The genus Populus L. (Salicaceae Mirb.) consists of 32 species worldwide, mostly distributed in the temperate zone of the Northern Hemisphere (Kim 2018). Poplars have an outstanding economic importance for production of wood of industrial purposes, furniture and firewood. Also, they prevent soil erosion, favor restoration of riverbank environments and carbon sequestration reducing the effects of climate change (Gordon 2001). In Mexico, there are 11 species, outnumbering those of the United States (eight spp.) The state of Jalisco, with five species of poplars, represents 45 % of Mexican species richness of the genus and 16 % worldwide (Vázquez-García & Cuevas-Guzmán 1989, Eckenwalder 1996, Martínez-González & González-Villarreal 2005, Dickmann & Kuzovkina 2014, Vázquez-García et al. 2017, 2019). The two newly described species of white poplars: Populus luziarum A. Vázquez, Muñiz-Castro & Padilla-Lepe and P. primaveralepensis A. Vázquez, Muñiz-Castro & Zuno, are endemic to central Jalisco, have a very restricted distribution along riparian forests and consist of few and small populations, thus, they are ranked as critically endangered following the criteria of the IUCN Red List, and urgently need a conservation strategy (Vázquez-García et al. 2017, 2019, IUCN 2019).
Seed production, dispersal and germination are important factors constraining population dynamics (Harper 1977). Germination, from imbibition to radicle emergence, is often the most critical phase, during the life cycle of any terrestrial plant (Vázquez-Yanez & Orozco-Segovia 1993).
In the family Salicaceae, there are species of temperate (mesothermal) and cold (continental, microthermal) climates with two seed dispersal periods: late spring-early summer (hereafter name as “early”) and late summer-early autumn (hereafter named as “late”) (Zasada & Viereck 1975, Zasada & Densmore 1980) in addition, some subtropical (megathermal) species are reported, where the relationship between germination and seed dispersal is scarcely known. Populus seeds do not develop a dormant state but have high viability and germination percentage (Karrenberg et al. 2002). Due to their characteristics, Populus luziarum and P. primaveralepensis seeds are small, measuring between 0.2 × 0.1 mm and 0.3 - 0.7 × 0.2 - 0.4 mm, long and width respectively (Vázquez-García et al. 2017, 2019), so they are considered “late seed-dispersing species” as in other species of the same genus, the seeds can be dispersed by wind or water (Karrenberg et al. 2002). Since Populus seeds lack endosperm, they contain little energy, and their longevity is limited to a few days or weeks and prevent the formation of long-term seed banks under natural conditions (Muller et al. 1982, Venable & Brown 1988, Karrenberg et al. 2002, Bonner 2008). Seed storage of Populus does well at low temperatures, near or below 0 °C, particularly in late seed-dispersing species from boreal to template regions of the Northern Hemisphere with subarctic, circumpolar or cold climates (Moss 1938, Zasada & Viereck 1975, Tauer 1979, Zasada & Densmore 1980, Fechner et al. 1981, Pence 1996, González et al. 2010, Popova et al. 2013, Kim 2018). Under ex-situ conservation conditions, seeds tend to increase their longevity by about 12 years or more, but their viability may deteriorate (Popova et al. 2013, Kim 2018). It is unknown whether cold conditions (4 °C) are favorable in late seed-dispersing subtropical species since no studies are known from subtropical climates (Holdridge 1967) as those of Mexican mountains and highlands. Also, the method of storing Populus seeds could depend on the species (Wyckoff & Zasada 2008). Optimal storage conditions for Populus luziarum and P. primaveralepensis seeds are yet to be determined, so studies are needed to develop short and long-term seed conservation protocols in subtropical species. We hypothesize that germinability behavior of Populus luziarum and P. primaveralepensis, subtropical species are like Salicaceae boreal and template species dispersed in spring or early summer, since they have characteristics such as high germination percentages, germinate faster and loss of viability when stored at ambient temperature. This study can contribute to the knowledge of ex-situ conservation of these two endemic and endangered species in western Mexico, providing new life history data of both species and allowing plant production to contribute to their population and restoration.
Materials and methods
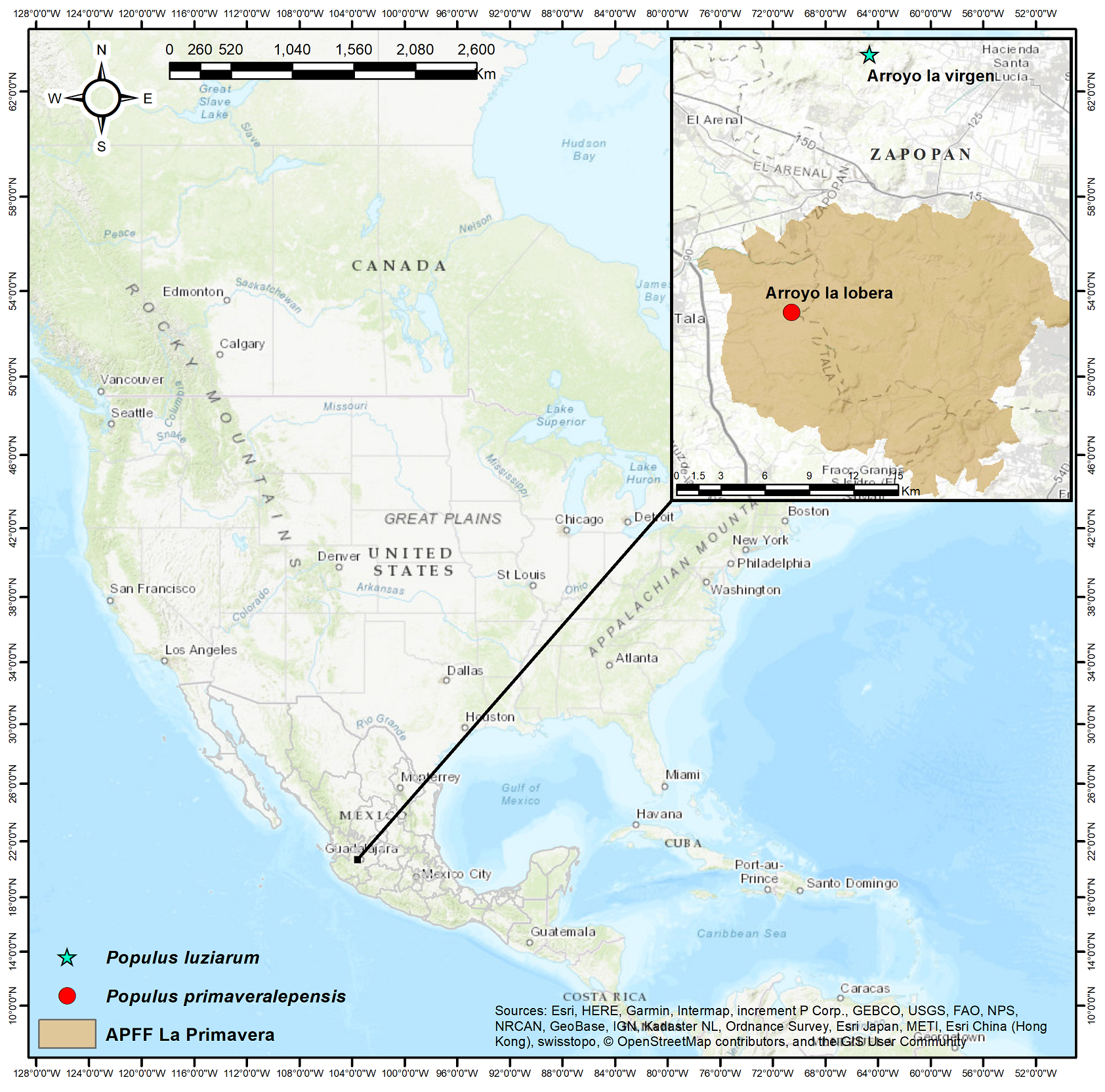
Seeds of the two species were collected on two riparian forests of western Mexico. Both species are in the municipality of Zapopan, Jalisco, Mexico (Vázquez-García et al. 2017, 2019). Populus luziarum is found in Arroyo La Virgen (20° 48' 53.22'' N, 103° 34' 51.30'' W), with an average annual temperature of 21.3 °C, and annual precipitation of 942 - 946 mm (Ruiz-Corral et al. 2018). Populus primaveralepensis in Arroyo La Lobera (26° 18' 0'' N, 103° 35' 59.9'' W) in the Área de Protección de Flora y Fauna La Primavera. Annual precipitation ranges from (899 - 910 mm), with an average annual temperature of 20.6 °C (SEMARNAP 2000, Ruiz-Corral et al. 2018) (Figure 1).
Source: Esri, HERE, Garmin, Intermap, increment P Corp., GEBCO, USGS, FAO, NPS, NRCAN, GeoBase, IGN, Kardaster NL, Ordnance Survey, Esri Japan, METI, Esri China (Hong Kong), swisstopo, © OpenStreetMap contributors, and the GIS User Community
Figure 1 Seed collection sites for Populus luziarum and P. primaveralepensis in central Jalisco in the western Trans-Mexican Volcanic Belt.
Seed collection and management. During October 2019, catkins with yellowish and brown capsules slightly opened showing cottoned seeds (mature capsules) (Bonner et al. 2008) were collected from five branches of at least 10 trees per species 10 - 400 m apart each other, for a total 250 catkins of Populus luziarum and 310 of P. primaveralepensis. They were taken to the Laboratory of Evolution of Ecological Systems of the Centro Universitario de Ciencias Biológicas y Agropecuarias (CUCBA) of the Universidad de Guadalajara, in airtight plastic bags. The seeds were immediately extracted from the capsules by hand using a strainer; about 1,200 seeds per species were obtained. The seeds were placed for (zero to eight weeks) in sterile plastic bottles with hermetic caps, in two storage conditions: average room temperature inside the laboratory (21 °C), and at 4 °C, placed inside a refrigerator (TOR-REY, Model R-14).
Physical integrity of the seeds. The physical integrity of freshly collected seeds of both species was evaluated according to ISTA (2020) using X-ray equipment (FAXITRON, Model MX-20, Specimen X-ray System), at the National Center for Genetic Resources (CNRG-INIFAP), in Tepatitlán de Morelos, Jalisco, Mexico. A total of 100 seeds of each species (five replicates with 20 seeds each), were placed on a transparent adhesive tape (double-sided) and fixed in transparent plastic foil. The plastic foil was placed inside the X-ray equipment and subject to radiation for 12 s at 26 kV. Internal morphological changes that could produce damage to seeds during the ripening process and that could influence germination were identified. The number of full and undamaged seeds was quantified and repotted in percentage.
Germination tests. An experiment with 100 seeds randomly selected per specie was carried out to obtain the percentages of germination, five replicates of 20 randomly selected seeds were used. Seeds were placed in sterile glass Petri dishes (60 × 15 mm), containing cotton pads, which were soaked just one time with 5 ml of distilled water, sealed with plastic wrap to prevent evaporation. They were placed in a germination chamber (LUZEREN, Model SPX250) with photoperiods of 12 hrs at 24 °C, based on Karrenberg et al. 2002 who cited that Populus germinate more than 80 % at 15 to 27 °C. The seed was considered germinated when the radicle was observed (Baskin & Baskin 2014) and compare them with the results of the physical integrity. In the second experiment, the storage time factor with nine levels (zero to eight weeks) was evaluated across two levels of storage temperature treatments: 21 and 4 °C, both temperatures were selected because they have been tested in Salicaceae species, in particular Populus (Moss 1938, Siegel & Brock 1990).
The experimental design was completely randomized, with a 2 × 9 factorial arrangement, with two storage temperatures, and nine storage time trials. For both storage temperatures, freshly collected seeds (week 0) and eight sequential stored seed lots were put to germinate every week, from zero to eight week. For each combination of treatments per species, similar conditions at germination test of first experiment, the number of seeds germinated was recorded daily until seventh day, after no further germination occurred. The number of germinated seeds were counted and expressed as germination percentage, that was calculated, as per the formula by Scott et al. (1984):
were, n
i
the number of seeds germinated in each experimental unit, k the last day of germination evaluation and N, the total number of seeds. Mean germination rate was calculated as the reciprocal of the mean germination time, according to Ranal et al. (2009). Mean germination time
where t i the time from the beginning of the experiment to the i th observation (in days); n i the number of seeds germinated in the i th time (not the accumulated number, but the number corresponding to the i th observation), and k, the last day of germination evaluation.
Statistical analysis. A Chi-square χ2 goodness of fit test was used between the number of seeds with good physical integrity and total germinated seeds from freshly collected seeds. Additionally, germination curves were analyzed through time with a logarithmic regression for both storage treatments, since this was the one that had the best adjustment according to its determination coefficient. Later, germinability and mean germination rate data were analyzed with analysis of variance (two ways ANOVA), where the storage time (weeks) and the storage temperature treatment were used as discrete explanatory factors. The germination percentage was transformed with
Results
It was observed that 96 % (Populus luziarum) and 98 % (P. primaveralepensis) of seeds were fully developed, with a well-defined embryonic axis (radius lucida region), and without insect, mechanic damage or malformations. The rest of the seeds showed entire seed cavity opaque, so the embryonic axis is not observed (Figure 2). We found 91 and 95 % of the seeds germinated in 24 h for P. luziarum and P. primaveralepensis, respectively, after this time no germination was observed (Figure 3). Furthermore, no statistically significant differences were found between the proportions of physical integrity test and germination of recently collected seeds (Table 1).
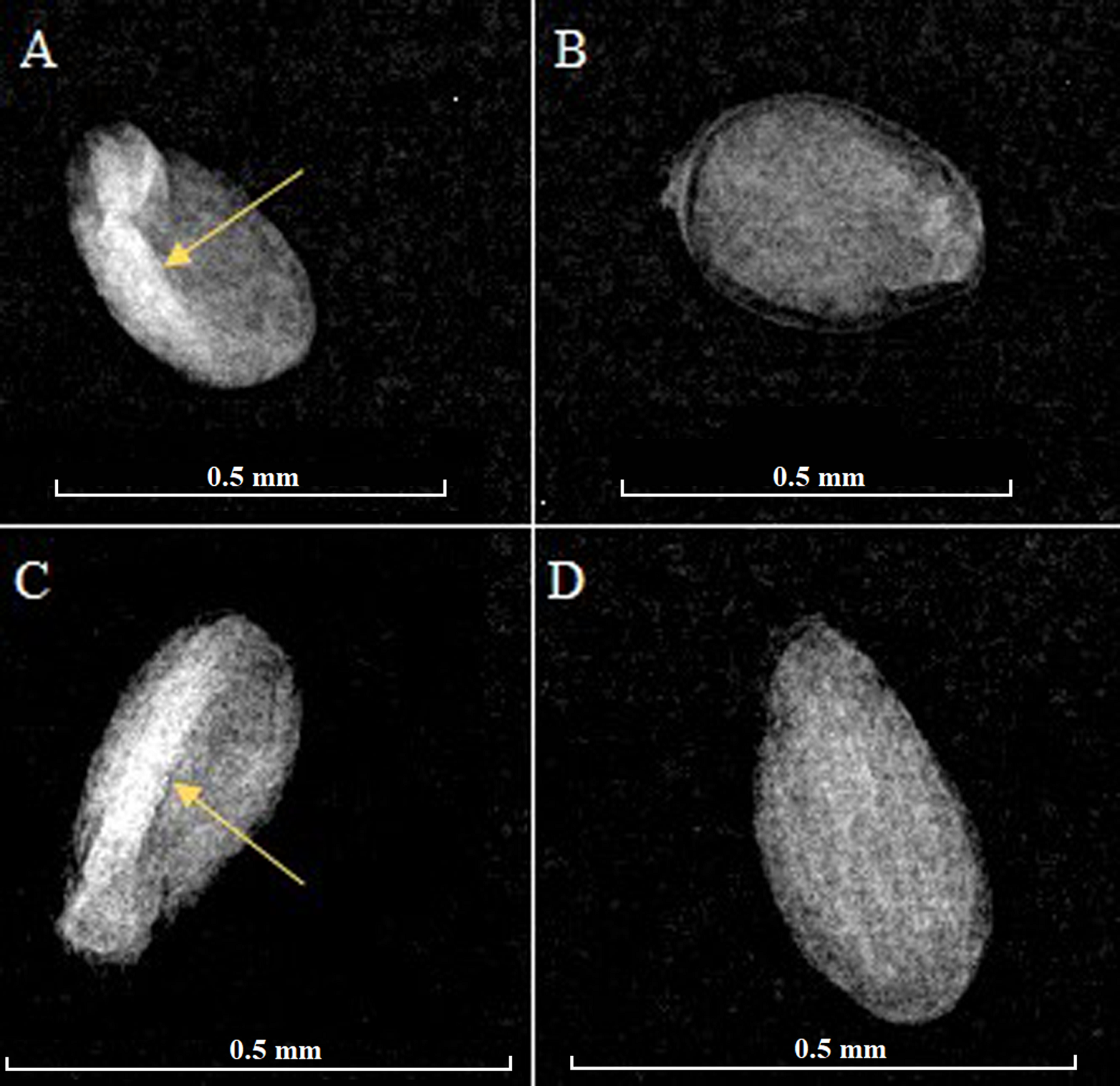
Figure 2 X-ray images of the physical integrity of Populus luziarum (A, B) and P. primaveralepensis seeds (C, D). The arrows indicate embryonic structures. (A, C) and full and undamaged seeds, (B, D) seeds without defined embryonic structures.
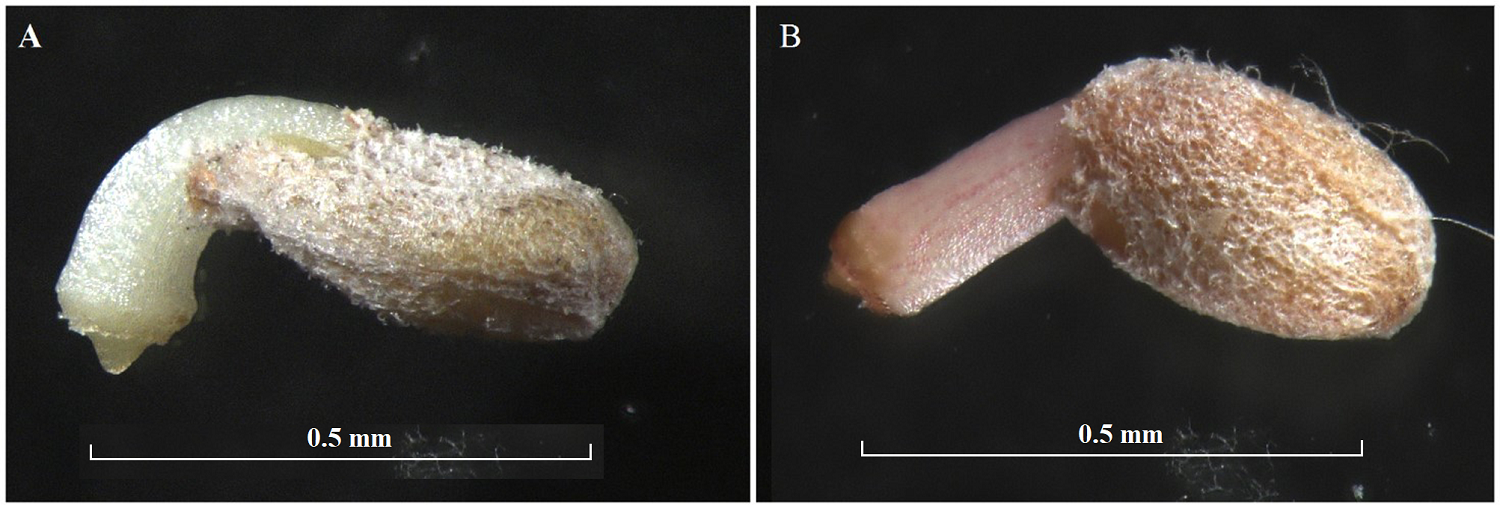
Figure 3 Germination after 24 hrs, at the emergence of the radicle. (A) Populus luziarum and (B) P. primaveralepensis.
Table 1 χ2 goodness of fit test between germination from freshly collected and physical integrity seeds (full and without damages) of Populus luziarum and P. primaveralepensis (n = 100).
| Species | Physical integrity (%) | Germination (%) | χ2 | P-value |
|---|---|---|---|---|
| Populus luziarum | 96 | 91 | 0.1336 | 0.7146 |
| Populus primaveralepensis | 98 | 95 | 0.0466 | 0.8290 |
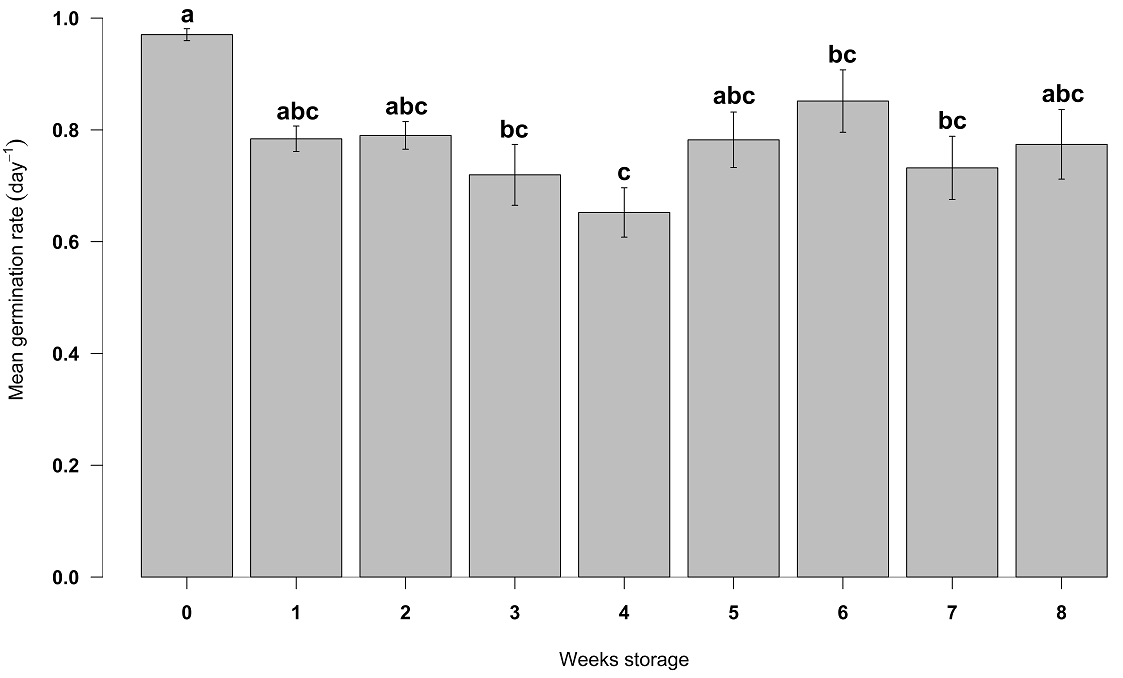
Seeds of Populus luziarum also showed a decrease in germination concerning increased time. Based on logarithmic adjustment, the coefficients of determination were R 2 = 0.86 and R 2 = 0.86 for seeds stored at 4 and 21 °C, respectively (Figure 4). If the conditions are kept constant, according to equations generated, in seeds stored at room temperature, germination will decrease 44 % before first week of storage, and will decrease 91 % after eight weeks of storage. In contrast, the refrigerated seeds decreased their germination until 50 % in seven weeks. ANOVA revealed significant differences between storage temperature levels (F = 44.06, P < 0.001) and weeks storage (F = 63.46, P < 0.001) for germination percentage. The highest germinability was observed at week 0 for both the 4 and 21 °C storage treatments (93 ± 2.0 and 91 ± 2.4 %), respectively. The lowest germination (14 %) was obtained after the fifth week for the 21 °C treatment and the eight weeks for the seeds stored at 4 °C, (Table 2). The mean germination rate was generally rapid but differed statistically among weeks storage (F = 4.78, P < 0.01). Week seven had the slowest mean germination rate of 0.6 ± 0.09 day -1, while the fastest germination was obtained at week 0 (0.9 ± 0.01 day -1) (Figure 5).
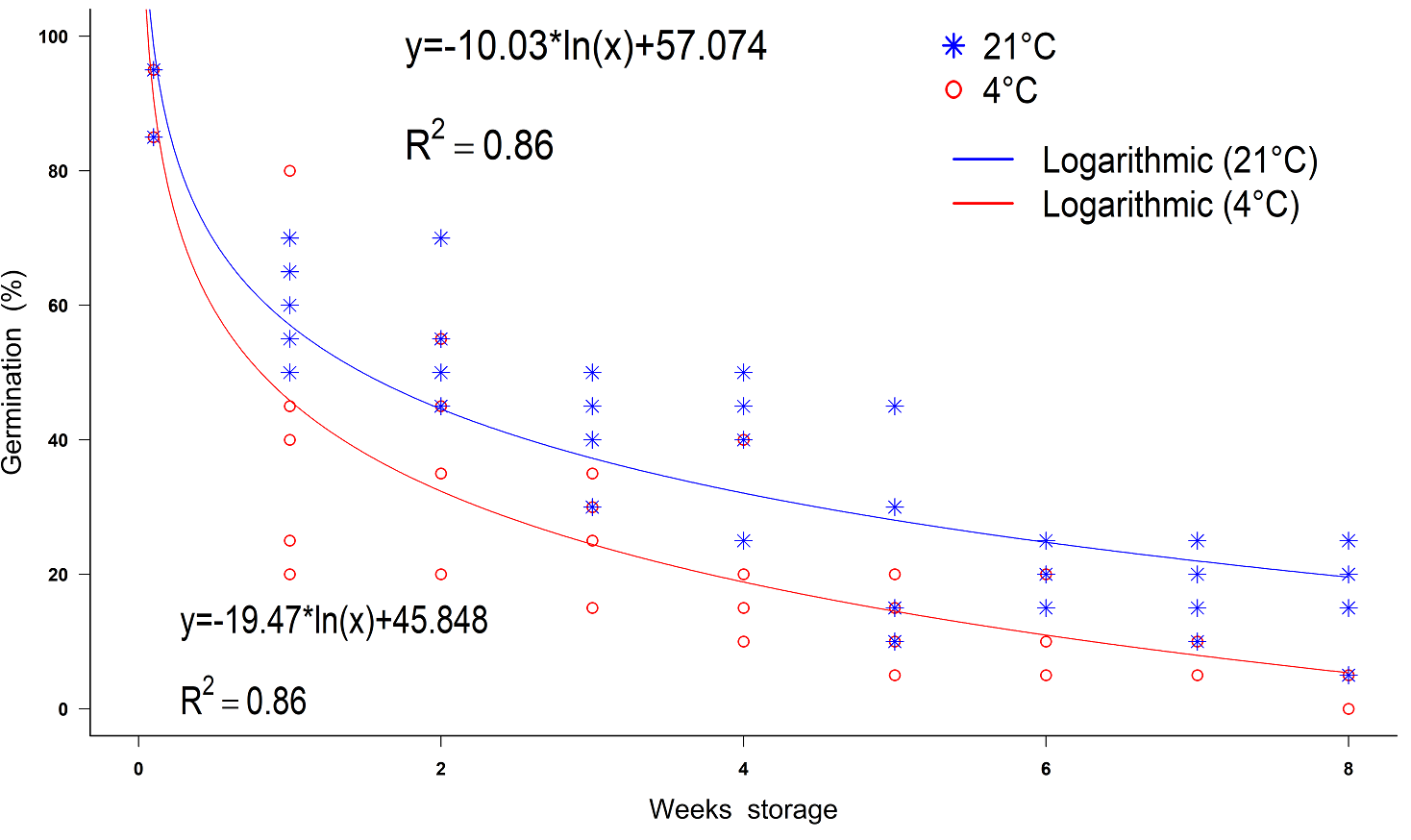
Figure 4 Decrease in germination rates of Populus luziarum seeds under different temperatures storage conditions over time.
Table 2 Effects of temperature and weeks storage on seed germination percentage of Populus luziarum. Different lower-case letters next to the standard error indicate significant differences between means (P < 0.05) according to Tukey test.
| Weeks storage | Temperature (°C) |
Germination (%) |
Weeks storage | Temperature (°C) |
Germination (%) |
|---|---|---|---|---|---|
| 0 | 4 | 93 ± 2.0 a | 5 | 4 | 26 ± 6.2 cdef |
| 21 | 91 ± 2.4 a | 21 | 14 ± 2.9 efgh | ||
| 1 | 4 | 60 ± 3.5 b | 6 | 4 | 21 ± 1.8 cdefg |
| 21 | 42 ± 10.5 bc | 21 | 11 ± 2.4 fgh | ||
| 2 | 4 | 54 ± 4.3 b | 7 | 4 | 17 ± 2.5 defg |
| 21 | 35 ± 6.8 bcde | 21 | 7 ± 1.2 gh | ||
| 3 | 4 | 42 ± 3.3 bc | 8 | 4 | 14 ± 4.0 efgh |
| 21 | 27 ± 3.3 cdef | 21 | 4 ± 1.0 h | ||
| 4 | 4 | 37 ± 5.1 bcd | |||
| 21 | 20 ± 5.2 cdefg |
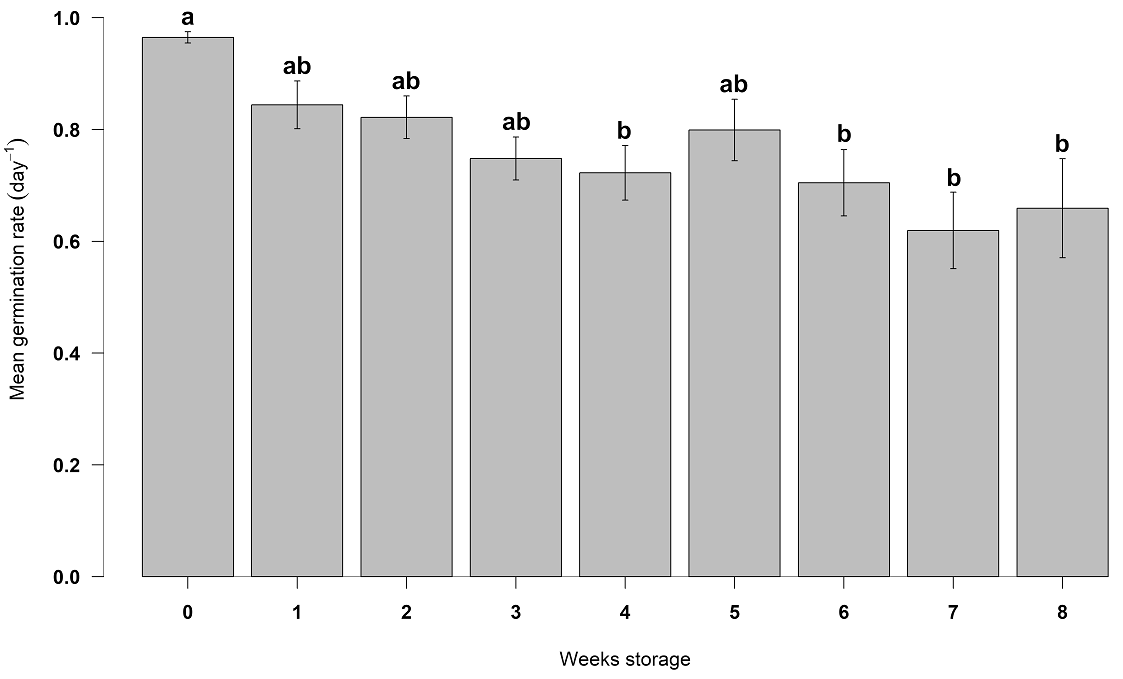
Figure 5 Mean germination rate (day -1) for Populus luziarum among different weeks storage. Means and ± SE, different lower-case letters denote significant differences, according to Tukey test (P < 0. 05).
The trends in germination percentages for the different storage times for the seeds of Populus primaveralepensis, with the treatments at 4 and 21 °C, had the best adjustment R 2 = 0.87 and R 2 = 0.70, respectively, to a logarithmic regression, in which a decrease in germination percentage was observed as storage time increases (Figure 6). Based on the equation obtained, if the seeds are stored at room temperature the germination will be reduce to 54 % in one week, and 94 % in eight weeks, while the refrigerated seeds reduce their germination until 55 % in two weeks. ANOVA of germinability revealed statistically significant differences between storage temperature treatments (F = 328.35, P < 0.001), storage times (F = 105.82, P < 0.001) and the interaction of factors (F = 13.13, P < 0. 001). The highest germination percentages were obtained in week 0 (95 ± 0.31 and 94.8 ± 0.37 %) at 4 and 21 °C respectively, and it decreased as the time of storage increased. From the fourth week on, there were significant differences in germination percentages among the storage treatments carried out per week. In the eighth week, the germination percentages were 4 ± 1.8 % for the storage temperature of 21 °C, while it was 41 ± 2.4 % for that of 4 °C (Table 3). On the other hand, the mean germination rate did not differ significantly among the storage temperature levels (F = 3.043, P > 0.001), but there were significant differences between weeks storage (F = 4.382, P = < 0.001). Mean germination rate was generally fast, values between 0.9 ± 0.01 day -1 and 0.6 ± 0.06 day -1 were obtained; however, there was no tendency for the mean germination rate to decrease with storage time (Figure 7).
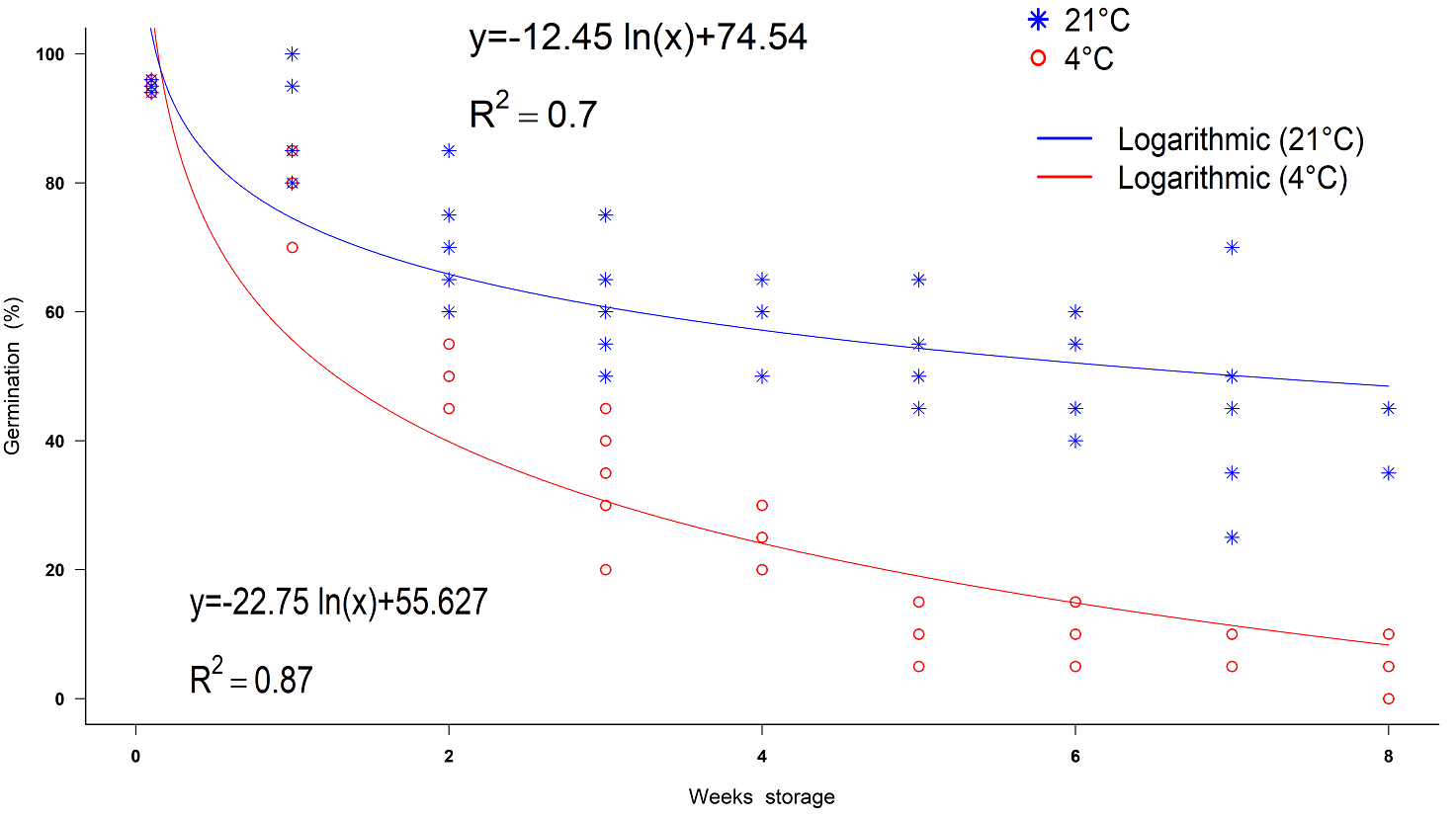
Figure 6 Decrease in germination rates of Populus primaveralepensis seeds under different temperature storage conditions over time.
Table 3 Effects of temperature and weeks storage on seed germination percentage of Populus primaveralepensis. Different lower-case letters next to the standard error indicate significant differences between means (P < 0.05) according to Tukey test.
| Weeks storage | Temperature (°C) |
Germination (%) |
Weeks storage | Temperature (°C) |
Germination (%) |
|---|---|---|---|---|---|
| 0 | 4 | 94.8 ± 0.4 a | 5 | 4 | 54 ± 3.3 def |
| 21 | 95 ± 0.3 a | 21 | 10 ± 1.6 h | ||
| 1 | 4 | 89 ± 3.7 ab | 6 | 4 | 52 ± 4.1 def |
| 21 | 77 ± 2.9 bc | 21 | 9 ± 1.9 h | ||
| 2 | 4 | 71 ± 4.3 cd | 7 | 4 | 45 ± 7.6 ef |
| 21 | 51 ± 1.9 def | 21 | 6 ± 1.0 h | ||
| 3 | 4 | 61 ± 4.3 cde | 8 | 4 | 41 ± 2.4 efg |
| 21 | 34 ± 4.3 fg | 21 | 4 ± 1.9 h | ||
| 4 | 4 | 59 ± 2.4 cde | |||
| 21 | 26 ± 1.9 g |
Discussion
The hypothesis of this work is accepted, since, that seeds dispersal for Populus luziarum and P. primaveralepensis occurs in autumn, however, seed gemination behavior is similar to that of species dispersed in spring or early summer, since they have characteristics such as high germination percentages, germinate faster and loss viability when stored at ambient temperature.
The results of this study showed a high percentage of germination during the first 24 h in freshly collected seeds. Very fast germination may be crucial because ants collect seeds but not seedlings, thus rapid transition from seeds to seedlings allows escape from ant predation (Parsons 2012). Very fast germination also indicates that seeds of both species are non-dormant, which coincides with findings by Karrenberg et al. (2002) for several Populus species. The lack of dormancy, as well as the fluctuations in temperature and humidity in the seeds, makes them have short longevity and a faster decrease of viability (Siegel & Brock 1990). Zasada & Viereck (1975) and Karrenberg et al. (2002) reported that late seed dispersing species of Salicaceae maintain viability at low temperatures (e.g. Salix glauca for eight to nine months), and even require stratification for germination after winter. In contrast, Populus luziarum and P. primaveralepensis reduced germination percentages at 4 °C after four weeks. Both species recorded high germinability (91 and 95 %) respectively without stratification, suggesting a distinct pattern of germination behavior for subtropical Populus species.
In this study, we found that temperature at 4 °C could maintain seed longevity and thus prolong the percentage of germination in both species until to four weeks with percentages greater than 50 %. Similar results have been obtained by Kim (2018) for Populus davidiana and P. koreana, species with early seed dispersal, registered germination percentages of 97 and 87 %, respectively, for non-stocked seeds, these percentages were significantly reduced in the four weeks of storage at room temperature of 25 °C. However, storage at low temperatures prolonged viability and maintained initial germination percentage for both species. Our results also coincide with Moss (1938), with high initial germination of almost 100 % for P. tremuloides, P. balsamifera, and P. petrowskyana, early seed dispersing temperate and boreal species, but in storage conditions at 21 °C, there was a gradual decrease to 45 % at eight weeks and an absence of germinating seeds at 15 weeks for all species. In addition, in storage condition at -5 °C, species showed an increase of longevity, even after two years some seeds obtained 70 % germination. But our results contrast with those obtained by González et al. (2010) for Populus alba, with 92 % of germination in recently collected seeds that remained viable for two weeks (> 80 %), at average room temperature of 22 °C, with seed longevity relatively long with average viability period of 30 days.
The mean germination rate for both species Populus luziarum and P. primaveralepensis (Figures 5 and 7) registered significant differences among storage times, but the differences are not large, this behavior coincides with the species of Populus without endosperm (Karrenberg et al. 2002, González et al. 2010). This also coincides with reports by Vandelook et al. (2012) and Chilpa-Galván et al. (2018) that endosperm reduction may have evolved as an adaptation to favorable conditions for rapid germination, thus allowing growth and establishment in a short time in favorable environments. A decline in germination percentage observed for Populus luziarum as storage time did increase could partly explain why its population size is smaller than that of P. primaveralepensis, however, this seems to be compensated with the clonal reproduction strategy through rhizomes, while in P. primaveralepensis, being more efficient in its germination, without clones (Vázquez-García et al. 2019).
According to Maroder et al. (2000) and Kim (2018), the correlation observed between low-temperature storage and germination in this study could be explained because low temperature maintains the moisture content in the seeds, which is the main factor controlling all cellular activities. Bonner (2008) and Pritchard & Nadarajan (2008) mentions that seeds can be kept viable for several years, when stored at sub-zero temperatures and in a dry atmosphere. For future research, we suggest to include treatments at temperatures below 0 °C and cryopreservation for more promising ex situ seed conservation of these species (Suszka et al. 2014), as it has been shown that these techniques could obtain favorable results in longevity and maintain viability for longer periods (Pawłowski et al. 2019).
The high percentages of physical integrity of seeds through the analysis of X-ray images, and seed germination for both Populus species, are consistent with those reported by Javorski & Moure (2017) and Javorski et al. (2018) who indicate a significant relationship between physical integrity and germination percentages. Gomes-Junior et al. (2013) and De Medeiros et al. (2018) obtained similar results for a wide variety of species in different genera. According to the results of the present research, X-ray image analysis for Populus luziarum and P. primaveralepensis seeds can be a competent, non-destructive alternative that allows a realistic assessment of physical integrity (Gomes-Junior et al. 2012). This coincides with the reported by Abud et al. (2018) and De Medeiros et al. (2020), about the positive and significant correlations, among morphology, internal physiological quality of the seeds, viability and germination percentage.
The results of this study provide valuable information, revealing that decrease of germination capacity is related to the storage conditions of the seeds. In general, our data contribute to the understanding of the processes associated with ex situ seed conservation and the strategies to increase the possibility of maintaining viable seeds for subtropical Populus species.











 text new page (beta)
text new page (beta)