Introduction
The mitochondrion is the cellular organelle that regulates metabolism, cell signaling, and immune response; therefore, it requires coordination with other organelles and cellular niches to maintain cellular homeostasis. Disease disrupts this homeostasis or balance, and mitochondria register these changes, defining useful elements in recognizing and managing different diseases1,2. This relationship directly impacts medicine, as understanding the mitochondria-health-disease axis could improve decision-making and generate solutions in the context of mitochondria-based medicine.
Mitochondria possesses their own genome (mitochondrial DNA [mtDNA]); in humans, it contains 37 genes, 13 of which encode proteins related to oxidative phosphorylation (OXPHOS, the adenosine triphosphate [ATP] synthesis process)3; however, it contains a significant diversity of proteins (approximately 2000), such that the majority are encoded and imported from the nucleus4. mtDNA can also vary: a cell contains between 100 and 10,000 copies3, which can have subtle differences and form populations (heteroplasmy) that could influence the health/disease phenotype. Furthermore, mitochondria communicate with virtually all cellular structures both intrinsically and extrinsically, as they make use of various extracellular structures (nanotubes, vesicles, and bloodstream) that allow them to establish long-distance communication; thus, they have also become attractive tools for the design of novel therapies5,6.
Mitochondrial medicine
Initially, mitochondrial medicine focused on mitochondriopathies (inherited metabolic disorders that affect ATP synthesis)7. These diseases primarily present in childhood, although they can also appear in adults; they lack well-defined diagnostic criteria and treatment protocols, their symptoms are nonspecific, and the molecular defect is difficult to pinpoint, thus requiring different methodological strategies to resolve these ambiguities8.
Mitochondriopathies are frequently confused with other pathologies (neuropathies, movement disorders, or heart diseases)9,10, which can delay their diagnosis. For example, in cardiac diseases (one of the most frequent pediatric problems), mitochondrial alterations can go unnoticed and be treated inappropriately; hypertrophic cardiomyopathy11 and tachycardias12 associated with mitochondrial disease represent 20-40% of cases and present significantly higher mortality (71%) compared to patients without mitochondrial alterations (26%)11. This reveals that mitochondrial alterations are of utmost importance because, although they appear rare, their consequences can be substantial.
One of the most effective traditional approaches for determining mitochondriopathies is through catalytic activity assays that allow the identification of functional defects. However, these require various resources and involve invasive procedures (biopsies). At present, some strategies improve the diagnosis of mitochondriopathies; the proteins FGF21 and GDF15 (two mitokines found circulating in the blood) reflect mitochondrial damage and identify disease carriers13. Meanwhile, omic platforms define molecules that could guide a more accurate diagnosis and provide better monitoring of these diseases14,15. The diagnosis of a mitochondriopathy requires understanding the defect at the genetic level; conventional molecular biology strategies such as Sanger sequencing and polymerase chain reaction have relative success (≈6%) in determining genetic defects, in contrast to massive genome/exome sequencing strategies that achieve close to 45-60%, positioning themselves as the best diagnostic strategy15,16.
On the other hand, proteomic analysis of primary human skin fibroblast cultures from patients with mitochondriopathy allows the detailing of the molecular mechanisms at the protein level17. Through metabolomics, easily accessible biomarkers (metabolites) have been proposed with potential clinical utility in the development and treatment response of these diseases. Implementing these approaches improves the diagnosis of mitochondriopathies; however, their widespread use still requires corresponding clinical validation.
Mitochondrial alterations beyond mitochondriopathies
Non-communicable chronic diseases (NCDs) are long-term conditions and are the leading cause of death worldwide (80%)18. In these conditions, mitochondrial alterations can be found from their onset19 and impact different processes that can be used to identify, monitor, or treat these diseases (Table 1), as well as extend diagnostic options and develop better mitochondria-targeted treatments (Table 2)19,20.
Table 1 ECNTs with mitochondrial defects in their pathogenesis
| Disease | Mitochondrial alteration | Cellular alterations |
|---|---|---|
| Obesity | Fragmentation, overproduction of mtROS, reduction of ETC components, decreased ATP synthesis, impaired membrane potential | Increased proliferation and differentiation, metabolic shift, and cell death. |
| Insulin resistance | Fragmentation, Ca+2 handling, mtROS production, MAM deregulation | Defective insulin-related signaling, decreased glucose uptake, GLUT4 translocation, defective insulin signaling, insulin resistance. |
| Type 2 diabetes mellitus | Respiratory uncoupling, decreased ATP synthesis, mitophagy, exacerbated fission/fusion | Decreased insulin secretion. |
| Cancer | Reduction of PGC-1α, increased mitophagy and glycolysis, moderate mtDNA mutations, metabolic hyperactivity | Metabolic shift, exacerbated biosynthesis, proliferation, tumorigenesis, aggressiveness, cell migration, and invasion. |
| Cardiovascular diseases | mtDNA damage, fragmentation, MAM deregulation, mtROS overproduction, Ca+2 handling defects, decreased oxygen consumption | Proliferation, hypertrophy, Ca+2 overload, energy crisis, impaired contractility, cell death. |
mtROS: mitochondrial reactive oxygen species; ETC: electron transport chain; ATP: adenosine triphosphate; MAM: mitochondria-associated membranes; mtDNA: mitochondrial DNA (modified from 19).
Table 2 Treatment of NCDs through mitochondria-targeted molecules (modified from 20)
| Type | Effect | Disease |
|---|---|---|
| Antioxidants | ROS neutralizers, SIRT activators based on CoQ | Neurodevelopment (Rett, Duchenne), Parkinsons, chronic kidney disease, among others. |
| Metabolism modulators | Niacin derivatives, metabolic (creatine, metformin), AMP kinase activators | Obesity, type 1 diabetes mellitus, myocardial infarction, liver disease (non-alcoholic). |
| PtPm inhibitors | Translocators, cyclophilin D | Acute kidney injury, Alzheimers disease. |
NCDs: non-communicable diseases; ROS: reactive oxygen species; SIRT: sirtuin; CoQ: Coenzyme Q; PtPm: permeability transition pore.
This includes pediatric patients, as their main health conditions (obesity, diabetes, infections, among others) are related to mitochondria; therefore, the mitochondrial approach is very important in developing diagnostic tests and effective treatments. Mitochondria-related biomolecules (nucleic acids, proteins, and metabolites) can define useful elements in the stratification and pathophysiology of these diseases21,22. For example, in intractable childhood epilepsy, there are various mitochondrial aspects23, such as variations between copy number and mtDNA damage24,25 that are involved.
The outer mitochondrial membrane contains transporter complexes that activate the immune system: the translocase of the outer membrane, the sorting and assembly machinery and import machinery, as well as proteins sensitive to Damage Associated Molecular Patterns26, whose alteration could indicate preferential signaling pathways associated with NCDs. Proteomic analysis of brain tumors in children and adults shows relevant participation of mitochondrial proteins; in fact, for glioblastomas, there are proposals for molecular classification that include mitochondrial categories21,22. Furthermore, in breast cancer, mitochondrial cristae are altered27, and their evaluation could improve diagnosis and clinical monitoring, as their integrity and size are associated with the degree or advancement of malignancy28; in contrast, in gastric cancer, some mitochondrial proteins involved in energy metabolism (OXPHOS, TCA), mitochondrial dynamics (fusion/fission), and mitophagy29 could function as early markers of the disease.
In cancer, energy metabolism (ATP synthesis) shifts in favor of anerobic glycolysis (Warburg effect) outside the mitochondria30,31, such that mitochondria redirect their functions (Fig. 1)30,32. Based on this change, a bioenergetic signature was established to categorize prognosis in various types of cancer33. Furthermore, analysis of the mitochondrial proteome can distinguish different stages of cancer, with specific molecular events additional to this change in energy metabolism34.
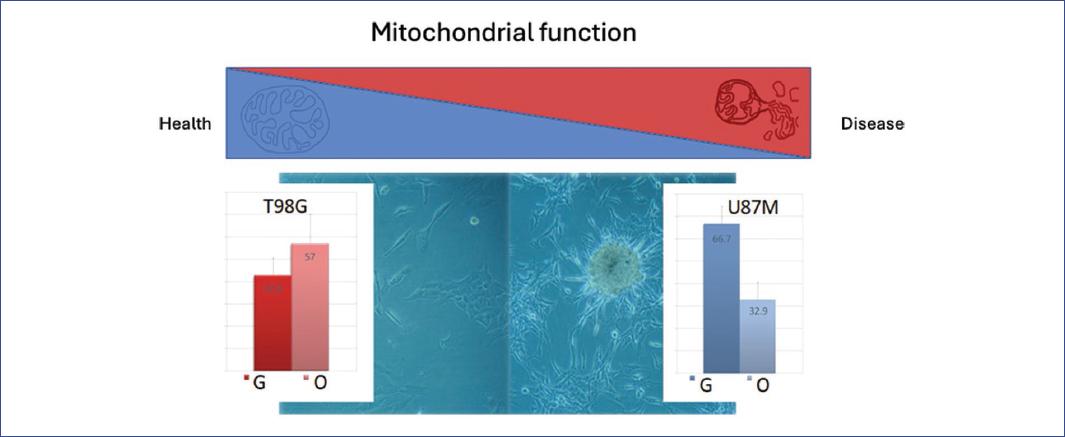
Figure 1 The transition to disease as a result of mitochondrial deterioration and resulting homeostatic loss. In cancer, mitochondria can follow the course of the disease and accompany disease progression (red triangle); the same occurs with non-communicable diseases, enabling the search for early markers of various diseases. In health, mitochondria are functional and structurally intact (blue triangle). In a glioblastoma cell model, less aggressive cancer (T98G cells) follows a more oxidative metabolism (O), as occurs in healthy cells, while advanced glioblastoma (U87 cells), where cells tend to form tumors, the metabolism becomes glycolytic (G), and mitochondrial structure is fragmented. This transition is associated with the dominance of certain metabolic pathways and structural changes in the mitochondria.
The distribution of mitochondria in the cell fluctuates and is found in different configurations, from bacteria-like units to tubular conglomerates extending and forming contact with other organelles35. Mitochondrial dynamics (fusion/fission processes) change notably in different pathologies, making their monitoring relevant in distinguishing health/disease. The mitochondrial arrangement reflects the metabolic-functional state and cellular health. A mitochondrial network increases oxidative capacity and is related to good health, while fragmentation into small units could indicate stress or the repair/degradation of damaged elements35.
Mitochondrial dynamics is a quality control mechanism that requires specific proteins; some, like dynamin-like protein and dynamin-related protein, are related to processes involved in multiple pathologies36. Mitochondria continuously fluctuate in number, function, and form in response to inter- and intracellular stimuli, whether due to changes in location or metabolic rate (Fig. 1), grouping them according to energy demands35.
Many pathologies involve mitochondrial dynamics; obesity, for example, has a particular interest in medical care as it is one of the main health problems in the pediatric population, and it implies and predicts other health problems such as infertility, hypertension, high cholesterol, and fatty liver, among others. Childhood obesity can lead to adult obesity with other health risks such as cardiovascular conditions and increased mortality rates37. It is interesting to note that these changes are described from the neonatal period and involve intrauterine processes since mothers with obesity during pregnancy have children who retain these changes after birth38,39. In this context, mitochondrial defects are usually found in energy metabolism, mtDNA mutations, increased oxidative stress, and, as mentioned, changes in mitochondrial dynamics. As a result, various therapeutic strategies targeting mitochondria have been proposed, such as the induction of thermogenesis or the use of natural molecules targeting mitochondria40, like MOTS-c, which, in addition to decreasing body fat, regulates insulin sensitivity and metabolic homeostasis41,42.
In this context, genomic variants and mitochondrial populations should be investigated considering their surroundings, as specific contacts with other organelles are established in the mitochondrial microenvironment. A mitochondrion can contact the endoplasmic reticulum (ER) and nucleus, with each interface involving different proteins43. The interaction with the ER is better understood where contacts occur through mitochondria-associated membranes (MAMs)44; the proteins located there are required for common activities (mitochondrial dynamics, calcium homeostasis, transport, among others) and become highly responsive metabolic platforms44. Regarding nucleus-mitochondria interaction, it has been observed that it regulates OXPHOS performance and determines somatic heteroplasmy dynamics45.
MAMs participate in signaling and maintenance of cellular homeostasis44,46, contain regulatory proteins and tumor suppressors47, and activate the immune system, as they harbor inflammasomes (Nlrp3: NOD-[nucleotide-binding oligomerization domain), LRR-Leucine rich repeats domain and pyrin domain-containing protein 3) which are activated during microbial infections or by interleukin secretion in the inflammatory response48,49. In common pediatric infections, such as Mycobacterium tuberculosis, Proteus mirabilis, Escherichia coli, Salmonella enterica, Staphylococcus aureus, Streptococcus pneumoniae, Neisseria meningitidis, and Plasmodium falciparum, there is generally an increase in reactive oxygen species, metabolic changes, and apoptosis, which occur through mitochondrial mediation; furthermore, it should be considered that various antibiotic treatments can damage mitochondrial function. Microbial pathogenicity is closely linked to mitochondrial damage, as many products derived from bacterial infections, such as nitric oxide, are respiratory complex inhibitors; the molecular and structural alterations of mitochondria caused by pathogens and treatments show great adaptability and therefore, their potential as therapeutic targets50.
Other proteins in MAMs are involved in neurodegenerative diseases: Presenilin 1 and 2 (PSEN1 and PSEN2) in Alzheimers; DJ1, Parkin (Park) 1 and 2, and Pink in Parkinsons; while AKT, mTORC2, GSK3b, and PTEN are involved in diabetes, and Sig1 in breast, liver, and colon cancer, among others44.
MAMs also contain chaperones generated during the unfolded protein response (UPR), which is involved in stress response and the biogenesis of various pathologies where nucleus-mitochondria communication is established51,52 and induces a response according to the stimulus. For example, when faced with a stressor, a transcriptional response can be activated, producing proteins safeguarded by chaperones while they achieve their functional structure. The UPR is distributed in MAMs53,54 and regulates synthesis/alarm (apoptosis) cycles (Figs. 1 and 2), which is exacerbated in various diseases and infections55. Therefore, the UPR proteins involved could serve as biomarkers or therapeutic targets.
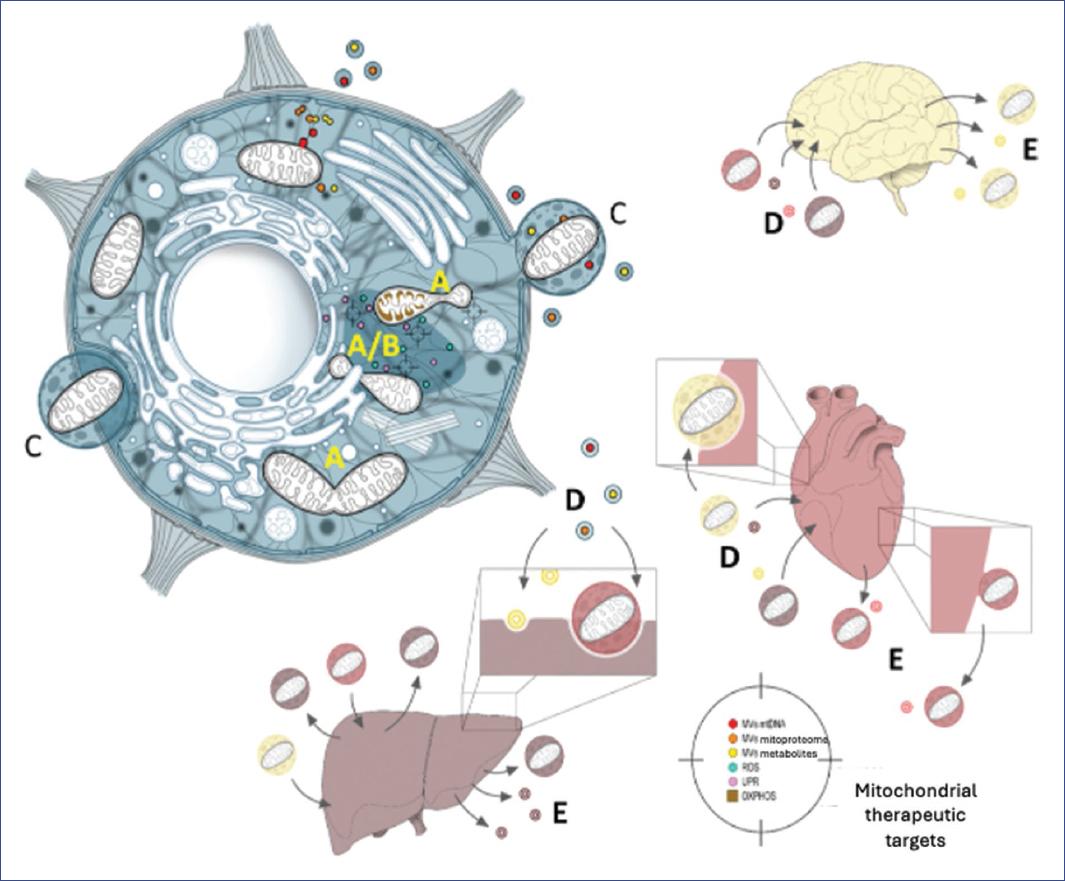
Figure 2 Mitochondria have great communication capacity at intra- and intercellular levels with various capabilities that can be used as therapeutic targets: At the intracellular level, fusion/fission (A/B) can signal different events in non-communicable disease pathogenesis as the damage caused leads to mitochondrial dysfunction. Likewise, mitochondria obtain information from the environment (unfolded protein response response, mitochondria-associated membranes), communicate with other cell organelles, and carry diverse information in extracellular vesicles (C, MVs) where they can even be contained completely and, in turn, can be directed to other targets or tissues such as brain, heart, or liver among others, through the bloodstream (D). The mitochondrial content they carry could activate (incoming arrows) a response to disease or release warning messages (E, outgoing arrows), a useful process in designing therapeutic strategies.
Therapeutic possibilities
There are several areas of interest for mitochondrial therapeutics, primarily focused on disease diagnosis, prognosis, and treatments. One of these approaches is through extracellular vesicles (EVs)56, which function in biomolecule transport and communication between cells and tissues57,58. Their content varies according to biological conditions5, and one advantage of EVs is that they are found in easily accessible fluids such as blood, saliva, and urine. The content of EVs can reach different tissues and influence the function, signaling, and regulation of various biological processes; they can also contain complete mitochondria or fragments, and there are even mitochondria-derived vesicles whose content has demonstrated diagnostic and therapeutic utility59-61. In experimental models, EVs correct the energy imbalance produced by ischemic, dilated, and hypertrophic heart diseases62. In disease, the number and diversity of EVs are exacerbated and affect different metabolic pathways34,58,59,63, so identifying their content could benefit the clinical approach to various diseases5,63,64.
Therapeutic targets for mitochondria-based medicine
Mitochondria-based medicine is founded on metabolic changes caused by disease32,65,66, thus functioning as a sensor for different clinical phases and stages, which could help define various biomarkers to facilitate treatment selection. Regarding treatment, there is a wide variety of antioxidant reagents, both natural and designed, that can target mitochondria to prevent, modulate, or treat NCDs67. For example, mitocans prevent cancer progression or drug resistance, and several of these can be easily manipulated68,69. Mitochondria have great inter- and intracellular communication capacity, making them novel therapeutic options, such as mitochondrial transplantation, which was conceived to restore or recover optimal functional and structural states of cells comprising the organ70,71. Mitochondrial transplantation emerges as a treatment strategy due to mitochondria's ability to transfer from one cell to another, particularly when mitochondrial damage exists. It could thus function as a treatment for diseases where typical therapies have not been effective. The process involves obtaining mitochondria (from adipose tissue, liver, or muscle) from a donor and transplanting them intranasally or by injection (systemic delivery) and has been tested primarily in murine models. Transfer can be facilitated using peptides that aid membrane absorption. At present, the technique's application focuses mainly on assisted reproduction to prevent transmission of mitochondrial diseases from mother to child; however, it has not been applied as a treatment for children who already have diseases72. Research is currently being conducted to evaluate its viability and safety as therapy for pediatric mitochondrial diseases73,74 such as Leigh syndrome, Mitochondrial Encephalopathy with Lactic Acidosis and Stroke-like episodes, Kearns-Sayre syndrome, Leber's Hereditary Optic Neuropathy, or mtDNA depletion syndrome.
There are still several challenges regarding mitochondrial transplantation for treating existing diseases, as ethical and biosafety considerations must be addressed. However, it is important to note that although the therapeutic potential is promising, widespread clinical application requires more research75. While the clinical potential of mitochondria is evident, there are limitations to their implementation. Much of our current information comes from biological models76 or has only been determined in small population samples and may vary. In addition, proteins with multiple functions require further studies about their role. Such is the case of ATP synthase and its intrinsic inhibitor (IF1)77 or sigma 1 receptor (a MAM chaperone)78, which perform multiple functions according to their location or interactions. Therefore, the identification and validation of mitochondria-related biomarkers, as well as the development of targeted therapies, offer new perspectives in disease diagnosis and treatment, making mitochondria-based medicine a field of research and clinical application of growing relevance. Thus, information about mitochondrial function and its byproducts becomes central aspects in the health/disease balance that can be approached as potential therapeutic targets (Fig. 2).
Conclusions
Mitochondria-based medicine emerges as a promising field of research and clinical application for treating diseases acquired in childhood or NCDs. This approach focuses on understanding how mitochondrial alterations can trigger and contribute to developing these diseases. Mitochondria-based medicine contributes to developing options targeting these organelles, offering new perspectives on disease understanding and treatment, including personalized approaches.
In a context where complex and multifactorial diseases represent a significant challenge for contemporary medicine, mitochondria-based medicine opens new opportunities to advance patient care and quality of life.











 nueva página del texto (beta)
nueva página del texto (beta)


