Introduction
Chest pain accounts for approximately 5-20% of the total visits to the emergency department (ED)1. As chest pain can be caused by life-threatening conditions, it is of utmost importance to identify which of those patients presenting to the ED with chest pain are at risk of major adverse cardiovascular events (MACE). A rapid and extensive workup is needed to exclude acute coronary syndromes (ACS) and to simultaneously avoid an excessively long length of stay and high ED costs2. Many clinical risk stratification tools to determine risk of MACE have been developed for patients presenting with chest pain, but none of these have been validated in Mexican patients.
The aim of this study is to compare the discriminative power and accuracy for the prediction of MACE of five commonly used chest pain scoring tools: ED assessment of chest pain score (EDACS), thrombolysis in myocardial infarction (TIMI) score, global registry of acute coronary events (GRACE) score, history, ECG, age, risk factors, and troponin (HEART) Score, and Vancouver chest pain rule in Mexican patients who presented to the ED of a tertiary-level hospital.
Materials and methods
This prospective, observational, and comparative study included patients admitted to the ED with chest pain. The study was performed in a tertiary-level teaching hospital which serves as a cardiovascular referral center in a large metropolitan area. The protocol received local research and ethics committee approval and complied with the Declaration of Helsinki principles. Written informed consent was obtained from all participants.
Any patient admitted to the ED of a tertiary level referral hospital in Mexico City with chest pain as the chief complaint between August 8, 2019, and March 12, 2020, was eligible for the study. Patients presenting with other symptoms that could be potentially caused by ACS, such as dyspnea or palpitations, were not included in the study. Patients with ST-elevation (defined as more than 1mm elevation in two or more anterior leads, or 0.5 mm elevation in posterior leads) on the initial electrocardiogram (EKG), with cardiac arrest during pre-hospital transfer or in the ED, or who lacked any of the necessary data required for calculation of the different scores were excluded from the study.
Research fellows from the study site acquired the information using a standardized case report form (CRF). The CRF consisted of entries for past medical history, cardiovascular risk factors, EKG findings, laboratory results, and contact information for further follow-up. Initial EKGs were digitized and stored in an anonymized database, after which they were reviewed, blinded, by two experienced cardiologists. Laboratory values, including high sensitivity cardiac troponin I values, were collected throughout the study period, and entered into the CRF. A secure database was constructed for the data input. Each score was prospectively applied to each patient in our study. We used the originally described cut-points of each score to distinguish low-risk from high-risk patients. An algorithm was constructed within the database to obtain automatic interpretation of the different chest pain scores. EDACS, TIMI, GRACE, HEART, and Vancouver chest pain rule scores were calculated and recorded as the main independent variables.
Patients who were enrolled in the study were contacted through telephone by research fellows at 30 days after their initial ED visit to determine if any of the study outcomes had occurred; if deemed necessary, on-site visit was planned for further evaluation. All collected information was added to the original database for later analysis.
The primary endpoint in our study was the incidence of MACE within 30 days of a patient's index presentation to the ED. MACE consisted of cardiovascular death, myocardial infarction, coronary intervention, coronary artery bypass grafting, or readmission for cardiovascular causes.
Statistical analysis
For the descriptive analysis, categorical variables were analyzed with frequencies and percentages. Comparisons were made using the χ2 or Fisher's exact test, as appropriate. Distributions of continuous variables were analyzed using Shapiro-Wilk's test for normality and were described as mean and standard deviation, with median and interquartile ranges as appropriate. For comparisons between continuous variables, we used Student's t or Mann-Whitney's test, according to the distribution of the variables. To assess the performance of chest pain scores for prediction of 30-day MACE, we used ROC (receiver-operating characteristic) analysis and for the differences among these scores, we used the integrated discrimination improvement (IDI) test. To know the exact predictive power for 30-day MACE, we performed a logistic regression analysis. A two-sided significance level of 0.05 was used for all analyses. Data were analyzed with STATA SE 14 (StataCorp College Station, Texas).
Results
A total of 199 patients met eligibility criteria. Of these, 17 patients (8.54%) were lost to follow-up. In 14 patients (7.03%), the complete scores could not be calculated; therefore, 168 patients were included in the final analysis. At admission, only two clinical findings with statistical significance between patients with and without MACE were history of diabetes mellitus (44.2% vs. 26.4, p = 0 0.02) and previous coronary intervention (44.2 vs. 28.5, p = 0 0.05) (Table 1).
Table 1 Baseline characteristic and clinical finding of patients admitted for chest pain
| Variable | Total (n = 198) | Without MACE (n = 144) | With MACE (n = 43) | p |
|---|---|---|---|---|
| Male (%) | 65.7 | 63.9 | 72.1 | 0.32 |
| Female (%) | 34.3 | 36.1 | 27.9 | 0.45 |
| Hypertension (%) | 59.6 | 59 | 65.1 | 0.47 |
| Diabetes (%) | 29.8 | 26.4 | 44.2 | 0.02 |
| Atrial fibrillation (%) | 5.1 | 5.6 | 4.7 | 0.81 |
| Dyslipidemia (%) | 49.5 | 47.2 | 57.1 | 0.20 |
| Prior smoking (%) | 42.9 | 44.4 | 39.5 | 0.56 |
| Active smoking (%) | 14.7 | 12.5 | 18.6 | 0.31 |
| Previous myocardial infarction (%) | 36.4 | 34 | 48.8 | 0.07 |
| Cancer (%) | 2 | 0.7 | 4.7 | 0.07 |
| Previous hospitalization (%) | 4 | 3.5 | 4.7 | 0.57 |
| Previous heart failure (%) | 43.4 | 41 | 51.2 | 0.23 |
| Previous coronary intervention (%) | 30.8 | 28.5 | 44.2 | 0.05 |
| Previous CABG (%) | 4.1 | 4.9 | 2.3 | 0.41 |
| Age (years) (median, IQR) | 61 (51-70) | 59 (49-68) | 63 (58-71) | 0.04 |
| Systolic blood pressure (mmHg) (median, IQR) | 138 (121-150) | 140 (123-150) | 136 (116-150) | 0.70 |
| Diastolic blood pressure (mmHg) (median, IQR) | 80 (70-90) | 80 (70-90) | 82 (70-92) | 0.16 |
| Heart rate (bpm) (median, IQR) | 73 (63-85) | 72 (63-84) | 78 (62-90) | 0.35 |
| Respiratory rate (rpm) (median, IQR) | 17 (16-18) | 16 (16-18) | 18 (16-18) | 0.54 |
| Oxygen saturation (%) (median, IQR) | 94 (93-96) | 94 (93-96) | 93 (90-95) | 0.00 |
| High-sensitivity troponin (pg/mL) (median, IQR) | 8 (4.3-24.1) | 6.9 (3.9-14) | 36.4 (7.8-744) | 0.00 |
| NT-pro BNP (pg/mL) (median, IQR) | 636 (105-1810) | 289 (66-1245) | 1115 (296-4380) | 0.00 |
| Hemoglobin (g/L) (median, IQR) | 15.3 (14-16.4) | 15.3 (14-16.3) | 15.5 (14-16.4) | 0.68 |
| Creatinine (mg/dL) (median, IQR) | 0.93 (0.75-1.13) | 0.91 (0.78-1.1) | 0.96 (0.72-1.15) | 0.42 |
MACE: mayor adverse cardiovascular events; CABG: coronary artery bypass graft; IQR: interquartile range; NT-pro BNP: N-terminal pro hormone brain natriuretic peptide.
A total of 43 MACE occurred within 30 days of discharge (25.5%) consisting of 1 death (0.6%), 28 myocardial infarctions (15.9%), 31 coronary interventions (17.5%), and 3 rehospitalizations (1.7%); no patient underwent CABG surgery. Figure 1 demonstrates the ROC curves of the HEART score, EDACS, Vancouver rule, TIMI, and GRACE scores to predict MACE within 30 days. The area under the curve (AUC) of the HEART score was highest (0.76, 95% CI: 0.70-0.85) among the five scores.
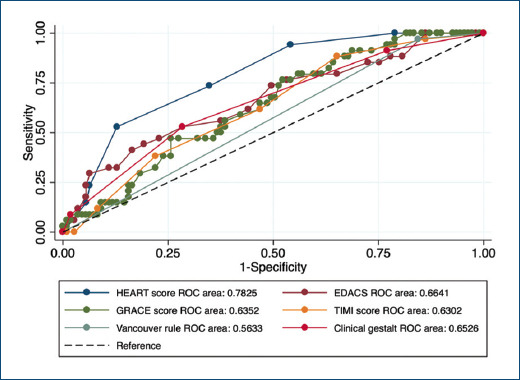
Figure 1 ROC curve analysis showing the association of the different chest pain prediction rules for 30-day MACE in patients with chest pain. ROC: receiver operating characteristic; MACE: mayor adverse cardiovascular events; HEART: history, ECG, age, risk factors, troponin (HEART) score; GRACE: global registry of acute coronary events score; EDACS: emergency department assessment of chest pain score; TIMI: thrombolysis in myocardial infarction score.
The median results for each of the tools among patients with and without MACE during follow-up are shown in table 2. To evaluate the incremental value of each score, we performed an integrated discrimination improvement (IDI) test for the HEART score, which demonstrated higher power for MACE prediction (IDI = 0.06, p = 0.001). Finally, we performed a logistic regression model for MACE prediction, which demonstrated that the HEART score, EDACS, and Vancouver rule had higher, statistically significant odds ratios (OR) (Table 3).
Table 2 Comparison of SCORES for chest pain
| Variable | Total (n = 198) | Without MACE (n = 144) | With MACE (n = 43) | p |
|---|---|---|---|---|
| HEART | 5 (4-6) | 5 (4-6) | 6 (5-7) | < 0.001 |
| EDACS | 14 (8-18) | 12 (6-17) | 14 (13-23) | < 0.001 |
| GRACE | 81 (54-103) | 77 (49-98) | 89 (74-107) | 0.02 |
| TIMI | 2 (1-3) | 2 (1-3) | 3 (2-4) | 0.02 |
| Abnormal Vancouver rule | 45.8 | 38.8 | 67.6 | < 0.001 |
| Residual risk | ||||
| Low | 21.4 | 25.7 | 7 | < 0.001 |
| Intermediate | 47 | 48.6 | 41.8 |
HEART: history, ECG, age, risk factors, troponin score; EDACS: emergency department assessment of chest pain score; TIMI: thrombolysis in myocardial infarction score; GRACE: global registry of acute coronary events score; MACE: mayor adverse cardiovascular events.
Table 3 Logistic regression model for the prediction of 30-day MACE adjusted by age and sex in patients with chest pain
| Variable | OR | SE | p | 95% CI |
|---|---|---|---|---|
| HEART > 6 points | 3.56 | 1.50 | < 0.001 | 1.55-8.14 |
| EDACS > 14 points | 3.01 | 1.35 | 0.01 | 1.25-7.26 |
| Vancouver rule | 2.97 | 1.22 | < 0.001 | 1.33-6.66 |
| TIMI > 4 | 1.21 | 0.45 | 0.60 | 0.8-2.51 |
| GRACE > 89 | 0.86 | 0.36 | 0.72 | 0.37-1.97 |
HEART: history, ECG, age, risk factors, troponin (HEART) score; EDACS: emergency department assessment of chest pain score; TIMI: thrombolysis in myocardial infarction score; GRACE: global registry of acute coronary events score; OR: odds ratio; SE sensibility; CI: confidence interval.
Discussion
In our study, we found the HEART score to be the most accurate tool to identify those patients with the highest risk of MACE within 30 days of their index visit to the ED in a Mexican population presenting to a tertiary level referral hospital.
The HEART score was developed in 2008 with the aim to stratify patients who presented to the ED with chest pain into low, intermediate, or high risk of MACE at 423 ± 106 days and has been widely validated3. The study recruited patients in a general community hospital where most of the population was Caucasian. It is important to emphasize that their population had a lower proportion of comorbidities compared with our study population4.
The other two scores which better identified those patients at high risk of MACE in our population were the EDACS score and Vancouver chest pain rule.
The EDACS score can reliably identify patients with low risk for MACE (< 1%) in caucasians; however, it does not stratify patients from other backgrounds into different risks5.
Furthermore, their population had a lower incidence of comorbidities than those in our study, while they reported a lower incidence of MACE (15.5% vs. 23%).
The Vancouver chest pain rule was derived from a prospective cohort in a tertiary care center with the disadvantage that it only recognizes myocardial infarction as its primary outcome6. It was further validated by Cullen et al. by including high sensitivity troponin assays in 20147. Similarly, to the EDACS score, it identifies those patients at low risk for myocardial infarction who can be safely discharged from the ED. The original study was performed in a tertiary level referral hospital whose population had the greatest incidence of comorbidities among original studies of the five clinical scores; however, they were still not as frequent as in our population. The proportion of patients with myocardial infarction was also lower than that in our study (21.6% vs. 24%)7.
Compared to the previous scores, when applying TIMI and GRACE risk scores in our population study did not perform as well and had a low prognostic value.
TIMI score is derived from selected clinical trial cohorts and is based on eight clinical indicators available on admission8. The ability of TIMI score to predict major adverse cardiac events (MACE) was assessed in TIMI 11B study, where a MACE was identified at 14 days noted in 31% of patients with a TIMI risk score of 6 or 79. However, the relationship between the events and the TIMI score vary according to the characteristics of the population in which is applied as occurred in our study.
Finally, the GRACE score is based on a large registry of patients across the entire spectrum of coronary syndromes and is designed to determine all cause mortality at 6 months10. Despite including components such as age, Killip classification, hemodynamic status, and creatinine, which have been shown to be strong predictors of mortality after STEMI, in our study, the GRACE score showed a poor performance that may be related to the exclusion of the component of cardiac arrest and ST-segment deviation, which are components of GRACE score, indicating myocardial damage in progress and causing further events such as cardiogenic shock and recurrent MI11.
One of the strengths of our study is that the chest pain classification tools employed were originally described to classify patients with fewer comorbidities and cardiovascular risk factors. Therefore, this favors them as useful tools for discriminating patients at higher risk for cardiovascular disease, as in our study population due to the higher incidence of comorbidities.
Another strength of our study is that the discriminative power of each score was analyzed separately; in addition, calculation of IDI further improved the discrimination and classification of patients. To the best of our knowledge, the previously mentioned scores had never been compared by this approach in this kind of population.
Study limitations comprised development at a single site as well as the small patient sample, so our results may not be generalizable to other hospital settings.
Conclusions
This prospective and observational study demonstrated that the HEART score showed the higher accuracy and discriminative power to identify patients with chest pain at a higher risk of major cardiovascular endpoints. These results are of particular interest for the proper stratification of patients with chest pain in ED s in Mexico.











 text new page (beta)
text new page (beta)


