INTRODUCTION
The reduction of aldehydes and ketones is one of the most important reactions to produce alcohols that are used to synthesize industrially important chemicals such as pharmaceuticals, agrochemicals, natural products, scented substances for cosmetic fragrances, and the flavor industry (Salvano, Cantero, Vázquez, Formica & Aimar, 2011).
The reduction of benzaldehyde is used to obtain benzyl alcohol (BA), that is widely used in a variety of end-use industries; for example, in cosmetic formulations as well as in non-cosmetic products such as household cleaners and detergents (Scognamiglio, Jones, Vitale, Letizia & Api, 2012;U.S. National Library of Medicine, 2015). BA itself confers a floral scent, and is frequently employed as a precursor to synthesize a variety of esters used as flavors or fragrances (U.S. National Library of Medicine, 2015).
In the area of health care, BA is employed as a preservative in intravenous medication and topical drugs (Acofarma, 2015), and at low concentrations it is applied as a local anesthetic and antipruritic (Acofarma, 2015). The main use of benzyl alcohol in pharmaceuticals is as pediculicide (Meinkinget al., 2010).
Due to its polarity, low toxicity, and low volatility, BA is a very useful, safe, and efficient solvent for paints, coatings, lacquers, and inks; as a solvent in special applications, like curing agent formulations for epoxy resins of coatings, flooring systems, and adhesives. It is also applied in photo developer systems and metal cleaning formulations. In the nanotechnological industry, BA serves as a dielectric solvent for the dielectrophoretic reconfiguration of nanowires (Snowhite Chemical Co., 2015;U.S. National Library of Medicine, 2015).
Benzyl Alcohol is one of the aroma constituents ofCamellia sinensis (Ogawaet al., 1995),Rhododendron simsii, R. scabrum(Godefroot, Sandra & Verzele, 1981),Nicotiana otophor, Nicotiana sylvestris, apple (Malus domestica, (Loughrin, Hamilton-Kemp, Andersen & Hildebrand, 1990),Vitis vinifera species(Williams, Strauss, Wilson & Massy-Westropp, 1983), and allium plants (Scognamiglioet al., 2012). In all the cases mentioned above, the BA contents have rarely been found above 30 mg/kg. Therefore, these natural sources are unsuitable for supporting a commodity scale of BA bioproduction (Pugh, McKenna, Halloum & Nielsen, 2015).
On the other hand, the chemical reduction of the benzaldehyde carbonyl group to the corresponding alcohol involves the use of heavy metals or their hydrides, such as NaBH4, LiAlH4, LiBHEt3, Li(Si)3BH, NaCNBH3, DIBAL, Zn(BH4)2(Abdel-Magid, 2014;Magano & Dunetz, 2012) or through the disproportionation of benzaldehyde over irreducible basic metal oxides (Cannizzaro reaction) like ZnO, Cr2O3, MgO, CaO, BaO, SrO (Haffad, Kameswari, Bettahar, Chambellan & Lavalley, 1997;Saadi, Rassoul & Bettahar, 2006). Some of the substances employed in the reduction are flammable and can constitute a health hazard (Cameo Chemicals, 2015). In addition to the use of intensive energy and harsh reaction conditions, this process suffers from sustainability concerns as it involves the use of non-renewable feedstocks (Cameo Chemicals, 2015;Environmental and Occupational Health Assessment Program, 2015).
However, comparatively few reduction methodologies have been developed considering the concept of green or sustainable chemistry, which is focused on making useful compounds and materials without causing harm to the environment. The principles of green chemistry encourage chemists to use alternative feedstocks, avoid hazardous reagents or products, and to minimize waste; as well as improve the efficiency of chemical processes while simultaneously reducing production costs. Ideally, the application of green chemistry principles renders regulation, control, clean-up, and unnecessary remediation. The resultant environmental benefit can be expressed in terms of economic impact (Anastas & Eghbali, 2010;Dichiarante, Ravelli & Albini, 2010).
The use of biocatalysts represents an enormous potential in organic synthesis, in addition, it is a very attractive alternative that follows the principles of green chemistry (Höfer & Bigorra, 2008;Ningqing, Lishan, Zhenming & Junhua, 2008). In recent years, the use of plants as biocatalysts has received considerable attention, some relevant characteristics of these biocatalysts are their low cost, accessibility, high versatility, and efficiency. These biocatalysts are eco-friendly since the reactions are performed in aqueous media, at room temperature, and generate only biodegradable waste (Cordell, Lemos, Monte & de Mattos, 2007). Besides this, the use of plant biocatalysts is an interesting alternative to promote the use of agricultural by-products and bio-waste; for example, banana and maize leaf wastes have been used as sources of reductase (Lunaet al., 2014); Prunus seeds like capulin, apricot, plum, and mamey (Pouteria sapota) are important hydroxynitrile lyase sources (Solís, Luna, Manjarrez & Pérez, 2004;Solís, Manjarrez, Pérez & Cassani, 2011); and the byproducts of orange juice processing are sources of lipases (Okino-Delgado & Fleuri, 2014).
A green alternative to the chemical reduction of carbonyls for the production of alcohols is biocatalytic reduction, because the use of metals and the generation of toxic waste are avoided. There are several reports on the possibility of using plants as biocatalysts for chemical transformations. The reduction of aromatic aldehydes has been carried out using broccoli, cauliflower, spinach beet, and spinach (Suárez-Franco, Hernández-Quiroz, Navarro-Ocaña, Oliart-Ros & Valerio-Alfaro, 2010),Conium maculatum(Salvanoet al., 2011),Aloe vera(Leyva, Moctezuma, Santos-Díaz, Loredo-Carrillo & Hernández-González, 2012), banana and maize leaf wastes (Lunaet al., 2014), coconut and palmyra palm juice (Misra, Maity, Chanda & Nag, 2012), passion fruit (Machadoet al., 2008), and lentil (Alveset al., 2012). However, the investigation into the use of waste from plants as enzyme sources for the reduction of carbonyls is very scarce.
The objective of this work was to investigate the use of plant wastes as a source of new biocatalyst sources for the reduction of benzaldehyde to BA with the purpose of developing economically viable and environmentally friendly reaction systems, which meet some of the principles of green chemistry.
EXPERIMENTAL
Chemicals and biological material
Benzaldehyde and BA, used as standard, were purchased from J.T. Baker and Jansen Chimica respectively. The biological materials used were bean pods (Phaseolus vulgaris), turnip rape (Brassica campestrisL.), lima bean (Phaseolus lunatus), broad bean (Vicia faba), and jinicuil (Inga edulis); mango (Mangifera indicaL. cv. manila), avocado (Persea americana), mamey (Pouteria sapota), papaya (Carica papaya), and green apple (Malus domestica) peel; mamey, capulin (Prunus serotina), green pepper (Capsicum annuum), chili (Capsicum annuumL. var.annuumL. cv. ‘Jalapeño’) and avocado seeds and chive leaves (Allium schoenoprasum), all of them were obtained from a local market and transported to the laboratory.
Preparation of the biocatalyst source
Peels and pods were thoroughly washed with deionized water, disinfected (10% v/v NaClO, 5 min) and rinsed with plenty of deionized water. The peels and pods were ground with distilled water (ratio w/v, peels 1:1, pods 1:3) in a blender, then the mixture was centrifuged, the supernatant was used as enzyme source.
Capulín and mamey seeds were ground with acetone in a blender then filtered. The process was repeated three times, the defatted powder was dried and sieved, and stored at 4 °C until use. The defatted powder (10 g) was stirred with 30 mL of distilled water. After one hour the mixture was centrifuged, the supernatant was used as enzyme source.
Chili and green pepper seeds were ground in a mortar with distilled water (ratio w/v 1:3 and 1:4 respectively), the mixture was centrifuged and the supernatant was used as the enzyme source.
General procedure for the enzymatic reduction
The benzaldehyde (1.5 × 10-5mol) was mixed with the corresponding aqueous extract (1 mL), the mixture was incubated at 25 ºC and stirred at 1300 rpm, for 24 h. Then, the mixture was extracted with diethyl ether (2 × 0.5 mL), dried over anhydrous sodium sulfate and analyzed by GC to determine the conversion. The analyses were carried out on a Hewlett-Packard HP 6890 gas chromatograph, equipped with a FID detector, a column Supelcowax-10, at 180 °C and flow of 1 mL/min; the injector and detector were at 250 °C , using N2as carrier. Each reaction was performed in triplicate.
RESULTS AND DISCUSSION
The study of the biocatalytic reduction of benzaldehyde to BA with different plant wastes as enzyme source aimed to find an environmentally friendly and economically viable reaction system, and to suggest a useful application of the biological waste that represents a severe pollution problem around the world. The bioreduction of benzaldehyde was carried out with the aqueous extract from different plant wastes as enzyme source, the reaction products were analyzed by Gas chromatography. Figure 1 shows the chromatograms of four bioreductions, the RT of benzaldehyde (B) was 2.74 min and of BA (A) 4.22 min.
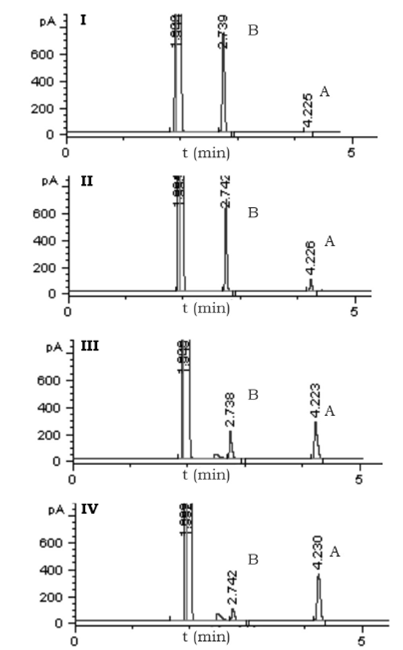
Source: Author's own elaboration.
Figure 1 Gas chromatographic analysis of the reduction of benzaldehyde (B) to BA (A) using different biocatalyst sources: I) Mamey peel (0% A), II) Avocado seed (17% A), III) Mamey seed (67% A) and IV) Capulin seed (87% A).
The results of the reduction of benzaldehyde using different biological material are shown in table 1, it can be observed that most of the biocatalyst sources were able to reduce benzaldehyde to BA. The most promising waste materials were the capulin and mamey seeds followed by the bean pods, turnip rape stalks, and chive leaves, with conversions between 45% and 54%. The rest of the seeds (green pepper, chili, and avocado), pods (broad bean, lima bean, and jinicuil) and papaya peel showed moderate activity towards the reduction of benzaldehyde (17% to 40% conversion to BA), and are also good candidates for further studies on sources of reductase. It is worth highlighting that the meals prepared from capulin and mamey seeds, that were stored at 4 °C for ten and seven years respectively, retained their ability to catalyze the reduction of benzaldehyde to BA. The reductase activity is preserved regardless of the storage time; this may be because the enzyme is retained in the defatted seed meal, hence it can be said that the defatted meal is an excellent support for the enzyme. The mango, avocado, mamey, and green apple peels were unable to reduce benzaldehyde.
Tabla 1 Percent conversion of benzaldehyde to BA using different aqueous extracts from plant waste as a reductase source
| Biological material | Family | Genus | % Conversiona |
|---|---|---|---|
| SEEDS | |||
| Capulin | Rosaceae | Prunus | 86 |
| Capulinb | Rosaceae | Prunus | 65 |
| Mamey | Sapotaceae | Pouteria | 67 |
| Mameyc | Sapotaceae | Pouteria | 77 |
| Green pepper | Solanaceae | Capsicum | 23 |
| Chili | Solanaceae | Capsicum | 17 |
| Avocado | Lauraceae | Persea | 17 |
| PODS | |||
| Bean | Fabaceae | Phaseolus | 54 |
| Turnip rape | Brassicaceae | Brassica | 49 |
| Broad bean | Fabaceae | Vicia | 40 |
| Lima bean | Fabaceae | Phaseolus | 30 |
| Jinicuil | Fabaceae | Inga | 30 |
| PEELS | |||
| Papaya | Caricaceae | Carica | 38 |
| Mango | Anacardiaceae | Mangifera | 0 |
| Avocado | Lauraceae | Persea | 0 |
| Mamey | Sapotaceae | Pouteria | 0 |
| Green Apple | Rosaceae | Malus | 0 |
| LEAF | |||
| Chives | Amaryllidaceae | Allium | 45 |
a: determined by GC;b: stored ten years;c: stored seven years.
Source: Author´s own elaboration
The process of reduction of benzaldehyde using the plant wastes can be improved by changing the reaction conditions (pH, temperature, time, and concentration of the substrate). Besides the biocatalysts, sources can be tested to reduce other aromatic aldehydes. Although we did not conduct studies to find the group of enzymes responsible for carrying out this transformation, the aldo-keto reductase (AKR) superfamily comprises of a large number of primarily monomeric protein members, which reduce a broad spectrum of substrates ranging from simple sugars to potentially toxic aldehydes/ketones. Aldehydes and have been identified in numerous plants (Narawongsanont, Kabinpong, Auiyawong & Tantitadapitak, 2012;Sengupta, Naik & Reddy, 2015). These enzymes display NADPH-dependent reduction of carbonyl compounds, typical of the superfamily, but will accept a very wide range of substrates, reducing aliphatic and aromatic aldehydes/ketones. AKR from plants are a good alternative to reduce the different types of aldehydes/ketones of the corresponding alcohols (Simpsonet al., 2009).
CONCLUSIONS
The use of plant wastes as a source of biocatalyst for the reduction of benzaldehyde to BA is a suitable biocatalytic methodology, avoiding the use of costly metal reducing agents commonly utilized in organic synthesis, reducing the generation of toxic waste. In addition, since the biocatalyst is present in the aqueous extract of the biological material, the residual plant waste can be used as fertilizer. With this methodology, the plant waste has gained an added value because, as its name indicates, it is a waste that has no further use.











 text new page (beta)
text new page (beta)


