Services on Demand
Journal
Article
Indicators
Related links
Share
Revista odontológica mexicana
Print version ISSN 1870-199X
Rev. Odont. Mex vol.16 n.3 Ciudad de México Jul./Sep. 2012
Original research
In vitro macroscopic analysis of dental tissues and some dental materials used in endodontics, submitted to high temperatures for forensic applications
Lucero Vázquez,* Patricia Rodríguez,§ Freddy MorenoII
* University of Nayarit, (Mexico) Scholarship, Delfin Program, Mexico
§ Endodontics specialist, Teacher , School of Dentistry Universidad del Valle, (Colombia).
II Teacher, School of Dentistry, Universidad del Valle, Colombia. Researcher, Maxillofacial and Oral Surgery Group, Universidad del Valle.
ABSTRACT
Objective: The present paper aims at describing macro-structural physical changes taking place in materials and tissues employed in endodontic treatments after their exposition to high temperatures. Material and methods: An in vitro experimental study was conducted to observe macroscopic physical changes in dental tissues (enamel, dentine and cement) as well as materials conventionally used in endodontic practice: gutta-percha, Maillefer Dentsply®, Eufar® cement with zinc oxide eugenol, Top Seal® Dentsply® endodontic cement with epoxy resin, Fuji® GC America® glass ionomer, GS80® SDI® silver amalgam, and Point 4® Kerr® resin, in 124 human teeth exposed to five temperature ranks: 200°C, 400°C, 600°C, 800°C and 1,000°C. Results: Tissues and dental materials studied in the present research offered great resistance to high temperatures, without exhibiting considerable macrostructure variation, in such a way that physical changes (dimensional stability, fissures, cracks, fractures, texture, color, carbonization and incineration) can be identified and associated to each specific temperature rank. Conclusions: Dental tissues and materials offer great resistance to the effect of high temperatures. Moreover, they present specific changes (color, texture, fissures, cracks fractures, fragmentation) which might contribute to the process of identifying a corpse, or burned, incinerated or carbonized human remains.
Key words: Forensic dentistry, human identification, dental tissues, dental materials, endodontic use, high temperatures.
INTRODUCTION
In Latin America, state-of-the art forensic dentistry is progressing, as well as other forensic science disciplines, in conjunction with political and social processes taking place in every country. Forced disappearances, massacres, terrorist attacks and everyday violence, have fostered research in forensic sciences, so as to achieve satisfactory identification of victims, even when offenders follow procedures aimed at avoiding identification of a corpse or humans remains. To that effect they employ individual or common graves, obliterate fingerprints, destroy teeth and cremate the remains. These vicious procedures become obstacles hindering all identification procedures. For the aforementioned reasons, the dental scientist, as an auxiliary of justice, and acting as expert according to Colombian legislation, and with full knowledge of human rights situation in Latin America, to gain knowledge on offenders behavior and their techniques for disappearing systems and destruction, must contribute with his knowledge when requested to do so by government agencies, prosecution, attorneys, advocacies, presidential counseling offices, judges of the Colombian Republic, non-government organizations (NGO s) as well as international agencies such as INTERPOL, International Amnesty Freedom and Responsibility Committee (UN) which might require the use of their knowledge.
Based on all the aforementioned factors, the Maxillofacial and Oral Surgery Research Group of the Del Valle University in Colombia, considers forensic dentistry as a special branch of dentistry. This science, through clinical, technical and scientific inter-disciplinary work, along with other forensic sciences, fosters the upright administration of justice. This can be achieved through evidence provided by teeth, anatomical structures, conditions, and dental treatments. All these elements must be assessed by facial, maxillary and oral health providers who must show professional responsibility during observation and stomatognathic system analysis when faced with the task of identifying living or dead human beings, in cases of special rulings, great disasters, exhumations, bite marks, personal injuries, child abuse, sexual abuse and professional responsibility.
All this is possible because teeth, as part of the stomatognathic system structures, even after the death of the subject, are the best kept structures of the human body.1,2 This can be due to the fact that enamel is the hardest tissue found in the human body and is preserved in extreme pH , salinity, humidity and high temperature situations.3,7 It must be borne in mind that teeth, articulated and physiologically in place in their corresponding sockets, in both upper and lower jaws, are protected by spongy and cortical bone tissue as well as by mucosal, epithelial and muscular soft tissues. These tissues isolate teeth for some time in cases when they are exposed to high temperatures.4
Historically, all the aforementioned factors have allowed teeth to be the first elements to study when establishing identity of corpses rendered unrecognizable through violent death or bodily destruction. Along these same lines, each country possesses different legislations to rule how to exercise forensic dentistry. AFBO (American Board of Forensic Odontology)8 has proposed basic guidelines to be taken into account when identifying a deceased subject, especially in cases when conventional means of recognition such as visual recognition and print matching cannot be performed as would be the case in burned, carbonized or incinerated bodies.
In general terms, dental identification is, as such, a comparative science. The forensic dentist can obtain sufficient distinctive characteristics to identify a subject based on matching of ante-mortem dental records (medical history, odontogram, periodontogram, x-rays, study models, cephalometric analysis, treatment plans, etc) as well as post-mortem records (information gathered from the body) as well as reconstructive processes recorded, corpses in advanced state of decomposition, bodies that are carbonized, in skeletal reduction, or human remains requiring facial reconstruction. This is also the case of the basic four elements for identification: gender, age, ethnic influence and height. Nevertheless, matching procedures performed by the forensic dentist are not necessarily based upon data obtained from medical history (indubitable evidence), they can also guide the identification process based upon gathering and comparison of biographic data, medical history, clothing items, documents and specific data signs though the use of non scientific methods (dubitable evidence).9,10
When faced with a carbonized or incinerated body, identifying processes are conducted according to the state of human remains. To this end, Norrlander,3 for forensic purposes, conducted a classification of bodily burns into five categories:
1. Superficial burns.
2. Areas of destroyed epidermis.
3. Destruction of epidermis, dermis, and necrotic areas in underlying tissues.
4. Total destruction of skin and deeper tissues.
5. Cremated remains.
Usually, dentists are called upon to assist in the identification processes of burned victims, in cases where there is extensive tissue destruction, and subjects cannot be identified through conventional means, digital recognition or fingerprints, (Norrlander categories 3, 4, and 5). Identification of a burned or incinerated subject through dental procedures is conducted comparing post-mortem records with ante-mortem dental clinical history. This is conducted through information provided by the stomatognathic system. It is evident that identity verification of burned victims is hindered by the post-mortem state of the victim and the amount and quality of ante-mortem data. The most usual matching process is conducted based upon dental treatment such as prosthetic restorations and dental fillings, distinctive osseous and dental morphological traits, distinctive pathological variations, and DNA collection from dental pulp not affected by high temperatures.5,8,11-14
Due to all the aforementioned factors, describing physical macro-structural changes which take place in tissues and dental materials used in conventional endodontic treatment, when subjected to high temperatures, could eventually assist in the assessment of incinerated, carbonized or burned human remains, and thus determine which dental treatments existed , as well as dental materials with which they were conducted. All this will probably, in the future, enable researchers to scientifically base matches performed pre-mortem based upon dental history, and thus establish reliable identification of a subject or his remains. Therefore, results presented in this research contribute to generation of new knowledge in the field of dental identification, within the scope of judicial, clinical technical and scientific fields of forensic dentistry.
MATERIAL AND METHODS
The present paper is an experimental In vitro study of the effect of high temperatures on dental tissues (enamel, dentine and cement) and of some dental materials (gutta-percha Maillefer Dentsply ®, endodontic-use cement made of zinc oxide and eugenol Eufar ® , endodontic-use cement made of epoxy resin Top seal ® Dentsply ® , glass ionomer Fuji ® GC America ® , silver amalgam GS80 ® SD®, and resin Point 4 ® Kerr ®) To this effect, a 124 recently extracted teeth sample was gathered. Teeth had been extracted for orthodontic or periodontal reasons, were free of caries, restorations, endodontic treatments, pulp disease, and congenital malformations.
Variables considered for this study obey macroscopic changes observed in dental tissues and materials used to fill pulp canals as well as access cavity in an endodontic treatment. To facilitate discussion, these changes will be grouped according to dental materials and tissues and to temperature rank. The following factors were considered: 1) filling material maladjustement, 2) fissures, cracks cracked appearance and fractures. 3) texture changes, 4) color changes, 5) incineration and carbonization levels.
SAMPLE GATHERING
Once the research was endorsed by the Ethics Committee for Human Beings of the Del Valle University, in agreement with Article 11 of Resolution 008430 of the Social Protection Ministry15 and with ethical principles for medical research in human beings dictated by the World Medical Association at the Helsinky Declaration,16 and having receive authorization from the Board of the School of Dentistry, and having as well obtained authorized consent from the patients, a sample was obtained. The sample was composed of teeth extracted at the oral surgery clinic of the School of Dentistry, Universidad del Valle. Teeth had to comply with inclusion criteria.
SAMPLE HANDLING AND CONSERVATION
Immediately after extraction, teeth were cleansed with non-sterile water to dispose of blood residues. Teeth were then immersed in a dark container with 5% Chloramine T solution (100 g sodium tosylchloramide diluted in two liters distilled water) for a week. After this period, teeth were placed in saline solution at 37°C temperature, with 100% relative humidity. Every two weeks, the saline solution was renewed, following ICONTEC 4882/200017 and ISO/TS 11405/2003 norms18 until initiation of sample processing.
CAVITY PREPARATION
One single operator placed each and every tooth in a wax base and proceeded into performing a Type I palatine cavity. This was conducted in concordance with access recommendations set out in endodontic literature, which aim at allowing pulp chamber exposition with a NSK ®high speed hand-piece with constant refrigeration, and pear-shaped (Diatech®) medium grain diamond burr. Once the cavity was completed, it received peroxide prophylaxis to disinfect the cavity and reduce dentine superficial tension and thus optimize composite resin adhesive properties.
ENDODONTIC TREATMENT
A conventional endodontic treatment was conducted following telescopic technique19 Once the canal was located, the master file was placed in an apical position. Measurement of canal was taken at 2 mm distance from the apical foramen. Instruments were used in the canal with first series files, one by one in proper order. Working length was decreased for each file in 2 mm with respect to the apical foramen. Irrigation was conducted with sodium hypochlorite. The master file was used apically between every usage of the other files. Once the canal was bio-mechanically prepared. cone measurement was undertaken. For this, a gutta-percha cone was used, of same diameter and working length as the file previously used. Finally, canal filling was undertaken following the condensation technique which employs condensation of gutta-percha cones and filling cement. Once the filling was assessed, the crest of the cone was cut at 2 mm below the line of cement-enamel junction. The preparation was the sealed with glass ionomer.
CAVITY FILLING
Cavity filling was performed in concordance with the group to which the samples belonged, and according to dental material used to fill the cavity. Teeth were then classified into three groups according to dental materials used.
GROUP 1
44 teeth were subject to the aforementioned canal treatment. Canals were filled with Dentsply Maillefer gutta-percha® as well as Eufar® zinc oxide-eugenol cement, to later be sealed with Fuji® GC America® glass ionomer. The cavity was filled with GS80® SDI® silver amalgam following the conventional technique which consists on packing, condensing, and polishing the restoration.
GROUP 2
44 teeth were selected, subject to the aforementioned canal treatment. Canals were filled with Dentsply Maillefer® gutta-percha as well as Eufar® zinc oxide-eugenol cement, to later be sealed with Fuji® GC America® glass ionomener. The cavity was filled with Point 4® Kerr® resin following the technique of enamel acid-etch for 15 seconds, and 10 seconds dentin conditioning with 37% phosphoric acid (GelEtchan® Kerr®). Adhesive was applied (OptiBond Solo Plus® Kerr®,) with micro-brush in two layers, allowing the first to impregnate 20 seconds with indirect air jet for five seconds, so that the adhesive could penetrate into the dentin tubules, and a second layer to homogenize the surface avoiding dry zones. The adhesive agent was polymerized for 40 seconds with a light-polymerizing lamp (Spectrum 800® Denstply®). Finally composite resin (Point 4® Kerr®) was packed, following the oblique increments technique, where each increment is polymerized for 40 seconds with a light-polymerization lamp (Spectrum 800®, Dentsply®) until the cavity was totally filled. Finally the restoration was polished and shined with Hiluster Plus and Oclubrush® systems so as to remove superficial inhibited layers.
GROUP 3
47 teeth were selected, subject to the aforementioned canal treatment. Canals were filled with Maillefer® Dentsply® gutta-percha and Top Seal® Dentsply resin cement, sealed with Fuji® GC America® glass ionomer, and Point 4® Kerr® resin. The cavity was filled following the same protocol as in Group 2.
In turn, teeth of all these groups were divided according to the temperature rank to which they were exposed (200°C, 400°C, 600°C, 800°C, 1,000°C) Table I.
USE OF HIGH TEMPERATURES
Once the fillings were completed, teeth were placed in individual trays made of refractory coating (Cera-Fina Whipmix®) material to facilitate handling according to the prototype proprietory of the Dental Materials Unit, Stomatological Department, University of Pavia (Italy). They were subject to direct heat in a muffle type furnace (Thermolyne®), previously calibrated to five different temperature ranks (200°C, 400°C, 600°C, 1,000°C) with a 10°C per minute rate of ascent, from an initial 34°C temperature (room temperature) until reaching every targeted rank. Once the rank specific temperature was reached, teeth were removed from the furnace, and they were left to cool until reaching room temperature. Once the teeth were cold, they were sprayed with hairspray so as to grant them a certain degree of resistance and ease their handling.4
Some specimens were soaked in clear acrylic (New Stethic®), and they were trimmed (WhipMix® trimmer) in a sagittal direction, so as to expose endodontic treatment and macroscopically observe changes in tissues and dental materials.
OBSERVATION AND STATISTICAL ANALYSIS
With the naked eye, one single examiner observed and described macro-structural changes in dental and filling materials. With the help of 20x digital photographs (digital camera Sony® Cyber shot® DSC- H50, 8.1 mega pixels, and 35 mm Carl Zeiss Lens) according to the variables of this study. These changes were recorded into electronic templates in Excel® Microsoft Office® software. They were later processed in SPSS® software to conduct frequency analysis.
RESULTS
ENAMEL
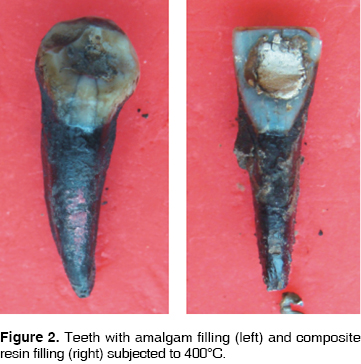
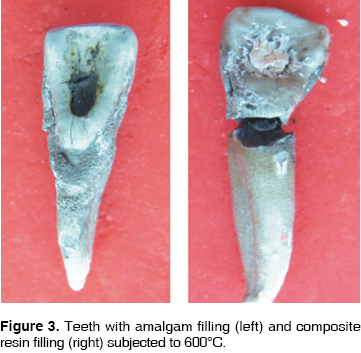
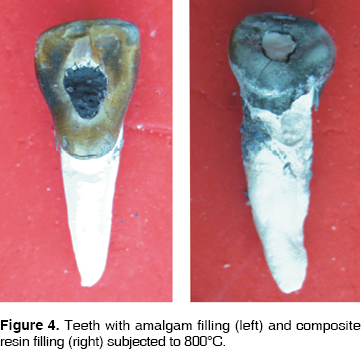
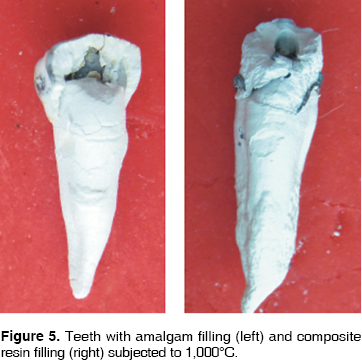
At 200°C, crowns of teeth filled with amalgam and resin turn brownish, with a whitish incisal edge, and the enamel loses its brightness (Figure 1). At 400°C, the crown becomes darker, remaining dental plaque is carbonized, the enamel cracks all over the crown, and bursts in the cervical area (Figure 2). At 600°C the crowns takes an intense brownish hue, associated to underlying dentine carbonization: cracked enamel with longitudinal cracks separates from dentin at the cervical area. In teeth filled with amalgam, in surrounding enamel, a yellowish halo is observed associated to oxides released by the heat (Figure 3). At 800°C, the crown is grayish and the incisal edge acquires a chalk-like white color, associated to incineration. In teeth filled with amalgam, oxides pigment enamel-amalgam inter-phase in brownish and golden hues (Figure 4). At 1,000°C, in most teeth, fragmentation and enamel loss could be observed (Figure 5).

DENTIN
At 200°C, at macroscopic sagittal section, the enamel dentine union inter-phase along all the crown surface, turns to a brownish hue. The crown therefore adopts that color since the enamel is translucent (Figure 1). At 400°C, dentine takes on a blackish hue due to carbonization, a separation between dentin and enamel is observed due to internal fractures (Figure 2). At 600°C, the most external dentine turns to a grayish hue, associated to the beginning of incineration, whereas, dentine located in more internal areas, in contact with endodontic materials, maintains its black color, which proves it is still carbonized (Figure 3). At 800°C root dentine is incinerated and white, while crown dentine, being protected by enamel, still presents black streaks product of carbonization (Figure 4). At 1,000°C, totally incinerated white dentine is observed (Figure 5).
CEMENT
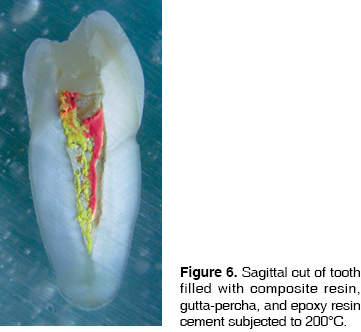
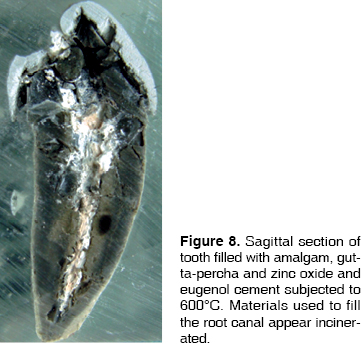
Cement is one of the tissues that macroscopically allows clear identification of the temperature rank to which teeth were subjected. At 200°C no significant changes were observed, although there was loss of brightness (Figure 1). At 400°C, cement turned darkish brown, this was associated to carbonization, from the cervical third to the root it took on a cracked appearance, and showed longitudinal cracks along the root (Figure 2). At 600°C cement, cracked along the root, adopted a light brown hue associated to incineration, with longer and deeper longitudinal cracks (Figure 3). At 800°C cement adopted a chalk-like whitish hue with transverse and longitudinal cracks which extended up to the dentine and could generate root fractures (Figure 4). At 1,000°C cement was white and completely incinerated (Figure 5). At the sagittal section, macroscopically, cement could only be differentiated from dentine at 200°C 400°C and 600°C, in the other two temperature ranks, both tissues were incinerated (Figures 6 - 8)(7).

AMALGAM
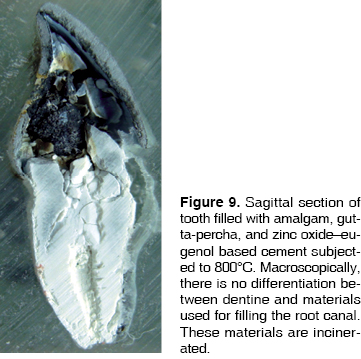
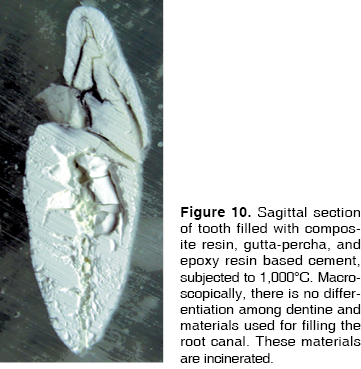
At 200°C amalgam experienced marginal maladjustment, it turned opaque and rough, due to nodule formation on the surface elicited by mercury evaporation (Figure 1). At 400°C, amalgam darkened and there was formation of superficial cracks (Figure 2). At 600°C, amalgam turned black, and man-created morphological characterizations were lost (Figure 3). At 800°C and 1,000°C amalgam continued black and presented rounded nodules called ''silver bullets'' (Figures 4 and 5). At sagittal section, amalgam marginal maladjustment was observed at 400°C, 600°C and 800°C (Figures 8 and 9). Teeth subjected to 1,000°C showed dislodged filling due to crown fragmentation (Figure 10). Amalgam discolorations occured only in the filling surface directly exposed to the furnace.
COMPOSITE RESIN
At 200°C, resin presented marginal retraction and there was no evidence of significant macroscopic changes (Figure 1). At 400°C, resins turned to a light brown hue, and presented marginal maladaptation as well as superficial cracks and fissures (Figure 2). At 600°C, resin, which then was of a white hue, was totally maladjusted, and in some teeth it had been dislodged from the cavity (Figure 3). At 800°C resin adopted a chalk-like white hue and at 1,000°C it was totally dislodged from the cavity due to the fragmentation of the crown (Figures 4 and 5). In sagittal sections, resin appeared with fissures and separated from dental tissues and from the glass ionomer used a coronary seal of the endodontic treatment (Figures 6, 7, 10).
GLASS IONOMER
This material could only be observed in fractured teeth or in sagittal sections. At 800°C, in teeth filled with amalgam, traces of material can be observed. They present a light brown hue, adhere to the filling, and in the crown section of the endodontic treatment adhered to the gutta-percha and dentinal walls (Figure 9). At 1,000°C since there is crown fracture and ionomer exposition to the furnace, the incinerated material can be seen. In sagittal sections, the ionomer can be seen as a thin layer, in concordance with its clinical function, therefore, macroscopically, it is very difficult to appreciate significant changes. At 200°C and 400°C it is observed intact, but separated from filling material and gutta-percha (Figures 6 and 7). At the other temperature ranks it is difficult to observe the material due to incineration and carbonization levels as well as to crown fragmentation and dislodgement of filling.
ENDODONTIC CEMENT BASED ON ZINC OXIDE-EUGENOL
In sagittal sections, at 200°C, material found between gutta-percha cones appears unchanged. From 400°C onwards, both cement and gutta-percha, of a whitish hue, incinerate, and therefore are impossible to differentiate macroscopically (Figure 8). At 800°C and 1,000°C they acquire a chalk-like whitish hue and are undifferentiated from incinerated dentin (Figure 9).
ENDODONTIC CEMENT MADE OF EPOXY RESIN
This material behaves in a similar manner to zinc oxide-eugenol cement, that is to say, at sagittal section, at 200°C, it can be observed among gutta-percha cones found within the canal (Figure 6), at 600°C, some traces of gutta-percha can be discerned, and finally, from 800°C onwards, the material experiences incineration and is macroscopically unrecognizable.
GUTTA-PERCHA CONES
This material could be observed in fractured teeth and in sagittal sections. Nevertheless, in all three teeth groups, a very interesting feature could be observed: at 200°C, heat-melted gutta-percha filtered through the apical foramen and preserved its original color. At 400°C, the same event took place, but, due to the higher temperature, the black colored material carbonized. At 600°C in some teeth, white gutta-percha appeared incinerated. In sagittal sections, changes could be assessed in a way that, at 200°C gutta-percha cones were observed, along with endodontic cements particular to the treated teeth, filing all the canal and preserving their original color (Figure 6). At 400°C, some white streaks were observed in the material and continuity with endodontic cement was lost (Figure 7). These changes were even more evident when reaching 600°C (Figure 8). At 800°C and 1,000°C gutta-percha acquired a chalk-like whitish hue and appeared completely incinerated (Figures 9 and 10).
DISCUSSION
This in vitro research exposes how tooth-related tissues and materials used for dental purposes resist the effect of high temperatures and present specific changes at each temperature rank to which they were subjected. Nevertheless, it is important to mention that In vivo, these changes can vary according to extrinsic factors such as exposition time to thermal attack, nature of the fire, participation of combustible substances, temperature elevation curve and materials employed to extinguish the fire. Intrinsic factors must also be considered; among these are tissue and materials' thermal expansion coefficient, as well as the materials' point of fusion. Notwithstanding, in situ , teeth are not directly exposed to fire (especially posterior teeth) since they are protected by tissues surrounding the mouth, facial muscles, and roots exhibit all the protection provided by the periodontium, and bone cortical sections of upper and lower jaws.1-3,20 In addition to all the aforementioned factors, when bodily temperature rises, the digestive system produces gas, which, upon arriving to the oral cavity, protrude the tongue so that teeth become immersed in it.4
CHANGES IN DENTAL TISSUES
One of the most characteristic changes in dental tissues is the bursting of enamel in the cervical region, and the later detachment of the crown from the rest of the tooth. This phenomenon occurs because dentin, when subject to high temperatures, with high organic and 12% water content21 contracts due to dehydration. This fact confers dentin a certain degree of resistance with respect to enamel. Enamel possesses a high content of inorganic matter (96% to 99%), its mineral structure is composed of great amounts of calcium phosphate in the form of apatite crystals.22 Therefore, when this tissue is subject to high temperatures, it loses the little water it had, along with the collagen matrix, thus eliciting a strong contraction. As a consequence of this process, fissures, cracks or fractures appear, which in turn generate a cracked appearance. This discrepancy in tissue behavior with respect to its dimensional stability causes that enamel, at the cervical third , fractures at 200°C , separates from dentin at 400°C and finally separates from the crown as a cap once dentin is carbonized, and considerably reduces root volume from 800°C onwards.23-25 All the aforementioned changes were reported in studies conducted by Günther and Schmidt and mentioned by Rötzcher & al,26 Merlati & al6 and Moreno & al.24
When dealing with dental tissues fissures, cracks fractures and cracked appearance we can inform that, from 400°C onwards, fissures appear in enamel and cement, at 600°C, cracks are formed which can deepen up to the crown and root dentin respectively. This can be observed in saggital sections which, from 800°C onwards, can generate root fracture in some teeth. This pattern of longitudinal and transverse fissures and cracks confer to enamel and cement surface a cracked aspect which will later disappear when enamel fragmentation and cement incineration occur at 1,000°C. This was described by Merlati & al6 and Moreno & al.24
Finally, we might state that, discoloration of dental tissues is directly caused by carbonization and incineration levels at each temperature rank. This was reported by Merlati & al6 and Moreno et al.24
CHANGES IN DENTAL MATERIALS
In amalgam, changes in structure and superficial texture are related to the fusion points of all metals forming the alloy. Between 200°C and 400°C, amalgam presents a rough occlusal surface associated with the apparition of nodules which arise between 400°C up to 850°C with the mercury evaporation process, in the shape of gasified bubbles. When temperature decreases, pressure forces minerals dragged by mercury to group in the shape of these nodules.1 This process was mentioned by Merlati & al,6 Moreno & al,24 Günther and Scmidt, and by Rötzscher & al.26 These authors described these structures as ''silver bullets'' due to the fact that mercury drags traces of silver from 800°C onwards, and thus generate porosities in the amalgam structure.27
Another amalgam behavior relevant characteristic is pigmentation. It occurs especially in enamel, as temperature increases. Merlati & al6 mention the emergence of pinkish pigmentation in the root and crown of amalgam-filled teeth and subjected to 1,000°C and 1,100°C, whereas Moreno & al24 describe the presence of the aforementioned pigmentations in enamel located around the amalgam filling when subject to temperatures from 600°C up to 1,200°C. Reddish-brownish pigmentations are associated to the oxidation caused by high temperatures sustained by copper at temperatures from 450°C onwards.28 The brownish-golden halo which surrounds the amalgam obturation at temperatures between 800°C and 1,200°C is associated to mercury vapors,29 to copper itself28 and to silver oxides.29 The present study has found that dissociation of amalgam components due to high temperatures discolors enamel producing a yellowish halo at 600°C, and a golden-brownish halo at 800°C.
When dealing with composite resin, the fact that draws attention is the change in color. This is associated to incineration and carbonization levels as well as texture alteration. Texture becomes rougher as temperature increases up to the incineration point found at 1,000°C. From 400°C onwards, resin initiates the process of carbonization through the combustion of the acrylic matrix; at 800°C it incinerates and acquires a chalk-white color. Merlati & al6 and Moreno & al24 informed of similar changes.
Glass inomener showed results similar to those reported by Moreno & al24 which were not evident in all samples, mainly due to the materials' film thickness according to their function in the endodontic treatment which could be pulp protector, cavity base and crown sealing.
With respect to materials used for endodontic filling, no reports were found in scientific literature to inform of the behavior of endodontic-use cements used in the present study, therefore, discussion focuses on gutta-percha. This thermo-plastic material is characterized by having a soft consistency between 25°C and 30°C and liquid consistency from 60°C onwards. These characteristics can be observed in vitro. Nevertheless, Lopez & al30 inform that in vivo gutta-percha is able to withstand high external temperatures. The present study agrees with these observations: we found that at 800°C gutta-percha, even incinerated, can differentiate from dental tissues within the root canal. Another interesting aspect of gutta-percha is that, in the Moreno & al24 study, a greater number of fractures and root bursting were reported in teeth which had not received endodontic treatment. This situation did not take place in the present study, therefore, it can be inferred that gutta-percha, within the canal, can confer structural soundness to carbonized and incinerated dental tissues. This same situation was reported in the Savio & al31 study.
CONCLUSIONS
Dental tissues (enamel dentine and cement) as well as different dental use materials studied in the present paper, show a series of specific changes (color, texture, fissures, cracks, fractures, fragmentations). Therefore, their macroscopic behavior reveals information on temperatures ranges to which they might have been subjected.
Results of this study show that being familiar with the behavior of dental tissues and materials used in dentistry to the effect of high temperatures is of paramount importance in forensic dentistry during identification processes of a subject whose remains have been burned, carbonized, or incinerated.
REFERENCES
1. Moya V, Roldán B, Sánchez JA. Odontología legal y forense. Primera edición. Barcelona: Editorial Masson SA; 1994. [ Links ]
2. Ferreira JL, Espina AL, Barrios FA, Mavaréz MG. Conservación de las estructuras orales y faciales del cadáver quemado. Cien Odontol 2005; 2: 58-65. [ Links ]
3. Norrlander AL. Burned and incinerated remains. In: Bowers CM, Bell GL, Editores. Manual of forensic odontology. Third edition. Colorado Springs: American Society of Forensic Odontology; 1997: 16-18. [ Links ]
4. Delattre VF. Burned beyond recognition: Systematic approach to the dental identification of charred human remains. J Forensic Sci 2000; 45: 589-596. [ Links ]
5. Sweet D. ¿Por qué es necesario un odontólogo para la identificación? En: Fixot RH Editor invitado. Clínicas odontológicas de Norteamérica: odontología forense. Volumen 2. México: McGraw-Hill Interamericana; 2001. [ Links ]
6. Merlati G, Savio C, Danesino P, Fassina G, Menghini P. Further Study of restored and unrestored teeth subjected to high temperatures. J Forensic Odontostomatol 2004; 22: 17-24. [ Links ]
7. Mazza A, Merlati G, Savio C, Fassina G, Menghini P, Danesino P. Observations on dental structures when placed in contact with acids: Experimental studies to aid identification processes. J Forensic Sci 2005; 50: 406-10. [ Links ]
8. American Board of Forensic Odontology (ABFO). Body identification guidelines. J Am Dent Assoc 1994; 125: 1244-1254. [ Links ]
9. Marín L, Moreno F. Odontología forense: identificación odontológica, reporte de casos. Rev Estomatol 2003: 11: 41-49. [ Links ]
10. Marín L, Moreno F. Odontología forense: identificación odontológica de individuos quemados, reporte de dos casos. Rev Estomatol 2004; 12: 57-70. [ Links ]
11. Pretty IA, Sweet D. A look at forensic dentistry. Part 1: The role of teeth in the determination of human identity. Br Dental J 2001; 190: 359-366. [ Links ]
12. Edgar HJ. Prediction of race using characteristics of dental morphology. J Forens Sci 2005; 50: 1-5. [ Links ]
13. Williams D, Lewis M, Franzen T, Lissett V, Adams C, Whittaker D et al. Determination by PCR analysis of DNA extracted from incinerated, deciduous teeth. Sci Justice 2004; 44: 89-94. [ Links ]
14. Urbani C, Lastrucci RD, Kramer B. The effect of temperature on sex determination using DNA-PCR analysis of dental pulp. J Forensic Odontostomatol 1999; 17: 35-9. [ Links ]
15. Ministerio de la Protección Social. Resolución N° 008430/1993 de octubre 4. Por la cual se establecen las normas científicas, técnicas y administrativas para la investigación en salud. (fecha de acceso febrero de 2009). URL disponible en http://www.minproteccionsocial.gov.co/vbecontent/library/documents/DocNewsNo267711.pdf [ Links ]
16. Asociación Médica Mundial. Principios éticos para las investigaciones médicas en seres humanos. Declaración de Helsinki. Finlandia, junio 1964 (fecha acceso febrero de 2009). URL disponible en: http://www.wma.net/s/policy/b3.html [ Links ]
17. Instituto Colombiano de Normas Técnicas. Materiales odontológicos: Ensayo de la adhesión a la estructura dental. Bogotá: ICONTEC 4882; 2000. [ Links ]
18. International Organization of Standardization. Dental materials: Testing of adhesion to tooth structure. ISO/TS 11405: 2003. [ Links ]
19. Cohen S, Burns RC. Pathways of the pulp. Eigth edition. St. Louis: Mosby, Inc.; 2002. [ Links ]
20. Bohnert M, Rost T, Pollak S. The degree of destruction of human bodies relation to the duration of the fire. Forensic Sci Int 1998; 95: 11-21. [ Links ]
21. Torneck CD. El complejo pulpodentinario. En: Ten Cate AR, Editor. Histología oral: desarrollo, estructura y función. Segunda edición. Buenos Aires: Editorial Médica Panamericana SA; 1986: 190-235. [ Links ]
22. Eisenmann DR. Estructura del esmalte. En: Ten Cate AR, Editor. Histología oral: desarrollo, estructura y función. Segunda edición. Buenos Aires: Editorial Médica Panamericana SA; 1986: 252-73. [ Links ]
23. Espina A, Barrios F, Ortega A, Mavaréz M, Espina O, Ferreira J. Cambios estructurales en los tejidos dentales duros por acción del fuego directo, según edad cronológica. Cien Odontol 2004; 1: 38-51. [ Links ]
24. Moreno S, León ME, Marín L, Moreno F. Comportamiento de los tejidos dentales y de algunos materiales de obturación dental sometidos a altas temperaturas con fines forenses. Colomb Med 2008; 39 (1): 28-46. [ Links ]
25. Moreno S, Marín L, Merlati G, Savio C, Moreno F. Effects of high temperatures on different dental restorative systems: experimental study to aid identification processes. Journal of Forensic Dental Sciences 2009; 1 (1): 17-23. [ Links ]
26. Rötzscher K, Grundmann C, Benthaus S. The effects of high temperatures on human teeth and dentures. Int Poster J Dent Oral Med 2004; 6: Poster 213. [ Links ]
27. Odanov Z, Djurdjev M. Investigation of the mechanism of mercury removal from a silver dental amalgam alloy. J Serb Chem Soc 2004; 69 (12): 1111-11120. [ Links ]
28. Mrowec S, Stokosa A. Oxidation of copper at high temperatures. Oxidation of Metals 1971; 3: 291-311. [ Links ]
29. Kerl B, Forbeck F. Mercurio. En: Stohmann F, Editor. Gran enciclopedia de química industrial. Tomo X. Barcelona: F. Soix; 1956: 68-155. [ Links ]
30. López L, Arimany J, Prieto L, Martínez M. Importancia de la endodoncia en la identificación de cadáveres carbonizados. A propósito de un caso. Tercera Jornada Catalana de Actualización en Medicina Forense. Departamento de Justicia de Cataluña: Barcelona; 1995: 215-223. [ Links ]
31. Savio C, Merlati G, Danesino P, Fassina G, Menghini P. Radiographic evaluation of teeth subjected to high temperatures: Experimental study to aid identification processes. Forensic Science International 2006; 158: 108-116. [ Links ]
 Mailing address:
Mailing address:
Dr. Freddy Moreno
E-mail: freddyodont@hotmail.com
Note
This article can be read in its full version in the following page: http://www.medigraphic.com/facultadodontologiaunam











 text in
text in 


