Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Agrociencia
On-line version ISSN 2521-9766Print version ISSN 1405-3195
Agrociencia vol.52 n.8 Texcoco Nov./Dec. 2018
Crop Science
Vascularization and starch grains during the morphogenesis of peach floral buds on different flowering date
1Universidad Intercultural del Estado de México. Libramiento Francisco Villa S/N, Centro, San Felipe del Progreso, Estado de México. 50640.
2Botánica. Unidad de Microscopía Electrónica, Colegio de Postgraduados. 56230. Montecillo, Estado de México.
3Edafología. Campus Montecillo. Colegio de Postgraduados. 56230. Montecillo, Estado de México.
4Campo Experimental Valle de México. INIFAP. Carretera los Reyes-Texcoco, Km 13.5. Coatlinchán, Texcoco, Estado de México. 56250.
The anatomical aspects of vascularization and distribution of starch during the morphogenesis of the floral bud are unknown for Prunus persica (L.), and this knowledge helps to predict the degree of vascularization and susceptibility of the bud to cold temperatures. The aim of the present study was to compare the differentiation of the xylem and the distribution of starch in flower buds, from the phenological stage (PS) “dormancy” to “visible stamens” in the Puebla, CP Precoz and Zacatecas cultivars (early, intermediate and late flowering). Sampling was carried out completely at random (two flower buds of the middle part of five mixed twigs (one year old), from 10 trees (per cultivar) from October 29th (autumn) to March 19th (winter), 2012. The tissues were fixed in a formaldehyde -alcohol-water (FAA) solution, and processed in Paraplast. Serial sections were stained using Safranin O and Fast green FCF. The anatomical changes observed were consistent for each PS studied, regardless of the cultivar and flowering date. At the start of the PS “dormancy” there was no vascular connection between the pedicel and the ovary. In the PS “swollen bud,” the xylem reached the base of the ovary and starch was found in the ovary and anthers. In the PS “buds with visible calyx,” the xylem reached the stigma and the starch remained in the ovary and anthers. In the PS “pink tip,” the anther matured and presented no starch. In the PS “visible stamens,” the stigma and the ovule matured and starch was almost absent. The presence of starch preceded the differentiation of the xylem. The vascularization in the vegetative whorls was delayed in the intermediate and late cultivars, in contrast with the early cultivar.
Keywords: Prunus persica; vascularization; starch; anthers; dormant buds
Los aspectos anatómicos de la vascularización y distribución de almidón durante la morfogénesis de la yema floral no se conocen en Prunus persica (L.), y este conocimiento ayuda a predecir el grado de vascularización y susceptibilidad de la yema al frío. El objetivo del presente estudio fue comparar la diferenciación de xilema y la distribución de almidón, en yemas florales, desde el estado fenológico (EF) “reposo” a “estambres visibles”, en los cultivares Puebla, CP Precoz y Zacatecas (floración precoz, intermedia y tardía). El muestreo fue completamente al azar (dos yemas florales de la parte media de cinco ramillas mixtas (de un año), de 10 árboles (por cultivar) del 29 de octubre (otoño) al 19 marzo (invierno) de 2012. Los tejidos se fijaron en solución de formaldehído-alcohol-agua (FAA) y procesados en Paraplast. Cortes seriados se tiñeron con Safranina O y Verde fijo FCF. Los cambios anatómicos observados fueron consistentes para cada EF estudiado, independiente del cultivar y fecha de floración. Al inicio del EF “reposo” no hubo conexión vascular entre el pedicelo y el ovario. En el EF “yema hinchada” el xilema llegó a la base del ovario y el almidón se observó en el ovario y anteras. En el EF “yemas con cáliz visible” el xilema llegó al estigma y el almidón persistió en el ovario y anteras. En el EF “punta rosada” la antera maduró y no presento almidón. En EF “estambres visibles” el estigma y el óvulo maduraron y el almidón fue casi ausente. La presencia de almidón precedió a la diferenciación del xilema. La vascularización en los verticilos vegetativos se retrasó en los cultivares intermedio y tardío en contraste con el cultivar temprano.
Palabras clave: Prunus persica; vascularización; almidón; anteras; yemas en reposo
Introduction
Deciduous fruit trees characteristically undergo a period of low hormonal and metabolic activity in the meristems, which is called “dormancy.” This period is an adaptation to cold winters (Horvath, 2009), which helps it to develop resistance mechanisms against damages from low temperatures (Elle and Sauter, 2000; Campoy et al., 2011). The acclimatization to low temperatures of flower buds in several species of Prunus L. is determined by genetic (Arora and Wisniewski, 1994; Yamane et al., 2011), physiological, biochemical (González-Rossia et al., 2008; Die and Rowland, 2014), morphological and anatomical (Ashworth, 1982; 1984) factors. Low temperatures are required to satisfy the physiological needs of the flower buds (Andreini et al., 2014). A deficiency of chilling hours causes deficient flowering, malformations in the vegetative and reproductive whorls (Nava et al., 2009; Reig et al., 2013). At the end of the “dormancy” stage, the flower bud of the peach tree has biochemical, physiological and anatomical changes related with the establishment of the vascular connection, which increases the risk of freezing (Ashworth, 1982; 1984; Wisniewski and Davis, 1989), although there is no exact knowledge regarding the exact moment and bud tissues in which the differentiation of the xylem, the tissue responsible for the accumulation of water in tissues, takes place. While “dormant,” enzyme activity increases and the concentration of sugars decreases, which increases the susceptibility to chilling of peach flower buds (Paloen, 1999) and the concentration of starch decreases in peach and pear trees (Pyrus communis L.) (Ito et al., 2002 and González-Rossia et al., 2008), yet there is no knowledge on the sequence of these events in relation to the phenological stage and date of flowering. Regarding the development of anthers in apricot (Prunus armeniaca L.), the change in color of the anthers is related to meiosis and to the end of the buds “dormant” stage (Julian et al., 2014). Reinoso et al. (2002) described the anatomical development of peach flower buds from a botanical point of view, but did not review the differentiation of the vascular tissue or the presence of starch grains. The xylem is responsible for conducting water to the buds and starch is an important source of energy for the morphogenesis; the detailed description of these events in peach trees is an efficient tool in agronomic management and reduction of damages caused by frost. The aim of the present study was to characterize the development of the xylem and the distribution of starch grains in five phenological stages (PS), from PS “dormant” to the PS “visible stamens” in three P. persica cultivars with different flowering dates. Assuming that the differentiation of the xylem is related to a particular phenological stage of the flower bud, this would help predict the degree of vascularization and susceptibility to low temperatures in cultivars with different flowering dates.
Materials and methods
Plant material and experimental site
The Prunus persica cultivars used and dates of anthesis were as follows: Puebla (January 12th, early), CP Precoz (February 20th, intermediate) and Zacatecas (March 17th, late) during the winter of 2011-2012. The five-year old trees were grafted onto creole peach trees. The orchard is located in the Campo Experimental Valle de México (CEVAMEX), el Horno, Chapingo, Texcoco, Estado de México, (19° 29’ 17” N y 98° 53’ 41” W, altitude 2280 m), with Mollic fluvisol soil (Govaerts et al., 2008) a temperate subhumid climate C (W0), temperature and annual rainfall of 15.5 °C and 644 mm (García, 1987).). The trees were structured according to the Modified Tatura method and the orchard was planted in the milpa intercropped with fruit trees (MIFT) system (Santiago-Mejía et al., 2015). Cultural practices were: fertilization 60-1560 (g of N-P2O5-K2O) and 3 kg of poultry manure per tree in April (1/3; 1-1-0 de N, P, K, poultry manure) and August (2/3; 0-0-1 de N, P, K, poultry manure); irrigation by gravity without limits of water.
Sampling and experimental design
Sampling was carried out completely at random and two central flower buds were taken, with five mixed one-year-old twigs, from the middle section of the canopies of 10 trees to be planted. In order to cover the phenology of the three cultivars, three sample collections were carried out: October 29th; November 18th; December 08th and 28th of 2011 and January 17th and 30th; February 06th, 23rd and 26th, and March 01st, 17th y 21st of 2012. The anatomic characteristics reviewed were qualitative, and therefore a statistical analysis was not carried out. The results are representative of all the buds analyzed in each phenological stage.
Phenological stages and anatomical stages
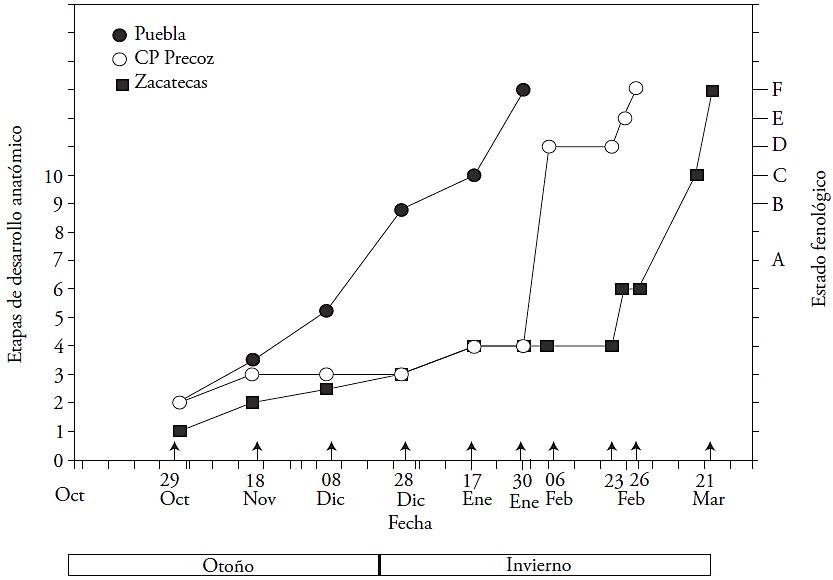
The flower buds were classified in the PS’s according to Baggiolini’s nomenclature (1952): A: “dormant bud”; B: “swollen bud”; C: “visible calyx”; D: “pink tip”; and E: “visible stamen.” For each PS, anatomical changes were described, which were grouped in “anatomical stages,” according to the nomenclature suggested by Reinoso et al. (2002). Seven anatomical stages (17) were described in PS “A”, two anatomical stages (8 and 9) in PS “B”, one anatomical stage (10) in PS “C”, and in PS “D” and ”F” only morphological changes are described, since they refer to the emergence of the anthers of the floral bud and the anthesis or floral aperture. The morphological characteristics of these phenological stages are illustrated in Figure 1.
Microtechnic for buds anatomy
The external morphology of the buds was documented in each phenological stage using a Moticam 2000® camera (Motic, China), attached to a stereoscopic microscope (Nikon SMZ800®, Japan). The buds were fixed in FAA (10 % formalin, 50 % ethanol, 5 % glacial acetic acid, 35 % deionized water) for 48 h and processes for their inclusion in Paraplast. Lengthwise cuts (rotary microtome, American Optical, USA) were dyed using Safranin O and Fast Green FCF. Safranin O is a cationic (basic) colorant that dyes lignified walls and nucleic acids red, which is why it was chosen to identify the mature xylem vessels. Fast Green FCF is an anionic (acid) green dye that stains cellulose, cytoplasm, sieve tube elements, parenchyma and meristematic cells, that in combination with Safranin provide a metachromatic stain (Baker, 1966). The anatomy of the buds was studied under a Axioskop 2 plus® microscope (Carl Zeiss, Germany) and an Axiocam 5 MRc camera (Carl Zeiss, Germany).
Vascularization and distribution of starch grains
The appearance of xylem vessels in the receptacle, calyx, corolla, anthers and gynoecium was registered using a Axioskop 2 plus® microscope (Carl Zeiss; Germany) in a bright field. The starch grains were viewed with epifluorescence (excitation BP 450-490 nm and emission LP 515 nm), and images were captured using the Axiocam 5 MRc camera (Carl Zeiss, Germany).
Results and discussion
Phenology of flowering
The first cultivar to leave the stage of “dormancy” (A) was Puebla (late December), followed by CP Precoz (late January), and finally, Zacatecas (late February). The development of the bud was fast in cv. Puebla, and by January, it was showing visible sepals (PS “C”) (Figure 2). The development of the bud for cv. CP Precoz was slower, since the calyx became visible (PS “D”) after it did in cv. Puebla. The slowest cultivar was Zacatecas; the bud remained dormant (PS “A”) until February, and then it developed quickly, displaying the pink tip (PS“D”) in March.
Vascularization of the flower bud
The differentiation of the xylem in the flower bud was acropetal (the maturation of vessel elements began at the base and moved towards the bud) in all three cultivars. During the dormant stage (A1 to A5), the bud of the three cultivars is not vascularized. The cv. Puebla was the first (December 28th) to present xylem in the receptacle, calyx and corolla (A6 and A7) (Figures 2 and 3). In contrast, the lack of vascularization was prolonged in CP Precoz (January 30th) and in Zacatecas (February 23rd) (Figures 2 and 3A). Bud vascularization begun after 2 months in cv. Puebla, after 3 months in cv. CP Precoz and after 4 months in cv. Zacatecas (Figures 2 and 3B). Only vascular procambial cells were observed in the receptacle (Figure 4A). In stages A3, A 4 and A5, the xylem was observed in the basal scales (Figure 3A). The vascularization of anthers and ovary took place first in cv. Puebla (swollen bud B8 and B9) on January 17th, and the latest (March 14th) was Zacatecas (Figures 2 and 3B). The vascularization of the style presented the greatest variation between cultivars: in cv. Puebla, it occurred on day 15 (January 17th to 30th), in CP Precoz, on day 27 (January 30th to February 26th), and in cv. Zacatecas, on day 4 (from March 17th to 21st).
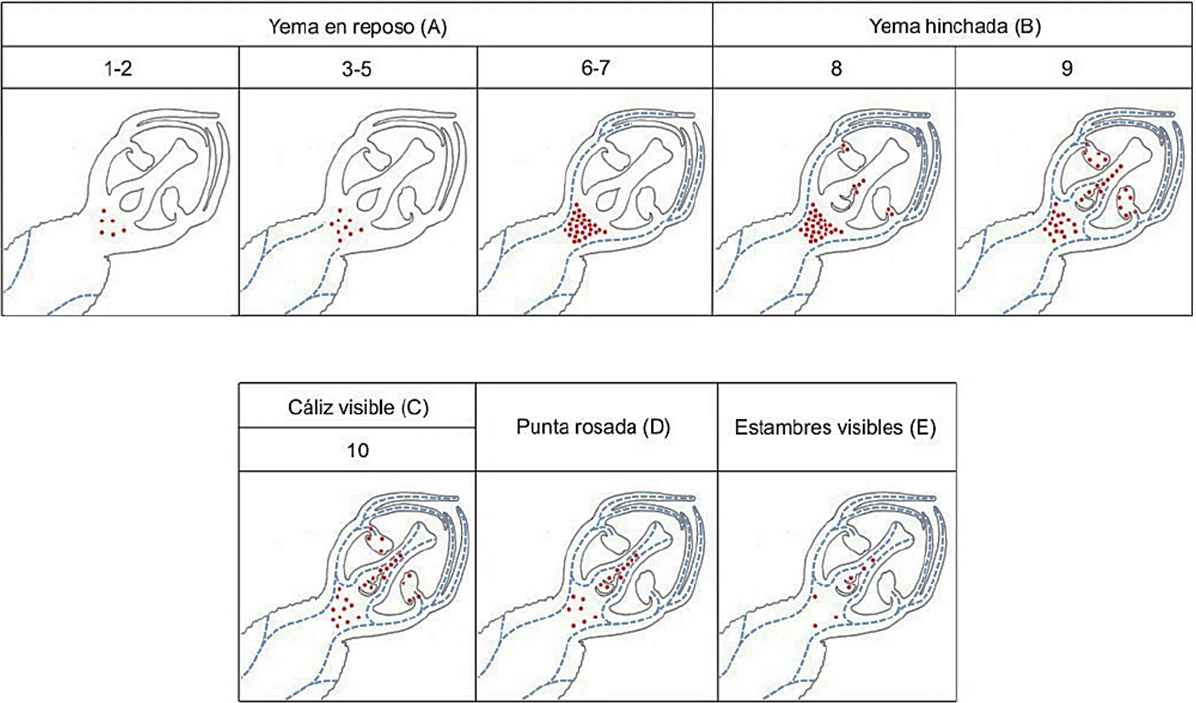
Figure 3 Distribution and abundance of starch grains and xylem in the vegetative and reproductive whorls of the flower bud in the phenological stages: dormant bud A); swollen bud B); visible calyx C); pink tip D); visible calyx E). Red dots indicate starch grains and blue dotted lines indicate xylem vessels.
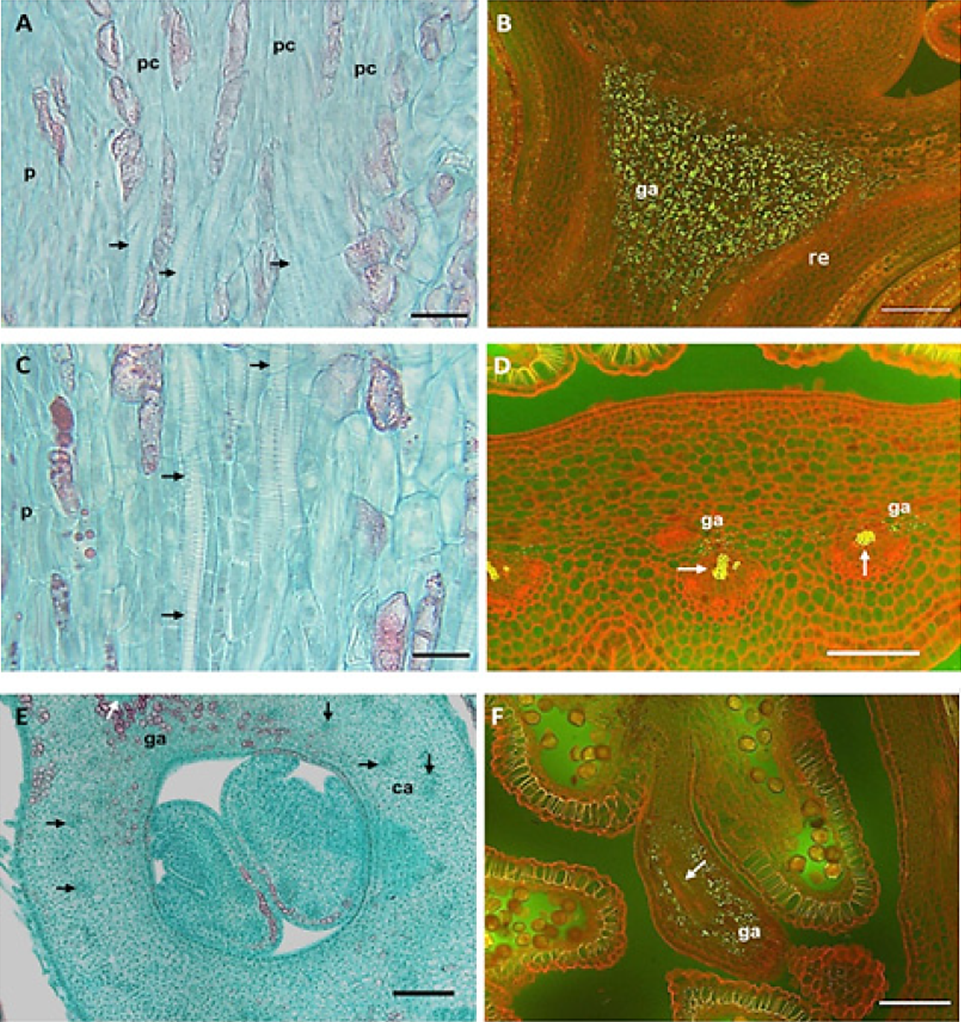
Figure 4 Anatomy of the vascularization and presence of starch grains in peach flower buds. A) Procambium in the receptacle, absence of xylem at dormancy (stages 3 to 5, cv. Zacatecas); B) starch grains at the receptacle of dormant bud (stages 6 to 7, cv. Puebla); C) xylem vessels in the vegetative whorls of swollen bud (stage 8, cv CP Precoz); D) xylem and starch in the base of anthers of swollen bud (stage 8, CP Precoz); E) xylem on the carpel wall, visible calyx bud (stage 10, cv. Puebla); F) vessels and starch grains in the anther (stage 10, cv. Puebla). A, C and E) bright field; B, D and F) epifluorescence. Arrows indicate vascular bundles (ga: starch grains; re: receptacle; pc: procambium; ca: carpel). Bars: A and C = 20 µm; B, D and F = 100 µm; E = 500 µm.
Vascularization was reported at the end of the stage of dormancy for P. armeniaca, P. avium, P. ceratus, P. saliciana and P. sargentii (Ashworth, 1984; Reinoso et al., 2002). However, our study is the first to describe the advancement of vascularization in parts of the bud, as well as the comparison between cultivars with different dates of flowering or precociousness.
The vascularization of the vegetative whorls was related to an increase of starch in the receptacle (Figure 3) in all three cultivars. The starch grains were visible with epifluorescence (Figure 4B) and the elements of the xylem vessel presented thickening and scalariform perforation plates (Figure 4C). According to Ashworth (1984), in apricots, the vascular connection between the twig and all the bud occurs at the end of the dormant stage. In contrast, in the Mexican cv. of peach trees, this event was gradual, as shown in Figure 3: the xylem appears first in the receptacle (A-5), then in the calyx and the corolla (A7), and later the xylem extends to the anthers in the swollen bud (B-8), it continued towards the ovary (B-9) and reached the style and the stigma up to the visible calyx stage (C-10).
Reinoso et al. (2002) only mentioned the presence of vascular tissue in all the whorls at the end of the dormant stage without including the anatomical and phenological details. Figure 4D shows vascular bundles and starch grains in the base of anthers of swollen buds (B-8). The ovary wall was vascularized (B-9) towards the end of the swollen bud (Figure 3). These events took place first (early January) in cv. Puebla, then in CP Precoz (February), and finally, in cv. Zacatecas (early March). In contrast, in apricot this took place before, at the end of the stage of dormancy (Julian et al., 2011). In the visible calyx stage (C-10) the xylem bundles moved to the style until they reached the stigma (Figure 3). The filaments of the anthers presented a central amphicribral bundle. The vascular bundles were collateral and abundant in the carpel walls (Figure 4 E). The complete vascularization of the pistil (C10) first took place in cv. Puebla (mid-January), then in cv. CP Precoz (early February), and finally, in Zacatecas (mid-March).
The latest event was the vascularization of the ovule, which explains why the primordium of the seed is the structure that requires protection from low temperatures, for reproductive reasons. During the year of the experiment, frosts took place when the flower buds were dormant (Santiago-Mejía et al., 2015) and our study showed the absence of vascular connections in this stage.
Distribution of starch grains
The presence and distribution of the starch grains was related to the differentiation of the xylem and development of the floral whorls. In cv. Puebla, the starch grains first appeared at the base of the receptacle (December 08th) and then they increased in number when the anthers began developing (Figure 4B). In cv. CP Precoz, this event took place on February 23rd, and later the starch appeared on the base of the anthers, while the pollen grains formed the generative and vegetative cell (Figure 5G) (Table 1). In swollen bud (B-8 and B-9), starch grains appears in the style and the anthers; which coincided with the differentiation of the xylem in the ovary, the beginning of the nucellus and the formation of the pollen wall (Table 1, Figure 5I). The xylem differentiation at the stamens and during the formation of the polen grains in cv. Puebla and CP Precoz was similar to observations in P. armeniaca, although the presence of starch in the intermediate cells occurred before the formation of microspores (Julian et al., 2011).
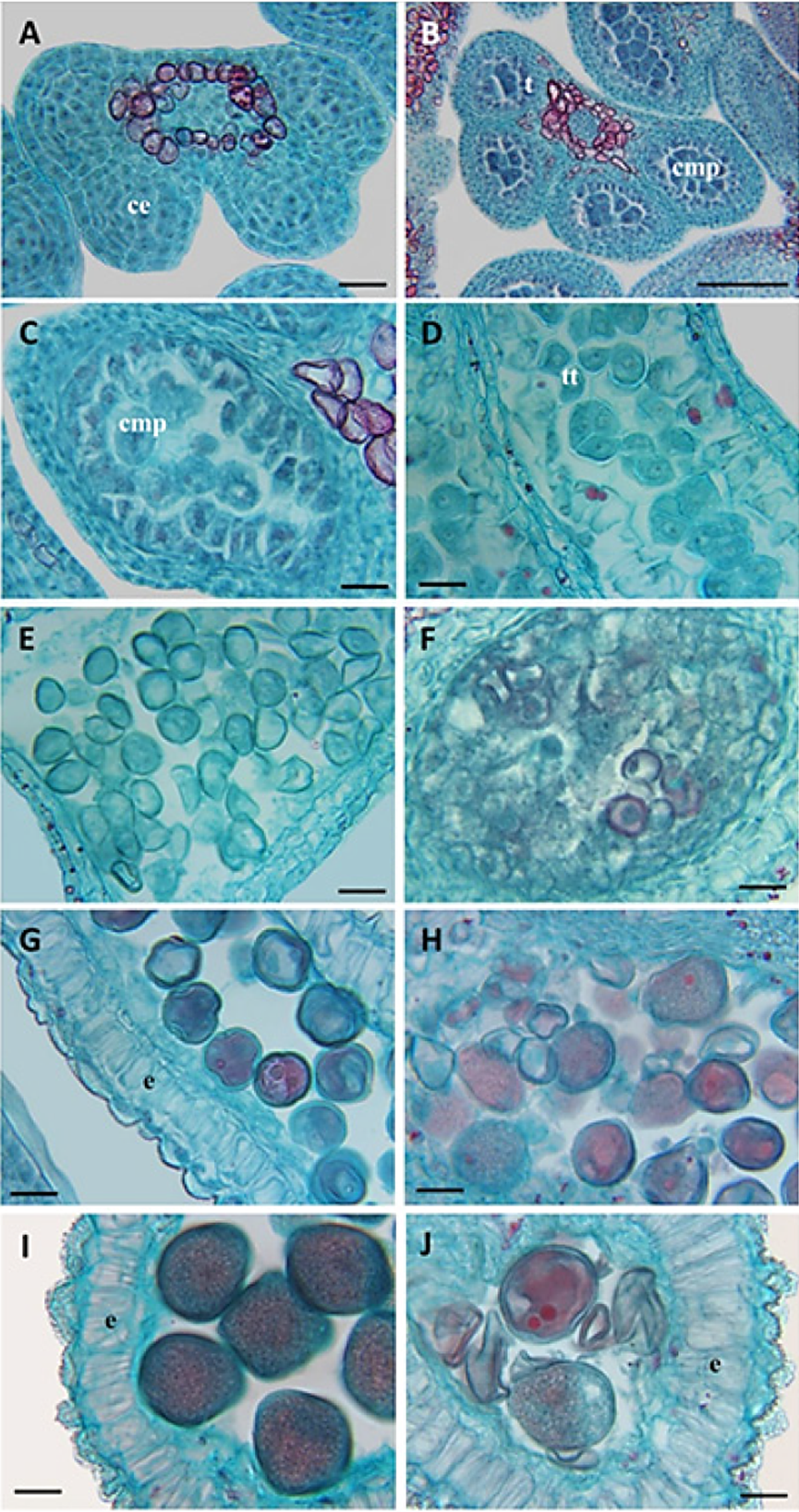
Figure 5 Development of the anther in three Prunus persica cultivars A) Tetrasporangiated anther with sporogenous cells (2n) cv. Puebla; B) pollen mother cells (PMC) and differentiated tapetum cv. CP Precoz; C) meiosis of the pollen mother cells (1n) Zacatecas; D) formation of tetrads with a tetrahedral arrangement cv. Puebla; E) release of microspores (1n) cv. CP Precoz; F) defect in the release of microsporas and abortion cv. Zacatecas; G) elongation of the endothecium and first mitosis of the pollen grain cv. Puebla; H) abortion of pollen grains cv. Zacatecas; I) thickening of the endothecium and the pollen wall cv. Puebla; J) collapsing of pollen grains, lack of thickening of the pollen wall cv. Zacatecas. ce: sporogenous cells; e: endothecium; cmp: pollen mother cells; tt: tetrades. Bars: A, C-J = 20 µm, B = 100µm.
Table 1 Anatomical characterization of the phenological stages of the floral peach buds (Prunus pérsica), from “dormant bud” (A) to “visible stamens” (E), observed in the cultivars of Puebla, CP Precoz and Zacatecas.
| Estado fenológico† | Etapa anatómica ¶ | Caracterización Anatómica |
| A: Yema en “reposo” | 1 | No existe vascularización de la yema (Figura 3 A ), presencia de algunos granos de almidón en la base del receptáculo. Anteras jóvenes tretra-esporangiada con dos lóculos en cada uno de los lóbulos unidos por el tejido conectivo (Figura 5A). En el gineceo solo se observa la cavidad locular. |
| 2 | No hay vascularización de la yema. Epidermis diferenciada en la pared del microesporangio y en el interior células esporógenas grandes con citoplasma denso, núcleos grandes y nucléolos prominentes (Figura 5B). Algunos granos de almidón en el receptáculo (Figura 3 A 1-2). | |
| 3 | No hay vascularización de la yema, se identifican algunos elementos de vaso en la base del receptáculo, y aumentan los granos de almidón en la base del receptáculo (Figura 3 A 3-5). Estratos identificables del microesporangio, epidermis, endotecio, tres estratos de células parietales, y el tapete. Inicia la diferenciación de las células madre de polen (CMP, 2n) (Figura 5 C). | |
| 4 | Presencia de tejido vascular llegando al centro del receptáculo (Figura 3 A 3-5). La pared interna del microesporangio se diferencia en tapete, con células de citoplasma denso que rodean a las CMP en desarrollo. Inicio de protuberancias meristemáticas del óvulo en la cavidad locular. | |
| 5 | Tejido vascular en el receptáculo (Figura 3A 3-5). Células del tapete con grandes vacuolas, algunas binucleadas. Las CMP agrandadas comienzan a separarse una de otra. | |
| 6 | El tejido vascular se comienza a diferenciar en los verticilos vegetativos, aumentan los granos de almidón en el receptáculo y éste crece arriba del ovario (flor periginia) (Figura 3 C), En el ovario se diferencia el obturador sobre cada óvulo (sólo ‘Puebla’). Meiosis asincrónica, algunas CMP inician la meiosis (Figura 5 C), pero en otras anteras las CMP forman tétradas (1n) (Figura 5 D). | |
| 7 | El tejido vascular avanza hacia los verticilos de cáliz y corola (Figura 3 A 6-7). Las microsporas (1n) recién se liberan de las tétradas (Figura 5 E). El tapete degenera gradualmente, las células parietales comienzan a comprimir al endotecio. | |
| B: Yema hinchada | 8 | Los granos de almidón aparecen en la base de las anteras y el tejido vascular se extiende a las anteras (Figura 4 F) y aparece almidón cerca del óvulo (Figura 3 B 8). Expansión de las células del endotecio y granos de polen en mitosis (Fig. 5 G), una célula generativa y una célula vegetativa. Se observan nucela del óvulo y obturador en CP Precoz y Zacatecas.. |
| 9 | Aumentan los granos de almidón en el pistilo y anteras, pero disminuyen en la base del receptáculo (Figura 3 B-9). La vascularización abarca el ovario (Figura 3 E). La nucela de los óvulos es prominente. Estratos intermedios de la antera comprimidos y los granos de polen forman una exina gruesa (Figura 5 I) | |
| C: Cáliz visible | 10 | Los granos de almidón disminuyen en toda la yema, pero su vascularización es completa, el xilema avanza hasta el estigma (Figura 3 C-10). En el ovario, los óvulos continúan su desarrollo. La pared del endotecio desarrolla bandas de engrosamiento de pared secundaria en la parte anticlinal y tangencial. En el estigma se inicia la diferenciación de una epidermis con papilas secretoras. |
| D: Punta Rosada | Granos de almidón ausente en las anteras y persistente en el pistilo (Figura 3 D). Grano de polen maduro. El ovulo continua su desarrollo. La corola se observa como un círculo rosa pequeño en el ápice de la yema (Figura 1 Punta rosada), los sépalos adquirieren su color verde-marrón. Adelgazamiento en el estomio. | |
| E: Estambres visibles | Granos de almidón muy escasos en el pistilo y base del receptáculo (Figura 3 E). Los pétalos duplican su longitud, las flores jóvenes se abren parcialmente, los estambres se hacen visibles hacia el exterior de la flor (Figura 1 E), el estomio se rompe. El estigma ha desarrollado una cubierta con papilas secretoras. |
No se incluye el estado fenológico de antesis por no presentar cambios anatómicos relevantes para este estudio.
† La nomenclatura usada de los estados fenológicos es según Baggliolini (1952).
¶ La nomenclatura usada de las etapas anatómicas es según Reinoso et al. (2002).
In the visible calyx stage (C-10), of the three cultivars, there was a reduction of starch grains in the base of the bud and an increase in the pistil, in coordination with the differentiation of the xylem in the style. However, in pink tip (D), the starch disappeared from the anthers, although it persisted in the ovary (Figure 4). This pattern was similar to the one observed in olive inflorescences (De la Rosa and Rallo, 2000). The changes in the partition of starch during the development of the floral buds have been shown in Prunus persica (Bonhomme et al., 2005; González-Rossia et al., 2008), Rubus idaeus (Palonen, 1999), Pyrus pyrifolia (Ito et al., 2002), Populus x canadensis (Elle and Sauter, 2000) and Olea europea (De la Rosa and Rallo, 2000), although our report is the first on the histological location of starch in the flower bud related to the morphogenesis and differentiation of the xylem in peach.
Anatomic characterization with emphasis on anther development
The anatomical changes observed in the bud in the phonological stage of dormancy (A) were grouped into seven anatomical stages (1-7), which are shown in Table 1. These events took place with differences in time between cultivars (Figure 2). The anatomical changes observed during the first four stages of dormancy (A-1 to A-4), occurred slowly in the flower bud of the cv Zacatecas (4 months), less slowly in cv. CP Precoz (3 months), and fast in cv. Puebla in 20 d (Figure 2 and Table 1).
In the three cultivars, during the “dormancy” (A), the early development of the pollen was normal and similar; the microsporangia presented sporogenous cells (2n) (Table 1 and Figure 5A); in stage A-2 the pollen mother cells (2n) were differentiated (Table 1); in stages A-3 and A-4 the tapetum was prominent (Table 1 and Figure 5C); in stage A-6 the pollen mother cells (PMC) were observed in meiosis and the microspores (1n) were organized in a tetrahedral arrangement (Figure 5D). In swollen bud (B8 and B-9), of the cv Puebla and CP Precoz, the microspores were separated into young pollen grains (1n) (Table 1 and Figure 5E), although in cv. Zacatecas, the microspores did not separate, and some were obliterated and disintegrated (Figure 5F). This meant a defect in the formation of the pollen grain in cv. Zacatecas. Later, cv Puebla and CP Precoz continued developing normally, although with differences in time; cv Puebla was the first to present the mitosis of the young pollen grain and the thickening of the pollen wall (Figure 5G), and cv. Zacatecas displayed obliterated pollen grains with a deficient thickening of the exine (Figure 5H).
In the following stage, visible calyx (10-C), the anticlinal walls of the endothecium were normally lignified in the three cultivars, although the maturation of the pollen grain only appeared in cv. Puebla and CP Precoz (Figure 5I).
In the pink tip stage (D), several microspores stopped developing and aborted in cv. Zacatecas (Figure 5H). The cv. Zacatecas presented the majority of abortive pollen grains with defects in the exine (Figure 5J).
Pollen abnormalities and abortions have been observed when the requirements of chilling have not been met (Bonhomme et al., 2005; Nava et al., 2009; Reig et al., 2013). In the context of global warming, temperate fruit trees of will face reduction in chilling hours, problems during “dormancy” related to abortion and reduction in the flow of carbohydrates towards the buds (Campoy et al., 2011; Marafon et al., 2011). According to the method by Richardson et al. (1974), cv. Zacatecas requires 550 Chill Units (CU), CP Precoz requires 400 CU, and Puebla, 350 CU (Santiago-Mejía et al., 2015). The average chilling accumulated between November 17th and March 31st in the area under study was below 550 CU (Santiago-Mejía et al., 2015). In Natural conditions cv. Zacatecas is at higher latitude and accumulate more chilling hours (Rumayor et al., 2009). The chilling deficiency causes anomalies in the male gametophyte, such as; the abortion of pollen, reduction in the polen production and germination problems (Nava et al., 2009), as well as an abnormal floral opening (Bonhomme et al., 2005). This explains the anomalies observed in the development of pollen in Zacatecas.
Conclusions
The differentiation of the xylem in the peach flower bud is acropetal (the maturation of the vessel elements begins at the base and moves towards the tip of the bud) and gradual. There are no signs of vascularization during the early dormancy in the three cultivars under study (Puebla, CP Precoz and Zacatecas). The presence of starch preceded the appearance of the xylem. The vascularization of the vegetative whorls began at the late dormancy: after two months for cv. Puebla, after three months for cv. CP Precoz, and after four and a half months for cv. Zacatecas. Vascularization moves up to the ovary in the swollen bud, and continues towards the stigma until calyx was visible. The vascularization of the style was the event that presented the greatest variation between cultivars: in cv. Puebla, 15 days; in CP Precoz, 27 days; and in cv. Zacatecas, 4 days. Vascularization was postponed according with the precociousness of the cultivar. Late vascularization of the female, part would be a strategy to protect its reproductive function in the presence of frost. This information can be used in programs of agronomic management and reduce damages caused by frost in peach trees.
Literatura citada
Andreini, L., I. García de Cortázar-Atauri, I. Chuine, R. Viti, S. Bartolini, D. Ruiz, J.A. Campoy, J. M. Legave, J. Audergon, and P. Bertuzzi. 2014. Understanding dormancy release in apricot flower buds (Prunus armeniaca L.) using several process-based phenological models. Agric. Forest Meteorol. 184: 210-219. [ Links ]
Arora, R., and M. Wisniewski. 1994. Cold acclimation in genetically related (sibling) deciduous and evergreen peach, Prunus persica. II. A 60-kilodalton bark protein in cold acclimated tissues of peach is heat-stable and related to the dehydrin family of proteins. Plant Physiol. 105: 95-101. [ Links ]
Ashworth, E. N. 1982. Properties of peach flower buds which facilitate supercooling. Plant Physiol. 70: 1475-1479. [ Links ]
Ashworth, E. N. 1984. Xylem development in Prunus flower buds and the relationship to deep supercooling. Plant Physiol. 74: 862-865. [ Links ]
Baggiolini M., 1952. Les stades repères de l’abricotier. Revue romande d’Agriculture et d’Arboriculture 8: 28-30 [ Links ]
Baker, J. R. 1966. Cytological Technique. The Principles Underlying Routine Methods. 5th Edition. London, Mehuen & Co. 321p. [ Links ]
Campoy, J. A., D. Ruiz, and J. Egea. 2011. Dormancy in temperate fruit trees in a global warming context: A review. Sci. Hort. 130:357-372. [ Links ]
Cortés, F., J. I., A. Turrent F., P. Díaz V., E. Hernández R., R. Mendoza R., y E. Aceves. 2005. Manual para el establecimiento y manejo del sistema milpa intercalada con árboles frutales (MIAF) en laderas. (SAGARPA, Ed.) México: Colegio de Postgraduados. < sagarpa.gob.mx/desarrolloRural/Documents/fichasCOUSSA/03%20MILPA%20INTERCALADA%20CON%20FRUTALES.pdf > (Consulta: abril 2014). [ Links ]
De la Rosa, R., and L. Rallo. 2000. Olive floral bud growth and starch content during winter rest and spring budbreak. HortScience. 35: 1223-1227. [ Links ]
Die, J. and L. Rowland. 2014. Elucidating cold acclimation pathway in blueberry by transcriptome profiling. Environ. Exper. Bot. 106: 87-98. [ Links ]
Elle, D., and J. Sauter. 2000. Seasonal changes of activity of a starch granule bound endoamylase and a starch phosphorylase in poplar wood (Populus x canadensis Moench “robusta”) and their possible regulation by temperature and phytohormones. J. Plant Physiol. 156:731-740. [ Links ]
García, E. M. 1987. Modificación al Sistema Climático de Köppen. Universidad Nacional Autónoma de México. México. 217 p. [ Links ]
González-Rossia, D., C. Reig, V. Dovis, N. Gariglio, and M. Agustí. 2008. Changes on carbohydrates and nitrogen content in the bark tissues induced by artificial chilling and its relationship with dormancy bud break in Prunus sp. Sci. Hort. 118: 275-281. [ Links ]
Govaerts, B., M. G. Barrera-Franco, A. Limón-Ortega, P. Muñoz-Jiménez, D .K. Sayre, y J. Deckers. 2008. Clasificación y evaluación edafológica de tres sitios experimentales del altiplano central de México. Tropicultura 26: 2-9. [ Links ]
Horvath, D. 2009. Common mechanisms regulate flowering and dormancy. Plant Sci. 177: 523-531. [ Links ]
Ito, A., H. Hayama, and Y. Kashimura. 2002. Sugar metabolism in buds during flower bud formation: a comparison of two Japanese pears [Pyrus pyrifolia (Burm) Nak.] varieties possessing different flowering habits. Sci. Hort. 96: 163-175. [ Links ]
Julian, C., J. Rodrigo, and M. Herrero. 2011. Stamen development and winter dormancy in apricot (Prunus armeniaca). Ann. Botany. 108: 617-625. [ Links ]
Julian, C., M. Herrero, and J. Rodrigo. 2014. Anther meiosis time is related to winter cold temperatures in apricot (Prunus armeniaca L.). Environ. Exper. Bot. 100: 20-25. [ Links ]
Luna, V., E. Lorenzo, H. Reinoso, M. Tordable, G. Abdala, R. Pharis, and R. Bottini. 1990. Dormancy in peach (Prunus persica L.) flower buds I. Floral morphogenesis and endogenous gibberellins at the end of the dormancy period. Plant Physiol. 93: 20-25. [ Links ]
Marafon, A., C. I. Citadir, L. Amarante, F. G. Herter, F. J. Hawerroth. 2011. Chilling privation during dormancy period and carbohydrate mobilization in Japanese pear trees. Sci. Agri. (Piracicaba, Braz.). 68: 462-468. [ Links ]
Nava, G., G. Dalmago, H. Bergamaschi, R. Paniz, R. dos Santos, and G. Marodin. 2009. Effect of high temperatures in preblooming and blooming periods on ovule formation, pollen grains and yield of ´Granada´ peach. Sci. Hort. 122: 37-44. [ Links ]
Palonen, P. 1999. Relationship of seasonal changes in carbohydrates and cold hardiness in canes and buds of three red raspberry cultivars. J. Am. Soc. Hort. Sci. 124: 507-513. [ Links ]
Reig, G., I. Iglesias, C. Miranda, F. Gatius, and S. Alegre. 2013. How does simulated frost treatment affect peach [Prunus persica (L.)] flowers of different varieties from worldwide breeding programmes? Sci. Hort. 160: 70-77. [ Links ]
Reinoso, H., L. Virginia, R. Pharis , and R. Bottini. 2002. Dormancy in peach (Prunus persica) flower buds. V. Anatomy of bud development in relation to phenological stage. Can. J. Bot. 80: 656-663. [ Links ]
Richardson, E. A., S. D. Seeley, and D. R. Walker. 1974. A model for estimating the completion of rest for `Redhaven` and `Alberta` peach trees. HortScience. 9: 331-332. [ Links ]
Rumayor, R. A., L. J. Llamas, M. V. Melero, y D. J. Zegbe. 2009. Descripción fenotípica de material genético de durazno para Zacatecas. INIFAP. Publicación especial 16. 26 p. [ Links ]
Santiago-Mejía, H., J. I. Cortés-Flores, A. Turrent-Fernández, M. Livera-Muñoz, E. García-Moya, H. A. Zavaleta-Mancera, y V. H. Volke-Haller. 2015. Respuesta de árboles de duraznero [Prunus persica (L.) Batsch] compuesto de dos cultivares en un ambiente con incidencia de heladas: floración y amarre de fruto. Agrociencia. 49: 573-592. [ Links ]
Wisniewski, M., and G. Davis. 1989. Evidence for the involvement of a specific cell wall layer in regulation of deep supercooling of xylem parenchyma. Plant Physiol. 91: 151-156. [ Links ]
Yamane, H., T. Ooka, H. Jotatsu, R. Sasaki, and R. Tao. 2011. Expression analysis of PpDAM5 and PpDAM6 during flower bud development in peach (Prunus persica). Sci. Hort. 129: 844-84. [ Links ]
Received: May 2018; Accepted: October 2018











 text in
text in