1. INTRODUCTION
Dyes are organic compounds used mainly in the textile, pharmaceutical, cosmetic and food industries (Shao, Zheng Chen & Liu, 2017). They are characterized by having complex molecular structures with several functional groups, presenting low biodegradability (Hassaan, El Nemr, & Madkour, 2017), and high stability to light and heat. Their presence in bodies of water can foster carcinogenic and teratogenic effects in species living there (Cotto-Maldonado, Duconge Morant, & Cotto-Maldonado, Duconge Morant, & Márquez, 2017; Hassaan et al., 2017; Shao et al., 2017) and avoid entry of solar radiation affecting the aquatic flora and fauna.
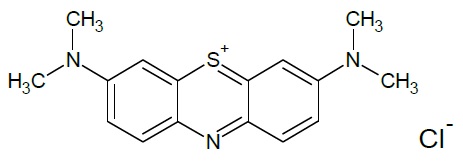
Methylene blue also called by IUPAC as 3,7-bis (Dimethylamino) phenothiazin-5-ium chloride is a dye which molecular structure is C16H18N3ClS (Figure 1). This dye is an aromatic heterocyclic cationic (Dawood & Sen, 2014). Used as medication, antiseptic and tissue colorant in some surgical processes (Cotto-Maldonado et al., 2017), and for dyeing cotton, wool, nylon, and other textile fibers (Turhan, Durukan, Ozturkcan, & Turgut, 2012). Its presence in effluents even at low concentrations becomes an important source of water pollution, contributing mainly to its eutrophication, affecting living beings. Physical, chemical and biological conventional methods are used for its degradation (Jian-Xiao, Ying, Guo-hong, Ling-yun, & Su-fen, 2011), however their application is not recommended, vis-à-vis the complexity of methylene blue chemical structure which demands large treatment times and energy, increasing degradation cost and sludge generation after treatment (Dawood & Sen, 2014; Hassaan et al., 2017; Lalnunhlimi & Krishnaswamy, 2016; Shao et al., 2017). For these reasons, it is essential to develop and evaluate new methods able to provide high pollutant removal efficiencies and to be environmentally friendly.
In the last decade, the Advanced Oxidation Processes (AOPs) have been used as technologies for removal of pollutants in water at room temperature and at atmospheric pressure (Hassaan et al., 2017; Turhan et al., 2012). These technologies are characterized by the production of active species mainly hydroxyl radicals (OH) (Li, Qiang, Pulgarin, & Kiwi, 2016) and for that some procedures are implemented such as: addition of oxidizing reagents and catalytic compounds. Specifically ultraviolet (UV) radiation, ozone (O3), hydrogen peroxide (H2O2), Fenton reactions (Fe2+/H2O2), photocatalysts (TiO2, ZnO, MnO, CuO, CuS) (Julkapli, Bagheri, & Bee Abd Hamid, 2014), and their combinations can be used. The degradation is possible by molecular reactions of the active species and the pollutant through direct and indirect pathways (Hassaan et al., 2017), where the mineralization of the contaminant can be achieved. The method choice mainly depends on pollutant characteristics, concentration and effluent volume (Liotta, Gruttadauria, Di Carlo, Perrini, & Librando, 2009).
AOPs have been applied for degradation of various water pollutants (Hossain et al., 2016; Ortiz et al., 2016; Rad et al., 2015). Diverse authors have used these methods for oxidation of methylene blue; some of them are summarized in Table 1.
Table 1 Summary of methylene blue degradation studies.
| Method used | Process conditions | Decolorization/ Degradation efficiency (%) | Reference |
| O3 | CMB = 3.12 ×
10−5 M CO3 =6.2 × 10−4 M Gas flow rate = 1 L/min T = 25 °C, pH 8 |
95.3% (liquid
circulation) 28.2% (without liquid circulation) |
(Al Jibouri, Wu, & Upreti, 2015) |
| O3 | CMB = 100
mg/L CO3 = input rate of 0.45 g/h V = 100 mL |
82.2% | (Zhang, Lee, Cui, & Jeong, 2009) |
| O3 | CMB = 200
mg/L CO3 = 5 g/h (A mixture of oxygen and ozone was introduced into the ozonation reactor) V = 450 mL pH = 11 |
90% | (Sumegova, Derco, & Melicher, 2013) |
| O3 carbon-assisted | CMB = 30
mg/L CO3 = input rate of 5 g/h V = 250 mL pH = 11 |
100% | (Zhang, Wang, Zhang, Zhang, & Fan, 2013) |
| H2O2/UV | CMB = 3, 5 and
10 mg/L CH2O2=0.25, 0.5, 1, 2.5, 5, 10, 20, 40, 80, 160 and 240 mmol Lamp = 8 W |
The best results for the
removal was performed with 1 Mmol of H2O2 |
(Masoumbeigi & Rezaee, 2015) |
| H2O2/UV | CMB = 30
mg/L CH2O2 = 3 mL (30% in H2O) V = 30 mL pH = 4-5 |
80% | (Jian-xiao et al., 2011) |
| H2O2/UV | CMB = 7.5 mg/L CH2O2 = 13.24 mmol (30% in H2O) = 50 min Lamp = 10 W |
85 % | (Xu, Tang, Liu, Jiang, & Tang, 2012) |
However, due to the undesirable presence of more and more different pollutants in water bodies around the world, it is important to increase and diversify studies for the treatment and removal of contaminants in water. The method to be applied for each case will be in function of the technical capabilities and the presence of necessary regional resources to try to reestablish the environmental equilibrium. In addition to the consideration of the use of the fewer chemical components quantity and keeping in mind the goal to obtain high removal efficiencies for great volumes of water.
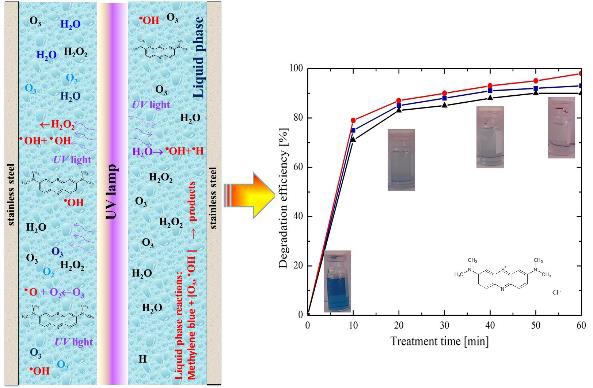
In this paper a comparative study of the degradation of methylene blue by combining various AOPs: O3, O3/H2O2, O3/UV, O3/UV/H2O2 and treatment time effect is presented. To evaluate qualitatively methylene blue degradation, color change of samples was considered as an indicator of degradation efficiency.
2. MATERIALS AND METHODS
2.1 MATERIALS
Methylene blue and hydrogen peroxide (30% W/W) from Fermont analytical grade were used. The ozone was provided by a generator as it is shown in next section.
2.2 EXPERIMENTAL SETUP
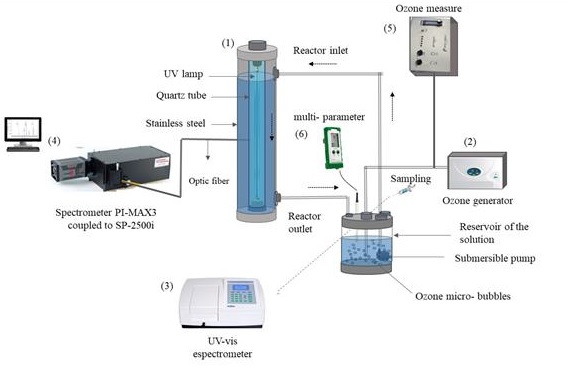
Figure 2 shows the applied experimental setup featured by: (1) a treatment reactor of stainless steel cylindrical configuration with a length of 35 cm and diameter of 7 cm, placed in the center a 12 W Philips ultraviolet lamp of 30 cm in length and shielded by a quartz tube, (2) an OEM-XEGZO-30N ozone generator, (3) an UV-visible spectrometer model VELAB, (4) an Acton SP2500 spectrometer with iCCD PI-MAX3, (5) a Teledyne Instruments 460L ozone meter and, (6) a Hanna-Instruments Gro Line HI9814 multi-parameter.
The degradation study of methylene blue was accomplished preparing an aqueous solution containing a 22 ppm initial concentration in 500 mL of distillated water. The aqueous solution was placed inside of the stainless steel cylindrical reactor. This solution was circulated continuously with a submersible pump located in a reservoir next to reactor lower part, this reservoir is filled with the solution coming from the reactor. The submersible pump send the aqueous solution to the upper inlet of reactor, flowing inside and collected again into the reservoir (see Figure 2). O3 micro-bubbles as an oxidizing reagent were introduced to the reservoir to increase the degradation efficiency. When it was necessary, H2O2 was added to the solution in the reservoir with one of three initial concentrations considered: 5 ppm, 10 ppm, or 20 ppm. The pH value was established by the addition of hydrochloric acid and sodium hydroxide both at 1 M and quantified with the multi-parameter device. The treatment time was set at 60 minutes, whereas the samples were collected and analyzed every 10 minutes using an UV-visible spectrometer. Previously, it was established a calibration curve at known concentrations of methylene blue, where the wavelength analyzed was 665 nm corresponding to the maximum absorbance of the dye (Goby, Penner, Lajoie, & Kelly, 2017). Coloration change of the solution was observed throughout the treatment as a qualitative parameter.
3. RESULTS AND DISCUSSION
3.1 CHARACTERIZATION OF THE OZONE GENERATOR AND UV LAMP
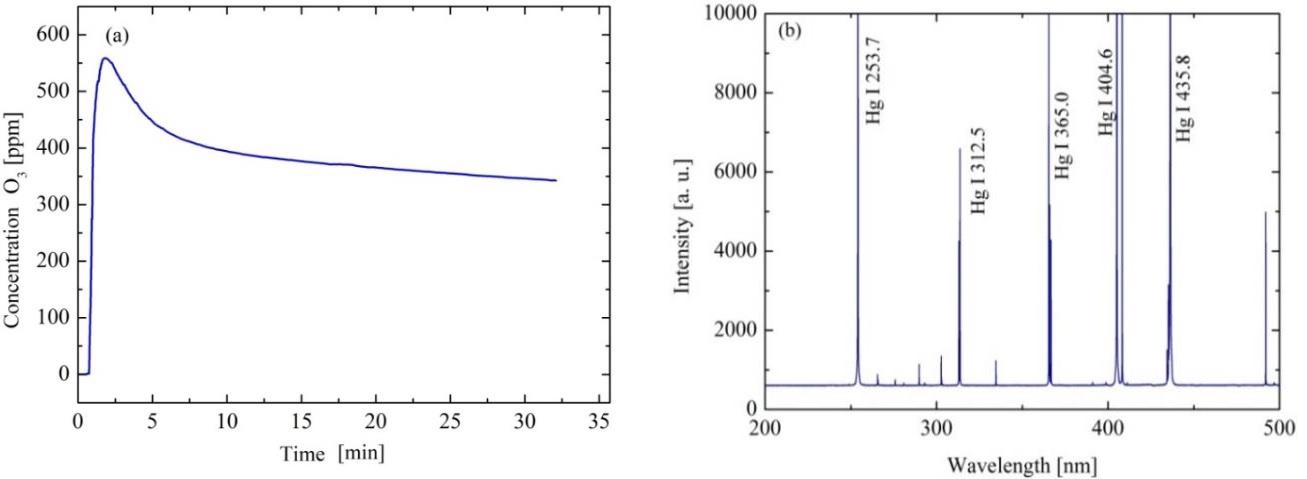
Figure 3(a) shows the O3 curve concentration at the output of the OEM-XEGZO-30N generator, which was feed with the atmospheric air as a precursor gas. From this figure, it can be observed that formed ozone concentration decreases progressively as the treatment time increases. From 30 minutes the resulting concentration was maintained quasi-constant at approximately 350 ppm. Ozone micro-bubbles were incorporated into the methylene blue solution to achieve a better diffusion favoring the degradation process of the contaminant. The aqueous solution and the diffused ozone are transported from the reservoir to the reactor by the submersible pump operation. Figure 3(b) shows the emission spectrum of the used lamp, where it can be noticed the characteristic Hg emission at 254 nm which produces the photolysis of molecules such as O3, H2O2 and H2O.
3.2 DEGRADATION OF METHYLENE BLUE BY ADVANCED OXIDATION METHODS
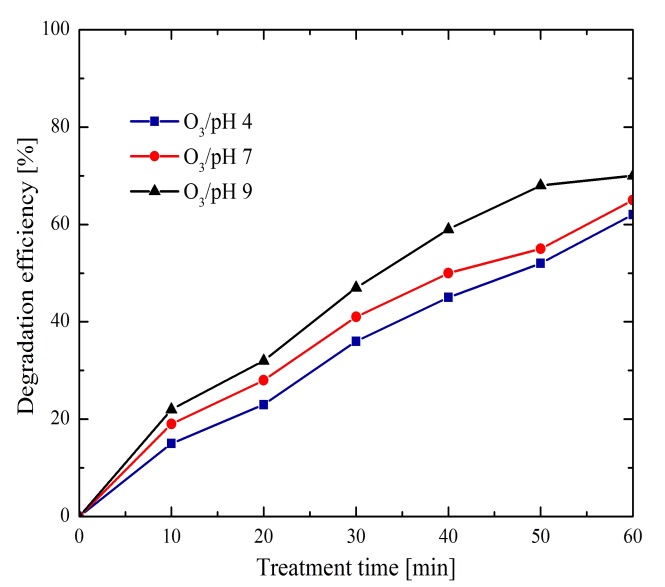
3.2.1 O3Effects
Figure 4 shows the results obtained from methylene blue treatment with an initial concentration of 22 ppm. It was analyzed the ozone effect in a solution with one of three different initial pH values (4, 7 and 9). Efficiencies for the three different pH values were obtained up to 70%, with a pH of 9 and 60 minutes of treatment time. As higher the treatment time the higher efficiencies are obtained. The ozone degradation mechanism of methylene blue can be performed by two different oxidation pathways. The first one consists in the direct reaction of O3 with the contaminant, see the reaction (1). Whereas the second one is due to the O3 decomposition with intermediate species generating active species, mainly OH, which in turns reacts with the pollutant promoting its degradation, see reactions (2)-(6):
Greater effectiveness was observed from resulting curves of Figure 4, when the pH was increased; some authors (Kusic, Koprivanac, & Bozic, 2006), have determined that the decomposition of O3 is reached at alkaline pH. Hence, the generation of OH radicals is augmented; as a consequence a better degradation of methylene blue is promoted. Also, the OH radicals are non-selective (Zhang et al., 2009), and their reaction with the pollutant is more significant than the O3 direct reaction process (Kusic et al., 2006).
However, the removal efficiency in Figure 4 can be considered relatively low, so other oxidizing agents in combination with ozone were evaluated as shown below.
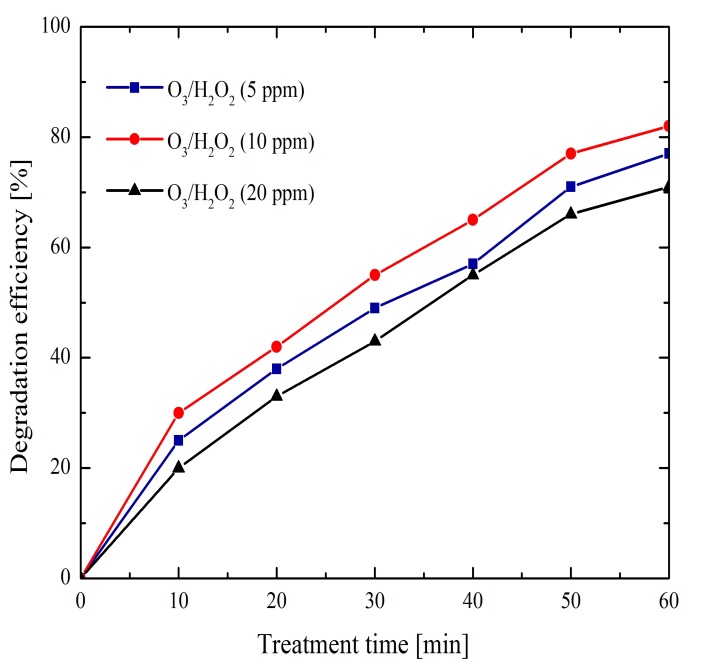
3.2.2 Effect of O3/H2O2
Figure 5 shows the obtained results of methylene blue degradation by the combination of ozone and hydrogen peroxide. In this analyzed case one of three different initial concentrations of H2O2 (5 ppm, 10 ppm, or 20 ppm) was added to the solution at the beginning of the treatment; for all cases, an initial value of 7 pH was set. Degradation efficiencies up to 82% were obtained with the addition of 10 ppm of H2O2, which represents an improvement compared to the previous study case results. This can be attributed to the combination of oxidizing agents that increase the amount of OH radicals mainly by the mechanism of reaction (7). Also, the H2O2 contributes to the generation of OH radicals, through the reactions (8)-(9). Therefore the methylene blue degradation process is improved.
It also can be noticed when 20 ppm H2O2 were added the degradation efficiency decreased. Because a higher OH radical production involves inhibitory reactions of reactive species avoiding their interaction with the pollutant. This inhibition processes can be attributed to:
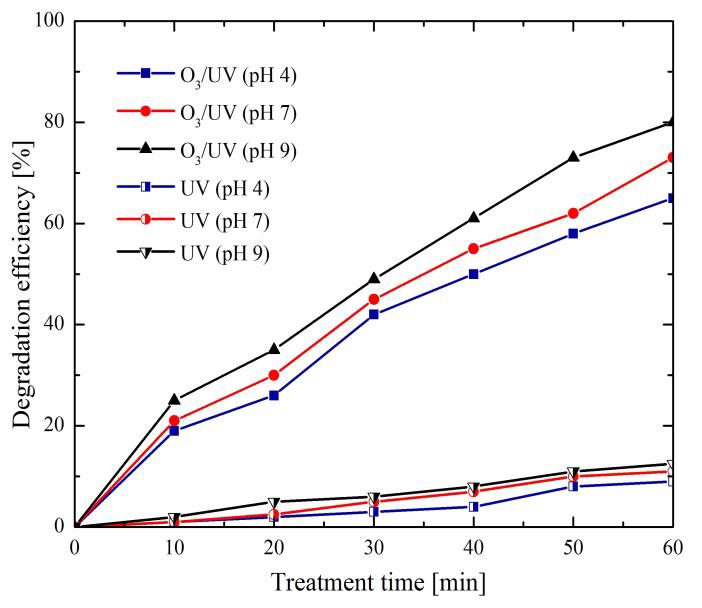
Figure 6 shows the results obtained from the degradation of methylene blue through of UV irradiation and O3 addition. The solution was prepared for each one of three different initial pH values (4, 7 and 9). A degradation efficiency of 12% was obtained when only UV radiation was supplied; this low efficiency was considerably increased up to 79% with the presence of O3 in solution with a pH 9. The UV radiation interaction with water and ozone allows the generation of several radicals following two different mechanisms. The first one, via the water photolysis at wavelengths between 200-280 nm defined by reaction (14); and the second one, by the molecular breakdown of chemical species including O3 and H2O2, see reactions (15)-(21).
As can be seen, a pH alkaline and the UV radiation contributed to increase the efficiency of degradation with regard to the case shown in Figure 4, where O3 was used only as a reactive agent. At pH alkaline O3 undergoes a fast decomposition.
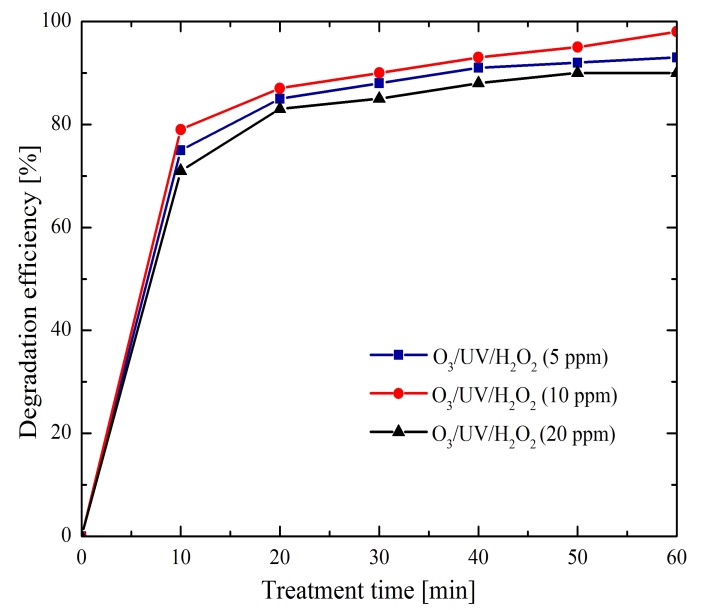
3.2.4 O3/UV/H2O2 effect
In this last case, the aqueous solution containing methylene blue was prepared only with an initial pH 7 and three different H2O2 concentrations were evaluated: 5 ppm, 10 ppm, or 20 ppm. And for all combinations a UV radiation was supplied. Where the respectively obtained degradation efficiencies were 94%, 98%, and 90%, which are depicted in Figure 7. Making a comparison of herein attained results, this last analyzed method provided the higher elimination efficiencies. Therefore, the combination of the two considered oxidizing agents and UV radiation made possible to obtain high efficiencies due to a synergistic effect. As it was observed in the implemented method of O3/H2O2 (Figure 5) the higher degradation efficiency was achieved when 10 ppm of H2O2 was added to the solution, confirming the inhibition processes effect.
For this last case, the set of reactions (14)-(21) were reinforced and even more the reaction (21), when adding directly to the system the H2O2 oxidizing agent and of which it is known that its decomposition is accelerated by UV radiation.
3.3 IDENTIFICATION OF BYPRODUCTS. OF OXIDATION: DECOLORATION OF METHYLENE BLUE IN SOLUTION
The decoloration of the methylene blue samples before treatment and each 10 minutes up to 60 minutes for a) O3 at pH 9, b) O3/H2O2 (10 ppm), c) O3/UV at pH 9 and d) O3/UV/H2O2 (10 ppm) are shown in Figure 8. It can be observed in all the cases a decoloration from the initial sample until the last one corresponding to the treatment end. When it was applied O3/UV/H2O2 (10 ppm), a better discoloration was obtained from the first ten minutes of treatment (case d). This can be attributed to the combined effect of the two oxidizing agents and the UV radiation, which generate an optimal quantity of OH radicals; these last oxidize the contaminant generating its decoloration.
4. CONCLUSIONS
The methylene blue degradation study was performed assessing some Advanced Oxidation Processes. In all the analyzed cases, it was observed as the higher treatment time; the higher degradation efficiencies were obtained. Furthermore, when H2O2 was added to the system, the increase in degradation efficiency allows to conclude that the degradation process occurs by an indirect mechanism, i.e. the electrophilic addition of the OH radical to the double bonds present in the compound molecule, this is still more reinforced when the pH is lightly alkaline and the UV radiation is supplied, since these last two conditions accelerate the chemical dissociation of O3 and H2O2 and OH radical formation. It was also observed that when H2O2 is added to the solution, it existed an optimal concentration and that excess was in detrimental to the degradation process. The combination of O3/UV/H2O2 generated a synergistic effect, and the highest elimination efficiencies were achieved (98%) without the addition of supplementary chemicals. Qualitatively a decrease in color was observed as the treatment time increased.











 text new page (beta)
text new page (beta)