Introduction
Mexico is considered the center of origin of the Mexican avocado (Persea americana Mill) race, although the Antillean race is also cultivated in the south of the country (Can-Alonzo et al., 2005). Likewise, Mexico is the world’s leading producer of ‘Hass’ avocado, with a volume of 2 million 29 thousand tons in 2017 (Servicio de Información Agroalimentaria y Pesquera [SIAP], 2019).
Avocado floral volatiles provide chemical signals to phytophagous and pollinating insects to locate food sources, habitats and oviposition sites (Bruce, Wadhams, & Woodcock, 2005; Sanders & van Doorn, 2004), in addition to providing taxonomic information on the species (Kim, Ha, Kim, Chang, & Kim, 2014). The chemical composition of these volatiles depends on the species, variety, physiological state and circadian rhythm of the plants (Kolosova, Sherman, Karlson, & Dudareva, 2001; Niinemets, Loreto, & Reichstein, 2004). In Mexico, avocado flowering periods depend on race, variety, climatic conditions and elevation. For example, the ‘Hass’ avocado flowering period is 85 days at 12 to 17 °C, and decreases to 15 days at 28 to 33 °C (Sanders & van Doorn, 2004). This period is similar to that of the Antillean race; however, it is shorter in temperate zones (Can-Alonzo et al., 2005).
In the state of Chiapas, Mexico, the commercial ‘Hass’ avocado is mainly grown in the uplands, and the Antillean race avocado is cultivated in the subtropical Soconusco (coast) region. There is little information on the aroma chemistry of the different phenological stages among avocado races and varieties. Therefore, the aim of this study was to identify and compare the floral volatiles of the ‘Hass’ and Antillean race avocados. This information could be useful for taxonomic purposes, and could eventually be used to find out if these volatile compounds influence the interactions of avocado with its pollinating and herbivorous insects.
Materials and methods
‘Hass’ avocado flowers were collected in orchards located in the municipalities of Margaritas (16° 18’ 1” N and 91° 57’ 18” W) and Comitán de Domínguez (16° 16’ 58.3” N and 92° 1’ 59.7” W), Chiapas, Mexico, which have an average temperature of 19.5 °C and an elevation of 1,600 masl. The Antillean race flowers were collected from trees in Canton Texcuntic, municipality of Frontera Hidalgo (14° 47’ 41.13” N and 92° 11’ 14.5” W) and in the municipality of Tuxtla Chico (14° 54’ 21” N, 92° 13’ 43.4” W), Chiapas, where the average temperature is 27 °C and the elevation is 92 masl. The collections were carried out between November and December 2018. These two types of avocado were selected due to their commercial importance in the region.
The volatiles were captured using the solid-phase microextraction technique. For this, 100 g of flowers were placed in 12 mL glass vials. The vial inlet was covered with aluminum foil and a 65 µm thick polydimethylsiloxane/divinylbenzene (PDMS/DVB) fiber (Supelco®, Mexico) was inserted into it, for a period of 16 h. The procedure was similar for the ‛Hass’ and Antillean avocado flowers, and five replications were performed in both cases.
The identification of the volatiles captured by the PDMS/DVB fiber was performed in a gas chromatograph (CP-3800, Varian, USA) coupled to a selective mass detector (Saturn 2200, Varian, USA). A methyl silicon column (DB5-MS) of 30 m in length by 0.25 mm in inner diameter, with helium as carrier gas, was used for the determination. The samples were injected in splitless mode and the temperature program used was: initial temperature of 50 °C (2 min) to a final temperature of 280 °C (2 min), with an increase of 15 °C per min. The volatile analysis was performed with the Saturn GC/MS Workstation software. Preliminary identification of the compounds was made by comparing retention time and mass spectrum with the NIST 2002 spectral library; later, they were confirmed with the synthetic standards. The relative abundance of the components was calculated considering the peak area of all of them.
Statistical analysis
Peak areas of floral compounds identified in ‘Hass’ and Antillean avocado were subjected to a principal component analysis (PCA), in which the correlation matrix was used to determine the compounds with the highest incidence in the two varieties. Differences in volatiles between varieties were tested by constructing confidence ellipses (α = 0.05). Confidence ellipses around the centroids allow observing the variation within the groups: similar correlation matrices denote a similar shape, while those having a common mean must overlap. Before the PCA, data were rescaled to have zero mean and one standard deviation, which reduces the influence of variables with very high or low magnitudes, in addition to normalizing the data (McCune & Grace, 2002). The statistical analysis was conducted with the R v3.6.3 software (R Core Team, 2020).
Results and discussion
Thirty-five volatiles were identified in ‘Hass’ and Antillean avocado flowers, which are mostly monoterpenes and sesquiterpenes. The average values of the percentage compositions are shown in Table 1, where linalool was the most abundant compound (30 %) in the Antillean race, while in ‘Hass’ the most abundant was (E)-β-ocimene (20 %).
Table 1 Mean relative abundance (± standard error) of ‘Hass’ and Antillean avocado floral volatiles.
| Number | Compound name | RI | Abundance (%) | |
|---|---|---|---|---|
| Antillean | ‘Hass’ | |||
| 1 | α-Pinene* | 927 | 0.00 | 0.39 ± 0.05 |
| 2 | Sabinene | 970 | 0.00 | 0.36 ± 0.36 |
| 3 | β-Pinene* | 977 | 0.00 | 0.43 ± 0.08 |
| 4 | β-Myrcene* | 985 | 0.93 ± 0.57 | 3.45 ± 0.07 |
| 5 | Limonene* | 1031 | 0.25 ± 0.08 | 0.49 ± 0.08 |
| 6 | (Z)-β-Ocimene* | 1034 | 1.45 ± 0.20 | 3.16 ± 0.81 |
| 7 | (E)-β-Ocimene* | 1045 | 9.99 ± 2.07 | 20.82 ± 3.92 |
| 8 | (E)-Linalool oxide* | 1078 | 0.00 | 0.30 ± 0.07 |
| 9 | (Z)-Linalool oxide* | 1093 | 3.98 ± 1.08 | 1.97 ± 0.40 |
| 10 | Linalool* | 1105 | 30.30 ± 6.05 | 0.43 ± 0.04 |
| 11 | Perylene (isomer) | 1117 | 24.36 ± 5.45 | 9.33 ± 4.76 |
| 12 | Benzyl nitrile | 1151 | 0.00 | 1.91 ± 0.63 |
| 13 | Lavandulol | 1170 | 0.00 | 0.43 ± 0.07 |
| 14 | Butyl caproate | 1193 | 7.65 ± 2.27 | 0.00 |
| 15 | Methyl salicylate* | 1206 | 0.00 | 2.74 ± 0.72 |
| 16 | n-Tridecane* | 1300 | 2.51 ± 1.45 | 0.36 ± 0.08 |
| 17 | α-Copaene* | 1362 | 0.00 | 2.41 ± 0.43 |
| 18 | β-Cubebene* | 1389 | 0.84 ± 0.20 | 1.94 ± 0.35 |
| 19 | β-Gurjunene | 1405 | 0.00 | 2.03 ± 0.60 |
| 20 | β-Caryophyllene* | 1443 | 3.62 ± 1.04 | 13.98 ± 1.22 |
| 21 | γ-Muurolene | 1447 | 0.00 | 0.55 ± 0.08 |
| 22 | Valencene | 1480 | 0.44 ± 0.10 | 1.58 ± 0.41 |
| 23 | β-Guaiene | 1494 | 1.04 ± 0.14 | 1.54 ± 0.34 |
| 24 | n-Pentadecane* | 1500 | 0.41 ± 0.20 | 2.40 ± 0.52 |
| 25 | γ-Cadinene | 1506 | 0.73 ± 0.30 | 12.85 ± 4.07 |
| 26 | β-Cadinene | 1535 | 1.45 ± 0.30 | 2.75 ± 0.58 |
| 27 | Dendrasaline | 1583 | 2.27 ± 1.21 | 1.34 ± 0.93 |
| 28 | n-Hexadecane* | 1600 | 0.29 ± 0.06 | 0.27 ± 0.04 |
| 29 | 8-Hexadecine | 1663 | 0.00 | 0.53 ± 0.05 |
| 30 | Cyclohexadecane | 1669 | 0.00 | 2.57 ± 0.48 |
| 31 | n-Heptadecane* | 1700 | 0.85 ± 0.18 | 3.36 ± 0.53 |
| 32 | n-Nonadecane* | 1900 | 2.53 ± 0.81 | 2.77 ± 0.70 |
| 33 | n-Icosane* | 2000 | 1.85 ± 0.51 | 0.00 |
| 34 | n-Heneicosane* | 2100 | 1.54 ± 0.30 | 0.50 ± 0.05 |
| 35 | n-Docosane* | 2200 | 0.730.18 | 0.05 ± 0.01 |
RI = retention index.* Compounds confirmed with synthetic standards.
The PCA between the compounds of the two varieties explains 66.1 % of the total variability of the data, which denotes high eigenvalues (Table 2). Of the 35 compounds included in the model, 26 were significantly correlated with PC1, and 2 with PC2, which had the highest contribution percentage in the ranking (Table 2; Figure 1).
Table 2 Eigenvalues and correlation coefficients of the first two components (PC1 and PC2) in ‘Hass’ and Antillean avocado floral volatiles.
| Compound | Correlation | |
|---|---|---|
| PC1 | PC2 | |
| C1 | 0.9566* | 0.0982 |
| C2 | 0.9343* | 0.1891 |
| C3 | 0.8648* | -0.3282 |
| C4 | 0.7430* | 0.4990 |
| C5 | 0.6105 | 0.1005 |
| C6 | 0.6517* | 0.5577 |
| C7 | 0.6840* | 0.0915 |
| C8 | 0.8248* | -0.2656 |
| C9 | -0.5702 | 0.0141 |
| C10 | -0.8251* | 0.0958 |
| C11 | -0.6115 | -0.4225 |
| C12 | 0.7247* | -0.3634 |
| C13 | 0.9117* | 0.0157 |
| C14 | -0.7980* | 0.0080 |
| C15 | 0.8466* | 0.3575 |
| C16 | -0.4690 | 0.4592 |
| C17 | 0.9152* | 0.0653 |
| C18 | 0.6688* | 0.1751 |
| C19 | 0.7548* | -0.2011 |
| C20 | 0.8965* | -0.0214 |
| C21 | 0.8975* | -0.2019 |
| C22 | 0.7481* | 0.5412 |
| C23 | 0.4453 | 0.4676 |
| C24 | 0.7716* | -0.0187 |
| C25 | 0.6807* | -0.2620 |
| C26 | 0.5736 | 0.2075 |
| C27 | -0.2147 | 0.6735* |
| C28 | -0.0725 | 0.8191* |
| C29 | 0.9646* | 0.0341 |
| C30 | 0.9076* | 0.0718 |
| C31 | 0.8142* | -0.3060 |
| C32 | -0.0228 | -0.2417 |
| C33 | -0.8107* | 0.3504 |
| C34 | -0.7999* | 0.2285 |
| C35 | -0.8391* | 0.2105 |
| Eigenvalues | 19.42 | 3.7 |
*Significant correlation (α = 0.05).
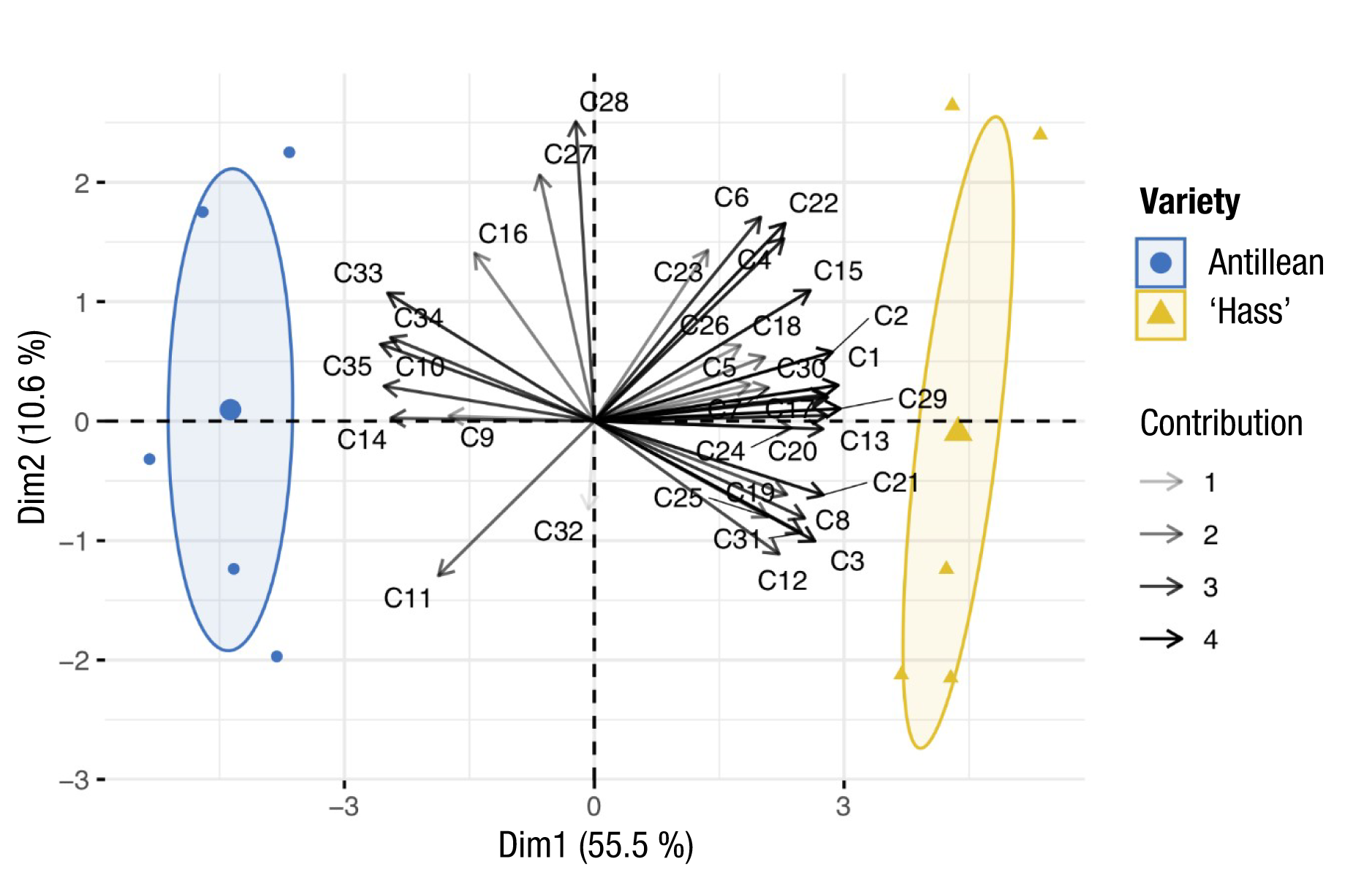
Figure 1 Principal component analysis of the areas of the floral compounds found in ‘Hass’ and Antillean avocado. The intensity of the arrows in the biplot indicates the contribution percentage of each compound in the construction of the first two dimensions of the ranking chart. From 1 to 35 correspond to the compound number in Table 1. Confidence ellipses (α = 0.05).
The confidence ellipses indicate high heterogeneity in the analyzed samples, with the greatest variation in the ‘Hass’ variety samples. However, differences in volatile composition are evident between varieties, which are explained to a greater extent by PC1, which indicates a positive association with the ‘Hass’ variety and a negative one with the Antillean race. Similarly, a greater amount of compounds associated with the ‘Hass’ variety was observed (Figure 1).
The results of this work allow identifying the compounds that were more abundant in ‛Hass’ and Antillean avocado, or that are only found in one of them. In this sense, the volatiles α-pinene, sabinene, β-pinene, (E)-linalool oxide, benzyl nitrile, lavandulol, methyl salicylate, α-copaene, β-gurjunene and γ-muurolene were only found in the ‘Hass’ avocado flowers. These differences in floral volatiles can help to taxonomically classify the avocado races analyzed, as in the study by Kim et al. (2014), where they classified different species of chrysanthemums (Chrysanthemum) in Korea according to the composition of floral volatiles.
Differences in the volatile compounds of the two types of avocados may also help explain the preference of some herbivorous species for these crops. Such is the case of the avocado seed weevil Conotrachelus perseae, which has only been reported in the ‘Hass’ avocado crop in the Comitan region, Chiapas (Vázquez, Cruz-López, & Chamé-Vázquez, 2015), while the seed borer moth Stenoma catenifer has been reported in Antillean race crops (Vázquez, Cruz-López, Gómez, & Castillo, 2017) in the Soconusco region, Chiapas. The same may occur with the attraction of several species of pollinators that have been reported for avocado (Castañeda-Vildózola et al., 1999), as happens with the floral volatiles of several species of pumpkin (Curcubita) and Cirsium arvense flowers that attract herbivores and pollinators (Andrews, Theis, & Adler, 2017; Theis, 2006).
Conclusions
Thirty-five compounds were identified in ‘Hass’ and Antillean avocado flowers. The floral volatiles α-pinene, sabinene, β-pinene, (E)-linalool oxide, benzyl nitrile, lavandulol, methyl salicylate, α-copaene, β-gurjunene and γ-muurolene were only found in ‘Hass’ avocado. In addition, differences were observed between the two types of avocados in terms of the proportions of the compounds.











 text in
text in 


