Services on Demand
Journal
Article
Indicators
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Polibotánica
Print version ISSN 1405-2768
Polibotánica n.28 México Sep. 2009
Antimicrobial activity of medicinal plants from the Huautla Sierra Biosphere Reserve in Morelos (Mexico)
Actividad antimicrobiana de las plantas medicinales de la reserva de la biosfera Sierra de Huautla en Morelos (México)
David O. Salinas Sánchez2,4, Gema L. Arteaga Najera2, Ismael León Rivera3, Oscar Dorado Ramírez4, Ma. Guadalupe Valladares Cisneros5, Víctor M. Navarro García1
1 Laboratorio de Microbiología, Centro de Investigación Biomédica del Sur, Instituto Mexicano del Seguro Social, Xochitepec Morelos, México.
2 Facultad de Ciencias Biológicas, Universidad Autónoma del Estado de Morelos, Cuernavaca, Morelos, México.
3 Centro de Investigaciones Químicas, Universidad Autónoma del Estado de Morelos, Cuernavaca, Morelos, México.
4 Centro de Educación Ambiental e Investigación Sierra de Huautla, Universidad Autónoma del Estado de Morelos, Cuernavaca, Morelos, México.
5 Laboratorio de Principios Fitoquímicos Bioactivos, Facultad de Ciencias Químicas e Ingeniería; Universidad Autónoma del Estado de Morelos, Cuernavaca, Morelos, México. Correo electrónico: vmanavg@yahoo.com.mx, osval1671@hotmail.com.mx
Recibido: 9 enero 2009
Aceptado: 7 agosto 2009
Abstract
Twenty–two extracts from nine Mexican medicinal plants of eight different families used for people neighbor to Huautla Sierra Biosphere Reserve (REBIOSH) in different infectious diseases were assayed in vitro to determine their antibacterial activity against Staphylococcus aureus, Streptococcus faecalis; Escherichia coli; Proteus mirabilis; Salmonella typhi and the yeast Candida albicans. Most plants showed antibacterial activity, while two plants showed activity against range 0.25 a 4 mg/mL almost one microorganism used. The extracts showed minimum inhibitory concentration (MIC) against S. aureus and S. faecalis, represents microorganisms Gram–positive were hexanic and acetonic extracts of Bur ser a copallifera and hexanic extract of B. grandifolia. The ethanol extract of Lippia graveolens showed a MIC value of 1 mg/mL against yeast Candida albicans.
Key words: antimicrobial activity, minimum inhibitory concentration (MIC), medicinal plants, Huautla Sierra Biosphere Reserve, Morelos.
Resumen
Se determinó la actividad antimicrobiana de veintidós extractos orgánicos de nueve especies vegetales medicinales de ocho diferentes familias, que son comúnmente empleadas por los habitantes de las comunidades vecinas a la reserva de la biosfera Sierra de Huautla (REBIOSH), Morelos, para tratar diferentes padecimientos infecciosos. Los microorganismos empleados fueron: Staphylococcus aureus; Streptococcus faecalis; Escherichia coli; Proteus mirabilis; Salmonella typhi y la levadura Candida albicans. La mayoría de los extractos de las plantas probadas inhibieron el crecimiento microbiano en un rango de 0.25 a 4 mg/mL de al menos uno de los microorganismos utilizados. Los extractos que mostraron las menores concentraciones mínimas inhibitorias (CMI) frente a los microorganismos S. aureus y S. faecalis, representativos de Gram–positivos fueron el extracto hexánico y acetónico de Bur ser a copallifera y el hexánico de B. grandifolia. El extracto etanólico de Lippia graveolens logró inhibir a C. albicans con una CMI de 1 mg/mL.
Palabras clave: Actividad antimicrobiana, concentración mínima inhibitoria (CMI), plantas medicinales, Reserva de la Biosfera Sierra de Huautla, Morelos.
INTRODUCTION
According to the World Health Organization, the population most susceptible to infectious diseases are children. In the whole world, a little over two million children under five years old died from these diseases from 2000–2003 (Lerberghe, 2005). In Mexico, the mortality statistics rank acute infectious respiratory diseases among the ten principal causes of death; seventh place with for women with 6 381 deaths, and in eighth place for men with 7 463 deaths (Estadísticas de Mortalidad en México, 2005). In the state of Morelos these diseases have high frequency rates, and in many cases are initially cared for by an herbalist (Taddei–Bringas et al., 1999, Monroy and Castillo, 2000; Romero et al., 2004; Maldonado et al., 2004).
In the Huautla Sierra Biosphere Reserve, Morelos–México (REBIOSH) (Figure 1), 1,025 species of vascular plants were documented to occur, and around 401 medicinal plant species were reported. Among them, are those used to control infectious diseases (Dorado et al., 2005). In the REBIOSH, the vegetation type is characterized as tropical deciduous forest (TDF), but in some areas temperate forest can be found mainly dominated by Quercus spp., and in some humid gullies the vegetation is similar to semi–deciduous forest. The TDF in the REBIOSH is found in general altitudes from 800 to 1500 m.a.s.l. Annual average temperatures are between 20° and 29°C, which is a determining factor in the distribution of the TDF (Rzedowski, 1978). A very important aspect is the fact that in this hilly region of south and southeast Morelos, more than 20 000 people live in about 29 communities inside the Reserve. Some of the species of wild medicinal flora of the REBIOSH are used by the people of the region to treat various sicknesses (Maldonado, 1997; Maldonado et al., 2004; Dorado et al., 2005).
At present, our team is investigating the field of medicinal plants with the potential to treat infectious diseases (Navarro et al., 1996; Navarro and Delgado, 1999; Rojas et al., 2001; Navarro et al., 2006). In the last few years, several plants that are used for various diseases have been studied, and the families–genera of plants that stand out are Boraginaceae–Cordia (Ioset et al., 1998; Ioset et al., 2000), Burseraceae–Bursera (Camporese et al., 2003; Yasunaka et al. 2005), Fabaceae–Prosopis (Salvat et al., 2004), Lamiaceae–Ocimum (Bozin et al., 2006), Passifloraceae–Passiflora (Nicolls et al., 1973), Rubiaceae–Randia (Jayasinghe et al., 2002), Urticaceae– Urtica (Gulcin et al., 2004) and Verbenaceae–Lippia (Pascual et al., 2001). As we continued the investigation of the aforementioned families–genera, we decided to perform antimicrobial bioassays on nine medicinal species belonging to these families of medicinal plants, whose habitat is the REBIOSH.
The present research paper is focused on the scientific validation of the use of certain medicinal plants for the treatment of infectious diseases by the communities next to the REBIOSH, who use this natural resource.
MATERIAL AND METHODS
Collecting the vegetable material
Plants material was collected in March and June, 2005, in the REBIOSH. The nine species were prepared for depositing in HUMO herbarium (Herbarium of the University of Morelos) of the Center of Environmental Education and Investigation of the Huautla Sierra of the Autonomous University of the State of Morelos. List the specimen accession number, as well as their common name and other ethnobotanical information (Table 1).

Preparation of extracts
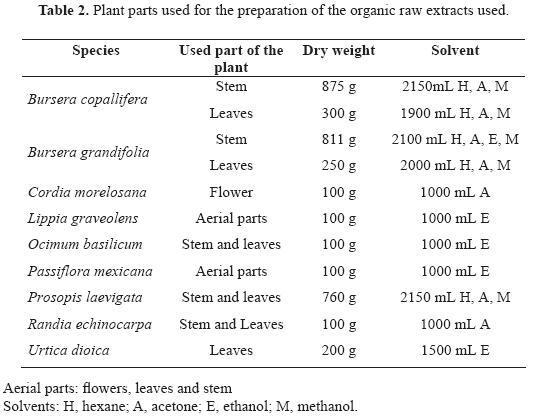
Plant materials (stem, leaves and flowers) were shade–dried at room temperature, powdered, and individually extracted sequentially with n–hexane, acetone, ethanol and methanol at room temperature (Table 2). Each solvent was replaced three–times with fresh solvent, remaining in contact with the plant material for 48 h on each occasion. After filtration, the extracts were concentrated under low pressure at 40°C. The resulting crude extracts were stored at – 20 °C until they were tested.
Microorganisms and media
The microorganisms used for the antimicrobial evaluation were purchased from the American Type Culture Collection (ATCC; Rockville, MD, USA). The following Gram–positive bacteria were used: Staphylococcus aureus (ATCC 6358), and Streptococcus faecalis (ATCC 10231). The following Gram–negative strains were used: Escherichia coli (ATCC 8937), Proteus mirabilis (ATCC 43071), Salmonella typhi (ATCC 06539) and the yeast Candida albicans (ATCC 10231). All bacteria were maintained on tryptic soybean agar (TSA; Merck, Darmstadt, Germany) and assayed on Mueller–Hinton agar (MH; Merck). Sabouraud 4% glucose agar (SGA; Merck) was used to maintain the yeast and as the assay medium.
Antimicrobial activity assays
The antibacterial and antifungal activities of the crude extracts were determined using the agar dilution method (Jorgensen et al. 1999). The hexane, acetone, ethanol and methanol extracts were individually dissolved in 2% dimethylsulfoxide (DMSO; Merck). Further two–fold dilutions of each extract were performed using sterile distilled water and were added to melted agar culture medium in 100 mm Petri dishes (Falcon Franklin Lakes, New Jersey, USA) at the following final concentrations: 0.125, 0.25, 0.50, 1, 2, 4, and 8 mg/mL. Agar culture medium and DMSO 2% were used as negative controls. The procedures followed for the antibacterial and antifungal assays are described below.
Antibacterial activity assay
Suspensions for each strain were prepared by transferring five or six colonies, chosen after overnight growth on tryptic soybean agar, to 5 mL of Mueller–Hinton broth (MHB; Merck). Cultures were incubated at 36°C until they were visibly turbid, and the suspensions were then diluted until the turbidity matched the 0.5 McFarland standard turbidity equivalents [108 colony–forming units (CFU)/mL]. Microbial suspensions were further diluted 1:10 to obtain a concentration of 107 CFU/mL. The diluted inoculum was applied with a loop calibrated to deliver 0.002 mL, resulting in a spot covering a circle of 5 to 8 mm diameter and containing 104 CFU/mL. The plates were incubated for 24 h at 36°C. Gentamicin (5 mg/mL; Sigma–Aldrich, St. Louis, MO, USA) was used as positive control. Observations were performed in duplicate, and results are expressed as the lowest concentration of plant extract that produced a complete suppression of colony growth, the minimal inhibitory concentration (MIC) (Ríos et al., 1988).
Antifungal activity assay
C. albicans inoculum was prepared by diluting the yeast suspension (adjusted to McFarland 0.5 scale) in 0.85% NaCl solution to a final concentration of 105 cell/mL. The filamentous fungi inoculums were prepared by diluting the scraped cells mass in 0.85% NaCl to a final concentration of 106 spores/mL. The diluted inocula were delivered on the top of the solidified agar with a loop calibrated to deliver 0.005 mL. The plates were incubated at 29°C. C. albicans growth was checked both in control plates prepared without any test sample and in experimental plates, C. albicans was cultured 48 h. Experiments were duplicated, and results were expressed as MIC. Positive control was prepared with Nystatin (5 mg/mL; Merck).
RESULTS AND DISCUSSION
Table 3 shows the plant part used, the percentage of yield, and the MIC obtained for the corresponding evaluations of each extract. The agar dilution method used in this investigation has been employed frequently and recommended as a good method for determining the relative potency of complex extracts and for establishing their antimicrobial spectrum (Ríos and Recio, 2005). In our experience and according to previous antimicrobial studies carried out in our laboratory, crude plant extracts with MIC values between 2.5 and 8 mg/mL (Lozoya et al., 1992; Navarro et al., 1996) have led to the isolation of strong antimicrobial compounds (Navarro and Delgado 1999). Under this criterion, and considering that in this study only crude extracts were employed, extracts with MIC values of 8 mg/mL or below against any of the microorganisms tested were considered active. Most of the extracts used showed antimicrobial activity with the Gram–positives bacteria being the more sensitive to the extracts.
The species which showed the highest activity was B. copallifera, of which the acetone stems extract and the methanol leaves extract were the most active against the S. aureus, both of them with a MIC value of 0.25 mg/mL. In addition these same extracts showed values of 1 and 0.25 mg/mL respectively against S. faecalis. On the other hand, the leaves acetone extract of this species showed a wide scale of inhibition against all the microorganisms, with MIC value of 2 mg/mL against the Gram–negatives and yeast. It is important to mention that the other species in this genus, that is to say B. grandifolia, also showed antimicrobial activity. The stems hexane and acetone extracts showed a MIC value of 0.5 mg/mL against the S. aureus. The hexane extract to B. grandifolia showed a MIC value of 4 mg/mL against all the microorganisms. The remainder of the plant species also showed notable antimicrobial activities. The ethanol extract of L. graveolens showed a MIC value of 1 mg/mL against S. aureus and the yeast C. albicans, and a MIC value of 4 mg/mL against S. faecalis. The ethanol extracts of O. basilicum and P. mexicana showed MIC values of 4 mg/mL against S. aureus.
On the other hand all the extracts of the medicinal plant P. laevigata showed antimicrobial activity. The hexane extract gave a MIC value of 0.5 mg/mL and the acetone extract showed a MIC value of 4 mg/mL, both against S. aureus. The ethanol extract of U. dioica showed antimicrobial activity with a MIC value of 1 mg/mL against the Gram–positive bacteria.
There were no literature reports of antimicrobial activity for most species studied, however, the chemical nature or the antimicrobial activity of some species of the same genera are described in the following research work.
The extract of Bursera simaruba (bark) was evaluated for its antibacterial properties. Activity was tested against standard strains of Escherichia coli ATCC 25922, Pseudomonas aeruginosa ATCC 27853, Staphylococcus aureus ATCC 25923 and Enterococcus faecalis ATCC 29212. The extract inhibited the growth of all of the bacterial strains, except that of Entero–coccus faecalis (Camporese et al., 2003). Other studies B. simaruba showed, higher activity against Staphylococcus aureus (methicillin–sensitive and methicillin–resistant) than against Escherichia coli (Yasunaka et al., 2005).
Two new natural products, characterized as 1–(3 '–methoxypropanoyl)–2,4,5–trimethoxybenzene and a prenylated hydroquinone, 2–(2Z)–(3–hydroxy–3,7–dimethylocta–2, 6–dienyl)–1,4–benzenediol, have been isolated from the root bark of Cordia alliodora. Both compounds exhibited antifungal properties against the phytopathogenic mold Cladosporium cucumerinum (Ioset et al., 2000). Also, two meroterpenoid naphthoquinones, the known cordiaquinone B and a new naphthoxirene, have been isolated from the roots of Cordia linnaei. The two showed activity against Cladosporium cucumerinum and Candida albicans (Ioset et al., 1998).
In a literature review of the genus Lippia, (Pascual et al., 2001) found that in vitro screening with extracts of L. alba, and L dulcis demonstrated an inhibitory activity against many of the Gram–positive bacteria most frequently responsible for infections in humans (Staphylococcus aureus. Streptococcus pneumoniae and Streptococcus pyogenes), and thus, may provide a scientific basis for the ethnomedical use of this species against bacterial respiratory infections (Cáceres et al., 1991).
Other studies report that the essential oils from genus Lippia have also been shown to exhibit antimicrobial activity against other microorganisms. For example L. sidoides shows a inhibitory activity against E. coli, S. aureus, Bacilus subtilis, and Mycobacterium smegmatis, and a low activity against Pseudomonas aeruginosa (Lemos et al., 1990).
The ethanolic extracts of L. palmeri and L. formosa exhibit activity against S. aureus, S. faecalis and B. subtilis (Dimayuga and Keer García, 1991). Extracts isolated from L. dulcis, shows antimicrobial activity against enteropathogenic Salmonella typhi and Shigella flexneri (Cáceres et al., 1993).
Some species of the genus Prosopis have been reported to exhibit antimicrobial activity. This is the case for P. juliflora. One alkaloid from this plant, which has been named juliprosinene, showed antibacterial activity against Escherichia coli strains 11303 HER 1024 and K–125 HER 1037, Klebsiella pneumoniae C3HER 1111, Pseudomonas aeruginosa PA 01 HER 1018, Staphylococcus aureus B–827, and Shigella sonnei YER HER 1048 (Ahmad et al., 1989). In another study of this plant, a mixture of alkaloids containing mainly juliprosine and isojuliprosine was tested for antifungal activity against dermatophytos, Aspergillus and Candida species in comparison with griseofulvin and econazole. Although econazole was found to be the most active one, the alkaloidal mixture was found more effective than griseofulvin (Ahmad and Khursheed, 1989).
Methanol extracts of the barks of two species of Prosopis were evaluated to determine their antimicrobial activity. Prosopis kuntzei exhibited antimicrobial activity against S. aureus ATCC 8095, INE 2213, and Ps. aeuruginosa ATCC 25617, with CMI below 1 mg/mL. On the other hand, Prosopis ruscifolia had activity against S. aureus ATCC 8095, INE 2213, Ent. faecium INEI 2464 and Kl. pneumoniae ATCC 25922, with MIC of 0.5, 0.38, 0.75 and 0.75 mg/mL respectively (Salvat et al., 2004).
Recent research has discovered a new potent anti–infective and antiparasitic compound, 2,3–dihydro–1H–indolizinium chloride, from Prosopis glandulosa var. glandulosa. It shows antibacterial activity against methicillin–resistant Staphylococcus aureus and Micobacterium intracellulare with values of IC50 0.35 and 0.9 ug/mL respectively (Samoylenko et al., 2009).
An investigation of Urtica dioica showed that a water extract exhibited antioxidant, antimicrobial, antiulcer and analgesic activities (Gulcin et al., 2004).
The results obtained in the current investigation indicate that B. copallifera, B. grandifolia, L. graveolens, P. leavigata and U. dioica possesses antibacterial and/or antifungal properties, showing a strong correlation between the reported uses of these plant species by the communities of the Huautla Sierra in the State of Morelos and in Mexican folk medicine and the obtained experimental data. It also supports the importance of ethnopharmacology as a guide in the selection of plants for the discovery of bioactive compounds.
Phytochemical bioguided studies are currently under way in order to isolate and characterize the compounds responsible for the antimicrobial activities of theses plants.
ACKNOWLEDGMENTS
The authors would like to thank Biol. Juan Carlos Juárez from the CEAMISH/UAE–Morelos for assistance in the collection and identification of plant specimens.
REFERENCES
Ahmad, A., Khursheed, A.K., 1989. "Antifungal activity of some hydrosoluble Prosopis juliflora alkaloids". Fitoterapia, 60(1):86–89. [ Links ]
Ahmad, V.U., Sultana, A., Qazi S., 1989. "Alkaloids from the leaves of Prosopis juliflora". Journal Natural Products, 52(3): 497–501. [ Links ]
Argueta, A., Cano, L., Rodarte, M., 1994. Atlas de las Plantas de la Medicina Tradicional Mexicana. Instituto Nacional Indigenista (1–3): 86–88, 999–1000, 1073–1074, 1086–1087. [ Links ]
Bozin, B., Mimica–Dukic, N., Simin, N., Anackov, G., 2006. "Characterization of the volatile composition of essential oils of some lamiaceae spices and the antimicrobial and antioxidant activities of the entire oils". Journal Agricultural and Food Chemistry. 54(5): 1822–1828. [ Links ]
Cáceres, A., Álvarez, A.V., Ovando, A.E., Samayoa, B.E., 1991. "Plants used in Guatemala for the treatment of respiratory diseases. 1. Screening of 68 plants against Gram–positive bacteria". Journal of Ethnopharmacology, 31: 193–208. [ Links ]
Cáceres, A., Fletes, L., Aguilar, L., Ramírez, O., Figueroa, L., Taracena, A. M., Samayoa, B., 1993. "Plants used in Guatemala for the treatment of gastrointestinal disorders. 3. Confirmation of activity against enterobacteria of 16 plants". Journal of Ethnopharmacology, 38: 31–38. [ Links ]
Camporese, A., Balick, M., Arvigo, R., Esposito, R., Morsellino, N., De Simone, F., Tubazo, A., 2003. "Screening of antibacterial activity of medicinal plants from Belize (Central America)". Journal of Ethnopharmacology, 87(1): 103–7. [ Links ]
Dimayuga, R.E., Keer García S., 1991. "Antimicrobial screening of medicinal plants from Baja California Sur, Mexico". Journal of Ethnopharmacology, 31: 181–192. [ Links ]
Dorado, O., Maldonado, B., Arias, D., Sorani, V., Ramírez, R., Leyva, E., Valenzuela, D., 2005. "Programa de Conservación y Manejo Reserva de la Biosfera Sierra de Huautla". Comisión Nacional de Áreas Naturales Protegidas, México; p. 3–4; 33–34; 54; 139–170. [ Links ]
Estadísticas de Mortalidad en México, 2005. "Muertes registradas en el año 2003". Salud Pública de México, Mar./Apr. 47(2): 178–179, 180,183. [ Links ]
Gulcin, I., Kufrevioglu, O., Oktay, M., Buyukokuroglu, M., 2004. "Antioxidant, antimicrobial, y antiulcera and analgesic activities of nettle (Urtica dioica L.)". Journal of Ethnopharmacology, 90(2–3): 205–215. [ Links ]
Ioset, J.R., Marston, A., Gupta, M., Hostettmann, K., 1998. "Antifungal and larvicidal meroterpenoid naphthoquinones and a naphthoxirene from the roots of Cordia linnaei". Phytochemistry, 47(5): 729–34. [ Links ]
Ioset, J.R., Marston, A., Gupta, M., Hostettmann, K., 2000. "Antifungal and larvicidal cordiaquinones from the roots of Cordia curassavica ". Phytochemistry, 53(5): 613–7. [ Links ]
Jayasinghe, U., Jayasooriya, C., Bandara, B., Ekanayake, S., Merlini, L., Assante, G., 2002. "Antimicrobial activity of some Sri Lankan Rubiaceae and Meliaceae". Fitoterapia, 73(5): 439–441. [ Links ]
Jorgensen, J.H., Turnidege, J.D., Washington, J.A., 1999. "Antibacterial susceptibility tests: Dilution and disk difussion nietbods". In: Annual of Clinical Microbiology, 7th ed. Washington, DC, ASM Press, pp. 1526–1530. [ Links ]
Lemos, T.L.G., Matos, F.J.A., Alencar, J.W., Craveiro, A. A., Clark, A.M., McChesney, J.D., 1990. "Antimicrobial activity of essential oils of Brazilian plants". Phytotherapy Research, 4: 82–84. [ Links ]
Lerberghe, W., 2005. Informe Sobre la Salud en el Mundo. Organización Mundial de la Salud. Cuadro 3 Número anual de defunciones por distintas causas en menores de 5 años en las regiones de la OMS, estimaciones para 2000–2003, p. 208. [ Links ]
Lozoya, X., Navarro, V., García, M., Zurita, M., 1992. "Solanum chrysotrichum (Schldl.) a plant used in Mexico for the treatment of skin mycosis". Journal Ethnopharmacology, 36(2): 127–32. [ Links ]
Maldonado, B., 1997. Aprovechamiento de los Recursos Florísticos de la Sierra de Huautla, Morelos. Tesis de Maestría. Facultad de Ciencias. UNAM. México DF. [ Links ]
Maldonado, B., Ortiz, S., Dorado, O., 2004. Preparados Galénicos e Imágenes de Plantas Medicinales, Una alternativa para promotoras de la salud en la Reserva de la Biosfera Sierra de Huautla, Centro de Educación Ambiental e Investigación Sierra de Huautla, Universidad Autónoma del Estado de Morelos, p. 7–11. [ Links ]
Monroy, C., Castillo, P., 2000. Plantas Medicinales Utilizadas en el Estado de Morelos, Centro de Investigaciones Biológicas. UAEM, p. 89, 90, 95, 136, 150, 203, 216, 228, 239, 258, 291, 298. [ Links ]
Navarro, V., Villareal, M., Rojas, G., Lozoya, X., 1996. "Antimicrobial evaluation of some plants used in Mexican traditional medicine for the treatment of infectious disease". Journal of Ethnopharmacology, 53(3): 143–147. [ Links ]
Navarro, V., Delgado, G., 1999. "Two antimicrobial alkaloids from Bocconia arborea". Journal of Ethnopharmacology, 66(2): 223–226. [ Links ]
Navarro, V., Rojas, G., Zepeda, L., Aviles, M., Fuentes, M., Herrera, A., Jiménez, E., 2006. "Antifungal and antibacterial activity of four selected Mexican medicinal plants". Pharmaceutical Biology, 44(4): 1–4. [ Links ]
Nicolls, J., Birner, J., Forsell, P., 1973. "Passicol, an antibacterial and antifungal agent produced by Passiflora plant species: qualitative and quantitative range of activity". Antimicrobial Agents Chemotherapy, 3(1): 110–7. [ Links ]
Pascual, M., Slowing, K., Carretero, E., Sánchez Mata, D., Villar, A., 2001. "Lippia: traditional uses, chemistry and pharmacology: a review". Journal of Ethnopharmacology, 76(3): 201–214. [ Links ]
Romero, C., Reyes, M., Herrera, A., Lozoya, X., Tortoriello, J., 2004. "Aceptación de los fitofármacos por médicos y pacientes en clínicas de atención primaria". Revista Médica IMSS; 42(2): 125–130. [ Links ]
Ríos, J. L., Recio, M. C., Villar, A. (1988) "Screening methods for natural products with antimicrobial activity: a review of the literature". Journal of ethnopharmacology, 23: 127–149. [ Links ]
Ríos, J.L., Recio, M.C., 2005. "Medicinal plants and antimicrobial activity". Journal of Ethnopharmacology, 100(1–2): 80–84. [ Links ]
Rojas, G., Lévaro, J., Tortoriello, J., Navarro, V., 2001. "Antimicrobial evaluation of certain plants used in Mexican traditional medicine for the treatment of respiratory disease". Journal of Ethnopharmacology, 74(1): 97–101. [ Links ]
Rzedowski, J., 1978. Vegetación de México. Ed. Limusa, México, p. 432. [ Links ]
Samoylenko, V., Ashfaq, M.K., Jacob, M.R., Tekwani, B.L., Khan, S.I., Manly, S.P., Joshi, V.C., Walter, L. A., Muhammad I., 2009. "Indolizidine, Antiinfective and Antiparasitic Compounds from Prosopis galdulosa var. glandulosa". Journal of Natural Products, 72: 92–98. [ Links ]
Salvat, A., Antonacci, L., Suarez, E., Godoy, H., 2004. "Antimicrobial activity in methanolic extracts of several plant species from northern Argentina". Phytomedicine, 11: 230–234. [ Links ]
Taddei–Bringas, G., Santillana, M., Romero, J., Romero, M., 1999. Aceptación y Uso de Herbolaria en Medicina Familiar. Salud Pública de México, 41(3): 216–220. [ Links ]
Yasunaka, K., Abe, F., Nagayama, A., Okabe, H., Lozada–Pérez, L., López–Villafranco, E., Estrada–Muñiz, E., Aguilar, A., Reyes–Chilpa, R., 2005. "Antibacterial activity of crude extracts from Mexican medicinal plants and purified coumarins and xanthones". Journal of Ethnopharmacology, 97(2): 293–9. [ Links ]














