Attention is the ability to focus on a particular source of information, either located externally in the environment or internally as a mental representation (Chun, Golomb, & Turk-Browne, 2011). Although this cognitive process is a preliminary requirement for explicit memory formation and retrieval, few studies have estimated how the orienting mechanisms of attention may influence episodic memory during encoding (Summerfield & Mangels, 2006; Turk-Browne, Golomb, & Chun, 2013; Uncapher, Hutchinson, & Wagner, 2011) or retrieval (e.g., Burianová, Ciaramelli, Grady, & Moscovitch, 2012; Ciaramelli, Grady, Levine, Ween, & Moscovitch, 2010). The control of orienting could be endogenous, i.e., controlled by voluntary “top-down” mechanisms, or exogenous, being directed by salient external information (“bottom-up”) (Posner, 1980). These mechanisms depend on different but interrelated networks, as revealed by neuroimaging studies (for a review, see Corbetta, Patel, & Shulman, 2008). The endogenous mechanisms rest on the dorsal frontoparietal network, which includes the intraparietal sulcus, where-as the exogenous mechanisms rely on the ventral frontoparietal network, which comprises the temporoparietal junction.
The effects of the orienting mechanisms for attention on episodic memory encoding were examined for the first time by a procedure in which endogenous attention was induced by controlling expectations of stimuli appearance in an electrophysiological study (Summerfield & Mangels, 2006). Words in different font colors were presented immediately after the previous word or after one or two crosshair displays. Top-down mechanisms were activated because after the presentation of two crosshairs, participants knew that a word was expected to be projected. Bottom-up mechanisms were activated when the words were presented immediately after the prior word. Their results revealed that the ability to subsequently recall the word and its font color was superior in the top-down trials compared to the bottom-up trials. This procedure has a disadvantage in that the bottom-up trials may be confounded with top-down mechanisms because there was a certain probability that the next word would appear immediately. Moreover, memory was affected in both orienting attention trials by the kind of event that followed each word; the immediate display of another word may interfere with the encoding process, whereas the appearance of a crosshair provided further encoding time.
Two functional magnetic resonance imaging (fMRI) studies (Turk-Browne, et al., 2013; Uncapher et al., 2011) used the Posner cueing paradigm (Posner, 1980) to assess the effects of orienting attention on episodic memory encoding. This classic paradigm consists of presenting a stimulus in one of two possible locations; the stimulus may appear in the position indicated by the preceding cue (valid cue) or it may not (invalid cue). Both studies found that memory performance was superior when the stimuli were encoded with valid cues rather than with invalid cues. Only one of these studies (Uncapher et al., 2011) examined recall processes by asking participants to indicate the position on the screen where the images appeared during encoding. The other study (Turk-Browne et al., 2013) used a recognition old/new task in which episodic memory performance may be supported by both familiarity and recollection processes (Cansino et al., 2015). Recollection is the ability to retrieve the contextual details of a previous experience, whereas familiarity is based on the impression that the experience has previously occurred without further details (Yonelinas, 2002). Contextual details are the specific circumstances in which an experience or episode took place, such as the moment, the place or the emotional state of the individual. Because this information is conceived as the origin of a memory, its recall is referred to as source memory. Hence, testing source memory allows the objective measurement of recollection because participants are required to judge in which specific context (source, such as its location or timing) an item was previously presented during an encoding phase.
According to numerous fMRI studies (for a review see Corbetta & Shulman, 2002), valid cuing trials generate brain activity in areas of the dorsal network, whereas invalid cues elicit activity in regions of the ventral network. These findings confirm that the cueing paradigm is suitable to distinguish the effects of endogenous and exogenous attention mechanisms on memory. However, to achieve this purpose, the changes in attention should be measured without eye or head movements (Posner, 1980), because the cueing paradigm is designed to measure covert attention, i.e., attention without eye movement, instead of overt attention. This is important because the cueing procedure’s main measure is based on reaction times; therefore, to avoid increasing reaction times due to time being spent on overt responses, such as shifting of the eyes, it is mandatory to control for eye movements, a procedure that was absent in some previous studies (Uncapher et al., 2011). As reported by Posner (1980), cover attention denotes the ability to shift attention without the support of the visual system and depends on central mechanisms such as those controlled by the parietal cortex; however, this central control is not entirely autonomous because it receives inputs from regions that control overt attention.
Likewise, the cueing method that is used to promote endogenous attention should be considered. For example, the use of arrows as cues is inconvenient because these conventional signs are likely to engender an automatic response; thus, they may produce an orienting response that contains both endogenous and exogenous mechanisms (Ristic & Kingtone, 2012). Consequently, symbolic cues, such as colors, are recommended to guarantee the induction of pure endogenous attention mechanisms. The effects of orienting attention on memory have so far been investigated using arrows (Uncapher et al., 2011) or faces looking to the left or right (Turk-Browne et al., 2013), which are over-learned symbols (Brignani, Guzzon, Marzi, & Miniussi, 2009), as cues.
Findings from these previous studies showed that endogenous orientation benefits the encoding of information into episodic memory more than does exogenous orientation. However, the effects of both kinds of mechanisms on memory performance have not been measured under equivalent conditions (Summerfield & Mangels, 2006) and symbolic cues have not yet been used. Therefore, whether endogenous or exogenous attention is a better predictor of episodic memory remains an open question. The current study builds on these previous findings by assessing which of these orienting mechanisms enhances recollection for spatial contextual details to a greater extent. To achieve this goal, we used the cueing paradigm with symbolic cues and controlled for eye movement. The encoding of recollection was examined under endogenous (valid cues) and exogenous (invalid cues) orienting attention mechanisms, and during neutral trials (uncued). Moreover, during encoding, we used a detection task that consisted of identifying whether the images that were used as stimuli were horizontally symmetrical, i.e., if their left and right sides had the same shape. We used this task because it relies on finer perceptual processes without generating the reorientation to internal structures, as occurs with complex tasks that demand semantic processes. In fact, these types of tasks tend to eliminate the effects of the cueing procedure (Posner, 1980). Previous studies have used detection tasks that only required the identification of stimuli that represent real objects (Uncapher et al., 2011) or real scenes (Turk-Browne et al., 2013), a procedure that may produce only shallow encoding processes. Enhancement of the perceptual processes increases the encoding of the stimuli and thus their memorability.
We hypothesized that recollection of information encoded under endogenous orientation would be superior to that of information encoded under exogenous orientation, as was observed in previous studies (Summerfield & Mangels, 2006; Uncapher et al., 2011). In turn, recollection of information encoded under neutral trials would be situated between that of information encoded under endogenous and exogenous orienting attentions; we expect this outcome because in the uncued trials, participants would be oriented to receive information from any of the two possible locations. As in the current study, one previous study (Turk-Browne et al., 2013) also used neutral cues, but for the purpose of assessing recognition rather than recollection.
Method
Participants
Thirty healthy right-handed adults (15 women) participated in the study. Participants were students recruited from the Psychology Faculty of the National Autonomous University of Mexico. Their mean age was 23.3 years (SD = 2.2) and their mean level of formal education was 15.3 years (SD = 1.9). All participants had normal or corrected-to-normal visual acuity, as measured with the Snellen chart, and no color-blindness, as measured with a short version of the Ishihara Color Blindness Test (Ishihara, 2003). The research protocol was performed in accordance with the principles stated in the Declaration of Helsinki. All participants provided written informed consent.
Stimuli
A total of 560 color images of common objects were used in the experiment. Half of the images (280) were symmetrical, while the other half were asymmetrical. An image was considered symmetric if its left and right sides were identical. The images had a horizontal visual angle ranging from 0.78º to 3.07º and a vertical visual angle between 0.35º and 3.07º. Four hundred images were randomly selected from the total set of images to be presented during the encoding task (200 were symmetrical and 200 were asymmetrical). From this set, 240 images were used during the valid trials, 80 were presented during the invalid trials and 80 during the neutral trials. The trials were divided into five blocks, each containing a total of 80 trials consisting of 48 valid trials, 16 invalid trials and 16 neutral trials. The same proportion of symmetrical images was included in each type of trial. During retrieval, 320 images that were previously presented during the encoding task (old images) were used; 160 were randomly selected from the 240 images displayed during the valid trials, 80 images were presented during the invalid trials, and 80 were used in the neutral trials. In addition, 160 new images were presented during retrieval. These trials were distributed throughout the five blocks for a total of 96 trials each, which included 32 valid old images, 16 invalid old images, 16 neutral old images, and 32 new images. Circles were used as cues (diameter visual angle of 0.5º), with blue and orange circles being used during valid and invalid trials to indicate if the stimuli would appear in the left or in the right side. The color used for each side was counterbalanced across participants. In addition, a blank circle with a black contour was used in the neutral trials.
Encoding task
Two black frames (horizontal and vertical visual angles of 3.29º) with a thickness of 0.07º were permanently projected on the screen. The frames were displayed to the left and to the right of the center of the screen and were separated by 2.57º. The images were projected inside one of these frames. The whole experiment was presented on a white background. Each trial began with a cross as a fixation point (diameter visual angle of 0.5º) placed at the center of the screen. The cross was displayed randomly for 200 ms, 300 ms or 400 ms. Immediately after, the cue was presented at the center of the screen for 700 ms, 800 ms or 900 ms, intervals that were also randomly selected. The color of the cue indicated the frame (left or right) where the image would be presented. Then, a cross replaced the cue, and the image was displayed inside one of the frames for 1000 ms, followed by a blank screen that was presented for 1000 ms. Afterwards, a black circle (diameter visual angle of 0.5º) was presented for 1500 ms to allow the participants to rest their sight. Trials could last a minimum of 4400 ms and a maximum of 4800 ms. The task consisted of indicating whether the image was symmetric or not as fast as possible. Participants were able to provide their response during the 2000 ms interval following the onset of the image presentation. Two response panels each with two buttons were used; one key was located in the superior part of the panels and was to be pressed by the index finger, and the other button was positioned in the inferior part of the response panels and was to be pressed by the thumb. Only the upper buttons were used during encoding, and they were assigned in a counterbalanced manner across participants; half of the participants used their left index finger to indicate that the images were symmetrical, and the rest used their right index finger.
Retrieval task
Each trial began with the presentation of a cross for 200 ms, as a fixation point. Then, a blank screen was displayed for 200 ms, followed by the presentation of an image at the center of the screen for 1000 ms. Then, a blank screen was displayed for 2000 ms. Afterwards, a black circle was presented for 1500 ms to allow the participants to rest. Participants could respond during the period of 3000 ms after the onset of the image. The task consisted of identifying whether the image was old or new; if it was old, participants had to indicate whether the image was presented in the left or in the right frame during encoding. Participants used their left index finger to signal that the images were presented to the left of the center of the screen and their right index finger if it was displayed to the right. If participants were unable to retrieve the side of the screen that the image was presented on but were sure that the image was old (previously seen at encoding), they indicated this by pressing one of the lower buttons on the response panel. To indicate that the image was new, participants pressed the lower button of the other response panel. The lower button assigned to each type of response was counterbalanced across participants.
Procedure
During the first session, which lasted approximately 30 min, the participants’ visual acuity and color-blindness were measured. If participants fulfilled all the inclusion criteria, they performed a task to learn the meaning of the color cues in a sound-dampened room. For the whole experiment, participants remained seated in an armchair at distance of 80 cm from the computer screen. The response panels were placed on platforms located on each armrest. During the learning task and the encoding phase, each participant’s head was placed in a metal structure where they positioned their forehead and chin to avoid head movements. The learning task was identical to the encoding task except that instead of presenting images, an asterisk was presented in one of the frames, and only valid cues were used. Participants were requested to peripherally view, without an overt response, the left or right frame, according to the cue’s color. Half of the participants learned that the blue cue indicated that the image would appear in the left frame and that the orange cue indicated that it would appear in the right frame. For the other half, the color of the cues indicated the opposite location.
During the second session, participants performed the encoding and retrieval phases in the same sound-dampened room. In the encoding phase, participants wore eyeglass frames containing an infrared light and a camera that served to register eye movements. Eye tracking was recorded with the EyeTracker system (Arrington Research Inc., Arizona, USA) and the ViewPoint EyeTracker PC-60 software from the same company. Before the encoding phase, participants performed a brief practice task to become familiar with the encoding task, the response panels and the eye-tracker recording. Participants were instructed to avoid head and eye movements during the encoding task, to fixate their sight on the events occurring at the center of the screen and to indicate whether the images were symmetrical or not by using their peripheral vision only. Once the encoding task was complete, participants performed the retrieval task after a delay of ten minutes. Participants did not know that their memory would be tested; thus, the retrieval task was incidental. During retrieval, participants were instructed to fixate their sight at the middle of the screen and to concentrate on the source memory judgment. The experiments were controlled with the E-Prime v2.0 software (Psychology Software Tools Inc., Pennsylvania, USA).
Statistical Analysis
Trials with horizontal eye movements greater than 1.28º from the fixation point were discarded from all analyses. Correct responses during the encoding task, correct source memory and correct recognition during the retrieval task were submitted to repeated measures analysis of variance (ANOVA) with the factor trial type (valid cues, invalid cues and neutral cues). The same analyses were conducted on reaction times for these responses. The Tukey honestly significant difference (HSD) test was used for post hoc comparisons. When necessary, the degrees of freedom were corrected using the Green-house-Geisser procedure. For these cases, the original degrees of freedom, the Greenhouse-Geisser co-efficient (ε) and the corrected probability levels are reported. The significance level was set to p < .05.
Results
The result of the repeated measures ANOVA conducted on the correct responses during encoding with the factor trial type (valid cues [mean ± SE]: 83.82% ± 2.48; invalid cues: 83.58% ± 2.45; neutral cues: 82.21% ± 2.44) was not significant, F (2, 58) = 0.74, p = .48. By contrast, the result of the analysis computed with the reaction times was significant, F (2, 58) = 17.90, p < .001, ε = .84, η2 =.38. Post hoc analyses revealed that the reaction times during the invalid trials differed significantly from those during the valid and neutral trials, whereas the reaction times during the valid trials did not differ from those during the neutral trials (Figure 2).
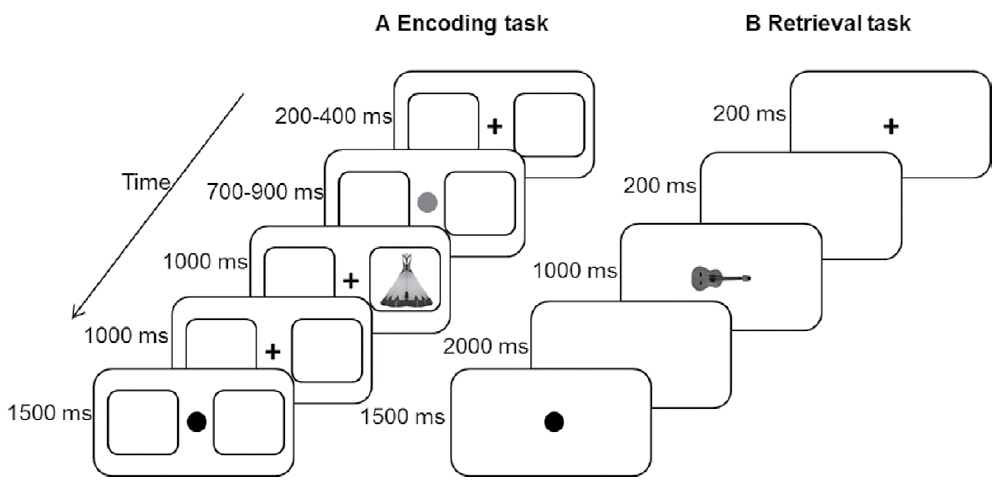
Figure 1 Events in each trial and their duration for the encoding (A) and retrieval (B) tasks. During encoding, participants could respond for a period of 2000 ms after the onset of the image, whereas during retrieval, the time to answer lasted 3000 ms after the onset of the image.
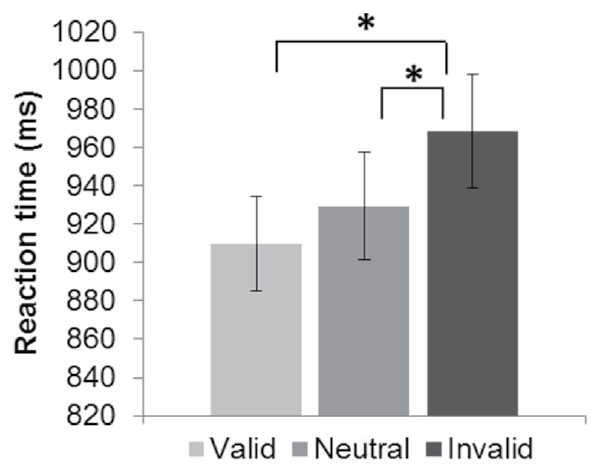
Figure 2 Reaction times (RT) during the encoding task for each type of trial: valid, neutral and invalid cues. Lines indicate significant differences (p < .05) between trial types. Error bars represent standard error
The percent of correct recognition responses during retrieval was computed for those stimuli that were identified as old (images that received an incorrect or correct source memory response and images whose context was forgotten). The result of the analysis conducted on these responses was significant, F (2, 58) = 6.63, p = .003, ε = .94, η2 =.19; post hoc analyses revealed that recognition for the images encoded in the neutral trials was superior to that for the stimuli encoded in the invalid trials, but recognition for the images encoded in the valid trials did not differ significantly from that for the stimuli encoded in the invalid and neutral trials (Figure 3). Reaction times during correct recognition responses did not differ significantly, F (2, 58) = 0.79, p = .46, between the images encoded in the different trial types (valid cues: 1318 ± 58; invalid cues: 1334 ± 62; neutral cues: 1334 ± 61).
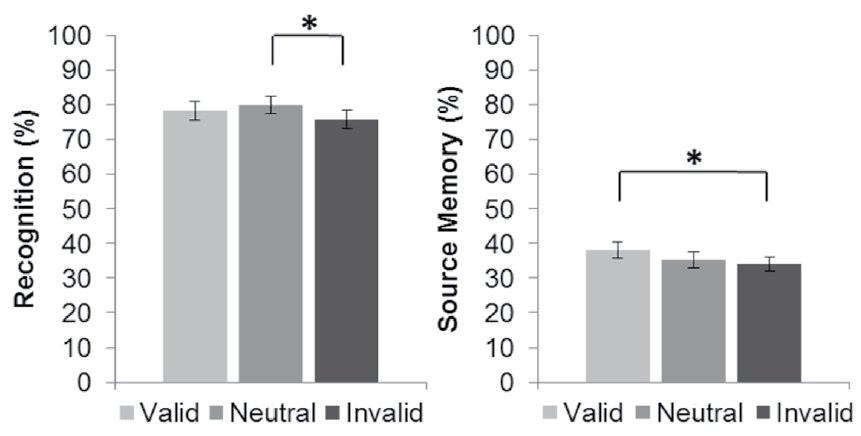
Figure 3 Recognition and source memory performance during the retrieval task for each type of trial: valid, neutral and invalid cues. Lines indicate significant differences (p < .05) between trial types. Error bars represent standard error
Source memory was measured as the percent of recognized stimuli that received a correct source response. The result of the analysis computed on correct source memory responses was significant, F (2,58) = 4.59, p = .01, ε = .98, η2 = .14; post hoc analyses revealed that source memory for the images encoded in the valid trials was superior to that for those encoded in the invalid trials, but source memory for the images encoded in the invalid trials did not differ significantly from that for the images encoded in the neutral trials (Figure 3). Reaction times during correct source responses did not differ significantly between images encoded in the different trial types, F (2, 58) = 0.39, p = .68, (valid cues: 1233 ± 56; invalid cues: 1248 ± 63; neutral cues: 1245 ± 58). The percent of images with an incorrectly retrieved source and those without any source retrieval differed across trial types, F (2,58) = 3.53, p = .04, ε = .94, η2 = .11 (valid cues: 31.21% ± 1.75; invalid cues: 33.62% ± 2.19; neutral cues: 35.44% ± 2.41). Post hoc analyses revealed that these responses were superior for the images encoded during the neutral trials to those for the images encoded during the valid trials, but for the images encoded during the invalid trials, these responses did not differ significantly from those for the images encoded during the valid and neutral trials.
The mean percent of correct rejections (correct identification of new images) and their reaction times were 64.16% ± 3.44 and 1317 ± 55, respectively. The mean percent false alarms (new image judged as old) and their reaction times were 35.84% ± 3.44 and 1497 ± 68, respectively. The percentage of trials that were eliminated due to eye movements was 3.02% for the valid trials, 2.88% for the invalid trials and 3.67% for the neutral trials. Additionally, images that received an incorrect response during encoding or no response, either during encoding or retrieval, were removed from all analyses (encoding: valid cues 3.86%; invalid cues 4.25%; neutral cues 4.29%; retrieval: valid cues 2.32%; invalid cues 3.03%; neutral cues 2.78%).
Discussion
The main finding of the current study was that information encoded under endogenous orienting attention enhances the ability to retrieve the spatial context in which the information was learned, whereas the encoding of information under non-orienting conditions (uncued trials) benefits the ulterior recognition of information without contextual details. Conversely, encoding information under exogenous orienting attention not only promotes less source accuracy and recognition but also increases the complete forgetfulness of information because the number of misses rose.
As expected, recollection is more likely to occur if our attention has been voluntarily oriented to the location where the event should arise. This out-come is in agreement with the two previous studies that had examined source memory under different orienting attention conditions (Summerfield & Mangels, 2006; Uncanpher et al., 2011). However, the current study provides further evidence on the relevance of endogenous attention for recollection formation because this orienting mechanism was generated by means of symbolic cues, which promote pure voluntary attention. In contrast, previous studies (Turk-Brown et al., 2013; Uncanpher et al., 2011) have used arrows or overlearned symbols that may have engaged a mix of endogenous and exogenous mechanisms. Moreover, the fact that incorrect source responses, which represented responses based on familiarity processes, were superior for information encoded during the neutral trials compared to those encoded during the valid trials indicates that endogenous attention is particularly relevant to achieve the encoding of episodic representations based on recollection but not for those based on familiarity.
The mere recognition of a previous experience without its spatial location was enhanced for information encoded in the neutral trials compared to that which was encoded in the invalid trials. This indicated that when orientation is freely set to all possible locations, the encoding of the item, but not the encoding of the details surrounding the item, is enhanced. By contrast, the need to reorient attention during the invalid trials clearly disturbed episodic memory encoding, since misses were superior for images encoded in these trials compared to those encoded in the neutral trials. These findings support our hypothesis that neutral trials would have an intermediate effect on episodic memory performance, between that generated by the valid and invalid trials. Conversely, Turk-Brown et al. (2013) did not find a recognition advantage during neutral trials compared to invalid trials, probably because the face used as the neutral cue was looking directly at the participants, inhibiting the orientation to any location, and was an issue discussed by the authors of this study.
The finding that endogenous orientation enhanced recollection whereas freely orienting attention (uncued trials) increased familiarity may indicate that different orienting mechanisms promote different episodic memory processes. This would be the case if recollection and familiarity are conceived as categorically different processes (Brown & Aggleton, 2001; Mandler, 1980), but not if they are considered as quantitatively different processes (Donaldson, 1996; Hirshman & Master, 1997); the latter would suggest that endogenous and exogenous orientation influence the encoding of different amounts of episodic information.
Moreover, the effects of orienting attention on episodic memory were observed after the occurrence of these mechanisms was clearly demon-strated during encoding, as reaction times were significantly longer during the invalid trials, which was expected in the cueing paradigm (Posner, Nissen, & Ogden, 1978; Posner, Snyder, & Davidson, 1980; Riggio, Bello, & Umilta, 1998). However, reaction times in the valid and neutral trials were equivalent, as was previously observed when symbolic cues were used with this paradigm (Botta, Lupiáñez, & Chica, 2014), indicating that the real cost on covert attention (without eye movements) occurs only during invalid cues. This outcome mirrors the effects observed on the retrieval task because items learned under exogenous orientation had a decreased probability of being subsequently remembered, suggesting that the cost of reorienting affected not only covert attention but also the encoding of information.
The outcome that exogenous orientation reduced the ability to encode episodic representations in the current study is not in agreement with the fact that salient stimuli may elicit greater attention, thereby increasing the memorability of their memory trace (Kamp, Brumback, & Donchin, 2013). One possibility that may explain the decreased memorability of items learned under bottom-up conditions could be that, in the present study, all the stimuli we used were equivalent and emotionless, whereas the salient effect on memory has mostly been observed with arousing (Sharot & Phelps, 2004) or emotional (Chainay, Michael, Vertpré, Landré, & Plasson, 2012) stimuli. Another possibility could be that after reorienting attention in the invalid trials, the remaining time was insufficient to successfully encode the images.











 text new page (beta)
text new page (beta)


