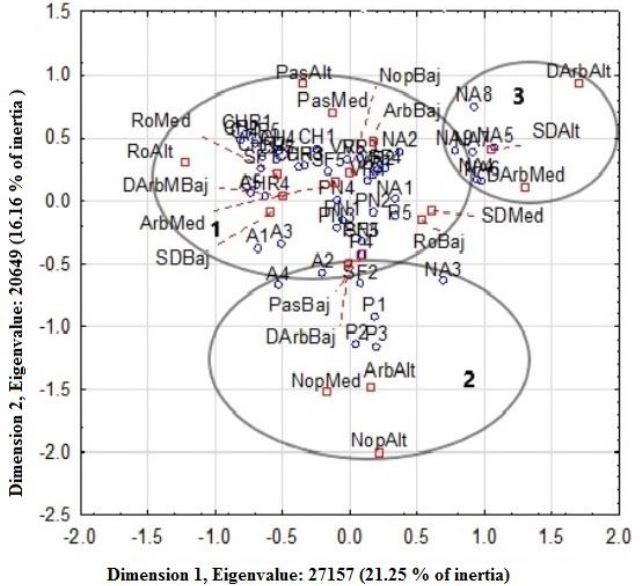
Figure 2 Dimensional representation of the SCA for white-toothed woodrat (Neotoma leucodon) presence and categorical habitat variables in the Potosino-Zacatecan high plateau. ° Sites with burrows and □ habitat variables; DArbMB= shrub density ha-1 very low, DArbBaj= shrub density ha-1 low, DArbMed= shrub density ha-1 medium, DArbAlt= shrub density ha-1high, ArbMed= shrub cover medium, ArbBaj= shrub cover low, ArbAlt= shrub cover high, RoBaj= rock cover low, RoMed= rock cover medium, RoAlt= high rock cover, PasBaj= low grass cover, PasMed= medium grass cover, PasAlt= high grass cover, NopBaj= low cactus density ha-1, NopMed= medium cactus density ha-1 ,SDBaj= low bare soil cover, SDMed= medium bare soil cover, SDAlt= high bare soil cover.
66.7 % of respondents mentioned that the white-toothed woodrat bases its diet on the intake of creeping cactus (O. rastrera), cardon cactus (O. streptacantha) and maguey (Agave spp.); therefore, it is considered as a healthy food free of contaminants. Thirty-six percent of the H-Gs stated that the rat can be hunted throughout the year; however, this activity has decimated its populations, as 42.8 % of the H-Gs stated, 38% mentioned that its abundance has remained the same and only 23.8% mentioned that it has increased. However, the abundance of this rat in the Potosino-Zacatecan high plateau is still unknown, as well as its level of consumption and rate of utilization by the local population.
71.4% of the H-G mentioned that there is one rat per burrow and the rest mentioned that when females are lactating this number increases. Therefore, the H-G mentioned that, in order to maintain an adequate abundance over time, it is important to let them reproduce, maintain the vegetation and establish hunting seasons (Figure 3). time they hunt the rats in their local common lands.
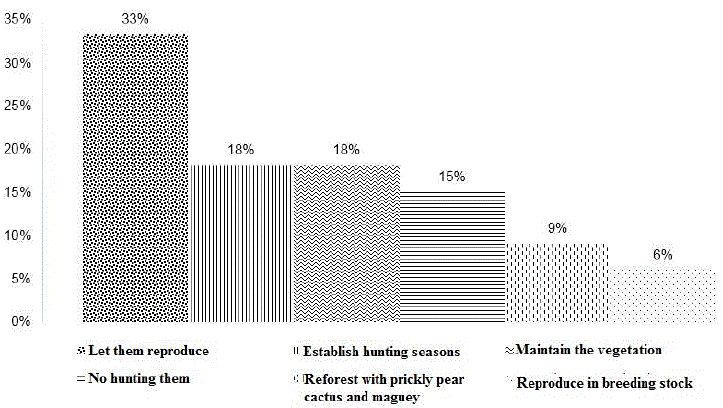
Figure 3 Aspects considered important by the local population to conserve populations of the white-toothed woodrat (Neotoma leucodon) in the Potosino-Zacatecan high plateau
It is important to mention that the H-G consider that hunting the white-toothed woodrat is not a sport; they do it out of necessity and because they consider it for medicinal use. The H-G mention that, even though the rats can be used throughout the year, they extract them when they order them for a sick person or when they wish to consume them.
In some places, rat hunting causes damage to vegetation; this happens when the rats are much hidden and it is necessary to dig deep to find them. However, hunters, aware of the impact of this activity, when they cut the cactus stalks to search for and collect rats, make sure to cover them with soil, which they consider to be reforestation. Likewise, in the field visits that were conducted, most of burrows were located in dry maguey where it is easier to obtain them. On the other hand, the H-G do not destroy the burrow completely, since another rat will nest in it; therefore, most of of the time they hunt the rats in their local common lands.
DISCUSSION
The construction of burrows at the base of its main food sources (Opuntia spp. and Agave spp.) guarantees proximity to its food and avoids antagonistic intraspecific encounters (De Haro & Martínez-Gutiérrez, 2017); in addition, it provides protection during foraging and the energy expenditure for displacement is lower (Atsatt & Ingram, 1983). Another survival strategy of this species is to protect the entrance of their burrows, as they nest, rest, store food and serve as shelter for other invertebrate species (Innes et al., 2007; Whitford & Steinberger, 2010). Protection of the burrow entrance consists of forming mounds of dry plant material, shrub branches and cacti with spines, which allows them to keep predators away, this behavior is also characteristic of N. lepida (Murray & Smith, 2012). N. magister, N. lepida and N. cinerea, select rocky outcrops to build their burrows, as it involves less work to protect their burrow and provides them with various forms of escape (Lombardi et al., 2017).
The density of shrubs and prickly pear cactus in the desert scrub proved to be an important component of white-toothed woodrat habitat. The prickly pear cactus is a source of food and water for the white-toothed woodrat, while shrubs provide it with building material and a diversity of food. In Arizona, N. albigula reached its highest populations at sites where prickly pear cactus is abundant, even when there was no shrub cover and even in open grasslands (Eldridge et al., 2009). Unlike N. leucodon, N. albigula does not associate with prickly pear cactus, but does associate with shrubs (Turkowski & Watkins, 1976) as does N. fuscipes riparia, which correlates positively with plant density, as it builds its burrows under shrub cover (Schooley et al., 2018). In contrast, in California it has been reported that even though shrub density is an important component of microhabitat, it does not determine the location of N. fuscipes (Innes et al., 2007).
Shrub, grasses, rock covers, and the presence of bare ground were important components of white-toothed woodrat habitat. Shrub cover not only provides protection from predation, but also improves moisture and temperature conditions for species survival (Slowik, 2015). Similarly, N. cinerea is associated with cover from conifers and aboveground logs (Lehmkuhl et al., 2006) unlike other studies that mention that Neotoma leucodon does not show an association with plant cover (Markovchick-Nicholls et al., 2008). However, to determine the habitat preference of the white-toothed woodrat, it is necessary to increase the sampling effort, identify shrub and prickly pear cactus species, and determine their importance value index. The cover of grasses is important, because when food is scarce, they function as a food substitute for the white-toothed woodrat (Eldridge et al., 2009). Bare ground cover may not be closely related to the presence of white-toothed woodrat in the habitat; however, this variable could be an indicator of human disturbances such as livestock grazing and erosive effects caused by wind and rain. It is very likely that the association of the white-toothed woodrat with grass and shrub cover is a strategy to avoid antagonistic encounters and competition in its niche.
A significant reduction of white-toothed woodrat was reported in north-central Mexico, where overharvesting reduced its populations to such a degree that local people believed it to be extinct (Martínez-Calderas et al., 2015). However, in the Potosino-Zacatecan high plateau, the abundance of rat is still unknown and there is no information on the exploitation level by the local population. Although internationally, N. leucodon and N. mexicana are in the category of Least Concern, due to their wide distribution and presumed large population (IUCN, 2016a; 2016b), in Mexico these species are not listed in NOM-059-SEMARNAT-2001 (SEMARNAT, 2010).
Even though the white-toothed woodrat is not under any protection status, in order to avoid overexploitation, it is necessary to establish management plans for sustainable harvesting, establish the harvesting season, and standardize hunting methods. In reference to the recommendations of the H-G to maintain white-toothed woodrats, a System of Management Units for the Conservation of Wildlife and reproduction and breeding centers should be established for reintroduction purposes in habitats such as those reported in the present study.
CONCLUSIONS
Local knowledge of the white-toothed woodrat habitat conditions and field research show that the presence of prickly pear cactus and shrubs (huizaches, gobernadora and rama blanca) are a key component of the white-toothed woodrat habitat, as it depends on them for food. Likewise, the prickly pear cactus provides the substrate for the construction of its burrow and the shrubs provide the material, as well as protection against predators and high temperatures. Although in this research maguey was the main substrate where white- toothed woodrats built their burrows, it was not an important component according to the PCA and SCA.
Undoubtedly, the survival success of the white-toothed woodrat is due to its food range, its adaptability to diverse ecosystems and the ability to build its burrows with the materials available in its habitat. The results of this research and considerations of hunter-gatherers suggest maintaining the vegetation in the white-toothed woodrat habitat, as well as elaborating management plans for a sustainable use of the species in the Potosino-Zacatecan high plateau.
ACKNOWLEDGMENTS
To the Postgraduate College Campus San Luis Potosí, for funding the research, to CONACyT for providing financial resources for the first author's master scholarship, to Biologist Alfredo Esparza Orozco for his support, and to the hunter-gatherers of the Potosino-Zacatecan high plateau for their support in the field and for their collaboration in answering the surveys.
REFERENCES
Atsatt PR, Ingram T. 1983. Adaptation to oak and other fibrous, phenolic-rich foliage by a small mammal, Neotoma fuscipes. Oecologia. 60:135-142. https://doi.org/10.1007/BF00379333 [ Links ]
Ceballos G. 2010a. Neotoma leucodon (Rata magueyera). Distribución potencial. http://www.conabio.gob.mx/informacion/metadata/gis/neo_leucgw.xml?_httpcache=yes&_xsl=/db/metadata/xsl/fgdc_html.xsl&_indent=no [ Links ]
Ceballos G. 2010b. Neotoma mexicana (Rata magueyera). Distribución potencial. http://www.conabio.gob.mx/informacion/metadata/gis/neo_mexigw.xml?_httpcache=yes &_xsl=/db/metadata/xsl/fgdc_html.xsl&_indent=no [ Links ]
Cranford JA. 1977. Home range and habitat utilization by Neotoma fuscipes as determined by radiotelemetry. Journal of Mammalogy. ISSN:0022-2372. 58:165-172. https://doi.org/10.2307/1379573 [ Links ]
De Haro S, Martínez-Gutiérrez GP. 2017. Relación espacial entre nopales y madrigueras de rata magueyera Neotoma leucodon en Mapimí, México. Galemys, Spanish Journal of Mammalogy. 29:1-6. https://doi.org/10.7325/84 [ Links ]
Edwards CW, Bradley RD. 2002. Molecular systematics and historical phylobiogeography of the Neotoma mexicana species group. Journal of Mammalogy. 83:20-30. ISSN: 0022-2372.https://doi.org/10.1644/1545-1542(2002)083<0020:MSAHPO>2.0.CO;2 [ Links ]
Edwards CW, Fulhorst CF, Bradley RD. 2001. Molecular phylogenetic of the Neotoma albigula species group: further evidence of a paraphyletic assemblage. Journal of Mammalogy. 82:267-279. ISSN: 0022-2372. https://doi.org/10.1644/1545-1542(2001)082<0267:MPOTNA>2.0.CO;2 [ Links ]
Eldridge DJ, Whitford WG, Duval BD. 2009. Animal disturbances promote shrub maintenance in a desertified grassland. Journal of Ecology. 97:1302-1310. ISSN: 1365- 2745. https://doi.org/10.1111/j.1365-2745.2009.01558.x [ Links ]
Giménez AJ, González CO. 2011. Pisos de vegetación de la Sierra de Catorce y territorios circundantes (San Luis Potosí, México). Acta Botánica Mexicana. 94: 91-123. ISSN: 0187-7151. http://www.scielo.org.mx/pdf/abm/n94/n94a4.pdf [ Links ]
Hall ER. 1982. The mammals of North America. 2nd ed. John Wiley and Sons, New York. Journal of Mammalogy. 63: 718-719. https://doi.org/10.2307/1380296 [ Links ]
Hornsby BS, Ruiz AM, Castleberry SB, Castleberry NL, Ford WM, Wood PB. 2005. Fall movements of allegheny woodrats in harvested and Intact stands in West Virginia. Northern Journal of Applied Forestry. 22:281-284. ISSN:0742-6348 https://doi.org/10.1093/njaf/22.4.281 [ Links ]
INEGI (Instituto Nacional de Estadística, Geografía e Informática). (2021). Mapas de climatología. https://www.inegi.org.mx/temas/climatologia/ [ Links ]
Innes RJ, Vuren V, Dirk H, Kelt DA, Johnson ML, Wilson JA, Stine PA. 2007. Habitat associations of dusky-footed woodrats (Neotoma fuscipes) in Mixed-Conifer Forest of the Northern Sierra Nevada. Journal of Mammalogy. 88:1523-1531. ISSN: 0022-2372. https://doi.org/10.1644/07-MAMM-A-002R.1 [ Links ]
Innes RJ, Vuren V, Dirk H, Kelt DA, Wilson JA, Johnson ML. 2009. Spatial organization of dusky-footed woodrats (Neotoma fuscipes). Journal of Mammalogy. 90: 811-818. ISSN: 0022-2372. https://doi.org/10.1644/08-MAMM-A-126.1 [ Links ]
Iucn Red List of Threatened Species (International Union to Conservation of Nature). 2016a. Neotoma leucodon: Timm R, Álvarez-Castañeda ST, Lacher T. 2016. The IUCN Red List of Threatened Species. International Union for Conservation of Nature. http://www.iucnredlist.org/details/136793/0 [ Links ]
Iucn Red List of Threatened Species (International Union to Conservation of Nature). 2016b. Neotoma mexicana: Linzey AV, Matson J, Pérez S. 2016. The IUCN Red List of Threatened Species. International Union for Conservation of Nature. http://www.iucnredlist.org/details/14590/0 [ Links ]
Ledesma R, Valero-Mora P, Young FW. 2002. Análisis de Homogeneidad en ViSta "The Visual Statistics System". Metodología de las Ciencias del Comportamiento. 4(1): 139-149. https://www.researchgate.net/profile/Ruben-Ledesma-2/publication/313758246_Analisis_de_Homogeneidad_en_ViSta_The_Visual_Statistics_System/links/58a4f93fa6fdcc0e07647da9/Analisis-de-Homogeneidad-en-ViSta-The-Visual-Statistics-System.pdf. [ Links ]
Ledesma R. 2008. Software de análisis de correspondencias múltiples: una revisión comparativa. Metodología de Encuestas. 10:59-75. ISSN: 1575-7803. http://casus.usal.es/pkp/index.php/MdE/article/view/987 [ Links ]
Lehmkuhl JF, Kistler KD, Begley JS. 2006. Bushy-tailed woodrat abundance in dry forests of eastern Washington. Journal of Mammalogy. 87: 371-379. ISSN:0022-2372. https://doi.org/10.1644/05-MAMM-A-053R1.1 [ Links ]
Lombardi JV, Mengak MT, Castleberry SB, Terrell VK. 2017. Mammal occurrence in rock outcrops in Shenandoah National Park: Ecological and anthropogenic factors influencing trap success and co-occurrence. Natural Areas Journal. 37:507-514. ISSN: 0885-8608. https://doi.org/10.3375/043.037.0407 [ Links ]
Markovchick-Nicholls L, Regan HM, Deutschman DH, Widyanata A, Martin B, Noreke L, Ann HT. 2008. Relationships between human disturbance and wildlife land use in urban habitat fragments. Conservation Biology. 22: 99-109. ISSN:1523-1739. https://doi.org/10.1111/j.1523-1739.2007.00846.x [ Links ]
Márquez-Olivas M. 2002. Características reproductivas de la rata magueyera (Neotoma albigula) en cautiverio. Acta Zoológica Mexicana. 86:139-144. ISSN: 0065-1737. http://www.scielo.org.mx/scielo.php?script=sci_arttext&pid=S0065-17372002000200008. [ Links ]
Martínez-Calderas JM, Palacio-Núñez J, Clemente-Sánchez F, Martínez-Montoya JF, Sánchez-Rojas G, Olmos-Oropeza G. 2015. Distribución potencial de la rata magueyera (Neotoma leucodon Merriam 1984) y densidad de madrigueras en el sur del desiertochihuahuense. Therya. 6(2):421-434. ISSN:2007-3364. https://doi.org/10.12933/therya-15-223 [ Links ]
Murray IW, Smith FA. (2012). Estimating the influence of the thermal environment on activity patterns of the desert woodrat (Neotoma lepida) using temperature chronologies. Canadian Journal of Zoology. 90(9):1171-1180. ISSN: 0008-4301. https://doi.org/10.1139/z2012-084 [ Links ]
Nathan N, Muller-Landau N. 2000. Spatial patterns of seed dispersal, their determinants and consequences for recruitment. Trends in Ecology & Evolution. 15:278-285. ISSN: 0169-5347. https://doi.org/10.1016/S0169-5347(00)01874-7 [ Links ]
R CORE TEAM. 2013. Methodology Reference. https://www.eea.europa.eu/data-and-maps/indicators/oxygen-consuming-substances-in-rivers/r-development-core-team-2006 [ Links ]
Riojas-López ME. 2012. Response of rodent assemblages to change in habitat heterogeneity in fruit-oriented nopal orchards in the Central High Plateau of Mexico. Journal of Arid Environments. 85:27-32. ISSN: 0140-1963 https://doi.org/10.1016/j.jaridenv.2012.04.004 [ Links ]
Schooley RL, Bestelmeyer BT, Campanella A. 2018. Shrub encroachment, productivity pulses, and core‐transient dynamics of Chihuahuan Desert rodents. Ecosphere. 9(7):1-18. ISSN: 2150-8925. https://doi.org/10.1002/ecs2.2330 [ Links ]
Schupp EW, Jordano P, Gómez JM. 2010. Seed dispersal effectiveness revisited: a conceptual review. New Phytologist. 188:333-353. ISSN:1469-8137. https://doi.org/10.1111/j.1469-8137.2010.03402.x [ Links ]
SEMARNAT (Secretaría de Medio Ambiente y Recursos Naturales). 2010. Protección ambiental-Especies nativas de México de flora y fauna silvestres-Categorías de riesgo y especificaciones para su inclusión, exclusión o cambio-Lista de especies en riesgo. Norma Oficial Mexicana NOM-059-ECOL-2010. https://www.gob.mx/profepa/documentos/norma-oficial-mexicana-nom-059-semarnat-2010 [ Links ]
Slowik TJ. 2015. Microhabitat and house use by dusky-footed woodrats (Neotoma fuscipes) in Northwestern California: Insight from trapping data. Western North American Naturalist. 75:380-384. ISSN: 1527-0904. https://doi.org/10.3398/064.075.0317 [ Links ]
Solis DM, Gutiérrez RJ. 1990. Summer habitat ecology of northern spotted owls in Northwestern California. The Condor. 92:739-748. ISSN: 0010-5422. https://doi.org/10.2307/1368693 [ Links ]
Sorensen JS, McLister JD, Dearing MD. (2005). Novel plant secondary metabolites impact dietary specialists more than generalists (Neotoma spp). Ecology. 86(1):140-154. ISSN:1939-9170. https://doi.org/10.1890/03-0669 [ Links ]
Turkowski FJ, Watkins RK. 1976. White-throated woodrat (Neotoma albigula) habitat relations in modified pinyon-juniper woodland of southwestern New Mexico. Journal of Mammalogy. 57:586-591. ISSN:1545-1542. https://doi.org/10.2307/1379311 [ Links ]
Villanueva-Hernández AL, Delgado-Zamora DA, Heynes-Silerio SA, Ruacho- González L, López-González C. 2017. Habitat selection by rodents at the transition between the Sierra Madre Occidental and the Mexican Plateau, México. Journal of Mammalogy. 98:293-301. ISSN: 0022-2372. https://doi.org/10.1093/jmammal/gyw173 [ Links ]
Whitford WG, Steinberger Y. 2010. Pack rats (Neotoma spp.): Keystone ecological engineers? Journal of Arid Environments. 74:1450-1455. ISSN:0140-1963. https://doi.org/10.1016/j.jaridenv.2010.05.025 [ Links ]
Received: March 09, 2021; Accepted: August 13, 2021











 text in
text in 



