Introduction
The Colección Nacional de Helmintos (CNHE) housed in the Instituto de Biología, Universidad Nacional Autónoma de México in México City, is the most important reference collection on helminth diversity in Mexico (Lamothe-Argumedo et al., 2010). The collection is composed by 10,000 specimen lots, 95% of them identified to generic or specific level, whereas the remaining specimens are completely processed, but not yet identified. As a part of an ongoing project in order to update the information harbored in the CNHE, we studied several trematodes collected in marine fishes between 1950 and 1980 in localities along the Pacific coast of Mexico. Among these specimens, we detected an undescribed species of the genus Pseudopecoelus von Wicklen, 1946. Thus, the main objective of this work is to describe and compare the new species with other congenerics including those previously recorded in Mexican fishes.
Materials and methods
Unidentified trematodes of fishes deposited in the CNHE were obtained for their taxonomic study. All specimens had been previously processed (stained with Meyer’s paracarmin and Gomori´s trichromic and mounted permanently in Canada balsam). Generic identification of trematodes follows Bray et al. (2008), Gibson et al. (2002), and Jones et al. (2005). Drawings of the holotype were made with the aid of a drawing tube attached to a microscope. Measurements were obtained with a calibrated ocular adapted to an optical microscope; all of them are given in millimeters (mm). We present length range, followed by mean and standard deviation in parentheses. Type and voucher specimens were deposited at CNHE; accession numbers are indicated in Table 1.
Table 1 Intestinal species of trematodes recorded in this study in marine fishes from the Pacific coast of Mexico.
| Trematode species | Host species | Site of collection | CNHE |
|---|---|---|---|
| Diplangus mexicanus Bravo-Hollis & Manter, 1957 | Gerreidae: Gerres cinereus | Mazatlán, Sinaloa | 9650 |
| (23°14’3” N, 106°27’40” W) | |||
| Genolinea laticauda Manter, 1925 | Kyphosidae: Girella nigricans | Ensenada, Baja California | 9652 |
| (31°51’14” N, 116°37’45” W) | |||
| Hypocreadium myohelicatum Bravo-Hollis & Manter, 1957 | Balistidae: Sufflamen verres | Mazatlán, Sinaloa | 9655 |
| (23°14’3” N, 106°27’40” W) | |||
| Lecithochirium microstomum Chandler, 1935 | Scombridae: Katsuwonus pelamis | Puerto Ángel, Oaxaca | 9656 |
| (15°39’35” N, 96°29’45” W) | |||
| Opecoelina scorpaenae Manter, 1934 | Sciaenidae gen. sp. | Puerto Ángel, Oaxaca | 9653 |
| (15°39’35” N, 96°29’45” W) | |||
| Opisthadena dimidia Linton, 1910 | Kyphosidae: G. nigricans | Ensenada, Baja California | 9651 |
| (31°51’14” N, 116°37’45” W) | |||
| Pachycreadium gastrocotylum (Manter, 1940) Manter, 1954 | Sparidae: Calamus brachysomus | Mazatlán, Sinaloa | 9657 |
| (23°14’3” N, 106°27’40” W) | |||
| Pseudopecoelus ibunami n. sp. | Serranidae: Epinephelus analogus | La Paz Baja California Sur | 9658-9659 |
| (24°14’30” N, 110°28’08” W) |
Results
A total of 8 species of trematodes were identified, including a new species of Pseudopecoelus. They were collected in the intestine of 7 species of fishes distributed in 4 localities along the Pacific coast of Mexico. The species registered belong to 4 families: Diplangidae: Diplangus mexicanus Bravo-Hollis & Manter, 1957; Haemiuridae: Genolinea laticauda Manter, 1925, Opisthadena dimidia Linton, 1910 and Lecithochirium microstomum Chandler, 1935; Lepocreadiidae: Hypocreadium myohelicatum Bravo-Hollis & Manter, 1957 and finally, Opecoelidae: Opecoelina scorpaenae Manter, 1934, Pachycreadium gastrocotylum (Manter, 1940) Manter, 1954, and one undescribed species, whose description is presented here (Table 1).
Description
Pseudopecoelus ibunami n. sp. (Fig. 1)
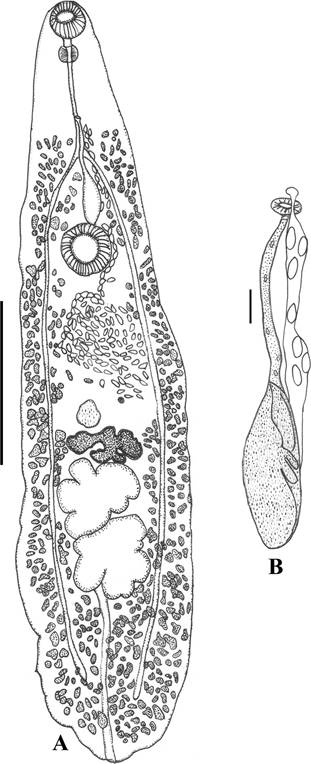
Figure 1 Psedopecoelus ibunami n. sp. A, Ventral view of the holotype (scale 1 mm); B, detail of terminal part of reproductive apparatus in a paratype (scale 0.25 mm).
Description (based in 5 specimens): Opecoelidae, Opecoelinae. Body elongate, narrow, with irregular end, 2.68-4.67 (3.61 ± 0.80) long × 0.5-1.2 (0.96 ± 0.26) wide. Forebody 0.84-1.37 (1.05 ± 0.23) long and hindbody 1.54-2.98 (2.27 ± 0.56) long. Forebody/hindbody ratio 1:2.16. Pre-oral lobe not seen. Tegument smooth. Oral sucker subterminal, 0.17-0.23 (0.20 ± 0.02) long × 0.16-0.25 (0.21 ± 0.03) wide. Pre-pharynx short, pharynx globular, 0.08- 0.11 (0.10 ± 0.01) long × 0.07-0.12 (0.10 ± 0.02) wide. Esophagus 0.30-0.53 (0.39 ± 0.10, n = 4) long × 0.01-0.02 (0.02 ± 0.01, n = 4) wide. Ventral sucker larger than oral sucker, 0.21-0.34 (0.29 ± 0.05) long × 0.17-0.34 (0.28 ± 0.07) wide. Sucker ratio 1:1.37. Intestinal bifurcation between pharynx and ventral sucker, at 0.23-0.47 (0.33 ± 0.10) from ventral sucker. Caeca long, narrow, reach close to posterior end of body, and end blindly. Testes 2, deeply-lobed (anterior and posterior testes with 3-6 lobules), oval with long axes oriented transversely, located contiguously in tandem, in the second third of hindbody. Anterior testis 0.25-0.56 (0.40 ± 0.13) long × 0.19-0.54 (0.37 ± 0.13) wide, at 0.52-1.11 (0.78 ± 0.24) from ventral sucker; posterior testis 0.24-0.55 (0.41 ± 0.14) long × 0.24-0.48 (0.36 ± 0.09) wide. Post-testicular area 0.57-1.01 (0.82 ± 0.19) long, approximately 25% of body length. Genital atrium not distinguished. Genital pore slightly sinistral, near to mid-body line, approximately halfway between pharynx and intestinal bifurcation. External seminal vesicle uncoiled, widened proximally, 0.17-0.34 (0.27 ± 0.87, n = 4) long × 0.06-0.13 (0.09 ± 0.03, n = 4) wide; does not invade hindbody, reaching the anterior margin of ventral sucker. Pars prostatica poorly developed, with few gland-cells. Cirrus-sac absent. Ovary deeply-lobed, with 4-5 lobes, pre-testicular, median, contiguous with anterior testis, 0.18-0.32-(0.26 ± 0.05) long × 0.24-0.48 (0.37 ± 0.10) wide, at mid-hindbody 0.34-0.91-(0.61 ± 0.23) from ventral sucker. Uterine seminal receptacle round, antero-lateral to ovary; Mehlis’gland and Laurer’s canal not seen. Uterine coils mostly pre-ovarian, in intercaecal space between ovary and ventral sucker, passes laterally to ventral sucker. Metraterm not distinguished. Eggs 0.04-0.05 (0.05 ± 0.004) long × 0.017-0.02 (0.02 ± 0.003) wide. Vitelline follicles extend from intestinal bifurcation to posterior end, some follicles overlapping caeca dorsally; vitelline fields mainly lateral to caeca, confluent in post-testicular region. Excretory pore terminal. Excretory vesicle I-shaped, partially hidden by the testes and ovary.
Taxonomic summary
Type host: Epinephelus analogus Gill, 1863
Type locality: La Paz, Baja California Sur (24°14’30” N, 110°28’08” W), Mexico.
Site of infection: intestine.
Material deposited: CNHE 9658 (holotype); 9659 (4 paratypes).
Etymology: this species is named after the Instituto de Biología of the Universidad Nacional Autónoma de México (IB-UNAM) where the Colección Nacional de Helmintos is housed.
Remarks
Pseudopecoelus was erected by von Wicklen (1946) to contain Pseudopecoelus vulgaris (Manter, 1934) von Wicklen, 1946; currently, this genus is composed by 39 nominal species distributed worldwide (Blend et al., 2017), parasitizing a wide range of marine fishes of many families (Cribb, 2005). Specific differentiation within the genus is difficult; for practical purposes, Bray (1987) divided it artificially into 6 morphological groups (A-F). Based on the extent of the vitelline follicles, which are distributed anteriorly to ventral sucker, our specimens are included in group A, along with 17 species (Table 2). Pseudopecoelus ibunami n sp. can be differentiated of all the species included in group A by having a combination of the following traits: 1) body elongate, narrow, with irregular posterior end, 2) testes and ovary deeply-lobed, and 3) external seminal vesicle reaching only the anterior margin of ventral sucker. Particularly, the extension of the external seminal vesicle of the new species allows to distinguish it from 13 of the 17 species included in the group A: in Pseudopecoelus ariusi Parukhin, 1983, Pseudopecoelus brayi Madhavi & Lakshmi, 2010, Pseudopecoelus dollfusi Ahmad & Dhar, 1987, Pseudopecoelus umbrinae Manter & van Cleave, 1951, and Pseudopecoelus vitellozonatus Pritchard, 1966 the external seminal vesicle reaches the posterior end of ventral sucker (Ahmad & Dhar, 1987; Madhavi & Lakshmi, 2010; Manter & van Cleave, 1951; Parukhin, 1983; Pritchard, 1966), while in Pseudopecoelus bilqeesae Ahmad & Dhar, 1987, Pseudopecoelus gibbonsia Manter & van Cleave, 1951, Pseudopecoelus gymnothoracis Nahhas & Cable, 1964, Pseudopecoelus manteri Sogandares-Bernal & Hutton, 1959, Pseudopecoelus minutus Nahhas & Cable, 1964, Pseudopecoelus pritchardae Gupta & Sayal, 1979, Pseudopecoelus pyriformis Prudhoe & Bray, 1973, and Pseudopecoelus sewelli Bray, 1990 overpass this structure (Ahmad & Dhar, 1987; Bray, 1990; Gupta & Sayal, 1979; Manter & van Cleave, 1951; Nahhas & Cable, 1964; Prudhoe & Bray, 1973; Sogandares-Bernal & Hutton, 1959). In addition, the body of P. pyriformis and P. umbrinae is pyriform (Manter & van Cleave, 1951; Prudhoe & Bray, 1973) (vs. elongate in the new species) and P. bilqeesae, P. dollfusi, P. manteri, and P. sewelli have pedunculated ventral sucker (Ahmad & Dhar, 1987; Bray, 1990; Sogandares-Bernal & Hutton, 1959) in contrast with the sessile ventral sucker of P. ibunami. Testes of P. gibbonsia and P. minutus are smooth (Manter & van Cleave, 1951; Nahhas & Cable, 1964) and medially constricted in P. gymonthoracis (Nahhas & Cable, 1964) rather than deeply-lobed as in our specimens. Genital pore is sub-median at level of intestinal bifurcation in P. ariusi and P. pritchardae (Gupta & Sayal, 1979; Parukhin, 1983) and is located to left of pharynx or at anterior end of esophagus in P. vitellozonatus (Pritchard, 1966), whereas in P. ibunami, the genital pore is situated at mid-body line, approximately halfway between pharynx and intestinal bifurcation. Finally, P. brayi also differs of P. ibunami by having a sinuous external seminal vesicle (instead of a straight vesicle, widened proximally as in the new species) and by having ovary and testes separated by vitelline follicles and not contiguous as in P. ibunami (Madhavi & Lakshmi, 2010).
Table 2 Species of Pseudopecoelus included in the group A according to Bray (1987).
| Pseudopecoelus species | Host family/distribution | Reference |
|---|---|---|
| P. ibunami n. sp. | Serranidae/Mexico | Present study |
| P. acanthuri Yamaguti, 1970 | Acanthuridae /Hawaii | Yamaguti (1970) |
| P. ariusi Parukhin, 1983 | Ariidae/Oman | Parukhin (1983) |
| P. bilqeesae Ahmad & Dhar, 1987 | Carangidae/India | Ahmad & Dhar (1987) |
| P. brayi Madhavi & Lakshmi, 2010 | Holocentridae/India | Madhavi & Lakshimi (2010) |
| P. brevivesiculatum Hanson, 1955 | Monacanthidae/Hawaii | Hanson (1955) |
| P. dollfusi Ahmad & Dhar, 1987 | Therapontidae/India | Ahmad & Dhar (1987) |
| P. gibbonsia Manter & van Cleave, 1951 | Clinidae/California | Manter & Van Cleave (1951) |
| P. gymnothoracis Nahhas & Cable, 1964 | Muraenidae/Curaçao | Nahhas & Cable (1964) |
| P. littoralis Caballero & Caballero-Rodríguez, 1976 | Sciaenidae/California | Caballero & Caballero-Rodríguez (1976) |
| P. manteri Sogandares-Bernal & Hutton, 1959 | Sciaenidae/Florida | Sogandares-Bernal & Hutton (1959) |
| P. minutus Nahhas & Cable, 1964 | Carangidae/Curaçao | Nahhas & Cable (1964) |
| P. pritchardae Gupta & Sayal, 1979 | Holocentridae/India | Gupta & Sayal (1979) |
| P. puhipaka Yamaguti, 1970 | Muraenidae/Hawaii | Yamaguti (1970) |
| P. pyriformis Prudhoe & Bray, 1973 | Callanthiidae/Tasmania | Prudhoe & Bray (1973) |
| P. sewelli Bray, 1990 | Trachichthyidae/Tasmania | Bray (1990) |
| P. umbrinae Manter & van Cleave, 1951 | Sciaenidae/California | Manter & Van Cleave (1951) |
| P. vitellozonatus Pritchard, 1966 | Acanthuridae/Hawaii | Pritchard (1966) |
The remaining 4 species of Pseudopecoelus included in the group A of Bray (1987), have the external seminal vesicle reaching only the anterior margin of ventral sucker as the new species described herein. Nonetheless, body of Pseudopecoelus littoralis Caballero & Caballero-Rodríguez, 1976, Pseudopecoelus acanthuriYamaguti, 1970 and Pseudopecoelus puhipaka Yamaguti, 1970 is pyriform rather than elongated as in P. ibunami and testes in P. littoralis, P. acanthuri and P. puhipaka are trapezoidal, indented and irregularly lobed, respectively (Caballero & Caballero-Rodríguez, 1976; Yamaguti, 1970), in contrast with the testes deeply-lobed of the new species. Pseudopecoelus ibunami most closely resembles Pseudopecoelus brevivesiculatum Hanson, 1955 in general morphology of body and internal organs arrangement. Notwithstanding, genital pore of P. brevivesiculatum is sinistral, located mid-way between esophagus and body wall and in the new species, genital pore is close to mid-body line, approximately halfway between pharynx and intestinal bifurcation. In addition, external seminal vesicle in the new species is uncoiled and widened proximally, while in the species described by Hanson (1955), external seminal vesicle has a sharp curve in the posterior half.
In addition to P. umbrinae collected in Umbrina xanti Gill, 1862 (Sciaenidae) from the Pacific coast of Mexico, which was compared with the new species above, Pseudopecoelus is represented in this country by 3 other species: Pseudopecoelus elongatus (Yamaguti, 1938) Von Wicklen, 1946 parasitizing Caranx sp. (Carangidae), Pseudopecoelus scorpaenae (Manter, 1947) Overstreet, 1969 in Scorpaena plumieri Bloch, 1789 (Scorpaenidae) and Pseudopecoelus priacanthi (MacCallum, 1921) Manter, 1947 recorded in Caranx caballus Günther, 1868 and Trachinotus rhodopus Gill, 1863 (Carangidae) (Pérez-Ponce de León, García-Prieto, & Mendoza-Garfias, 2007). However, P. elongatus and P. scorpenae are included in the group F of Bray (1987), characterized by having smooth testicular margins (rather than deeply-lobed as in P. ibunami) and external seminal vesicle extending posteriorly to ventral sucker (instead of only reaching the anterior margin of the ventral sucker, as in the new species); P. priacanthi, included in the group E by Bray (1987), also have smooth testicular margins and a large ventral sucker pedunculated and with lateral notches, characteristics not presented by our material.
Previous to the present study, the only species of Pseudopecoelus recorded in serranid fishes of the genus Epinephelus Bloch, 1793 was Pseudopecoelus epinepheli Wang, 1982, parasitizing Epinephelus akaara (Temminck & Schlegel, 1842) from Fujian, China (Wang, 1982). The slightly sinistral position of the genital pore of this species, included in the group B of Bray (1987), is similar to that observed in the new species described herein; however, in our material the genital pore is situated approximately halfway between pharynx and intestinal bifurcation, whereas in P. epinepheli it is located just before the division of the intestine. In addition, P. epinepheli can be distinguished from P. ibunami because vitelline field does not reach into the forebody and the external seminal vesicle surpasses extensively the posterior end of the ventral sucker (Wang, 1982).
Discussion
Studies on trematodes associated with marine fish from Mexico began almost 70 years ago; the first species of trematode described from marine fishes from this country was Homalometron mexicanum (Manter, 1937) Cribb & Bray, 1999 (= Apocreadium mexicanumManter, 1937) as a parasite of Labrisomus xanti Gill, 1860 from Oaxaca State, on the Mexican Pacific coast (Manter, 1937). Since a great amount of information has been accumulated according to García-Prieto, Mendoza-Garfias, and Pérez-Ponce de León (2014), these platyhelminthes are the most well-known group of helminths in this country. Only for Actynopterigians, these authors listed 305 nominal species ± 95 unnamed taxa collected in 395 fish species of marine, brackish and freshwater environments. The majority (205) of these 305 nominal species have been recorded parasitizing marine fishes (data not published). With the present study we add 2 species to the inventory of the trematodes associated with marine fishes distributed in Mexican waters: P. ibunami n. sp., and P. gastrocotylum. For 3 of the remaining 6 registered species of trematodes (H. myohelicatum, O. scorpaenae, and L. microstomum) their geographical distribution range is extended. Finally, fish species parasitized by Diplangus mexicanus, Opisthadena dimidia, and Genolinea laticauda in this study represent new host records for those species in Mexico (see Pérez-Ponce de León et al., 2007).











 text new page (beta)
text new page (beta)


