Genetic and phenotypic variation can occur between and within species because of the evolutionary and demographic history of species (Salgotra & Chauhan 2023). Phenotypic variation is influenced by genetic variance and by the interactions of the genotype with biotic and abiotic factors (Miner et al. 2005, Nicotra et al. 2010, Moles et al. 2014, Cocoletzi Vásquez et al. 2022), while genetic variation results mainly from the action of evolutionary forces such as mutation, natural selection and genetic drift (Cocoletzi Vásquez et al. 2022). Therefore, a pattern of morphological and genetic variation can be expected throughout the geographic distribution of any species.
In tree species used in agroforestry systems and valued for their social, cultural and economic importance, artificial selection may shape phenotypic and genetic variation (Souto et al. 2009, Nicotra et al. 2011, Fischer et al. 2016, Matesanz & Milla 2018, Carvajal-Arroyo et al. 2022). Continuous artificial selection may result in the fixation of desirable traits, such as disease and drought resistance or higher nutritional value; but domestication may also result in loss of genetic variation and fixation of detrimental alleles that could hinder the ability of species to respond to environmental change. Furthermore, a reduction in genetic diversity can occur through genetic drift if the population size under management is small, which can result in low reproductive rate due to the combined effect of inbreeding and selection (Nicotra et al. 2010, Meyer & Purugganan 2013, Gaut et al. 2015, Schierenbeck 2017, Matesanz & Milla 2018).
The genus Theobroma is constituted by 23 species which distribute from South America to Mexico (Richardson et al. 2015, Santamaría-Aguilar et al. 2023). Some species within this genus are currently undergoing domestication (Bretter & Daly 2009, Tinajero-Carrizales et al. 2021). Such is the case of pataxte (T. bicolor Humb. & Bonpl.). This species has cultural importance, as it is used to make beverages for special events such as gatherings and religious festivals. In addition, it has secondary agroforestry uses (used as shade for cacao trees [Theobroma cacao L.], fruit trees and coffee) (Rendón et al. 1998). This species could become economically relevant (Rendón-Aguilar et al. 1998, Chaves et al. 2021) as raw materials to produce beverages (Rendón-Aguilar et al. 1998, Ponce-Sánchez et al. 2021, Tinajero-Carrizales et al. 2021). However, little is known about its genetic and phenotypic variation (Gálvez-Marroquín et al. 2016, Joya Davila 2018).
Pataxte can be found from Brazil to Mexico (Richardson et al. 2015, Benlloch-Tinoco et al. 2024, Figure S1), and it occupies a vast range of environmental conditions that correspond to its wide geographical distribution (Cuatrecasas 1964, Joya Davila 2018). The species grows between 1,000 and 1,500 m asl at temperatures that oscillate between 25 °C and 30 °C in non-floodplain areas (Cuatrecasas 1964). In Mexico, its distribution includes the cacao-growing regions in the states of Chiapas, Tabasco, Oaxaca, and Guerrero (Rendón-Aguilar et al. 1998, INEGI 2001, Motamayor et al. 2002).
Pataxte is considered an outcrossing species, and incompatibility systems have been reported in other species of the same genus such as T. speciosum, T. cacao and T. grandiflorum (Melgarejo et al. 2006, Joya Davila 2018, de Souza & Venturieri 2010). The most abundant floral visitors reported for pataxte are insects from Ceratopogonidae, Chironimidae and Sciaridae, which may be its main pollinators (Ponce-Sánchez et al. 2021).
Sexual maturity in pataxte is reached at 4 to 6 years of age (Kufer & McNeil 2009). In western Amazonia, flowering occurs in the months of higher temperature and higher solar radiation, and each tree produces 7 ± 5 fruits (Ponce-Sánchez et al. 2021). Seeds take between nine and 28 days to germinate, depending on the environmental conditions (Joya Davila 2018). Seeds are short lived, which limits storage to create a seed collection, and, as in cacao, trees should be preserved in collections and botanical gardens (Joya Davila 2018, www.catie.ac.cr [CATIE]).
In pataxte, morphological variation has led to the description of four varieties in Mexico based on qualitative characteristics of the fruit, particularly the roughness and color of the pod shell, the shape of the basal constriction and the shape of the apex of the pod shell (Gálvez-Marroquín et al. 2016). These varieties were recorded in the states of Tabasco, Oaxaca and Chiapas, Mexico (Gálvez-Marroquín et al. 2016). In addition, morphological variation in fruits, leaves, flowers, and the chemical composition of the seeds of pataxte have been reported (Gálvez-Marroquín et al. 2016, Joya Davila 2018, Tinajero-Carrizales et al. 2021). To our knowledge, there is no information available about the origin of pataxte’s varieties, whether they are the result of artificial selection for characteristics that producers consider desirable, and/or if recorded morphological variation is related to its evolutionary trajectory. Also, levels of genetic diversity in pataxte have not been evaluated; this is key in understanding the relationship between morphological and genetic variation in this species.
The present study aimed to evaluate the patterns of genetic and morphological variation of pataxte found in cacao agroforestry systems from Mexico. We carried out the study guided by the following question: What levels of genetic and phenotypic variation can be found in pataxte from cacao regions in Mexico? We obtained morphological data from pataxte fruits and leaves collected in cacao agroforestry systems from the states of Chiapas, Guerrero, Oaxaca and Tabasco, Mexico. We also amplified four chloroplast DNA and five nuclear DNA regions to assess levels of genetic variation. This semidomesticated species shows incompatibility mechanisms and an outcrossing mating system, and previous studies have reported morphological variability; therefore, we expect to find high levels of genetic and morphological variation in pataxte from cacao regions from Mexico.
Materials and methods
Study area. To our knowledge, there are no reports of wild pataxte populations in Mexico. Instead, in Mexico, pataxte is reported as a semidomesticated species associated to cacao agroforestry systems (Rendón-Aguilar et al. 1998, Kufer & McNeil 2009, Gálvez-Marroquín et al. 2016), as can be appreciated from occurrence records of pataxte from the GBIF database (Supplementary material, Figure S1, GBIF www.gbif.org ). The main cacao growing regions in Mexico are found in the states of Chiapas (three regions: Soconusco, Norte and Selva Norte), Oaxaca (one region: Papaloapan Basin), Tabasco (three regions: Chontalpa, Centro and Sierra), and Guerrero (two regions: Costa Chica and Costa Grande) (Avendaño et al. 2011, Secretaría de Economía 2016, INTAGRI 2020, Suárez Venero et al. 2021, Acéves-Mares et al. 2024, El Universal Oaxaca 2024). These cacao-producing areas are characterized by their cultural value and stand out for their historical significance in cacao cultivation (Solís Montero et al. 2022). However, pataxte has been recorded in five of these regions: the Soconusco in Chiapas, the Chinantla in the Papaloapan Basin in Oaxaca, Chontalpa and Sierra in Tabasco and Costa Chica in Guerrero (Figure 1).
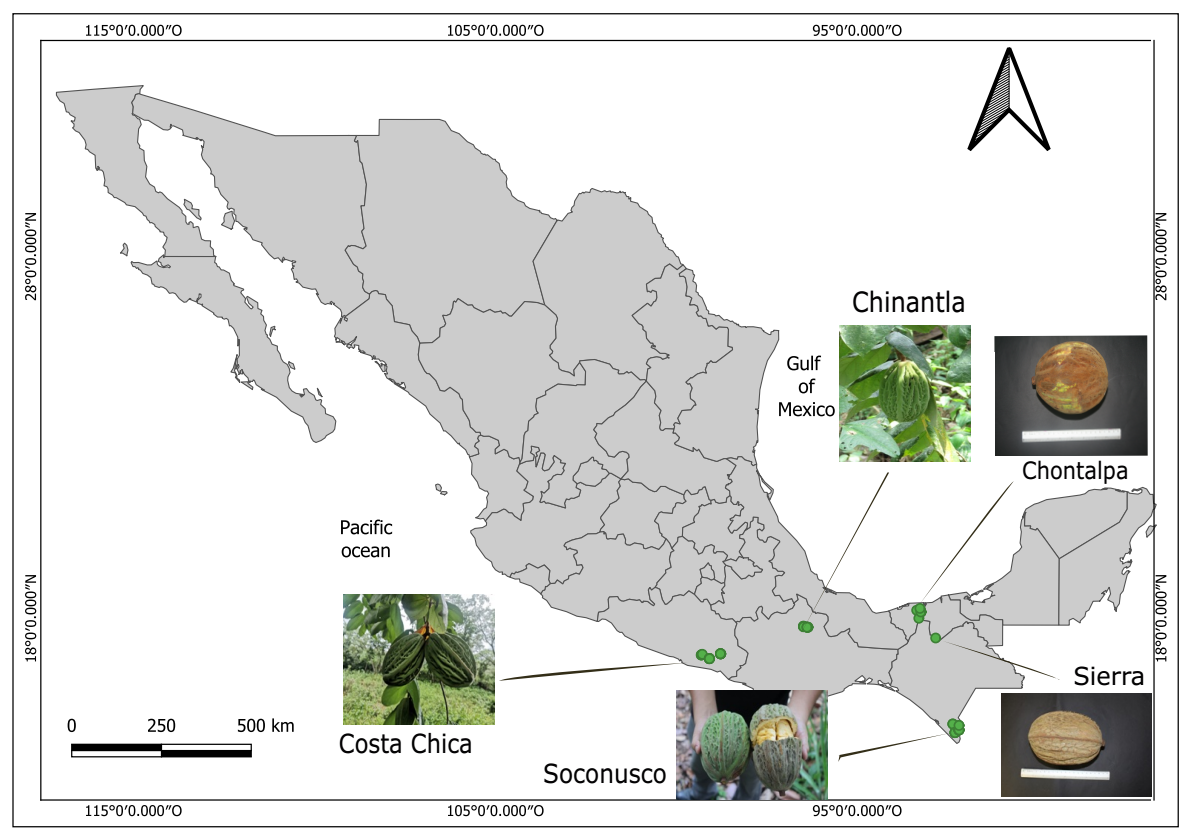
Figure 1 Sampling sites of Theobroma bicolor Humb. & Bonpl. in cacao agroforestry systems located in five cacao growing regions of Mexico and photographs of some of the collected fruits. The coordinates for each sampling site can be found in Table S1 (Supplementary material).
In the state of Tabasco we visited two areas of cacao agroforestry systems; (1) the Chontalpa region, in the occidental part of the state; it is a culturally significant area, and it is renowned for its long-standing tradition in cacao production (Ayuso de Dios 2023); and (2) Sierra region located in the southern area of this state, it is a mountainous transition region in the border between Tabasco and Chiapas, it is distinguished by its sustainable agroforestry production systems, where local producers integrate biodiversity crops using traditional agriculture techniques (Estañol Vidrio 2023).
The Chinantla region is located in the northern area of the state of Oaxaca, in the Papaloapan Basin. It is characterized by its historical and cultural importance due to its ancestral agricultural practices (González 2000). It is a region where people cultivate pataxte (T. bicolor) and cacao (T. cacao) (Contreras, D. pers. com).
In Chiapas, Soconusco is one of the most economically significant regions in Chiapas. It is a coastal area located in the southern part of the state, near the border with Guatemala. It is noted for its history of coffee production, fine and aromatic cacao cultivation, and its proximity to the Mexican Pacific coast (Rasgado Bonilla & Renard Hubert 2022).
Finally, the Costa Chica, located in the south coast of Guerrero. It is an emerging region in cacao production that combines traditional agricultural methods with modern sustainable techniques. This region presents great biocultural diversity. The warm, humid climate has supported the integration of cacao crops into sustainable agroforestry systems and the development of small farms (Gobierno del Estado de Guerrero 2021).
Sampling: We visited a total of 53 agroforestry systems within cacao-growing regions to collect pataxte fruits and leaves. Samples of pataxte were collected from 2021-2024 during the fruiting period within cacao agroforestry systems, during the months of September to November (Table 1). Pataxte is not abundant; in some areas it was removed from cacao agroforestry systems as it was believed that it could be a potential disease vector for cacao, so currently not all cacao producers have pataxte in their agroforestry systems (Rendón-Aguilar et al. 1998, Kufer & McNeil 2009, Sterling Cuéllar et al. 2015, Gálvez-Marroquín et al. 2016, Parada-Berríos et al. 2019).
Table 1 Sampling regions, number of agroforestry systems visited in each region, number of foliar tissue samples collected for genetic analyses (NDNA), number of collected fruits (Nfruits) and leaves (Nleaves) per cacao growing region from Southeast Mexico.
| Cacao region | Number of agroforestry systems included |
NDNA | Nfruits | Nleaves |
|---|---|---|---|---|
| Chontalpa | 10 | 37 | 28 | 15 |
| Chinantla | 10 | 22 | 8 | 14 |
| Soconusco | 13 | 33 | 3 | 31 |
| Sierra | 2 | 24 | 30 | 46 |
| Costa Chica | 18 | 26 | 21 | 28 |
| Total | 53 | 142 | 90 | 134 |
For genetic analyses, we collected young leaves for DNA extraction. To increase our sample size for genetic analyses, we sampled adult and young individuals per agroforestry system (Table 1). At agroforestry systems with more than 10 individuals, we randomly selected 10 individuals approximately 50 to 100 meters apart. We obtained 142 tissue samples for DNA extraction (Table 1). Tissue samples were cleaned with 70 % ethanol and cut into small pieces, the veins were removed from the leaves, and the samples were stored in individually labeled paper bags. To dehydrate and transport the tissue, the samples were preserved in hermetic bags with silica gel (CAMEN Mexico), which was constantly changed until arrival at the genetics laboratory. The samples were stored in a freezer at -20 °C until processing.
For morphologic analyses, we collected fruits (according on the availability; see details in the following section), as reproductive characteristics such as seed number may be indicators of fitness (Nicotra et al. 2010). We also collected leaves because leaf size may be crucial for growth and carbon balance (Nicotra et al. 2010).
For fruit morphological analyses, as the fruits detach from the pataxte tree when ripe, we picked from the ground three to four ripe fruits (greenish fresh color) at each cacao agroforestry system, depending on their availability and how many fruits the owners would give us. In addition, to increase sample size, mature fruits were purchased directly from producers at fairs and markets (six from Chontalpa) if the locality or area of origin was precisely known. Because of the latter, it was impossible to assess the parent tree, so we assumed that each collected fruit came from a different tree. We obtained a total of 90 fruits (Table 1).
For leaf morphological analysis, we obtained samples from 5 to 10 adult individuals by cacao agroforestry system, according to availability (Table 1). From each tree, we randomly selected two or three branches that were collected and placed in a botanical press. Botanical samples were dehydrated in a dryer for four days, herborized, and deposited in the herbarium of El Colegio de la Frontera Sur - San Cristóbal de Las Casas, (voucher numbers 26359 to 26382). Some of the specimens collected were not included in the herbarium because they did not meet the requirements of showing reproductive structures such as flowers and fruits. We collected a total of 134 leaves. The plant tissues were collected from fruit-bearing trees; however, producers mentioned that some of the trees came from the same mother tree, so it was not necessary to collect from all the individuals as this would be replicates from the same mother tree.
DNA extraction and molecular data collection and analysis: We took approximately 200 mg of dehydrated plant tissue from each sample and grounded it with liquid nitrogen. DNA extraction was performed using the protocol described by Doyle & Doyle (1990) modified by Vázquez-Lobo (1996), with modifications by the Genetics Laboratory of ECOSUR-Unidad San Cristóbal, including the buffer temperature (previously heated to 60 °C), centrifugation conditions (two spins at 12,700 rpm at 4 °C for 8 min), incubation time (4 h), and number of washes with 24:1 chloroform:isoamyl alcohol (two washes).
As molecular markers, we selected four chloroplast regions (cpDNA) and five nuclear regiones (ncDNA). These DNA regions were previously reported as variable for T. cacao (Supplementary material, Table S2) and have been amplified in pataxte for reference; but they have not been used to evaluate genetic diversity in pataxte at a population level. For each marker, adjustments to the PCR amplification program were made in terms of annealing time and temperature (Supplementary material,Table S2). For the PCR, we prepared a master mix with a final volume of 25 µl, which contained 12 µl of Master Mix (Thermo Fisher Scientific), 6 µl of ddH2O, 2 µl each of the forward and reverse primers [10 µM], and 3 µl of DNA (3:100 dilution to obtain a concentration of ~ 20 ng/µl). The PCR products were verified on 2 % agarose gels stained with ethidium bromide. To verify that there was no contamination in the reactions, a blank was included in each batch. The PCR products were sequenced via the Sanger ABI 3730xl System at Macrogen Korea (dna.macrogen.com). To evaluate whether these chloroplasts and nuclear regions presented genetic variation to perform population genetics analyses in pataxte, we sequenced a representation from 6 to 26 samples (Supplementary material, Table S2) taken at random representing the previously mentioned cacao-growing regions.
Sequence alignment. The quality of the electropherograms was manually checked, and a BLAST search (Zhang et al. 2000) was performed to verify that the sequences were from the targeted regions. Sequences for each molecular region were aligned with BioEdit Sequence Alignment Editor version 7.7.1 (Hall 1999) and Unipro UGENE version 49.1 (Okonechnikov et al. 2012). For the alignments, we downloaded reference sequences available in GenBank for the chloroplast and nuclear regions for T. bicolor and T. cacao (Supplementary material, Table S2).
Genetic data analysis. The WRKY12 and ITS markers did not amplify from the T. bicolor samples. The sequences of the remaining markers, both chloroplast and nuclear, were not variable. Therefore, it was not possible to perform genetic analyses, such as haplotype network and analysis of molecular variance (AMOVA), to obtain estimators of genetic diversity and genetic structure in pataxte samples. The consensus sequences obtained for each molecular marker were deposited in GenBank (Accession chloroplast marker: psbA-trnH (PQ045860), trnT-psbD (PQ045861), psbJ-petA (PQ045862), trnL-trnF (PQ045863); accession nuclear markers: WRKY11 (PQ632188), WRKY13 (PQ632189), WRKY14 (PQ632190)). Only for locus WRKY14 there were five segregating sites between the reference sequence from Peru and all our sequences.
Morphological variation. Linear measurements were taken using the morphological descriptors of Engels et al. (1979) and Phillips Mora et al. (2012) for cacao: fruit length (Lf), fruit width (Wf). The weights of the fruits were obtained using a 1000 g spring scale (PESOLA, Switzerland). Linear measurements of the fruits were determined with a precision electronic Vernier caliper with an accuracy of 0.01 mm (electronic digital caliper 0-155 mm).
The number of seeds (NS), seed wet weight (SWW), and seed dry weight (SDW) per fruit were obtained. Seeds were washed to remove mucilage and weighed on a wet basis with a portable electronic scale (Electronic Compact Scale SF-400 C). Then we measured six seeds per fruit. Seeds were randomly selected, and their width (Ws), length (Ls), and thickness (Ts) were determined with a precision electronic Vernier caliper with an accuracy of 0.01 mm (electronic digital caliper 0-155 mm). Then, the seeds were subsequently dehydrated in an electric dryer (Hamilton Beach Mod. 32100) for 18 h; once dry, the seeds were weighed again on a portable electronic scale.
For the leaf morphological measurements, from each herborized botanical sample, we measured the leaf of the third node. We procured two or three branches per tree depending on tree height, and we measure one leaf per duplicate. We used descriptors proposed for cacao by Restrepo Quiroz & Urrego Posso (2018): the length of the leaf blade (Lh), the width of the leaf blade (Wh), the length of the base to the widest point of the leaf (LBA), the width of the base (WB), the length of the apex (LA), and the length of the petiole (PL). Measurements were taken with a precision electronic Vernier caliper with an accuracy of 0.01 mm (electronic digital caliper 0-155 mm).
Analysis of morphological data. A small number of samples were obtained per cacao-growing region. Therefore, to characterize morphological variation, data were analyzed by cacao-growing region.
From a biological perspective, as there is intraindividual variation (Martin et al. 2017), measuring multiple leaves and fruits per individual helps avoid underestimating morphological variation. To approximate intrapopulation variation and to ensure that the results of morphological characterization are robust, it is recommended to sample and measure multiple leaves per individual. Along these lines, we collected two or three leaf samples per individual in each agroforestry system, but in some agroforestry systems there are few mature pataxte individuals or individuals are very tall precluding our ability to collect the leaves; these facts limited sample availability to conduct robust statistical analyses; therefore, we only performed descriptive analyses. For each morphological variable, the mean, standard deviation, and the minimum and maximum values were estimated for each cacao-growing region (Table 4).
In the case of Soconusco, due to the small number of fruit samples (N = 3), only the minimum and maximum values of measurements are reported, as they cannot be incorporated into other analyses. Additionally, we constructed violin plots to visualize the distribution and density of our data. Plots were prepared with vioplot (Adler et al. 2025) and ggplot2 (Wickhman 2016) libraries in R (R Core Team 2020) and R Studio (RStudio Team 2020).
We obtained principal component analysis (PCA) plots to visualize the distribution of samples among cacao-growing regions for the morphological variables of pataxte fruits and leaves. Plots were created using ggplot2 (Wickham 2016) library in R (R Core Team 2020) and R Studio (RStudio Team 2020).
Results
Two molecular markers did not amplify from the nine molecular markers tested. For cpDNA (psbA-trnH, trnL-trnF, psbD-petA) and ncDNA molecular markers (WRKY11, WRKY13, WRKY14), we found only one haplotype for each marker for the subset of samples tested, thus no genetic variation was detected.
In contrast, we detected morphological variation. Leaves collected in the Chinantla showed high mean values for leaf length (351.64 ± 105.74) and width (217.14 ± 88); followed by the Costa Chica region (length 220.75 ± 38.83, and width 113.18 ± 22.29). The leaves collected in the Soconusco region showed the smallest values (Table 2, Figure 2, Table 4). It is worth noticing that the data distribution for these attributes overlap (Figure 2).
Table 2 Mean and standard deviation for the morphological variables taken for collected leaves of Theobroma bicolor grouped by cacao growing region from southeast Mexico. Chinantla = Oaxaca, Chontalpa and Sierra = Tabasco; Costa Chica =Guerrero; Soconusco = Chiapas.
| Morphological variables | Chinantla | Chontalpa | Costa Chica | Sierra | Soconusco |
|---|---|---|---|---|---|
| N | 14 | 15 | 28 | 45 | 31 |
| Leaf width (mm) | 351.64 ± 105.74 | 214.93 ± 55.63 | 220.75 ± 38.83 | 220.46 ± 63.12 | 191.46 ± 45.31 |
| Petiole length (mm) | 190.52 ± 155.79 | 66.43 ± 88.48 | 19.75 ± 4.02 | 17.26 ± 5.52 | 21.85 ± 20.97 |
| Leaf length (mm) | 217.14 ± 88.6 | 111.05 ± 29.09 | 113.18 ± 22.29 | 108.52 ± 24.68 | 101.24 ± 30.75 |
| Leaf base width (mm) | 147.68 ± 75.55 | 50.31 ± 16.17 | 47.57 ± 16.51 | 50 ± 17.56 | 56.8 ± 29.14 |
| Apex length (mm) | 12.84 ± 3.16 | 9.33 ± 3.87 | 10.29 ± 6.83 | 7.68 ± 4.45 | 5.72 ± 5.1 |
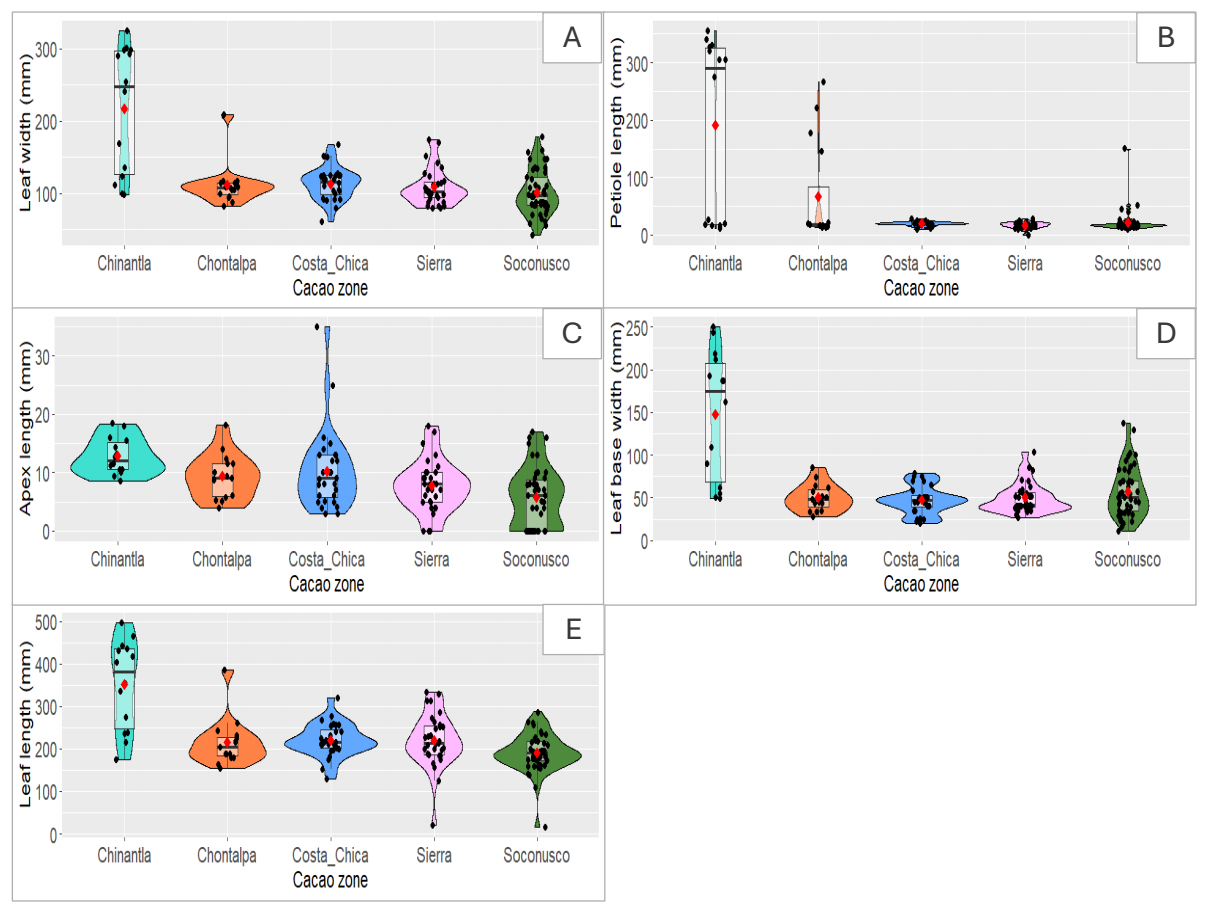
Figure 2 Violin plots showing data distribution and boxplot for pataxte’s (Theobroma bicolor Humb. & Bonpl.) morphological characteristics for leaves collected in cacao growing regions (Chinantla = Oaxaca, Chontalpa and Sierra = Tabasco; Costa Chica = Guerrero; Soconusco = Chiapas). A) Leaf width (mm), B) Petiole length (mm), C) Apex length (mm), D) Leaf base width (mm), E) Leaf length (mm).
Regarding the fruits, the heaviest and largest fruits (in terms of length, width of fruit size) were found in Chinantla agroforestry systems. The Chinantla also showed the highest values for seed fresh weight; followed by the Costa Chica region. Fresh fruit weight in Chinantla was 855.5 ± 180.47 while the Costa Chica showed an average of 704.52 ± 226.7. We observed that fruit length overlaps between regions, but the fruits from Chontalpa show the highest average values in fruit width and number of seeds (Table 3, Table 4). The highest mean value for seed fresh weight is found in the Chontalpa, but the mean value for seed dry weight was found in the Costa Chica region of Guerrero (Figure 3, Table 4). The Costa Chica region showed the widest distribution for fruit attributes (Figure 3). Although we collected fruits from the Soconusco region in Chiapas, the small number of samples (N = 3) does not allow us to calculate the average and standard deviation; the minimum and maximum values of fruit weight were 502 and 805 grams.
Table 3 Mean and standard deviation of morphological traits of collected fruits of Theobroma bicolor by cacao growing regions of southeast Mexico. In this table, the fruits collected in Sonocusco are not included because of small sample size (N=3). Chinantla = Oaxaca, Chontalpa and Sierra = Tabasco; Costa Chica = Guerrero.
| Morphological variables | Chinantla | Chontalpa | Costa Chica | Sierra |
|---|---|---|---|---|
| N | 8 | 28 | 21 | 30 |
| Fresh weight (gr) | 855.5 ± 180.47 | 454.96 ± 103.07 | 704.52 ± 226.7 | 650.67 ± 191.74 |
| Fruit length (mm) | 168.3 ± 18.5 | 131.25 ± 12.64 | 131.39 ± 40.85 | 146.83 ± 25.49 |
| Fruit width (mm) | 103.51 ± 9.92 | 86.84 ± 7.95 | 72.3 ± 38.74 | 95.8 ± 10.93 |
| Number of seeds (units) | 37 ± 7.33 | 31.29 ± 5.27 | 27.76 ± 7.13 | 29.53 ± 6.13 |
| Seed fresh weight (gr) | 205.25 ± 55.18 | 66.21 ± 24.25 | 108.13 ± 39.18 | 89.66 ± 44.75 |
| Seed dry weight (gr) | 40.12 ± 17.14 | 22.14 ± 12.99 | 44.61 ± 18.5 | 33.04 ± 16.67 |
Table 4 Table of maximum and minimum values of morphological values of fruits and leaves of pataxte samples collected in the cacao regions of Southeastern Mexico. Chinantla = Oaxaca, Chontalpa and Sierra = Tabasco; Costa Chica = Guerrero; Soconusco = Chiapas.
| Morphological variables | Chinantla | Chontalpa | Costa Chica | Sierra | Soconusco | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Leaves | min | max | min | max | min | max | min | max | min | max |
| Leaf length (mm) | 175 | 496 | 155 | 386 | 130 | 320 | 20.20 | 333 | 16 | 287 |
| Petiole length (mm) | 11.60 | 335 | 13 | 266 | 10 | 29 | 8 | 29 | 10 | 150 |
| Leaf width (mm) | 9.10 | 325 | 82.10 | 209 | 61 | 167 | 80 | 174 | 42 | 178 |
| Leaf base width (mm) | 48.90 | 250 | 27.40 | 86 | 20 | 79 | 27 | 103 | 11 | 137 |
| Apex length (mm) | 8.50 | 18.40 | 3.90 | 18.20 | 3 | 35 | 3.8 | 18 | 5 | 17 |
| Fruits | ||||||||||
| Fruit fresh weight (gr) | 540 | 1104 | 260 | 620 | 350 | 1400 | 390 | 1000 | 502 | 805 |
| Fruit length (mm) | 148.90 | 192 | 106.80 | 149.10 | 45 | 190.70 | 121.40 | 250.90 | 15 | 150 |
| Fruit width (mm) | 89.60 | 117.60 | 71.20 | 103.10 | 4.70 | 112.40 | 79.40 | 121 | 92.60 | 108.60 |
| Number of seeds (units) | 24 | 45 | 19 | 41 | 16 | 43 | 19 | 41 | 32 | 44 |
| Seed fresh weight (gr) | 82 | 263 | 49 | 166 | 44.95 | 192.76 | 34.10 | 239.59 | 16.82 | 20.05 |
| Seed dry weight (gr) | 9 | 61 | 2 | 61 | 15.89 | 88.95 | 9.56 | 71.86 | 10.42 | 15.30 |
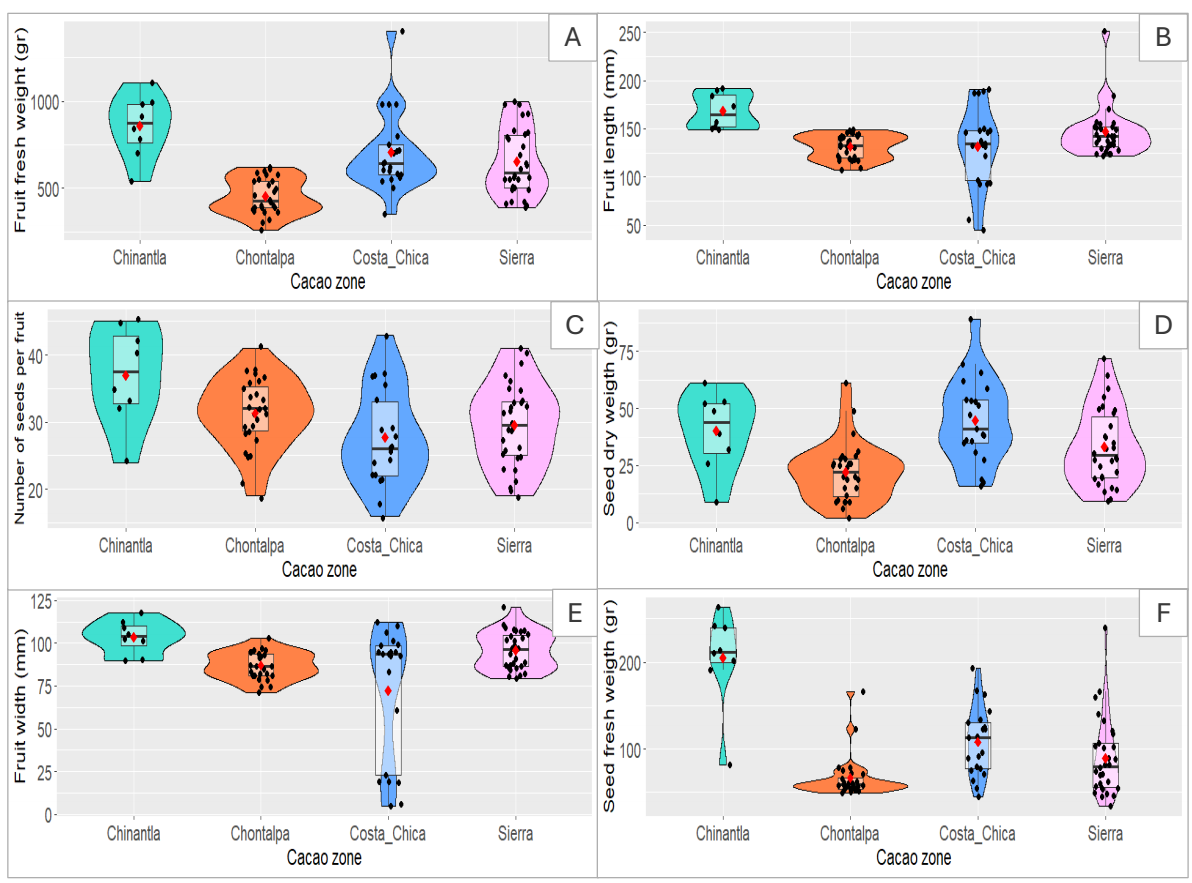
Figure 3 Violin plots showing data distribution and boxplot for pataxte’s (Theobroma bicolor Humb. & Bonpl.) morphological characteristics for fruits collected in cacao growing regions (Chinantla = Oaxaca, Chontalpa and Sierra = Tabasco; Costa Chica = Guerrero; Soconusco = Chiapas). A) Fresh fruit weight (gr), B) Fruit length (mm), C) Number of seeds (units), D) Seed dry weight (gr), E) Fruit width (mm), F) Seed fresh weight (gr).
The PCA for leaves’ morphological variables shows that the first two principal components explain 90.4 % of the total variance, but most variance is explained by PC1 (73.8 %). Leaf samples from each cacao-growing region overlap, suggesting that leaves share morphological characteristics. Nonetheless, the leaves from Chinantla, in Oaxaca, show differentiation as some samples cluster to the right of the graph on PC1, while leaves from the cacao regions from Chiapas, Tabasco and Guerrero overlap (Figure 4A). This suggests higher variance in foliar characteristics in pataxte’s from the Chinantla region (Figure 4A).
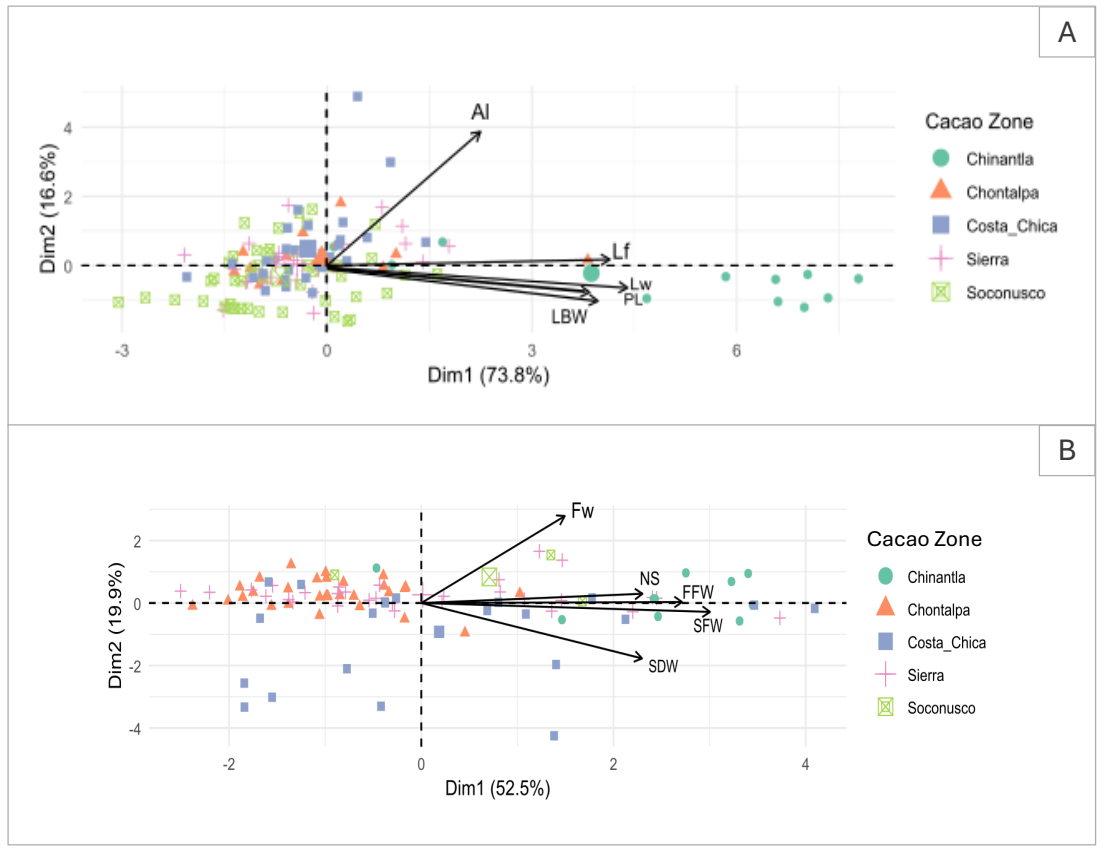
Figure 4 PCA with ordination plot on morphological traits of the pataxte (T. bicolor Humb. & Bonpl.). A) PCA of leaf: Al = Apex length, Lf = Leaf length, Lw = Leaf width, PL = Petiole length, LBW = Width of the leaf base. B) PCA of fruit: Fw = Fruit width, NS = Seed number, FFW = Fres Fruit Weight, SFW = Seed Fresh Weight, SDW = Seed Dry Weight. Larger symbols represent the means for each cacao zone in the space represented by the principal components.
The PCA plot for fruit morphological characteristics explains 72.4 % of variance. The distribution and morphological characteristics of fruit samples in cocoa-growing regions suggest that the samples from the Chinantla, Oaxaca, show displacement to the right area of the graphic on PC1, while fruits from cacao regions from Chiapas and Tabasco overlap (Figure 4B). Samples collected in Costa Chica, Guerrero, exhibit variance in fruit morphology on both PCs (Figure 4A).
In general, our data shows that the higher values for leaf and fruit attributes were found in Chinantla, Oaxaca, but there is a lot of overlap in pataxte’s morphological characteristics between cacao growing regions. We observed variance within each region, particularly in pataxte from the Chinantla in Oaxaca and Costa Chica in Guerrero.
Discussion
In the present study, no genetic variation was observed in pataxte amplified samples. The selected chloroplast and nuclear markers have been used and reported as variable in other genera, such as Quercus, to identify subspecies (Yang et al. 2017). In the genus Theobroma, they have been used for phylogenetic reconstruction and for the identification of varieties and genetic groups in T. cacao (Borrone et al. 2007, Gutiérrez-López et al. 2016, Ramos Ospino et al. 2020). For example, the cpDNA trnH-psbA region showed a haplotype diversity of 0.66 and a nucleotide diversity of 0.0032 in T. cacao from Chiapas (Gutiérrez-López et al. 2016), and the WRKY gene family for ncDNA showed between 61 to 173 variable sites in the same species (Borrone et al. 2007). In contrast, our results suggest that T. bicolor in these regions either lack genetic variation or have very low genetic variation. It has been reported that the chloroplast genome in Theobroma is conserved in length and organization (Tineo et al. 2024).
To assess intraspecific genetic variation in pataxte it is necessary to test other molecular markers, such as nuclear microsatellite loci, single-nucleotide polymorphisms (SNPs), or complete genomes (Allendorf et al. 2010, Gaut et al. 2015). In this sense, ISSRs have been successfully used to conduct population genetics studies in T. subincanum (Rivas et al. 2013) and T. speciosum (Giustina et al. 2014), microsatellite loci have been used for population genetics studies in T. speciosum in Brazil (Dardengo et al. 2016, 2018) and in T. cacao in Mexico (Vázquez-Ovando et al. 2014). Also, genomic resources have already been developed for T. cacao and T. grandiflorum, and there is an available chloroplast genome for T. bicolor (Abdullah et al. 2020, Bustamante et al. 2022, Izzah et al. 2023, Alves et al. 2024, Tineo et al. 2024).
Low levels or the lack of genetic diversity found in this study could relate to low mutation rates in tested genomic regions which result in highly conserved sequences. This, together with the biological and ecological characteristics of pataxte, the selective management conducted by producers and its evolutionary history may explain low levels of genetic variation. Few pataxte individuals, probably a single lineage, may have arrived in Mexico along with cacao, which was introduced to Mesoamerica approximately 2,000 years ago (Schultes 1984, Motamayor et al. 2002, O. Cornejo pers. comm.). Therefore, the species may have experienced a founder effect during its dispersal from South America to Mexico, and the time passed since pataxte was brought to Mexico is not enough for genetic variation to accumulate in this geographical area.
The importance of pataxte is mainly cultural and is currently used as a shade plant (Rendón-Aguilar et al. 1998, Sandoval Meda 2017), so a small number of scattered plants are usually found per agroforestry system, thus its effective population sizes in Mexico may have always been relatively small. Pollination by small insects may limit pollen dispersal between agroforestry systems. Although it is artificially propagated, it is unknown if it can be dispersed by birds, mammals, or other organisms. Therefore, it is possible that individuals within each agroforestry system are closely related.
Furthermore, pataxte populations in Mexico may have undergone one or more bottlenecks, which would have eroded its genetic diversity. Pataxte was considered a vector for the transmission of moniliasis, a disease caused by a pathogenic fungus (Moniliophthora roreri), and other phytopathogens affecting cacao production, and, at least in Mexico, programs to eliminate pataxte from cacao plantations were implemented (E. Hernández-Maldonado, pers. comm.). The latter favored a population reduction in pataxte populations in Mexico, which may have promoted loss of genetic diversity (Gálvez-Marroquín et al. 2016, Parada-Berríos et al. 2019, Rendón-Aguilar et al. 1998).
Pataxte maintains low population sizes (Rendón-Aguilar et al. 1998) and, according to the results of this study, likely has low genetic diversity. This condition increases the probability of inbreeding and thus the risk of inbreeding depression. To avoid this risk, it is important to assess genetic diversity with other molecular markers, as previously mentioned, and it may be necessary to promote seed exchange between producers within regions, and to establish in situ conservation areas and pataxte germplasm banks. Such measures would preclude the loss of genetic variation which is necessary to respond to long-term environmental changes. Currently, producers from Chinantla in Oaxaca are recovering pataxte populations from the few individuals still available (J. Jiménez, pers. comm.).
The morphological data of fruit and leaf variables show size variation, particularly in fruits. In the Chinantla, located in the state of Oaxaca, some fruits exhibit higher weight compared to the other cacao growing regions (540 - 1,104 grams per fruit). In contrast, the lightest fruits were found in the Chontalpa region (260 - 620 grams per fruit). There was also variation in fruit length and width in the Chinantla (Figure 3, Table 4). This variation in fruit size and weight may be related to differences in management employed by producers at each site or to the presence of different varieties of pataxte previously reported in Mexico (Gálvez-Marroquín et al. 2016). Nonetheless, the sample size used in the present study is limited and more samples are needed to formally test for statistically significant differences between regions.
Leaf morphology showed a similar trend to that of fruits. Higher values for leaf length were recorded in the Chinantla (175 - 496 mm) (Figure 2). These results suggest that morphological variability still exists in Mexico. In a study conducted in Colombia, the average leaf width and length recorded were 126.5-141.1 mm and 245.5-263.7 mm, respectively, with an average fresh fruit weight of 1,194 g and an average of 37 to 50 seeds per fruit (Melgarejo et al. 2006); these values are within the range we found for pataxte in Mexico. In Colombia, there were statistical differences in leaf width and petiole length between three pataxte ecotypes (Melgarejo et al. 2006). Nonetheless, a small sample size will preclude the adequate assessment of the variability found within and between populations, thus promoting a bias in the interpretation of the results (Quinn & Keough 2002, Zar 2010). Therefore, future studies should increase per region sample size and consider collecting individual replicas to control for intraindividual variation, to test if fruit and leaf morphological differences between cacao growing regions from Mexico suggested by our data are statistically significant.
Observed variation in fruits and leaves between cacao growing regions may be influenced by artificial selection, which promotes populations with low genetic diversity but with morphological differences. These differences could also result from plastic responses or adaptations to environmental conditions, such as temperature, precipitation, soil nutrient content and moisture, among the cacao regions of southeastern Mexico (Tavili & Jafari 2009, Ramírez-Guillermo et al. 2018, Cocoletzi Vásquez et al. 2021, Sauvadet et al. 2021). Therefore, it is necessary to evaluate the effects that management and artificial selection have had on the morphological characteristics of these plants.
Phenotype-environment interactions have also been observed in cacao (Kichenin et al. 2013, Nabais et al. 2018, Sauvadet et al. 2021, Pérez-Alva et al. 2022). Variation in environmental factors such as temperature, altitude, humidity, soil moisture and amount of light, interferes in fruit and leaf growth and development, and in the case of domesticated or semi-domesticated plants in fruit production and yield (Asitoakor et al. 2022, Pérez-Alva et al. 2022); such effects have been reported in cacao (Daymond & Handley 2008, Cocoletzi Vásquez et al. 2021). For example, an increase in temperature promotes a higher respiration rate that results in a higher demand for carbohydrates and consequently there are less nutrients available for fruit development (Daymond & Handley 2008, Osorio et al. 2017, Saavedra et al. 2020).
Variation in precipitation may affect fruit’s morphological characteristics, i.e., a dry environment produces fruits that are flatter, smaller, with less pulp and fruits with few seeds. In contrast, very humid environments can affect the degradation of organic compounds present in the soil, which impacts nutrient availability and results in larger fruits (Garay-Peralta et al. 2024). Likewise, in humid environments fruits and seeds may present early degradation related to the rapid degradation of organic compounds (Nicotra et al. 2011, Fischer et al. 2016).
Although it is well known that within the genus Theobroma pataxte is the species with the largest fruits (Cuatrecasas 1964, Sterling Cuéllar et al. 2013) no quantitative data on morphological variation within the species has been reported in Mesoamerica. However, qualitative data on fruit variation does exist, such as that reported by Gálvez-Marroquín et al. (2016), which describes four morphotypes in Mexico: four for the state of Chiapas, two in Oaxaca, and one in Tabasco. If phenotypic variation is a response to the environment, then by developing in different environments the attributes of the fruit will be different from those of the ancestral population. In contrast, those attributes with a genetic basis will be maintained regardless of the environment (Ruíz Corral et al. 2008, Lobo Arias & Medina Cano 2009). To determine the proportional importance of environment-genotype-phenotype interactions, a genome-wide association study (GWAS) is recommended, which allows identifying adaptive genetic variation and its relationship with phenotypes.
In general, our results suggest that the morphological variation in pataxte may be determined by multiple factors, including environmental factors, and possible agronomic management. Importantly, future work should incorporate environmental variables (e.g., amount of light, type of soil, organic matter, and soil nutrients), and include genomic tools to elucidate phenotype‒environment interactions. Similarly, increasing the number of samples per agroforestry system will allow detailed analyses regarding genetic and morphological variation at a local scale. Expanding sampling area to include Central and South America can provide a broader understanding of the adaptability, dispersal, gene flow, and evolutionary history of pataxte for its conservation in the short, medium, and long term.
Supplementary material
Supplemental data for this article can be accessed here: https://doi.org/10.17129/botsci.3693.











 nueva página del texto (beta)
nueva página del texto (beta)



