Services on Demand
Journal
Article
Indicators
Related links
Share
Revista mexicana de fitopatología
On-line version ISSN 2007-8080Print version ISSN 0185-3309
Rev. mex. fitopatol vol.39 n.1 Texcoco Jan. 2021 Epub May 07, 2021
https://doi.org/10.18781/r.mex.fit.2008-1
Scientific articles
Gains in recurrent selection cycles for grain yield and resistance to head smut in maize
1Programa de Fitosanidad-Fitopatología, Colegio de Postgraduados, Carretera México-Texcoco Km 36.5, Montecillo, Texcoco, Estado de México, C.P. 56230, México.
2Postgrado de Recursos Genéticos y Productividad-Genética, Colegio de Postgraduados, Carretera México-Texcoco Km 36.5, Montecillo, Texcoco, Estado de México, C.P. 56230, México.
The basidiomycet Sporisorium reilianum f. sp. zeae infects the maize plant causing important losses in regions with adequate conditions for disease development. In this work, the response to selection of six and five cycles of selection in a white and a yellow endosperm maize populations improved through S1 recurrent selection for desirable agronomic characters, grain yield and resistance to head smut were evaluated. In a separate trial, same cycles were planted to measure progress in disease resistance and to evaluate a new method of inoculation were artificially inoculated. A randomized complete block design with three replications was used for the yield trials and two replications in the inoculated trial. Cycles evaluated in the two populations showed an increase in disease resistance of 0.94 y 1.2% cycle-1, and a genetic gain in grain yield of 272.9 y 620 kg ha-1 cycle-1 for the white and yellow endosperm populations, respectively. Results showed that recurrent S1 selection was efficient in improvement of grain yield and resistance to head smut in both populations.
Key words: S1 families; polygenic resistance; genetic resistance; head smut
El basidiomiceto Sporisorium reilianum f. sp. zeae infecta al cultivo de maíz y causa importantes pérdidas en regiones donde existen condiciones propicias para el desarrollo de la enfermedad. En este trabajo se evaluó la respuesta a la selección de seis ciclos de selección recurrente de maíces de endospermo blanco y cinco ciclos de maíces de endospermo amarillo para caracteres agronómicos deseables, rendimiento de grano y resistencia al carbón de la espiga. Los diferentes ciclos en las dos poblaciones se obtuvieron siguiendo el método de selección recurrente de familias S1, los cuales fueron evaluados para determinar el avance genético. En un ensayo anexo, las poblaciones fueron inoculadas artificialmente para medir el progreso de la resistencia a la enfermedad y evaluar un nuevo método de inoculación. Se utilizó un diseño experimental de bloques completos al azar con tres repeticiones para el ensayo de rendimiento y dos repeticiones para el ensayo inoculado. Los ciclos evaluados en las dos poblaciones mostraron un incremento en la resistencia de 0.94 y 1.2% ciclo-1, una ganancia genética para rendimiento de grano de 272.9 y 620 kg ha-1 ciclo-1 para la población blanca y amarilla, respectivamente. Los resultados mostraron que la selección recurrente de familias S1 fue eficiente para mejorar el rendimiento de grano y resistencia al carbón de la espiga en ambas poblaciones.
Palabras claves: Familias S1; resistencia poligénica; resistencia genética; carbón de la espiga
Maize (Zea mays) is widely planted worldwide, surpassing wheat and rice in production (FAOSTAT, 2018). It is the cereal with the highest demand as a food product for both human and animal consumption, as well as for raw material in industries (Eckhoff et al., 2003; Kaul and Olakh, 2019). However, its production is affected by pests and diseases. One of the diseases with great economic importance is head smut, caused by the basidiomycete Sporisorium reilianum f. sp. zeae (Basidiomycota, Ustilaginaceae), sin. Sphacelotheca reiliana, which is distributed in several regions of the world where this cereal is grown (Stromberg, 1981; Lynch et al., 1980; Martínez de la Parte et al., 2016; Bernardo et al., 1992; Fullerton et al., 1974). In Mexico, there are reports of its presence in areas of the Bajío and the Highlands (Aquino et al., 2011; Ramírez et al., 2011).
The inoculum is found in the soil and in the initial stages of infection the infective hypha penetrates the seedling through the root, invades the vascular bundles and grows systematically in the phloem (Kruger, 1962; Qi et al., 2019). In the stage of vegetative growth, symptoms are difficult to observe, although some authors have reported development of chlorotic spots on the leaves (Matyac and Kommendhal, 1985; Martínez et al., 2002; Quezada-Salinas et al., 2017), as well as stunting (Stromberg et al., 1984; Halisky, 1963). In the reproductive stage of the plant, signs and symptoms become visible in both inflorescences, the flowers of which are replaced by a dusty carbonlike mass, composed of teliospores or producing phyllodia in the affected structures, causing a reduction in yield (Stromberg et al., 1984; Matyac, 1985; Martínez et al., 2002; Qi et al., 2019). In Mexico, the disease has been controlled using mainly chemical products, cultural practices (CESAVEM, 2015) and with the selection of tolerant genotypes using natural infection (Pérez-Camarillo and Bobadilla-Meléndez, 2003 and 2007; Pérez-Camarillo et al., 2009; Aquino et al., 2011). Most genetic breeding programs focus on increasing grain yield and there are few reports of programs designed to select resistance to maize diseases.
An efficient genetic breeding scheme to generate pest and disease-resistant cultivars is the recurring selection of S1 families. This method is well-known for increasing the frequency of favorable alleles of one or more agronomic traits which are quantitatively inherited, and because it eliminates 50% of undesirable alleles after every self-pollination, helping to set traits of interest which are additively inherited (Márquez, 1985; Hallauer et al., 2010). This method is recommended for low-heritability traits, since it improves the population mean and maintains the genetic variability of the population, which helps continue the selection through breeding cycles. The following steps are taken to carry out a recurring S1 family selection scheme: 1) S1 families are generated by self-pollinating 400 healthy and vigorous plants from a widely based gene population, 2) The S1 families are evaluated in different environments, and 3) The best S1 families are recombined with superior traits of interest to the breeder (Márquez, 1985; Hallauer et al., 2010).
There is also the possibility that the evaluation and recombination of the S1 families are done in one cycle, reducing costs and time. However, it is important to consider if the trait of interest is expressed before flowering (Dhillon and Khehra, 1989). With this information, the aim of this investigation was to evaluate six cycles of recurrent selection of maize with white endosperm and five cycles with yellow endosperm to determine their grain yield, resistance to head smut and agronomic traits of interest, including severity of common rust (Puccinia sorghi) by natural infection, floral synchrony, relationship between plant and ear height, plant aspect, and others, as well as to evaluate an inoculation method to facilitate the adherence of teliospores in the seed and ensure the uniform distribution of inoculum in the field.
MATERIALS AND METHODS
Description of the study area
The evaluation of gains in the cycles of selection was done in plots at the Cinta Larga Technological Innovation and Research Center, in Mixquiahuala, State of Hidalgo (20° 11’ N, 99° 14’ W, 2100 masl). The crop was sown on March 28, 2019 in the spring-summer planting cycle, under rainfed conditions. The field evaluation was carried out in the state of Hidalgo and the laboratory activities described were performed at the Colegio de Postgraduados, Campus Montecillo, State of Mexico.
Maize germplasm evaluated
The genetic material evaluated in this study consisted of six breeding cycles (C0 to C5) of a population with white endosperm and five cycles (C0 to C4) of a population with yellow endosperm. The genetically broad based maize populations of white and yellow endosperm were developed including 45 and 55 collections of diverse germplasm, including commercial hybrids, improved varieties and native populations collected in the Mexican Highlands. The different components included in the formation of the base populations were recombined in isolated fields for two consecutive cycles in the Ejido Santa Teresa Tiloxtoc, in Valle de Bravo, State of Mexico (19° 13’ N, 100° 107’ W, 1740 masl), planting each component in two rows, each one 10 m long, which were used as females, alternating with one row as male which included a mechanical balanced composite of all the materials included as females.
After two cycles of recombination for their homogenization, the method for the intrapopulational selection of S1 families began in both populations. Each cycle was completed in two years. Year one: in each white or yellow grain population, 400 plants were self-pollinated to generate S1 families of each population. During the growing cycle and until harvest, plants with undesirable characters were eliminated. Year two: seeds from the best 60-70 S1 selected families were inoculated with head smut (S. reilianum) teliospores, as described by Quezada-Salinas et al. (2013) and were planted ear-to-row in the field. Out of the total S1 families under evaluation, a selection pressure of 33% was applied and selected S1 families from each population were recombined. The best plants of each family selected, with desirable characters, healthy, with no symptoms of head smut and common rust (P. sorghi) were manually recombined with a pollen mixture. A balanced, mechanical mixture of seeds of the recombined families formed the next cycle (C1). This sequence was repeated to obtain the six cycles of the white endosperm population and the five of the yellow endosperm population evaluated in this study.
A completely randomized block design with three replications was used in the evaluation trials of the different cycles of selection, in 3m long rows, 80 cm between rows, 16 cm between plants and 44 plants per row to adjust to 22 and at a density of 80 000 plants ha-1. Each row constituted a plot. Soil preparation and crop management were carried as done at the Cinta Larga Technological Innovation and Research Center, in Mixquiahuala, State of Hidalgo. Furrow irrigation was applied after planting, followed by once or twice monthly depending on rainfalls. Weed control done with four applications of herbicide (potassium salt of 3,6-dichloro-2-methoxybenzoic acid and 6-Chloro-N2-ethyl-N4-isopropyl-1,3,5,triazine-2,4 diamine). There was no fertilizer application or pest and disease control.
Trials of artificial inoculation in white and yellow endosperm maize populations
To evaluate genetic gains by cycle of improvement of head smut resistance, two replications were included with seeds inoculated with S. reilianum teliospores were included. The inoculum was collected in September 2018 from infected maize plants from the previous planting at the Cinta Larga field, in Mixquiahuala, Hidalgo. The inoculum was collected from infected tassels and ears dried for seven days in a greenhouse, collecting the teliospores after cleaning with a 117 µm sieve and storing them at 20 ± 2 °C for their later use. Seeds from each cycle of both populations were treated using a suspension of 1x106 teliospores mL-1, immersed in white glue (Polyvinyl acetate = Resistol 85®) diluted to 20% with water (v/v) and drying them for 2 h at 35 ± 2 °C. Smut incidence was evaluated in the six cycles of the white endosperm population and the five cycles of the yellow endosperm population. The purpose of this trial was to evaluate the incidence of head smut with artificial inoculation and to evaluate the efficiency of white glue used to stick the teliospores to the seeds. The same conditions, design and management were applied as in the yield trial.
To evaluate the differences and gains obtained between the different cycles of selection, the following variables were determined:
1) Days to male flowering (anthesis): Registered as the number of days between the day of planting until 50% of plants began sheding pollen. 2) Days to female flowering (emission of silks): Recorded as the number of days between date of planting to 50% silking. 3) Plant height (cm): height between the basis of the stalk and the insertion of the tassel. 4) Ear height (cm) was measured from the base of the stalk to the basis of the main ear. 5) Grain moisture (%): determined by plot at harvest time, taking 500 g of seeds from the central part of five good ears. It was measured using a Dickey John miniGAC plus moisture tester. For the analysis, grain yield was adjusted to 15% moisture and transformed to t ha-1. 6) Rust severity: Data were taken at flowering stage using an arbitrary scale of 1 to 5 where 1: No damage (foliar tissue 100% healthy), 2: few lesions (90% healthy), 3: regular (80% healthy), 4: bad (60 - 70% healthy) and 5: very bad (0 - 60% healthy). 7) Head smut incidence: The response to infection was determined a few days after the emission of both inflorescences. Incidence was determined as a percentage (Num. of diseased plants divided by the total number of diseased plants per plot x 100). The variables studied were determined following IBPGR (1991) and Edmeades et al. (2000).
Statistical analysis
All data were analyzed using the software SAS (Statistical Analysis System, version 9.4) (SAS Institute Inc., 2013), using the procedure PROG GLM and PROG REG to obtain the linear regression. An analysis of variance was used along with a DMS comparison of means with α ≥ 0.05.
RESULTS
Yield trial in white and yellow populations
Due to low germination problems, cycle two (C2) of the populations was not included. Table 1 shows the measurements for the different traits described. For the variables of plant and ear height and head smut incidence, there were no significant differences based on the DMS (0.05) in both populations. For the trait of days to male and female flowering, significant differences were observed between cycles of selection in both populations. For the yellow endosperm cycles, the variables of rust severity and grain yield showed significant differences but not in the white endosperm population. In the variables that did not present a statistically significant response, the advanced selection cycles express an improvement in comparison with the initial cycles. To better appreciate the behavior of the traits evaluated in the different cycles of both populations, data were analyzed by linear regression to get the average gain cycle-1 (Table 2). For the ear height, an increase of 3.5 and 4.3 cm cycle-1 was observed, and for ear height there was also an increase of 1.7 and 2.2 cm cycle-1 in the white and yellow endosperm populations, respectively. For days to tassel, a reduction of -1.51 and -1.6 day cycle-1 was observed, and for days to silking -1.8 and -2.2 days cycle-1 was recorded in both populations, indicating that, as the selection cycles in the populations advanced, populations tend to be earlier in flowering. A reduction of -0.2 and -0.6 cycle-1 was found for common rust severity (Figure 1). For head smut incidence, a reduction of -0.9 % cycle-1 was found in the population of white endosperm and -1.2 % cycle-1 in the yellow endosperm population (Figure 1). For grain yield, there was an increase of 0.3 and 0.6 t ha-1 cycle-1 for the white and f yellow populations, respectively (Figure 1).
DISCUSSION
The S1 recurrent selection was effective for most of the traits evaluated in the white and yellow endosperm populations, mainly for grain yield and resistance to head smut (S. reilianum). Similar results have been found in other investigations, where the breeding method was also the recurring selection of S1 families. Rodríguez and Hallauer (1988) evaluated 10 populations using different selection models and concluded that the method of S1 families was better than other methods evaluated. The response to the selection for grain yield was lower than that obtained by Ruiz de Galarreta and Álvarez (2007), but similar to those reported by Tanner and Smith (1987) and Maya-Lozano and Ramírez-Díaz (2002), who reported at least one negative response to the selection, similar as in this study where ear and plant heights showed no responses to selection.
There are also reports indicating that, depending on the size of the base population, the evaluation of the progeny and the recombination method used are variables that modify their response to selection (Hallauer et al., 2010). Kebede (2001) suggests that toavoid genetic drift, 30 families should be used in case five or more selection cycles are desired. Weyhrich et al. (1998) mention that genetic drift is stronger when less than 10 families are used in the recombination. In this investigation 60-70 families were used per cycle, which ensures the maintenance of genetic variability.
Table 1 Means of the different characters evaluated in the white and yellow endosperm maize populations.
| Población endospermo blanco | |||||||
|---|---|---|---|---|---|---|---|
| Ciclos de selección | Días a floración masculina | Días a floración femenina | Altura de planta (cm) | Altura de mazorca (cm) | Severidad en roya (1-5) | Incidencia carbón de la espiga (%) | Rendimiento t ha-1 |
| C0 | 87.3 a | 91.0 a | 180.0 b | 113.3 a | 2.8 a | 12.6 a | 11.2 a |
| C1 | 79.3 b | 82.6 b | 195 ab | 113.3 a | 1.6 b | 10.7 a | 11.4 a |
| C3 | 77.0 b | 81.6 b | 190 b | 108.3 a | 1.5 b | 10.2 a | 12.2 a |
| C4 | 79.6 b | 82.3 b | 196.6 ab | 116.6 a | 1.5 b | 8.5 a | 11.6 a |
| C5 | 79.6 b | 83.3 b | 196.6 ab | 120.0 a | 1.8 ab | 9.0 a | 12.4 a |
| DMS (0.05) | 2.9 | 2.6 | 19.9 | 23 | 1 | 16.6 | 4.5 |
| Población endospermo amarillo | |||||||
| C0 | 87.3 a | 91.3 a | 208.3 a | 121.6 b | 3.6 a | 14.2 a | 12.5 b |
| C1 | 79.0 bc | 83.3 bc | 206.6 a | 121.6 b | 2.0 b | 8.3 a | 12.3 b |
| C3 | 78.6 bc | 81.0 bc | 215.0 a | 138.3 a | 1.5 b | 12.5 a | 12.2 b |
| C4 | 82.0 b | 84.6 b | 220.0 a | 123.3 b | 1.6 b | 8.8 a | 14.6 a |
| DMS (0.05) | 3.6 | 4 | 35 | 13.4 | 1 | 28 | 2 |
Table 2 Response to selection in the white and yellow endosperm populations.
| Carácter | Población endospermo blanco | Población endospermo amarillo | ||||
|---|---|---|---|---|---|---|
| xb | yI | zR2 | b | I | R2 | |
| Días a floración masculina | -1.51 | 85.0 | 0.4 | -1.63 | 85.8 | 0.30 |
| Días a floración femenina | -1.8 | 88.9 | 0.4 | -2.24 | 90.6 | 0.50 |
| Altura de planta (cm) | 3.48 | 181.2 | 0.61 | 4.35 | 201.6 | 0.82 |
| Altura de mazorca (cm) | 1.67 | 109.3 | 0.4 | 2.18 | 120.7 | 0.20 |
| Severidad roya (1-5) | -0.21 | 2.47 | 0.4 | -0.65 | 3.80 | 0.75 |
| Incidencia del carbón (%) | -0.94 | 13.02 | 0.86 | -1.2 | 13.95 | 0.30 |
| Rendimiento (t ha-1) | 0.2729 | 10.9 | 0.63 | 0.62 | 11.35 | 0.5 |
xRegresion coefficient (b); yinterception (I); zDetermination coeficiente (R2).
Regarding resistance to head smut (S. reilianum f. sp. zeae), it is inherited additively and is partially dominant (Whythe and Gevers, 1988). In this research, there was a gain in resistance of 0.94% cycle-1 in the white population and 1.2% cycle-1 in the yellow population, indicating that, as the breeding cycles progress, genes are accumulated additively with each cycle. Resistance is given by the genes zmWAK, zmNL and GRMZM2G047152, which are expressed in the first stages of the infection process, where they inhibit the spread of the infective hypha of the fungus S. reilianum f. sp. zeae (Zuo et al., 2014; Qi et al., 2019).
CONCLUSIONS
The S1 families recurrent selection was effective to increase the grain yield in 272.9 kg ha cycle-1 and resistance to head smut in 0.94% cycle-1 in the white endosperm population and 620 kg ha cycle-1 and 1.2% cycle-1 for resistance to head smut in the yellow endosperm population.
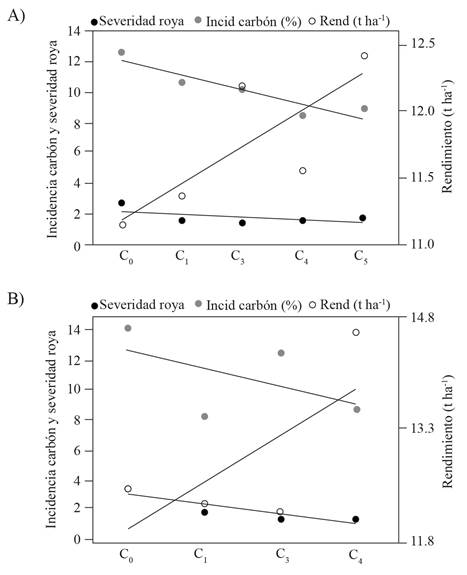
Figure 1. Linear regression for the variables of rust severity, head smut incidence and grain yield. Population of white endosperm maize (A) and population of yellow endosperm maize (B).
All other variables considered in the present research showed a positive response to selection, except for plant and ear heights in both populations, therefore, greater selection pressure must be applied on these traits to advance the populations to the following cycles.
Artificial seed inoculation using white glue 20% v/v as an adherent was efficient to produce a uniform infection in the field.
LITERATURA CITADA
Aquino-Martínez JG, Sánchez-Flores A, González-Huerta A y Sánchez-Pale JR. 2011. Resistencia de variedades e híbridos de maíz (Zea mays L.) a Sporisorium reilianum y su rendimiento de grano. Revista Mexicana de Fitopatología 29: 39-49. http://www.scielo.org.mx/scielo.php?script=sci_arttext&pid=S0185-33092011000100004 [ Links ]
Bernardo R, Bourrier M and Oliver JL. 1992. Generation means analysis of resistance to head smut in maize. Agronomie 12(4): 303-306. https://doi.org/10.1051/agro:19920403 [ Links ]
CESAVEM (Comité Estatal de Sanidad Vegetal del Estado de México). 2015. Campaña manejo fitosanitario del maíz. 8p. http://www.cesavem.mx/img/fitosanitariodelmaiz/maiz2.pdf [ Links ]
Dhillon BS and Khehra AS. 1989. Modified S1 recurrent selection in maize improvement. Crop Science 29: 226-228. https://doi.org/10.2135/cropsci1989.0011183X002900010049x [ Links ]
Eckhoff SR, Paulsen MR and Yang SC. 2003. Maize. pp 3647-3653. In: Caballero B, Finglas P and Toldra F. Encyclopedia of Food Sciences and Nutrition. 2nd Edition. Academic Press. 6000p. https://doi.org/10.1016/B0-12-227055-X/00725-2 [ Links ]
Edmeades GO, Bolaños J, Elings A, Ribaut JM, Bänziger M and Westgate ME. 2000. The role and regulation of the anthesis silking interval in maize. In: Westgate M, Boote K, Knievel D and Kiniry J (eds.). Physiology and modeling kernel set in maize. Crop Science Society of America and the American Society of Agronomy 48: 65-80. https://doi.org/10.2135/cssaspecpub29.c4 [ Links ]
FAOSTAT (Food and Agriculture Organization Corporate Statistical Database). 2018. Producción mundial de maíz. http://www.fao.org/faostat/es/#data/QC. [ Links ]
Fullerton RA, Scott DJ and Graham GJ. 1974. Effect of fungicides on the control of head smut of maize and the relationship between infection level and crop yield. New Zealand Journal of Experimental Agriculture 2: 177-179. https://doi.org/10.1080/03015521.1974.10425757 [ Links ]
Hallauer AR, Carena MJ and Miranda JB. 2010. Quantitative Genetics in Maize Breeding, Handbook of Plant Breeding. New York: Springer Science and Business Media. 663p. http://dx.doi.org/10.1007/978-1-4419-0766-0 [ Links ]
Halisky PM. 1963. Head smut of sorghum, sudan grass, and corn, caused by Sphacelotheca reiliana (kühn) Clint. Journal of Agricultural Sciencie 34(8): 287-304. http://dx.doi.org/10.3733/hilg.v34n08p287 [ Links ]
IBPGR. 1991. Descriptors for Maize. International Maize and Wheat Improvement Center, México City/International Board for Plant Genetic Resources, Rome. 85p. [ Links ]
Kaul J, Jain K and Olakh D. 2019. An overview on role of yellow maize in food, feed and nutrition security. International Journal of Current Microbiology and Applied Sciences 8(2): 3037-3048. https://doi.org/10.20546/ijcmas.2019.802.356 [ Links ]
Kebede PM. 2001. Response to S1 recurrent selection and estimation of genetic parameters in effective population sizes of the BS11 maize population. Retrospective theses and dissertations. 663p. https://doi.org/10.31274/rtd-180813-14332 [ Links ]
Kruger W. 1962. Sphacelotheca reiliana on maize. I. Infection and control studies. South African Journal of Agricultural Science. 5: 43-56. https://hdl.handle.net/10520/AJA05858860_650 [ Links ]
Lynch KV, Edgington LV and Busch LV.1980. Head smut, a new disease of corn in Ontario. Canadian Journal Plant Pathology 2: 176-178. https://doi.org/10.1080/070606680095014371B [ Links ]
Márquez SF. 1985. Genotecnia vegetal: Métodos, teoría, resultados. Tomo I. México D.F. AGT Editor, S. A. 357p. [ Links ]
Matyac CA and Kommedahl T. 1985. Factors affecting the development of head smut caused by Saphacelotheca reiliana on corn. Phytopathology 75: 577-581. https://www.apsnet.org/publications/phytopathology/backissues/Documents/1985Articles/Phyto75n05_577.PDF [ Links ]
Martínez CA, Roux A, Jauneau A and Dargent R. 2002. The biological cycle of Sporisorium reilianum f.sp. zeae: an overview using microscopy. Mycologia 94: 505-514. http://dx.doi.org/10.1080/15572536.2003.11833215 [ Links ]
Martínez- de la Parte E, Wilson BD, Lorenzo ME, Guerrero BD, García RD, Rodríguez GG, Sierra RP y Gómez LY. 2016. El carbón de la espiga del maíz causado por Sphacelotheca reiliana (J|G. Kühn) GP. Clinton en Cuba. Fitosanidad 20(1): 3-38. http://www.redalyc.org/articulo.oa?id=209157223005 [ Links ]
Maya-Lozano JB y Ramírez-Díaz JL. 2002. Selección recurrente en tres poblaciones de maíz para el subtrópico de México. Revista Fitotecnia Mexicana 25(2): 201-207. https://www.revistafitotecniamexicana.org/documentos/25-2/11a.pdf [ Links ]
Pérez-Camarillo JP y Bobadilla-Meléndez M. 2007. Carbón de la espiga de maíz. Síntesis de resultados del ciclo agrícola P.V. 2006. Valle del Mezquital, Hgo. Desplegable Técnica Número 15. Centro de Investigación Regional Centro. INIFAP-Hidalgo. 2 p. [ Links ]
Pérez-Camarillo JP y Bobadilla-Meléndez M. 2003. Carbón de la espiga de maíz. Síntesis de resultados del ciclo agrícola P.V. 2002. Valle del Mezquital, Hgo. Desplegable Técnica Número 6. Centro de Investigación Regional Centro. INIFAP-Hidalgo. 4 p. [ Links ]
Pérez-Camarillo JP, Martínez-Ruiz E y Bobadilla-Meléndez M. 2009. Carbón de la espiga de maíz. Síntesis de resultados del ciclo agrícola P.V. 2008. Valle del Mezquital, Hgo. Desplegable Técnica Número 21. Centro de Investigación Regional Centro. INIFAP-Hidalgo. 4 p. [ Links ]
Quezada-Salinas A, De León-García De Alba C, Hernández-Anguiano AM y Nava-Díaz C. 2013. Evaluación de métodos de inoculación de semillas de maíz con Sporisorium reilianum f. sp. zeae (Kûhn) Langdon & Fullerton. Revista Mexicana de Fitopatología 31(2): 80-90. http://www.scielo.org.mx/pdf/rmfi/v31n2/v31n2a1.pdf [ Links ]
Quezada-Salinas A, Moreno-Velázquez M, De León-García de Alba C, Nava-Díaz C, Solano-Báez AR. 2017. Resistencia genética a Sporisorium reilianum f. sp. zeae en líneas seleccionadas de maíz (Zea mays L.) con endospermo blanco y amarillo. Revista Mexicana de Fitopatología 35(3): 534-548. http://dx.doi.org/10.18781/R.MEX.FIT.1705-2 [ Links ]
Qi F, Zhang L, Dong X, Di H, Zhang J, Yao M, Dong L, Zeng X, Liu X, Wang Z and Zhou Y. 2019. Analysis of Cytology and Expression of Resistance Genes in Maize Infected with Sporisorium reilianum. Plant Disease 103(8): 2100-2107. https://doi.org/10.1094/PDIS-09-18-1687-RE [ Links ]
Ramírez Dávila JF, Sánchez Pale JR y De León C. 2011. Estabilidad espacio temporal de la distribución del carbón de la espiga del maíz (Sporisorium reilianum) en el Estado de México, México. Revista Mexicana de Fitopatología 29: 1-14. http://www.scielo.org.mx/pdf/rmfi/v29n1/v29n1a1.pdf [ Links ]
Rodríguez OA and Hallauer AR. 1988. Effects of recurrent selection in corn populations. Crop Science 28: 276-280. https://doi.org/10.2135/cropsci1988.0011183X002800050015x [ Links ]
Ruiz de Galarreta JI and Álvarez RA. 2007. Six cycles of S1 recurrent selection in two Spanish maize synthetics. Spanish Journal Agriculture Research 5(2): 193-198. http://dx.doi.org/10.5424/sjar/2007052-239 [ Links ]
SAS (Statistical Analysis System). 2013. The SAS system. Version 9.4. SAS OnlineDoc. HTML. Format, SAS Institute, Cary, NC, USA [ Links ]
Stromberg EL, Stienstra WC, Kommedahl T, Matyac CA, Windels CE and Geadelmann JL. 1984. Smut expression and resistance of corn to Sphacelotheca reiliana in Minnesota. Plant Disease 69: 880-884. https://doi.org/10.1094/PD-68-880 [ Links ]
Stromberg EL. 1981. Head smut of maize, a new disease in Minnesota. Phytopathology 71: 906. [ Links ]
Tanner AH and Smith OS. 1987. Comparison of half-sib and S1 recurrent selection in the Krug yellow dent maize populations. Crop Science 27: 509-513. https://doi.org/10.2135/cropsci1987.0011183X002700030016x [ Links ]
Weyhrich RA, Lamkey KR and Hallauer AR. 1998. Effective population size and response to S1 progeny selection in the BS11 maize population. Crop science 38(5): 1149-1158. https://doi.org/10.2135/cropsci1998.0011183X003800050008x [ Links ]
Whythe IV and Gevers HO. 1988. Diallel analysis of resistance of eight maize inbred lines to Sphacelotheca reiliana. Phytopathology 78: 65-68. https://www.apsnet.org/publications/phytopathology/backissues/Documents/1988Articles/Phyto78n01_65.PDF [ Links ]
Zuo W, Chao Q, Zhang N, Ye J, Tan G, Li B, Xing Y, Zhang B, Liu H, Fengler K, Zhao J, Zhao X, ChenY, Lai J, Yan J and Xu M. 2015. A maize wall-associated kinase confers quantitative resistance to head smut. Natural Genetic 47: 151-57. https://doi.org/10.1038/ng.3170 [ Links ]
Received: August 06, 2020; Accepted: November 20, 2020











 text in
text in 


