INTRODUCTION
The aquatic environment represents about 71% of the Earth’s surface (Seymour 2014). Life in these environments is conditioned by different factors such as temperature, salinity, the presence of sunlight, or the amount of oxygen dissolved in the water that determine the establishment of different biological communities. In the case of microorganisms, these represent around 70% of marine biomass (Bar-On and Milo 2019) and have a great metabolic diversity, especially in prokaryotic organisms, whose participation in biogeochemical cycles is essential for the correct functioning of the planet (Delong 2009, Seymour 2014, Deutschmann et al. 2023). Recently, the analysis of marine microbiota has been promoted to better understand its role in ecological processes and to investigate its possible biotechnological applications, such as the production of new bioactive compounds or the remediation of contaminated environments (Dash et al. 2013, Acosta-González and Marqués 2016, Hamdan et al. 2019, 2023). However, it is still less characterized than the terrestrial microbiota, especially that present on the ocean floor (Sanz-Sáez et al. 2020). Oil spills, as well as their derivatives, are one of the main causes of pollution in these environments, such as the destruction of the Deepwater Horizon oil platform in the Gulf of Mexico (2010), or the Prestige catastrophe in Galicia, Spain in 2002, in which thousands of tons of crude oil were released into the sea (Acosta-González and Marques 2016). Similar environmental disasters have also been recorded in the Mediterranean Sea, such as the release of large quantities of fuel off the coast of Jiyeb (Lebanon) in 2006 (Little et al. 2021, Hamdan et al. 2023).
The Mediterranean Sea is a continental sea that connects to the Atlantic Ocean through the Strait of Gibraltar and to the Indian Ocean through the Suez Canal. Surrounded by the continents of Europe, Africa, and Asia, this sea, with an area of approximately 2.5 million km² and a length of 3860 km, is the second-largest inland sea on the planet, surpassed only by the Caribbean Sea. Its pollution is not only due to occasional accidents, but also to commercial and tourist activity. In the case of coastal areas of the Mediterranean Sea, such as Costa del Sol (Málaga and Granada, Spain), a large part of this pollution is due to the increase in tourist and recreational activities, due to the use of motor boats (cruises, yachts, sport fishing boats, etc.). Malaga is one of the main tourist destinations on the Iberian Peninsula, welcoming approximately 2.6 million travelers between June and August 2023, with tourism increasing by 4.8% since 2022 (CSM 2023, DPM 2024). In addition, in relation to tourism revenue, an increase of 10.1% was estimated from 2022. Currently, the province of Malaga has 11 marinas along its coast (Fig. 1).
One of the best known is Muelle Uno (Málaga, Spain), located in the heart of the city, which stands out for its high presence of motor-boats for tourist and recreational purposes (PM 2024) As of 2021, the economic development of this city encouraged the construction of port infrastructures capable of accommodating the docking of luxury mega yachts and commercial cruise ships on this promenade. This area recorded an average temperature of 24.8 ºC on the Malaga coast in August 2023, which favors the proliferation of microorganisms facilitated by the presence of hydrocarbons.
This situation is similar in other parts of the Mediterranean Sea, where biological diversity is highly threatened by these fuels, since their chemical composition, which consists mainly of a mixture of aliphatic and aromatic hydrocarbons, can have harmful effects (Chandra et al. 2013, Varjani 2017). Despite being a minority, monocyclic aromatic hydrocarbons (MAHs) such as benzene, toluene, ethylbenzene, and xylene (BTEX), and polycyclic aromatic hydrocarbons (PAHs) are the main causes of these environmental catastrophes (Varjani and Upasani 2017, Honda and Suzuki 2020, Goveas et al. 2022). Acute or chronic exposure to these discharges significantly alters ecosystems and poses a risk to living organisms (White et al. 2017, Arockiaraj and Kankara 2018, Cariello et al. 2020).
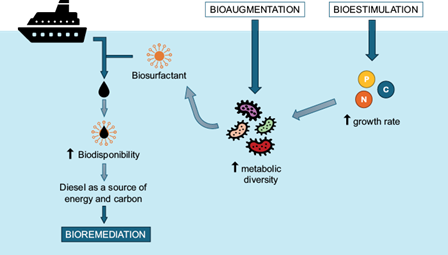
For this reason, the scientific community is investing great human and economic efforts in solving these problems. One of these strategies is based on remediation processes, which are addressed through physical, chemical, or biological methods (Varjani 2017). Among all of them, the biological method, commonly known as bioremediation, has proven to be the best alternative to eliminate hydrocarbons in these media, due to its low cost, low environmental impact, and high efficiency (Hamdan et al. 2019, Gao et al. 2023). Furthermore, the application of techniques such as biostimulation, bioaugmentation and the use of biosurfactants has further improved the efficiency of decontamination in these habitats (Fig. 2).
Biostimulation increases biodegradation rates by adding essential nutrients, such as nitrogen and phosphorus, to the medium (Luo et al. 2024), while bioaugmentation leverages the degradative capacities of different microorganisms to eliminate all compounds of the pollutant in question (Luo et al. 2024). Furthermore, the use of bacterial biosurfactants increases the availability of hydrocarbons in the marine environment as a potential energy and carbon source (Varjani and Upasani 2017, Goveas et al. 2022).
Hydrocarbon-oxidizing bacteria, or hydrocarbonoclastic bacteria, are able to metabolize these compounds via aerobic or anaerobic pathways. The degradation of aliphatic and aromatic hydrocarbons follows different metabolic pathways; therefore, different bacterial genera have genes encoding the enzymes required for each pathway. Some strains of the genera Alcanivorax sp. and Vibrio sp. have been shown to be capable of oxidizing aliphatic hydrocarbons (Imron et al. 2019, Sinha et al. 2021), while other strains of the genera Acidovorax sp., Bacillus sp., Burkholderia sp., and Sphingomonas sp. are capable of degrading aromatic hydrocarbons present in petroleum (Seo et al. 2009).
Other studies also highlight the degradative potential of genera such as Petrimonas sp. and Desulfovibrio sp., which appear to be able to metabolize PAHs vis anaerobic pathways on the ocean floor (Qian et al. 2020).
For this reason, the marine microbiota of waters contaminated by hydrocarbons from motor boats should be studied, as it may harbor hydrocarbonoclastic bacteria that could enhance the degradation of the pollutants present. In addition, biostimulation can promote the growth of specific cultivable isolates with the capacity to metabolize hydrocarbons such as benzene and pentane.
Therefore, in this work we will make an approach by characterizing the marine microbiota present in waters contaminated by hydrocarbons in Muelle Uno (Málaga, Spain), using molecular and culture techniques. Subsequently, we will evaluate the effect of biostimulation on the growth and composition of the bacterial microbiota under controlled experimental conditions with and without the presence of hydrocarbons (benzene and pentane).
MATERIALS AND METHODS
Marine samples
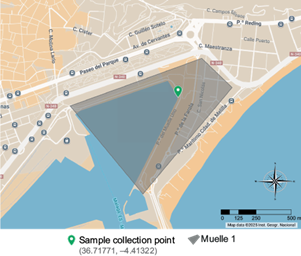
The sampled water comes from an area of Muelle Uno that contained a fuel slick, close to the docking area for recreational motorboats (coordinates 36.717713, -4.413229) (Fig. 3). The water temperature was 15 ºC (ST 2024). A total of 1 L of water was sampled from three different points. They were then transported to the laboratory at a cold temperature for processing.
Microbiological characterization of water and isolation of bacteria with potential biotechnological interest
In order to promote bacterial growth in the sampled water, and to select bacteria of biotechnological interest, the following bacterial culture media were prepared: nutrient-rich medium, tryptone soy broth (TSB, Oxoid, Hampshire, UK), and minimum nutrients medium (M9, Sigma-Aldrich, San Luis Missouri, USA), both supplemented with 15% NaCl (Honeywell, North Carloina, USA) to select marine bacteria. All culture media had a volume of 0.5 L, and different experimental conditions were performed (Table I). The hydrocarbons used in this study were benzene and pentane (ITW Reagents, Monza, Italy). The corresponding cultures had concentrations of 25 mL of benzene or pentane in 0.5 L of medium to exert greater pressure and select bacteria resistant to these hydrocarbons. To each condition, 50 mL of water from the samples mentioned in the previous section were added. In addition, the media in which there was no bacterial growth were supplemented with 5 mL of sterile 20% casamino acids (BD, New Jersey, USA) and/or 5 mL of sterile 50% D-glucose (PanReac AppliChem, Darmstadt, Germany). All media were tested in duplicate.
TABLE I EXPERIMENTAL CONDITIONS USED IN THIS STUDY INCLUDED TRYPTONE SOY BROTH (TSB) AND MINIMAL MEDIA (M9)* AS CONTROLS (C_TSB AND C_M9), AS WELL AS VERSIONS OF EACH SUPPLEMENTED WITH EITHER BENZENE (TSB_B, M9_B) OR PENTANE (TSB_P, M9_P). ALL MEDIA WERE SUPPLEMENTED WITH 15% NaCl.
| +Condition | Culture medium |
| C_TSB | Triptone soy broth |
| TSB_B | Triptone soy broth + benzene |
| TSB_P | Triptone soy broth + pentane |
| C_M9 | Minimal media (M9) |
| M9_B | M9 + benzene |
| M9_P | M9 + pentane |
*TSB and M9 are described in detail in section 2.
All media were incubated at 20 ºC throughout the experiment. The incubation period for each condition ranged from the addition of water until the appearance of turbidity, or up to a maximum of 26 days. In order to recover and isolate culturable bacteria, different tryptone soy agar plates (TSA, Oxoid) and supplemented with 15% NaCl, were streaked from two different types of samples: seawater samples and samples from media (TSB or M9) with added benzene (B) or pentane (P), designated as TSB_B, M9_B, TSB_P, M9_P. One hundred microliters of each sample at different dilutions were spread on plates and incubated for up to 72 h at 20 ºC.
After the incubation period ended, visually distinct colonies were isolated in pure culture. The strains were then stored in TSB + 15% NaCl + 20% glycerol at -80 ºC until analysis. For identification, DNA was extracted from each colony using the GeneJet DNA Purification Kit (Thermo Fisher Scientific, Massachusetts, USA) following the manufacturer’s protocol. The 16S rRNA region was amplified by PCR (Muyzer et al. 1998) and subsequently sent to Macrogen for sequencing. These sequences were identified by comparing them with the sequences available in the Genbank database (NCBI).
Taxonomic characterization after biostimulation
The appearance of turbidity in the media marked the end of its incubation period. From this moment, 1 mL of the surface of each medium was collected for the extraction of its total genomic DNA as described in the previous section. The quantification of the extracted DNA was performed using fluorometric methods, following the protocol of the commercial Qubit dsDNA HS Assay Kit (Thermo Fisher Scientific, Waltham, Massachusetts, USA).
All extracted DNA was stored at -20 ºC for further processing. DNA samples from each experimental condition were sent for massive sequencing using new-generation sequencing (NGS) techniques. Total DNA obtained from the media samples was sent for sequencing using the V3-V4 fragment of bacterial 16S rRNA using the Illumina MiSeq technology (Ilumina, San Diego, California, USA), using the paired-end sequencing technique (2 × 300 pb). This procedure was carried out by Novogene (Cambridge, United Kingdom) using the primers 5’CCTAYGGGRBGCASCAG3’ and 5’GGACTACNNGGGTATCTAAT3’. The analysis was carried out by the Ultrasequencing Service of the Bioinnovation Center of the University of Malaga (SCBI, UMA), using the technology of the Picasso supercomputer to carry out a sample quality control process with the FastQC software. In addition, for the purification of the obtained sequences, a workflow based on the MOTHUR software (v. 1.13.0) was used, which allowed the alignment of the paired reads to form the amplicon. The removal of adapters and chimeras from the sequencing was performed using the vsearch software (v. 2.16.0).
From the filtered data, the taxonomic assignment of the representative sequences of each amplicon sequence variant (ASV) was performed to obtain information about the taxa and their relative abundance. This process was carried out by comparing sequences with the SILVA database (v. 138.1) under 97% similarity and a threshold defined at 0.005%. The taxonomic analysis of the microbiota present in these samples was performed through graphical representations generated using the RStudio software (v. 4.4.0).
The study of bacterial communities in each sample was carried out using the Chao1, Shannon and Simpson indices, which described taxonomic richness, diversity and dominance, respectively. The graphical representation of these results allowed comparisons to be made between the mean α-diversity values of different experimental conditions. Variations in the composition of microbial communities between different samples were measured through β-diversity.
RESULTS
Isolation and characterization of bacterial strains in the presence of pentane and benzene
The incubation period for each condition ranged from the addition of the collected water to the presence of turbidity in the medium, indicative of microbial growth. Not all media developed this appearance, so a maximum incubation period of 26 days was established. In the other media, the incubation time varied according to the experimental condition (Table II).
TABLE II INCUBATION PERIOD (DAYS) FROM THE ADDITION OF WATER TO THE MEDIUM UNTIL THE DEVELOPMENT OF TURBIDITY. THE ADDITION OF CASAMINO ACIDS AND/OR GLUCOSE IS INDICATED WITH THE SYMBOL (+), AND THEIR ABSENCE WITH THE SYMBOL (-). TSB AND M9 AS CONTROLS (C_TSB, C_M9), AND EACH MEDIA SUPPLEMENTED WITH BENZENE (TSB_B, M9_B) OR PENTANE (TSB_P, M9_P).
| Condition | Incubation time (days) | Casamino acids | Glucose |
| Triptone soy broth (C_TSB) | 1 | - | - |
| Triptone soy broth and benzene | 1 | - | - |
| Triptone soy broth and pentene (TSB_P) | 2 | - | - |
| Minimal media (C_M9) | 6 | + | - |
| Minimal media and benzene (M9_B) | NC | + | + |
| Minimal media and pentene (M9_P) | 3 | + | - |
TSB: tryptone soy broth; M9: minimal media; NC: absence of growth during the incubation period.
TSB and M9 are described in detail in section 2.
The media corresponding to conditions C_TSB and TSB_B were the first to show turbidity after 24 h of incubation, whereas TSB_P required 48 h to develop turbidity. Conditions C_M9 and M9_P developed turbidity 24 h after supplementation with 20% casamino acids. However, in the M9_B condition, no growth was observed after the addition of casamino acids or after the subsequent addition of glucose (Table II).
From the media that presented turbidity, four colonies corresponding to the media containing benzene (TSB_B and M9_B) (14.29%) and three colonies from the media containing pentane (TSB_P and M9_P) (10.71%) were isolated. In addition, 21 colonies were obtained from the sampled seawater (75%). All of them were identified. Those colonies isolated from seawater belonged to the Proteobacteria phylum and the Gammaproteobacteria class. In all cases, the genera identified were Vibrio sp. and Pseudoalteromonas sp., with the isolation of a strain of Vibrio alginolyticus being noteworthy (Table III).
TABLE III COLONIES ISOLATED ON TRYPTONE SOYA AGAR PLATES FROM SEAWATER SAMPLES AND HYDROCARBON CONDITIONS: TSB AND MINIMAL MEDIUM (M9) MEDIA SUPPLEMENTED WITH BENZENE (TSB_B, M9_B) OR PENTANE (TSB_P, M9_P).
| Sample | Genus | % of genus per sample |
| Sea water | Pseudoalteromonas sp. | 28.57% |
| Vibrio sp. | 71.43% | |
| Triptone soy broth + bezene (TSB_B) | Bacillus sp. | 100% |
| Minimal media + benzene (M9_B) | Bacillus sp. | 100% |
| Triptone soy broth + pentane (TSB_P) | Vibrio/Photobacterium sp. | 50% |
| Vibrio sp. | 50% | |
| Minimal media + pentane (M9_P) | Vibrio sp. | 100% |
The colonies isolated from benzene conditions (TSB_B and M9_B) belonged entirely to the genus Bacillus sp., while those in the pentane groups (TSB_P and M9_P) corresponded mostly to the genus Vibrio sp. (Table III).
Post-bioestimulation taxonomic analysis of the microbiota
An abundance table was obtained from the sequences obtained. Graphical representations described the abundances of the phyla, families and genera present under the different experimental conditions. In all groups, the estimated Good’s coverage was 100%, so that all sequencing was representative of the bacterial groups present in each group.
All M9 media were mainly represented by sequences belonging to the Proteobacteria phylum (> 95%), whereas the TSB media supported the growth of different phyla depending on the experimental condition.
In the TSB control (C_TSB), a similar abundance was described for the phyla Proteobacteria and Fusobacteriota (> 40%), as well as the presence of the phylum Bacteroidota in a lower proportion (< 5%). However, the addition of benzene in TSB_B caused a considerable increase of the Proteobacteria phylum (> 95%), causing the displacement of Fusobacteriota below 10%, as well as the absence of Bacteroidota in this condition. The TSB media with pentane (TSB_P) were also represented almost entirely by the Proteobacteria phylum (> 95%), displacing the remaining phyla in its control (Fig. 4).
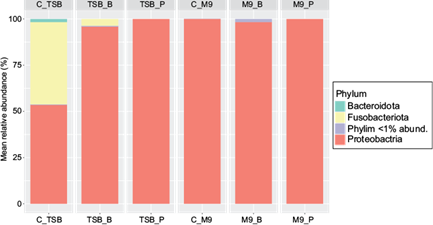
Fig. 4 Mean relative abundance values (%) of the resulting phyla in each experimental condition. Triptone soy broth (TSB) and minimal media (M9) as controls (C_TSB, C_M9), and each media supplemented with benzene (TSB_B, M9_B) or pentane (TSB_P, M9_P).
In figure 5, the M9 control (C_M9) is mainly represented by the Vibrionaceae family (> 75%), along with others such as Pseudomonadaceae (> 5%) and Pseudoalteromonadaceae (> 5%) to a lesser extent. In the TSB control (C_TSB), the presence of the families Fusobacteriaceae (> 35%), Pseudoalteromonadaceae (> 25%), and Vibrionaceae (< 20%) was observed. Benzene limited the growth of the Vibrionaceae family in both media M9_B and TSB_B. Moreover, the presence of benzene increased microbial diversity, with a greater number of bacterial families being represented. Among them, the Pseudomonadaceae family was dominant in both M9_B and TSB_B. This hydrocarbon also promoted the growth of the Sphingomonadaceae and Burkholderiaceae families to a lesser extent, whose relative abundances were not influenced by the media’s nutritional composition. The families Rhodobacteraceae (< 10%) and Xanthomonadaceae (< 10%) appeared exclusively in M9_B, whereas Comamonadaceae grew only in TSB_B (< 10%). The presence of pentane increased the abundance of the Vibrionaceae family by over 90%, both in the M9 (M9_P) and TSB (TSB_P) media.
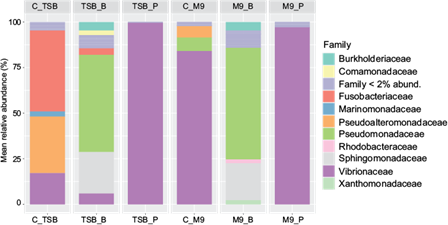
Fig. 5 Average relative abundance values (%) of the resulting families in each experimental condition. Triptone soy broth (TSB) and minimal media (M9) as controls (C_TSB, C_M9), and each media supplemented with benzene (TSB_B, M9_B) or pentane (TSB_P, M9_P).
The C_M9 control was mainly represented by the genera Vibrio sp. (> 40%) and Aliivibrio sp. (> 30%), both belonging to the Vibrionaceae family, together with Pseudomonas sp. (< 15%) and Pseudoalteromonas sp. (< 15%) to a lesser extent. The abundance of the genera corresponding to this family decreased below 15% in the TSB control, in favor of the genera Fusobacterium sp. (< 40%) and Pseudoalteromonas sp. (> 30%). The addition of benzene inhibits the growth of Aliivibrio sp. and decreases the relative abundance of Vibrio sp. and Fusobacterium sp. in TSB_B to below 10%. In minimal media, benzene prevents the development of all genera of the Vibrionaceae and Fusobacteriaceae families. Benzene conditions are mainly represented by the genus Pseudomonas sp. (> 40%), as well as others such as Blastomonas sp., Burkholderia sp. and Sphingopyxis sp. to a lesser extent. This hydrocarbon also promotes the minor appearance of Acidovorax sp. in TSB media, as well as that of Pseudoxanthomonas sp. in M9 media. M9 media with pentane (M9_P) were mostly represented by Vibrio sp. (>90%), while the presence of this hydrocarbon in TSB media (TSB_P) also allowed an abundance greater than 5% of the genus Aliivibrio sp. (Fig. 6)
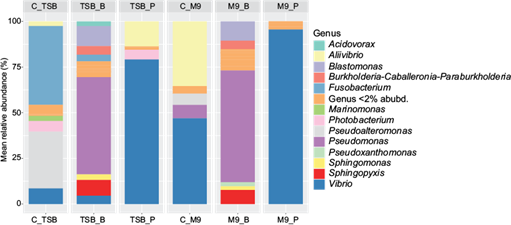
Fig. 6 Average relative abundance values (%) of the resulting genera in each experimental condition. Triptone soy broth (TSB) and minimal media (M9) as controls (C_TSB, C_M9), and each media supplemented with benzene (TSB_B, M9_B) or pentane (TSB_P, M9_P).
Table IV shows the average values of the number of ASVs sampled, as well as the Chao1, Shannon, and Simpson indices for the alpha-diversity analysis by experimental condition. One of the samples corresponding to the TSB_B condition failed the quality control, so the average value and deviation of this treatment could not be represented.
TABLE IV AVERAGE ALPHA-DIVERSITY VALUES BY EXPERIMENTAL CONDITIONS, BASED ON AMPLICON SEQUENCE VARIANTS (ASVs), CHAO1, SHANNON, AND SIMPSON INDICES. TRIPTONE SOY BROTH (TSB) AND MINIMAL MEDIA (M9) AS CONTROLS (C_TSB, C_M9), AND EACH MEDIA SUPPLEMENTED WITH BENZENE (TSB_B, M9_B) OR PENTANE (TSB_P, M9_P). MEAN VALUES ± STANDARD DEVIATION ARE REPRESENTED.
| Condition | ASVs | Chao1 | Shannon | Simpson |
| C__TSB | 113.50 ± 12.02 | 113.50 ± 12.02 | 2.28 ± 0.10 | 0.76 ± 0.01 |
| TSB_P | 96.00 ± 35.36 | 96.00 ± 35.36 | 2.72 ± 0.46 | 0.87 ± 0.05 |
| C_M9 | 136.50 ± 4.95 | 136.50 ± 4.95 | 3.02 ± 0.09 | 0.92 ± 0.00 |
| M9_B | 200.00 ± 8.49 | 200.00 ± 8.49 | 2.45 ± 0.13 | 0.85 ± 0.02 |
| M9_P | 60.00 ± 7.07 | 60.00 ± 7.07 | 1.93 ± 0.10 | 0.80 ± 0.01 |
Principal coordinate analysis (PCoA) allowed us to study how distinct the bacterial communities were between the different experimental groups, as well as possible differences within samples of the same experimental condition (Fig. 7).
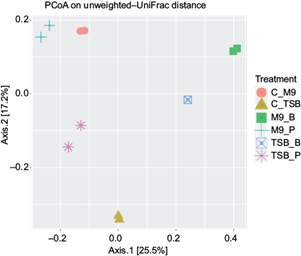
Fig. 7 Principal coordinate analysis (PCoA) between experimental conditions. Each symbol represents a sample of a given treatment. The legend relates each symbol to its corresponding treatment: triptone soy broth (TSB) and minimal media (M9) as controls (C_TSB, C_M9), and each media supplemented with benzene (TSB_B, M9_B) or pentane (TSB_P, M9_P).
The control groups (C-M9 vs C-TSB) differed greatly from each other. However, benzene produced more similar bacterial communities between TSB_B and M9_B, while pentane led to greater similarity between C_M9 and M9_P, as well as greater distance between these communities and that of TSB_P (Fig. 7).
DISCUSSION
The presence of hydrocarbons in the marine environment is a significant environmental problem that alters microbial communities and other resident organisms. The environmental impact of these organic compounds in aquatic environments has been extensively documented in previous research (White et al. 2017, Arockiaraj and Kankara 2018, Pereira et al. 2018). This study reveals how hydrocarbon pollution affects the growth and composition of marine microorganisms in an experimental setting, highlighting the importance of nutrients such as nitrogen for the proliferation of bacteria with bioremediation potential. Limitation of essential nutrients in contaminated environments prolonged incubation times under all conditions, this effect being particularly pronounced in the presence of benzene and pentane, where slower microbial growth was observed compared to controls. This is in line with previous studies that also highlight nitrogen and phosphorus as key elements in the biostimulation of contaminated waters (Varjani and Upasani 2016a, Luo et al. 2024).
In the case of water samples, members of the genera Vibrio sp. and Pseudoalteromonas sp. were isolated, with V. alginolyticus standing out, a species previously identified as a potential diesel degrader (Imron et al. 2019, Muralidharan et al. 2023). Other species of biotechnological interest within this genus are V. natrigiens (Thoma and Blombach 2021, Walton and Buchan 2024) and V. cyclotrophicus (Hedlund and Staley 2001). Regarding the genus Pseudoalteromonas, some strains stand out, such as Pseudoalteromonas sp. JSTW, capable of degrading aliphatic and aromatic compounds (Zan et al. 2021), and P. agarivorans SDRB-Py1, which is able to produce effective biosurfactants for the emulsification of hydrocarbons such as n-hexane and benzene, increasing their bioavailability (Lee et al. 2018, Ganesan et al. 2022).
Regarding the colonies isolated from the media supplemented with benzene, all the strains belonged to the genus Bacillus sp., which also encompasses some hydrocarbonoclastic species such as B. algicola 003-Phe1, capable of completely degrading aliphatic compounds of nine to 14 carbons, and others of higher molecular weight (Lee at al. 2018). P. agarivorans and B. subtilis have the ability to produce biosurfactants effective in the emulsification of benzene and n-hexane (Lee et al. 2018). B. subtilis is capable of producing lipopeptides and rhamnolipids as biosurfactants (Parthipan et al. 2017, Lee et al. 2018) and also presents genes encoding enzymes involved in the alkane degradation pathway, such as alkane hydrolase enzymes and alcohol dehydrogenases (Parthipan et al. 2017). The strains B. amyloliquefaciens VBS03 and B. pumilus VBS37 use hexadecane molecules as the sole carbon source in artificial marine culture media, degrading 99.6 and 86.9% of this compound over 28 days, respectively (Ferrari et al. 2019). The formation of a bacterial consortium consisting of strains of B. pumillus, P. stutzeri, and A. calcoaceticus was able to catabolize PAHs such as pyrene and anthracene (Marzuki et al. 2021).
Pentane present in the culture media selected or promoted the growth of Vibrio sp. again, suggesting that these organisms are particularly resilient to the presence of low molecular weight aliphatic hydrocarbons (Hedlund and Staley 2001, Fernandes et al. 2020).
Metagenomic analysis of the bacterial groups present in each culture medium again confirms the dominance of the Proteobacteria phylum in all media with benzene and pentane. The absence of the Bacteroidota and Fusobacteriota phyla in minimal media indicates that their nutritional requirements are more demanding than those of Proteobacteria, which mainly require a nitrogen source to grow. Furthermore, benzene inhibits the growth of Bacteroidota and limits Fusobacteroidota, decreasing its abundance compared to the control. Pentane also shows a negative influence on both phyla, benefiting Proteobacteria. However, a similar study carried out on the coasts of Lebanon positioned Bacteroidota as the second most abundant phylum behind Proteobacteria in the presence of oil spills (Hamdan et al. 2023). In addition, the class Bacteroidia has been identified in marine sediments contaminated by hydrocarbons (Dell’Anno et al. 2021).
It should be noted that the presence of the Fusobacteriota phylum in the control is unusual compared to similar studies, where it is not even a representative group (Hamdan et al. 2023). Gutiérrez et al. (2016) identified the genus Fusobacterium sp., which belongs to this phylum, on the ocean surface of the Gulf of Mexico after the Deepwater Horizon environmental accident. The identification of this genus in surface layers of the marine environment, where oxygen availability is high, is atypical due to its obligatory anaerobic metabolism, which allows it to be commonly found in marine sediments (Gutiérrez et al. 2016). These results lead to future research to explain the development of this genus under aerobic conditions.
Regarding the identified genera, the presence of Vibrio sp. increases significantly in media supplemented with pentane. The abundance of this genus has already been described in marine environments contaminated with tar balls (Fernandes et al. 2020). Meanwhile, media with benzene increased the abundance of the genus Pseudomonas sp. This fact does not correspond with the results obtained in similar studies (Hamdan et al. 2023). However, its degradative potential was studied in different species previously mentioned (Mittal and Singh 2009, Rocha et al. 2011, Sajna et al. 2015, Varjani and Upasani 2016a, b).
This study also shows how benzene promotes the growth of other genera, such as Burkholderia sp., Sphingomonas sp., Blastomonas sp. Sphingopyxis sp., and Acidovorax sp., which were previously classified as PAH degraders according to this study (Seo et al. 2009). The genomic study of different strains of the genus Sphingopyxis sp. reveals the appearance of genes related to the degradation of aromatic hydrocarbons through benzoate metabolism (Sharma et al. 2021, Yang et al. 2020). The genus Blastomonas sp. is also representative only in treatments with benzene, however, no previous studies have been found that study the influence of benzene on the growth of this genus. On the other hand, media enriched with pentane reflected the abundance of the genus Vibrio sp.
However, environmental factors such as temperature, oxygen availability, and the presence of additional contaminants can modulate microbial metabolic pathways and, consequently, affect their hydrocarbon degradation efficiency. Although this study did not expose the bacterial communities to temperatures above the Mediterranean average, it is important to consider that climate change may alter microbial dynamics and functionality. These shifts could either enhance or impair the effectiveness of bioremediation processes. Therefore, future research should evaluate the metabolic stability and performance of these candidate strains under variable environmental conditions that simulate projected climate scenarios.
Although this study focused on microbial characterization in the presence of benzene and pentane, it is important to consider the ecological implications of future field applications. Benzene degradation can generate intermediate compounds such as catechols or organic acids, which may retain some toxicity if not fully metabolized. Pentane degradation mainly produces short-chain alcohols, aldehydes, and carboxylic acids, which are less toxic but could still disrupt microbial balance or water quality at high concentrations. Additionally, the bioestimulation approach used, adding nutrients like casamino acids and glucose, could cause ecological imbalances if applied in natural environments without proper control. Therefore, nutrient selection and dosing must be carefully evaluated in future studies to minimize potential environmental impacts.
Finally, this study also highlights how the combined use of molecular and culture techniques can provide a more accurate view of microbial communities in contaminated environments. Alpha-diversity data and differences in beta-diversity show a clear response of the marine microbiota to different experimental conditions and types of hydrocarbons. These results highlight the importance of a comprehensive approach in the study of marine microbiota and its role in bioremediation, showing that each hydrocarbon induces specific variations in the composition and diversity of bacterial communities (Seymour 2014, White et al. 2017).
CONCLUSIONS
The addition of hydrocarbons such as benzene and pentane proved to be a limiting factor, prolonging incubation times and reducing bacterial growth in all experimental media, even those enriched with carbon and nitrogen sources. However, these contaminants favored the development of bacterial genera such as Pseudomonas sp., Bacillus sp., and Vibrio sp., which have been previously identified as hydrocarbon degraders. Specifically, benzene promoted the abundance of Pseudomonas sp. and facilitated the isolation of Bacillus sp., while pentane significantly increased the presence of Vibrio sp. These discoveries highlight the importance of these bacteria in the bioremediation of hydrocarbons in contaminated marine ecosystems, pointing to future research on their specific degradative capabilities in the presence of different types of hydrocarbons.











 nueva página del texto (beta)
nueva página del texto (beta)