Introduction
Optimal timing for reperfusion in ST-segment elevation myocardial infarction (STEMI) reduces infarct size, prevents, and delays ventricular remodeling and increases survival1-3. In spite of this, a big proportion of patients, especially in low-income countries do not receive reperfusion promptly or receive it lately.
Clinical practice guidelines recommend two main strategies for reperfusion in STEMI: primary percutaneous coronary intervention (PPCI) and pharmacoinvasive strategy (PS)1-4. PPCI consists in an urgent recanalization of the obstruction in the lumen of the affected coronary artery by means of a balloon or stent, without previous administration of a fibrinolytic agent. It has been demonstrated that coronary flow can return to normal in 90% of the cases, while for fibrinolysis flow is only restored in about 50-60%5-12. The success of reperfusion depends on many factors such as the time of onset of symptoms, the ability and experience of the operator and resource availability for the procedure12-15.
There is a problem with the availability of PCI centers in many parts of the world, as it has been stated in other studies16-18. To overcome these difficulties, PS consists in the administration of a fibrinolytic agent at first moment after diagnosis of STEMI, followed by a coronary intervention in the next 3-24 h. This practice has reduced reinfarction and recurrent ischemia compared to medical treatment alone18. The STREAM trial demonstrated that the PS, together with contemporary antithrombotic therapy (clopidogrel, aspirin, and enoxaparin), has the same efficacy and safety than PPCI19. Other trials stand out the ability to overcome the social and geographic limitations16,20-22.
Mexico City and its metropolitan area constitute one of the most populated urban areas in the world23. The peculiar social and economic problematics of our country, together with a high population density, makes it difficult to treat STEMI in optimal timing so to accomplish guidelines goals represents a big challenge24. Therefore, the means of this study were to compare in-hospital mortality in patients taken to PPCI against those taken to PS in Mexico City and a large metropolitan area with the goal to establish the efficacy of PS in a real life setting.
Methods
A cohort from the PHASE-MX trial was taken, which included all patients from both genders, between 18 and 80 years of age, with a diagnosis of STEMI, that were admitted to the Emergency Department and Coronary Care Unit of the Instituto Nacional de Cardiología, from April 1, 2018, to March 31, 2019. The myocardial infarction definition used in this study was the one proposed by the European Society of Cardiology and Thygesen et al.1,2
At admission, the following data were collected: age, gender, date of admission, the presence of diabetes mellitus, systemic arterial hypertension, smoking, chronic kidney disease, obesity, previous history of myocardial infarction, previous revascularization, vital signs, TIMI, GRACE, and CRUSADE scores, blood biometrics, blood glucose, troponin, NT proBNP, total ischemic time, first medical contact time, door-to-needle time, door-to-wire crossing or device time, medical treatment before reperfusion, time to PS, and treatment success. In this study, all the patients from the PS group received fibrinolysis at their first medical contact center and angiography was done at Instituto Nacional de Cardiología, both decisions were made according to the medical staff’s discretion. Afterward, we made an in-hospital follow-up where mortality and date of home discharge were registered.
Statistical analysis: all tests were done in STATA v13 (StataCorp LP, College Station, Tx). Quantitative variables were analyzed with descriptive methods depending on their distribution, corroborated by the ShapiroWilk test. Variables with a normal distribution were described with mean value and standard deviation. Otherwise, median and interquartile ranges were used. Taking into consideration the normality of each quantitative variable, an analysis with Student’s t and U MannWhitney tests was performed. Qualitative variables were described through frequencies and percentages, while for the bivariate analysis c2 or Fisher’s test were performed depending on the number of events. For the survival analysis, tables and KaplanMeier curves were made to describe mortality in both groups. Differences between survival times for both treatment groups were compared with log rank test. Cox regression models, adjusted, by sex and age, were built to determine the main predictors of in-hospital mortality in patients treated with both strategies. A p < 0.05 was considered as statistically significant.
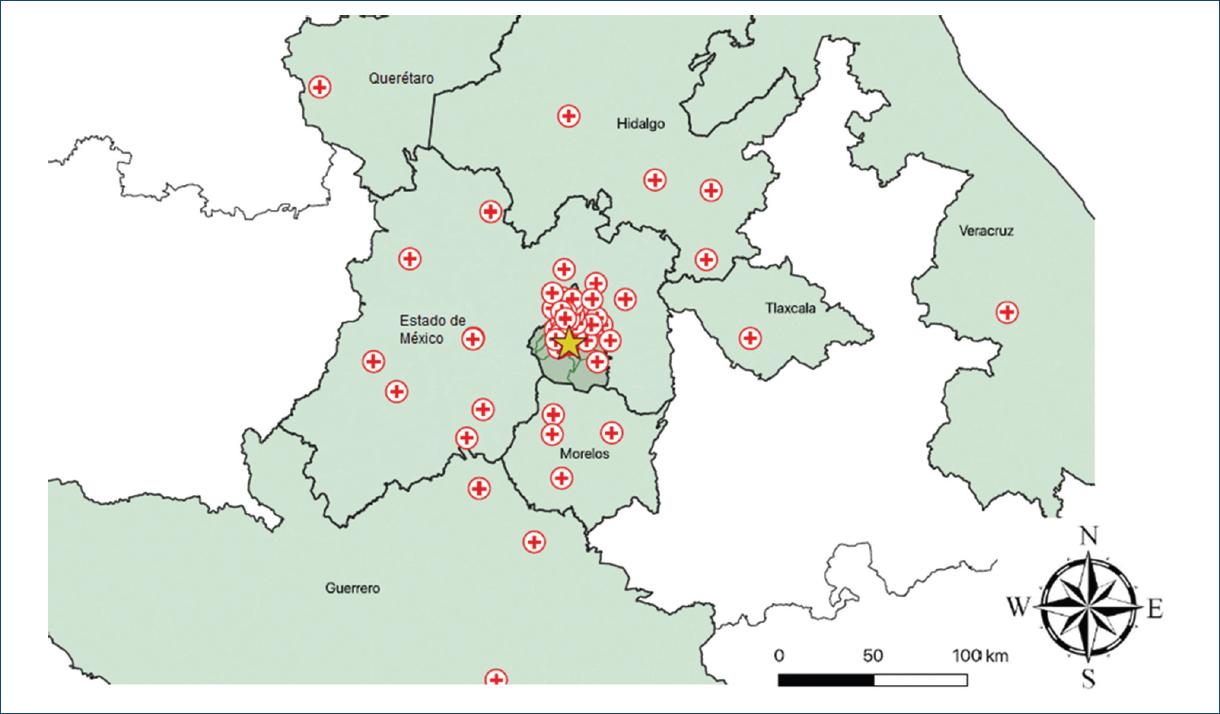
Furthermore, for the descriptive analysis, we created a map of the hospitals that are part of our STEMI network. All hospitals were geocoded by finding their latitude and longitude using Google Maps. The coordinates were recorded in a separate datasheet. We used QGIS 3.10 (2019, QGIS Geographic Information System. Open Source Geospatial Foundation. URL: http://qgis.org) to create our maps. From the University of California at Berkeley library for GeoData (https://geodata.lib.berkeley.edu/), we downloaded polygon shapefiles of the states and municipalities where the hospitals are located and added the hospitals (points) as another layer. Finally, we showed our center with a different mark to highlight the distances between the different hospitals in the network and our center.
Results
A total of 340 patients were included, 166 for PS and 174 for PPCI. The mean age was 59 ± 10.8 years, 87.1% were male and 12.9% were female. Regarding medical history, 35% had diabetes mellitus, 46.8% hypertension, 17.1% dislipidemia, 46.2% were current smokers, and 9.7% had a previous myocardial infarction (Table 1).
Table 1 Demographic characteristics of patients with STEMI
| Variable | Total (n = 340) | PS (n = 166) | PPCI (n = 174) | p | |||
|---|---|---|---|---|---|---|---|
| n | % | n | % | n | % | ||
| Male | 296 | 87.1 | 148 | 89.2 | 148 | 85.1 | 0.26 |
| Female | 44 | 12.9 | 18 | 10.8 | 26 | 14.9 | |
| Diabetes | 119 | 35 | 58 | 34.9 | 61 | 35.1 | 0.98 |
| Hypertension | 159 | 46.8 | 72 | 43.4 | 87 | 50 | 0.22 |
| Dyslipidemia | 58 | 17.1 | 21 | 12.7 | 37 | 21.3 | 0.03 |
| Current smoking | 157 | 46.2 | 88 | 53 | 69 | 39.7 | 0.01 |
| Previous smoking | 56 | 16.5 | 23 | 13.9 | 33 | 18.97 | 0.2 |
| Chronic kidney disease | 7 | 2.1 | 4 | 2.4 | 3 | 1.72 | 0.47 |
| Obesity | 77 | 22.7 | 35 | 21.1 | 42 | 24.1 | 0.50 |
| Previous myocardial infarction | 33 | 9.7 | 14 | 8.4 | 19 | 10.9 | 0.43 |
| Previous PCI | 23 | 6.8 | 7 | 4.3 | 16 | 9.2 | 0.05 |
| Previous CABG | 5 | 1.5 | 1 | 0.6 | 4 | 2.3 | 0.20 |
| Heart failure | 3 | 0.9 | 0 | 0 | 3 | 1.7 | 0.08 |
| Valvular heart disease | 2 | 0.6 | 0 | 0 | 2 | 1.2 | 0.26 |
| Atrial fibrillation | 1 | 0.3 | 0 | 0 | 1 | 0.6 | 0.32 |
| n | Mean ± SD | n | Mean ± SD | n | Mean ± SD | p | |
| Age (years) | 340 | 59 ± 10.8 | 166 | 58.5 ± 10.9 | 60 ± 11 | 0.08 | |
PS: pharmacoinvasive strategy; PPCI: primary percutaneous coronary intervention; CABG: coronary artery bypass grafting; SD: standard deviation.
As to the place of residency, 55.29% came from Mexico City, 29.41% came from Estado de México, and 5.29% came from Morelos. We received patients from 60 different hospitals, with a mean distance of 25.2 km, the smallest distance was 1.3 km and the longest was 312 km. The mean estimated transfer time was 53 min, being the minimum and maximum of 8 and 263 min, respectively. Moreover, 23.35% of the patients had their first medical contact at the Instituto Nacional de Cardiología and hospitals who referred the majority of patients to our center were Hospital General Dr. Manuel Gea González, Hospital General Balbuena, Hospital General La Perla, Hospital General de Cuernavaca, and Cruz Roja Mexicana (Table 2). Figures 1 and 2 show the geographic distribution of the PS network in the Instituto Nacional de Cardiología.
Table 2 Description of the place of first medical contact in patients with STEMI
| Place of first medical contact | n | % | Distance to NHI (km) | Estimated time to NHI (hh:mm) |
|---|---|---|---|---|
| Instituto Nacional de Cardiología | 76 | 22.35 | - | - |
| Médico particular | 55 | 16.17 | - | - |
| Hospital General Dr. Manuel Gea González | 42 | 12.35 | 1.3 | 0:08 |
| Hospital General Balbuena | 18 | 5.29 | 19.7 | 0:41 |
| Hospital General La Perla | 17 | 5 | 25.1 | 0:53 |
| Hospital General de Cuernavaca Dr. José G. Parres | 13 | 3.82 | 67.7 | 0:55 |
| Cruz Roja de México | 9 | 2.65 | 21.9 | 1:40 |
| Hospital General La Villa | 9 | 2.65 | 28.3 | 0:55 |
| Instituto Nacional de Ciencias Médicas y Nutrición | 6 | 1.76 | 0.85 | 0:06 |
| Hospital de Especialidades Dr. Belisario Domínguez | 6 | 1.76 | 12 | 0:50 |
| Instituto Nacional de Enfermedades Respiratorias | 5 | 1.47 | 1.4 | 0:08 |
| Hospital General de Texcoco Guadalupe Victoria | 5 | 1.47 | 46.5 | 1:06 |
| Hospital General de Tulancingo | 4 | 1.18 | 131 | 2:07 |
| Hospital General Dr. Enrique Cabrera | 4 | 1.18 | 20.8 | 0:39 |
| Hospital General de Naucalpan Dr. Maximiliano Ruiz Castañeda | 4 | 1.18 | 28.8 | 1:10 |
| Centro Médico de Toluca Adolfo López Mateos | 4 | 1.17 | 67.6 | 1:12 |
| Hospital General Dr. Gustavo Baz Prada | 3 | 0.88 | 25.3 | 0:53 |
| Hospital General de Cuatitlán José Vicente Villada | 3 | 0.88 | 56.2 | 1:22 |
| Hospital ISSEMYM Tlalnepantla | 3 | 0.88 | 39.6 | 1:10 |
| Hospital General Dr. Nicolás San Juan | 2 | 0.59 | 68.1 | 1:13 |
| Clínica 25, IMSS | 2 | 0.59 | 22.4 | 0:44 |
| Hospital General de Huichapan | 2 | 0.59 | 183 | 2:41 |
| Hospital General de Chilpancingo Dr. Raymundo Abarca Alarcón | 2 | 0.59 | 251 | 2:36 |
| Clínica Médica Mardán | 2 | 0.59 | 16.6 | 0:36 |
| Hospital General de Jilotepec | 2 | 0.59 | 114 | 2:07 |
| Hospital General Gregorio Salas | 2 | 0.59 | 19.2 | 0:45 |
| Hospital General de México | 2 | 0.59 | 17.3 | 0:36 |
| Hospital General del Valle del Mezquital | 2 | 0.59 | 168 | 2:50 |
| Hospital General de Tláhuac Dr. Miguel Lima Ramírez | 2 | 0.59 | 120 | 1:55 |
| Hospital General de Taxco | 2 | 0.59 | 161 | 2:06 |
| Hospital General de Valle de Bravo | 2 | 0.59 | 144 | 2:01 |
| Hospital General de Tlaxcala | 1 | 0.29 | 120 | 1:55 |
| Hospital General de Milpa Alta | 1 | 0.29 | 22.6 | 0:54 |
| Instituto Nacional de Cancerología | 1 | 0.29 | 1.2 | 0:06 |
| Hospital General de Cuautla Dr. Mauro Belauzarán Tapia | 1 | 0.29 | 90.2 | 1:16 |
| Hospital General de Huitzuco | 1 | 0.29 | 180 | 2:07 |
| Hospital General de Jojutla Dr. Ernesto Meana San Román | 1 | 0.29 | 128 | 1:37 |
| Hospital Escandón | 1 | 0.29 | 21.2 | 0:38 |
| Hospital General de Ixtapan de la Sal | 1 | 0.29 | 122 | 1:46 |
| Hospital Regional 1° de Octubre | 1 | 0.29 | 24.9 | 1:02 |
| Hospital General de Tenancingo Miguel Hidalgo y Costilla | 1 | 0.29 | 96.3 | 1:49 |
| Hospital General Darío Fernández Fierro, ISSSTE | 1 | 0.29 | 17.5 | 0:30 |
| Clínica Materno Infantil Sagrada Familia | 1 | 0.29 | 30 | 0:55 |
| Hospital General del Altiplano | 1 | 0.29 | 104 | 2:10 |
| Instituto Nacional de Neurología y Neurocirugía | 1 | 0.29 | 6.8 | 0:16 |
| Hospital General de San Felipe del Progreso | 1 | 0.29 | 147 | 2:05 |
| Hospital General de Ticomán | 1 | 0.29 | 29.1 | 1:20 |
| Hospital General de Zona #8 | 1 | 0.29 | 11.3 | 0:30 |
| Hospital General de Atizapán | 1 | 0.29 | 40.5 | 1:06 |
| Hospital General Ajusco Medio | 1 | 0.29 | 8.9 | 0:29 |
| Hospital General de Las Américas | 1 | 0.29 | 43.1 | 1:08 |
| Hospital General de Temixco | 1 | 0.29 | 87.6 | 1:09 |
| Hospital General Rubén Leñero | 1 | 0.29 | 28.8 | 0:48 |
| Hospital General de Querétaro | 1 | 0.29 | 231 | 2:59 |
| Hospital General de Tacuba, ISSSTE | 1 | 0.29 | 27.7 | 0:49 |
| Hospital Municipal de Temascaltepec | 1 | 0.29 | 130 | 2:13 |
| Hospital General de Coatepec | 1 | 0.29 | 312 | 4:23 |
| Hospital General de Pachuca | 1 | 0.29 | 114 | 2:08 |
| Hospital General de Ecatepec | 1 | 0.29 | 34.9 | 0:57 |
| Hospital Ángeles del Pedregal | 1 | 0.29 | 14.9 | 0:25 |
| Hospital Durango | 1 | 0.29 | 25 | 0:43 |
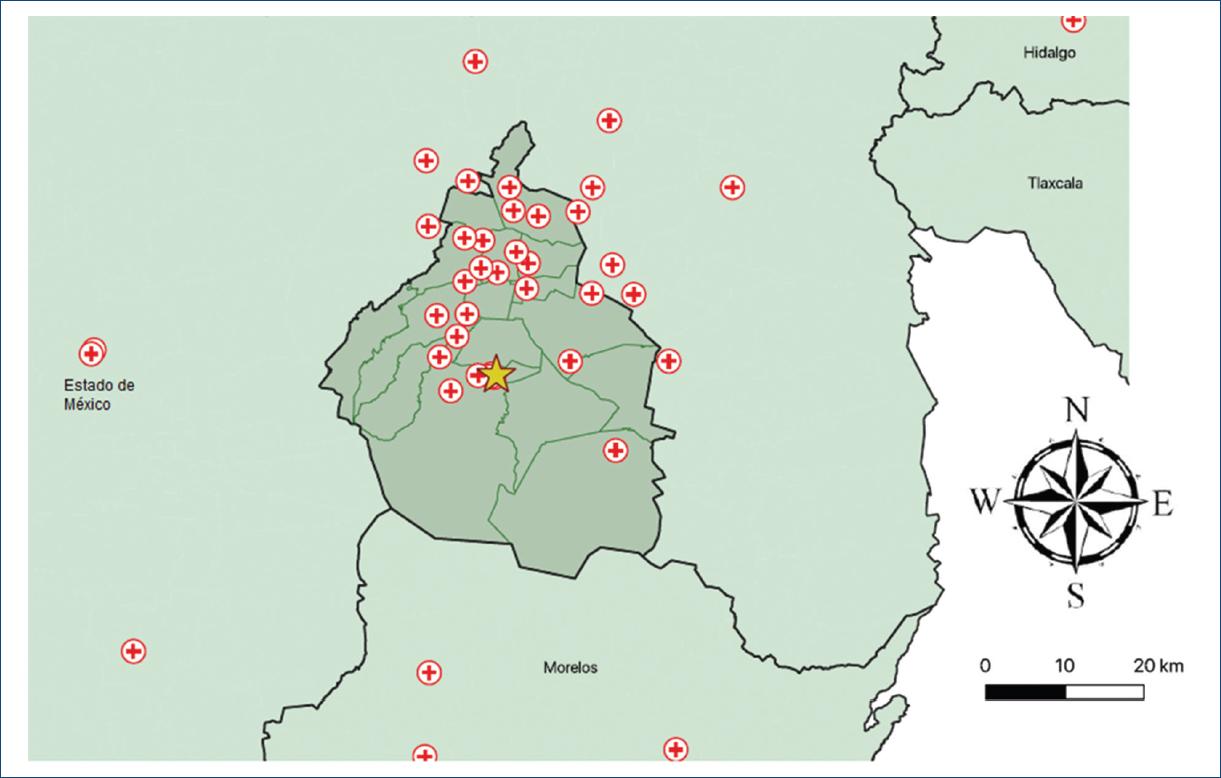
Figure 2 Regional map with a close-up to Mexico City and its metropolitan area indicating the places of first medical contact in patients with STEMI.
Regarding the clinical characteristics at admission, no overt differences were found between PS and PPCI groupseven though PPCI presented significantly higher blood pressure levels, these were not clinically relevant. Furthermore, a greater proportion of patients in the PPCI group presented a better KillipKimball score compared to the PS group (p < 0.001) (Table 3). Likewise, as shown in table 4, laboratory profiles between both groups were similar and most of the tests were among reference values, still some differences were found. For instance, C reactive protein was higher among PPCI patients, while cardiac dysfunction (NT-PROBNP) and damage (troponin I) markers were significantly higher among PS patients (p < 0.001).
Table 3 Clinical characteristics in patients with STEMI
| Variable | Total (n = 340) | PS (n = 166) | PPCI (n = 174) | p |
|---|---|---|---|---|
| n (%) | n (%) | n (%) | ||
| Killip-Kimball I | 181 (54.4) | 74 (45.1) | 108 (63.5) | 0.00 |
| Killip-Kimball II | 132 (39.6) | 78 (47.6) | 54 (31.8) | |
| Killip-Kimball III | 10 (3) | 5 (3.1) | 5 (2.9) | |
| Killip-Kimball IV | 10 (3) | 4.3 | 3 (1.8) | |
| Variable | Median (IQR) | Median (IQR) | Median (IQR) | p |
| Heart rate (bpm) | 75.5 (68.5-90) | 75.5 (70-90) | 75.5 (68-90 | 0.59 |
| Respiratory rate (bpm) | 18 (16-19) | 18 (16-19) | 18 (16-20) | 0.87 |
| Systolic pressure (mmHg) | 127 (114-147) | 126 (112-140) | 130 (117-150) | 0.01 |
| Diastolic pressure (mmHg) | 80 (70-90) | 76.5 (70-86) | 80 (70-93) | 0.00 |
| Pulse oximetry (%) | 92 (90-95) | 92 (90-95) | 92 (90-95) | 0.94 |
| TIMI score | 4 (2-5) | 4 (2-5) | 3.5 (2-5) | 0.36 |
| GRACE score | 125 (101-150) | 126 (106-153) | 123 (99-147) | 0.19 |
| CRUSADE score | 26 (18-35) | 27 (19-35) | 26 (18-37) | 0.72 |
PS: pharmacoinvasive strategy; PPCI: primary percutaneous coronary intervention; CABG: coronary artery bypass grafting; SD: standard deviation; IQR: interquartile range.
Table 4 Laboratory tests in patients with STEMI
| Total (n = 340) Median (IQR) | PS (n = 166) Median (IQR) | PPCI (n = 174) Median (IQR) | p | |
|---|---|---|---|---|
| Hemoglobin (g/L) | 15.6 (14.4-16.7) | 15.35 (14.4-16.3) | 15.8 (14.5-16.9) | 0.08 |
| Creatinine (mg/dL) | 1 (0.8-1.2) | 1 (0.8-1.2) | 1 (0.8-1.1) | 0.99 |
| Urea nitrogen (mg/dL) | 17 (14-23) | 18.1 (15-25) | 16.6 (14-21) | 0.04 |
| Na (mEq/L) | 136 (134-138) | 136 (134-138) | 136 (134-137) | 0.30 |
| C reactive protein (mg/L) | 6.9 (2.7-28.7) | 13.06 (4.26-46) | 4.5 (2-19) | 0.00 |
| Leukocytes (103/µL) | 11.7 (9.3-14.4) | 11.4 (9.3-14.7) | 11.9 (9.2-14.2) | 0.82 |
| NT-PROBNP (pg/mL) | 793.5 (222.5-3284.5) | 1445 (421-3643) | 389 (100.5-2644) | 0.00 |
| Troponin I (ng/mL) | 12.7 (0.9-52.8) | 35 (12-80) | 1.85 (0.3-14.9) | 0.00 |
| Maximum Troponin I (ng/mL) | 64 (24-80) | 67.9 (23.8-80) | 62.9 (26-80) | 0.46 |
| Glucose (mg/dL) | 162.5 (1278-238.5) | 150 (115-230) | 174 (136-246.8) | 0.00 |
| K (mEq/L) | 4.1 (3.8-4.4) | 4.1 (3.86-4.5) | 4.1 (3.8-4.4) | 0.07 |
| Cl (mEq/L) | 103 (100-105.52) | 103 (101-107) | 103 (100-105) | 0.30 |
| Glycated hemoglobin (%) | 6.1 (5.65-8.2) | 6.1 (5.6-7.6) | 6.1 (5.7-8.3) | 0.41 |
| Albumin (g/dL) | 3.6 (3.3-3.9) | 3.6 (3.4-4) | 3.7 (3.4-3.9) | 0.88 |
| Uric acid (mg/dL) | 6.7 (5.6-7.86) | 6.8 (5.8-8.1) | 6.4 (5.4-7.8) | 0.03 |
| Platelets (103/µL) | 217 (183-259) | 209.5 (177-257) | 221.5 (192-263) | 0.10 |
| Cholesterol (mg/dL) | 154.9 (130-188.9) | 153 (129.5-186) | 157 (131-189) | 0.36 |
| LDL Cholesterol (mg/dL) | 98.3 (75-121.8) | 99.9 (74.4-121) | 97.2 (75.8-122.6) | 0.72 |
| HDL Cholesterol (mg/dL) | 34.4 (29.7-40) | 34.3 (29.2-40.3) | 34.5 (30.7-40) | 0.33 |
| STH (mIU/L) | 1.4 (0.7-2.8) | 1.35 (0.9-2.8) | 1.4 (0.7-2.9) | 0.88 |
PS: pharmacoinvasive strategy; PPCI: primary percutaneous coronary intervention; SD: standard deviation; IQR: interquartile range; LDL: low density lipoprotein; HDL: high density lipoprotein; STH: stimulant thyroid hormone.
In relation to the time of first medical contact, it was 120 min (IQR: 60-225) for PS and 150 min for PPCI (IQR: 60-270), without significant differences (p = 0.11). Moreover, the mean total ischemic time was 347.5 min (IQR: 200-600) for PS versus 310 min (IQR: 205-557) for PPCI (p = 0.52). Furthermore, for PS patients the mean door-to-needle time was 54 min (IQR: 30-103), and the mean time for pharmacoinvasion was 1440 min (IQR: 600-2880), while among PPCI patients the time to door-to-device was 72.5 min (IQR: 60-95). Finally, hospital stay was similar with both strategies, with a mean time of 6 days, and an interquartile range of 3-9 days.
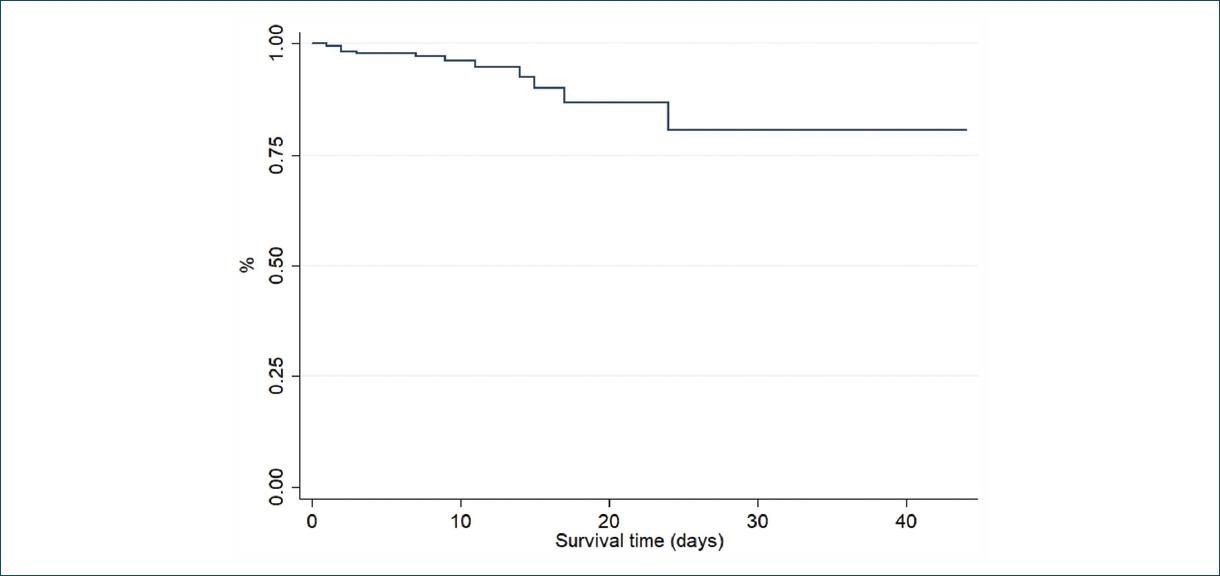
With regard to in-hospital mortality, a total of 20 patients died during the follow-up 11 patients (6.3%) who underwent PCI and nine (5.4%) who underwent PS (p = 0.82). A further subanalysis was made according to the time taken to pharmacoinvasion where the cutoff point was set according to the median value of 1440 min, without differences in mortality (6.56 vs. 5.73 %, p = 0.49). Moreover, 96% of patients from this cohort survived after 6 days of follow-up (Fig. 3). As to the type of post-infarction treatment, mean survival after 6 days was essentially the same between PPCI and PS patients (95% vs. 96%; p = 0.54) as shown in the KaplanMeier curves from figure 4.
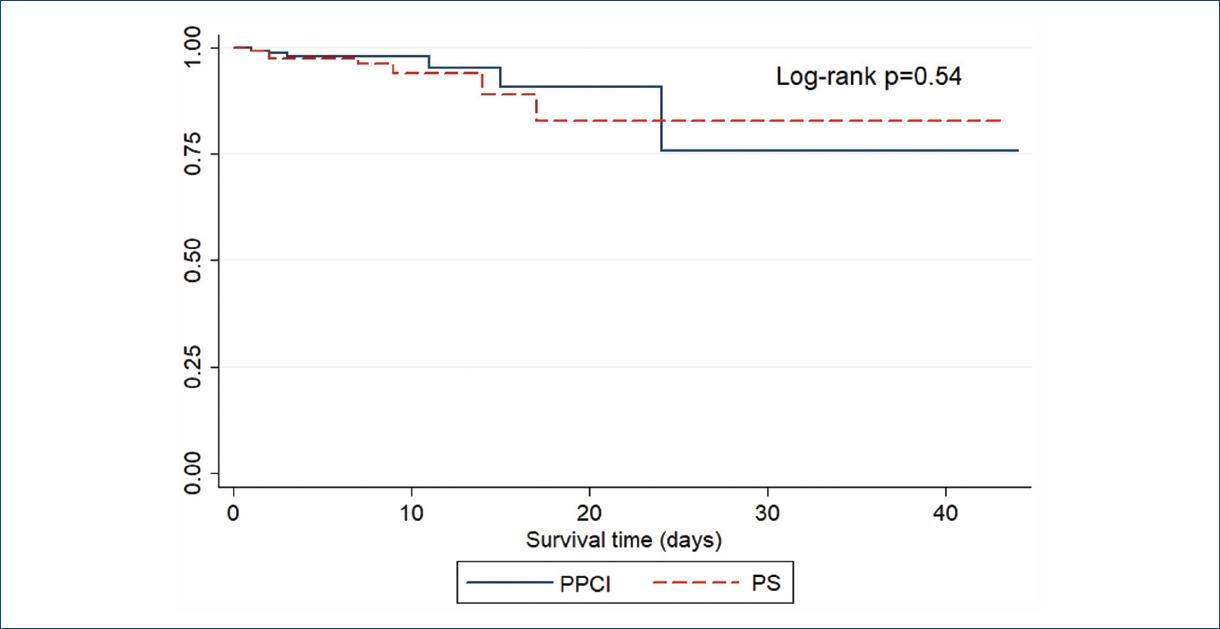
Figure 4 Survival according to the reperfusion strategy in patients with STEMI. PS: pharmacoinvasive strategy; PPCI: primary percutaneous coronary intervention.
Finally, variables associated with in-hospital mortality for both strategies were assessed through a Cox regression model, wherein blood glucose > 180 mg/dl (HR 3.73, IC 95% 1.02-13.56), total ischemic time > 420 min (HR 3.18, IC 95% 1.01-10.2), heart rate > 90 bpm (HR 5.46, IC 95% 1.69-17.59), Killip and Kimbal > II (HR 11.03, IC 95% 1.42-85.15), and left ventricular ejection fraction < 40% (HR 3.21, IC 95% 1.03-10.01) determined a greater mortality risk in the whole cohort (Table 5).
Table 5 Cox regression model for prediction of inhospital mortality in STEMI
| Variable | HR | SE | p | 95% CI |
|---|---|---|---|---|
| Male gender | 2.03 | 2.11 | 0.49 | 0.26-15.55 |
| Diabetes | 1.75 | 0.94 | 0.29 | 0.61-5.02 |
| Hypertension | 1.03 | 0.55 | 0.95 | 0.36-2.94 |
| Chronic kidney disease | 4.26 | 4.48 | 0.16 | 0.54-33.4 |
| CRP > 5 mg/L | 1.57 | 0.93 | 0.44 | 0.49-5.02 |
| Glucose >180 mg/dl | 3.73 | 2.45 | 0.04 | 1.02-13.56 |
| Total ischemia time > 420 min | 3.18 | 1.89 | 0.04 | 1.01-10.20 |
| First medical contact > 50 min | 1.28 | 0.98 | 0.74 | 0.28-5.75 |
| Heart rate > 90 bpm | 5.46 | 3.26 | 0.04 | 1.69-17.59 |
| Systolic pressure < 90 mmHg | 6.75 | 7.12 | 0.07 | 0.85-53.41 |
| Killip & Kimball > II | 11.03 | 11.5 | 0.02 | 1.42-85.15 |
| GRACE score > 140 | 3.04 | 1.84 | 0.06 | 0.93-9.98 |
| LVEF < 40% | 3.21 | 1.86 | 0.04 | 1.03-10.01 |
CRP: C reactive protein; LVEF: left ventricular ejection fraction; HR: hazard ratio; SE: standard error; 95% CI: 95% confidence interval.
Discussion
Optimal treatment for STEMI is timely reperfusion. The European Society of Cardiology (ESC) proposes that PCI is the treatment of choice; however, if the catheterization laboratory is beyond 2 h, the recommended treatment is PS2. Unfortunately, Mexico has many limitations, not only in urban infrastructure but also in social and economic development which keeps us away from achieving the proper timing for PCI. As an example of this, we received patients that came from 9 states and 60 different health facilities, being the most distant the Coatepec General Hospital (312 km and a mean estimated transfer time of 4 h and 23 min); moreover, it must be acknowledge that this network covers a big area with a median radius of 25.2 km. This pharmacoinvasive network covers a big area of the Mexican territory and constitutes one of the biggest in the world.
There was a higher proportion of men, diabetes mellitus, hypertension, and active smoking that seemed to surpass the national statistics, and it is striking that there was a lower prevalence of obesity compared to Mexico’s National Health and Nutrition Survey (ENSANUT in Spanish) 201225.
The time of the first medical contact was higher for PCI than for PS, however, there were no differences among the total ischemic time even though there was a slight increase in the latter one probably explained by the time of transfer.
The door-to-needle time was found 5 times higher than the time proposed by the ESC guidelines, which emphasizes the fact that training of the medical staff is necessary, because the lower the total ischemic time, the higher the proportion of rescued myocardial tissue2. On the other side, the door-to-device time was 72.5 min, which goes hand in hand with the established international guidelines. Furthermore, the time to pharmacoinvasion calculated in our study was 1440 min, equivalent to 1 day, the same time according to the recommendations of the ESC guidelines2,26.
It is important to acknowledge that our patients have longer hospital stays than usual, approximately 6 days, independently of the strategy. The ESC STEMI guidelines recommend a rapid home discharge, especially between the next 48-72 h, in low risk infarctions and when the assurance of a rehabilitation program and follow-up is reliable2.
Within the most relevant data of this study is the in-hospital mortality, represented by 11 patients taken to PPCI and 9 patients taken to PS, 6.3% and 5.4% (p = 0.82), respectively. The percentage of survival after STEMI was 94.1%, similar to what Sierra-Fragoso et al. reported in their study, where authors reported in-hospital mortality of 5.1% in PS and 5.3% in PCI22. On the other hand, the RENASCA registry reported a cardiovascular mortality of 14.9% higher than what we described27. Meanwhile, in other international registries, similar mortality has been reported among both strategies with a follow-up to 1 year after the index event, demonstrating that PS is a safe and effective method19,21,28,29.
Finally, the data obtained by the Cox regression model for predictors associated with in-hospital mortality were blood glucose >180 mg/dl, total ischemic time > 420 min, heart rate > 90 bpm, Killip-Kimball > II, and left ventricular ejection faction < 40%. This is relevant since we do not have tools to predict in-hospital mortality in our population.
Epidemiological transition has left its mark. Nowadays chronic diseases have a high prevalence and most of them will develop an acute myocardial infarction as a final outcome. Mexico has up to 3 times more in-hospital mortality than the rest of the countries belonging to the Organization for Economic Cooperation and Development (OECD) due to the lack of strategies that does not allow optimal access to medical services and proper timing for treatment. In the national context, the PS headed by the National Heart Institute has proven to be equally effective than PCI at least for in-hospital mortality, which by now will improve cardiovascular outcomes in the future. This model should keep growing and spreading to the health care centers that have the capacity to perform PCI.











 text new page (beta)
text new page (beta)