Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Revista latinoamericana de química
Print version ISSN 0370-5943
Rev. latinoam. quím vol.38 n.2 Naucalpan de Juárez Aug. 2010
Antibacterial activity of Ibicella Lutea glycosides
María E. García Da Rosa1, Cristina Olivaro1, María Pía Cerdeiras2 and Álvaro Vázquez1*
1 Cátedra de Farmacognosia, Departamento de Química Orgánica, Facultad de Química, Universidad de la República (UDELAR), Montevideo, Uruguay. *Corresponding author: Dr. Álvaro Vázquez, Cátedra de Farmacognosia, Facultad de Química, UDELAR, CC1157, Montevideo 11800, Uruguay. Phone: + 598 2 9244069 Fax: + 598 2 9241906 e–mail: avazquez@fq.edu.uy
2 Cátedra de Microbiología, Departamento de Biociencias, Facultad de Química, Universidad de la República (UDELAR), Montevideo, Uruguay.
Received March 2010.
Accepted August 2010.
ABSTRACT
In spite of the great advances in chemotherapeutics, infectious diseases are still one of the leading causes of death in the world. Amongst the most problematic clinically relevant pathogens at present, methicillin–resistant Staphylococcus aureus (MRSA) ranks as one of the most difficult bacteria to treat. Interest in plants with antimicrobial properties has revived as a consequence of current problems associated with the use of antibiotics.
The objective: was to evaluate the antibacterial activity of a compound isolated from Ibicella lutea (Lindl.) Van Eselt. (Martyniaceae) and some of its derivatives, against methicillin–sensitive and MRSA.
Compound 1 was isolated and identified as previously described and minimal inhibitory concentration (MIC) of 1 and simple analogs were determined by the microdilution technique.
Compound 1 (11–hydroxystearic acid 11–0–(6–0–acetyl–β–D–grucopyranoside) showed a MIC of 0.06 mM for S. aureus ATCC 6538p. All the synthetic analogs and precursors presented higher MIC values. Compound 1 showed antimicrobial activity against different strains of MRSA.
These results suggest that the glycosidic and acidic moieties are necessary for the antimicrobial activity under study.
Keywords: Martyniaceae, S. aureus, MRSA.
RESUMEN
A pesar de los grandes avances en la quimioterapia de las enfermedades infecciosas estas están lejos de ser controladas y más aún erradicadas y son aun una de las principales causas de muerte en el mundo. En especial Staphylococcus aureus meticilino–resistente (MRSA) se ha mostrado como una bacteria extremadamente problemática desde el punto de vista terapéutico.
A consecuencia de los problemas encontrados en la terapéutica con antibióticos, existe hoy en día un renovado interés en productos naturales antimicrobianos.
En este trabajo se presentan los resultados del estudio de la actividad antibacteriana de compuestos aislados de Ibicella lutea (Lindl.) Van Eselt. (Martyniaceae) y algunos derivados sencillos del mismo contra cepas sensibles y resistentes de Staphylococcus aureus.
El compuesto 1 fue aislado e identificado según las técnicas anteriormente descritas y la concentración mínima inhibitoria (MIC) de 1 y sus análogos fue determinada por una técnica de microdilución.
El compuesto 1 (11–hydroxiestearil 11–0–(6–0–acetil–β–D–glucopiranósido) mostró una MIC de 0.06 mM para S. aureus ATCC 6538p. También mostró actividad antibacteriana contra diferentes cepas de MRSA mientras que todos los análogos de 1 tuvieron MICs mayores que el producto natural. Estos resultados sugieren que tanto la función acido como la estructura glicosídica son necesarias para la actividad.
Palabras clave: Martyniaceae, S. aureus, MRSA.
INTRODUCTION
Infectious diseases are still one of the leading causes of death in the world, in spite of the great advances in chemotherapeutics. The World Health Organization (2003) states that infectious and parasitic diseases accounted for nearly 11 million out of the total of 57 million deaths in 2003.
Although there are a great array of antibacterial and antifungal drugs in clinical use, the increase of resistant organisms sometimes makes them ineffective or allows disease recurrence (Michel and Gutmann, 1996).
Among the most problematic clinically relevant pathogens at present, methicillin–resistant Staphylococcus aureus (MRSA) ranks as one of the most difficult bacteria to treat (Cosgrove et al., 2003).
The use of higher plants and preparations made from them to treat infections is an age–old practice in a large part of the world population, especially in developing countries, where traditional medicine is used for a variety of diseases. Interest in plants with antimicrobial properties has revived as a consequence of current problems associated with the use of antibiotics (Cherigo et al., 2009; Po Shiu and Gibbons, 2006).
Ibicella lutea (Lindl.) Van Eselt. (Martyniaceae) is the sole species of the genus Ibicella from the small Martyniaceae family comprising just 3 genera (Martynia, Proboscidea and Ibicella) with 13 species (Van Eseltine, 1929; Bretting, 1984).
In Uruguay, where the plant grows freely, it is called "Cuerno del Diablo" (Devil's horn) and its infusion is used in popular medicine as an antimicrobial for the treatment of eyes and skin infections (Alonso Paz et al., 1995 and references therein).
In previous work we studied the antimicrobial activity of aqueous and ethanol extracts of several Uruguayan medicinal plants. We found that I. lutea indeed has an important activity, deserving further study (Olano et al., 1996), and we isolated one of the active principles, 11–hydroxystearic acid 11–0–(6–0–acetyl–β–D–glucopyranoside) (Cerdeiras et al., 2000).
In this paper we report the antibacterial activity of this compound along with some of its derivatives against methicillin–sensitive and MRSA.
MATERIALS AND METHODS
General analytical methods
Gas chromatography (GC) analysis was performed in a Shimadzu GC 14 apparatus with an SE–52 column using a temperature program from 100° C to 280° C with a 5° C min–1 gradient. A Brucker micrOTOF–Q–TOF with ESI source in positive mode was used for mass spectra (MS) and a Shimadzu QP 5050 with a SE 52 column was used for the GC–MS analysis.
1H NMR spectra were obtained at 400MHz and 13C NMR spectra at 100MHz on a Brucker Advance DPX 400 spectrometer, using CD3OD or CDCl3 as solvents, and TMS (δH 0.00) and acetone (δC 31.00) as references. Two dimensional (different H, H–COSY, H, C–COSY, HMBC, HMQC and NOESY) experiments were carried out with programs available for the Brucker software.
Plant material
Plant material was collected in Pajas Blancas, 6 Km outside Montevideo at . 34° 51" 58.33"S, 56° 2005.38" W. The material was identified by Lic. E. Alonso Paz, Botany Department, Facultad de Química, UDELAR and voucher specimens (Leg. Alonso Paz et Bassagoda N° 2546) were kept in the MVFQ Herbarium, Montevideo.
Compounds
Compound 1 was isolated and identified as previously described (Cerdeiras et al., 2000). Compound 2 and 3 were obtained by acetylation of 1 with Ac2O/Pyridine and mild alkaline hydrolysis respectively. Compound 4 was synthesized from α–bromo–tetra–acetyl–glucose and methyl 11 –hydroxyoctadecanoate by the classical Koenigs–Knorr method using Ag2O as catalyst (Talley, 1963). The resulting compounds were identified by different GC–MS, HRMS and NMR techniques. Compounds 5 (12–hydroxy–9–octadecenoic acid) and 6 (methyl 11–hydroxyoctadecanoate) were purchased from Sigma.
Microbiological analysis
The strains used were Staphylococcus aureus ATCC 6538p and the clinical mul–tiresistant isolates S. aureus CCMC (Colección Cátedra Microbiología Clínica) 15027, 16369 and 19016
Minimal inhibitory concentration (MIC) was determined for the extracts that inhibited growth by the microdilution technique according to Elloff (1998) using 100 μl of Mueller–Hinton broth (Difco), 100 |il of twofold dilutions of the extracts (10 mgmL–1) and 10 μl of a suspension (108/mL) of the microorganisms. The trays were incubated (24 h, 37C) and developed with p–iodo–nitrotetrazolium violet (INT) (Sigma) 0.1% solution. Bacterial growth was indicated by development of a red formazan color. Gentamicin was used as control.
RESULTS AND DISCUSSION
Compound 1 had previously demonstrated very promising antimicrobial activity (Cerdeiras et al., 2000) against reference strains, in particular against susceptible S. aureus ATCC 6538p.
To see if the antimicrobial activity could be enhanced by some simple structural modifications, we prepared derivatives 2, 3 and 4 (Fig. 1). We also tested the free acid 5 and the methyl ester 6.
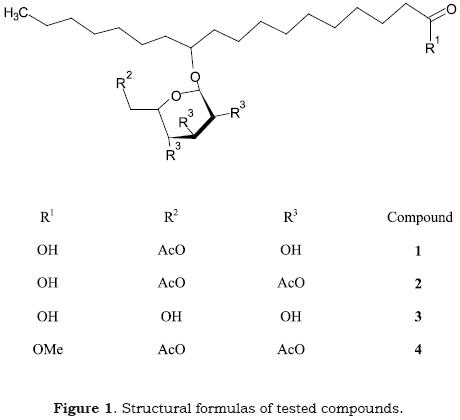
The results of the antibacterial activity of the compounds against S. aureus ATCC 6538p are depicted in Table 1.
The analysis of these results suggests that both the free acid function and the glycosidic moiety are indispensable for the activity. Compounds 4 and 6 which are methyl esters lack any antibacterial activity. Also the free acid 5 has a MIC that is more than 15–fold greater (on a mol/L basis) than compound 1 .
When MICs for compounds 1 , 2 and 3 are compared it is also clearly seen that the natural monoacetyl compound is 10 times more active than the tetra–acetyl and desacetyl derivatives.
More studies are needed to determine if this is due to solubility properties of the molecules or to a specific interaction with the bacterial target.
Compound 1 was also tested against S. aureus clinical isolates that showed different antibiotic resistance profiles, but were all MRSA. The phenotypic susceptibility profile of these strains is shown in Table 2. Strain 16369 is a hospital acquired isolate and strains 15027 and 19016 are community acquired infection isolates. The MICs obtained with these strains were 250, 500 and 62.5 μg/mL respectively.
CONCLUSIONS
The present work clearly shows that higher plants are still a primary source in the search of lead molecules for novel antimicrobial compounds.
The original compound 1 was the one that presented the highest activity against the S. aureus ATCC 6538p strain. But, more studies are needed to determine if this action is due to solubility properties of the molecule or to a specific interaction with the bacterial target.
Even though the MIC results obtained with the MRSA strains were relatively high, compound 1 is still of interest as a lead compound given that it is a simple and easily synthesized molecule.
REFERENCES
Alonso Paz, E., Cerdeiras, M.P., Fernández, J., Ferreira, F., Moyna,P., Soubes, M., Vázquez, A., Vero, S., Zunino, L: (1995) Screening of Uruguayan medicinal plants for antimicrobial activity. Journal of Ethnopharmacology. 45: 67–70. [ Links ]
Bretting, P.K. (1984) Folk names and uses for martyniaceous plants. Economic Botany. 38: 452–463. [ Links ]
Cerdeiras, M.P., Fernández, J., Soubes, M., Vero, S., Ferreira, F., Moyna, P., Olano, I., Vázquez, A. (2000) A new antibacterial compound from Ibicella lutea. Journal of Ethnopharmacology. 73: 521–525. [ Links ]
Cherigo, L., Pereda–Miranda,R., Gibbons, S. (2009) Bacterial resistance modifying tetrasaccharide agents from Ipomoea murucoides. Phytochemistry. 70: 222–227. [ Links ]
Cosgrove, S.E., Sakoulas, G., Perencevich, M.J., Schwaber, A.W., Karchmer, L., Carmeli, Y. (2003) Comparison of mortality associated with methicillin–resistant and methicillin–susceptible Staphylococcus aureus bacteremia: A meta–analysis. Clinical Infectious Diseases. 36: 53–59. [ Links ]
Michel, M., Gutmann, L. (1996) Methicillin–resistant Staphylococcus aureus and vancomycin–resistant enterococci: Therapeutic realities and possibilities. Lancet. 349: 1901–1906. [ Links ]
Olano, I., Alonso Paz, E., Cerdeiras, M.P., Fernández, J., Ferreira, F., Moyna, P., Soubes, M., Vázquez, A., Vero, S., Bassagoda, M.J. (1996) Screening of Uruguayan medicinal plants for antimicrobial activity. Part II. Journal of Ethnopharmacology. 53: 111–115. [ Links ]
Po Shiu, W.K., Gibbons, S. (2006) Anti–staphylococcal acylphloroglucinols from Hypericum beanii. Phytochemistry. 67: 2568–2572. [ Links ]
Van Eseltine, G.P. (1929) A preliminary study of the unicorn plants (Martyniaceae). Technical Bulletin 149, NYS Agricultural Exp. Station Geneva, Switzerland pp. 3–41. [ Links ]
WHO. (2003) The world health report 2003–Shaping the future. World Health Organization, Geneva, Switzerland pp. 3–22. [ Links ]














