Services on Demand
Journal
Article
Indicators
Related links
Share
Revista fitotecnia mexicana
Print version ISSN 0187-7380
Rev. fitotec. mex vol.34 n.3 Chapingo Jul./Sep. 2011
Artículos científicos
Humic substances and moisture content in the production of biomass and bioactive constituents of Thymus vulgaris L.
Sustancias húmicas y contenido de humedad en la producción de biomasa y constituyentes bioactivos de Thymus vulgaris L.
Cecilia R. Juárez R.1*, Lyle E. Craker 2, Ma. de las Nieves Rodríguez Mendoza3 y Juan A. Aguilar–Castillo1
1 Unidad Académica de Agricultura, Universidad Autónoma de Nayarit. Km. 9.5 Carretera Tepic–Compostela. Xalisco, Nayarit, México. * Autor para correspondencia (cecirjr_uan@hotmail.com)
2 Department of Plant Soil and Insect Sciences, University of Massachusetts. Amherst, MA, USA, 01003–0910.
3 Programa de Edafología, Colegio de Postgraduados–Campus Montecillo. Km 36.5 Carretera México–Texcoco. 56230, Montecillo, Texcoco, Estado de México.
Recibido: 25 de Mayo del 2010.
Aceptado: 13 de Julio del 2011.
Abstract
Thyme (Thymus vulgaris L.) is an aromatic and medicinal plant with antioxidant, antimicrobial and antinfungical properties that have attracted the interest of pharmacological and food industries. Humic substances (HS) are a natural product that has shown beneficial effects on soil and on plant growth. These positive effects are explained by the direct interaction of HS with physiological processes. But it is unknown their effect on the composition of the thyme bioactive constituents. To test the effect of humic substances, seeds of T. vulgaris were germinated and grown at different rates of HS (100, 200, 300 and 400 mg L–1). Thyme plants were also grown at three levels of moisture (20, 40 and 60 % of field capacity). At harvest plant height and shoot fresh and dry weight were measured. Bioactive constituents were determined in terms of yield and principal components of essential oil (by distillation in Clevenger apparatus and gas chromatography). Antioxidant activity, total phenolic and flavonoid content were determined with spectrophotometric techniques. Morphological variables, except for shoot fresh weight, showed significant differences (P ≤ 0.05) among treatments. The essential oil yield and composition, as well as antioxidant activity and total phenolic and flavonoid contents, were higher at the highest levels of HS and substrate moisture.
Key words: Thymus vulgaris, essential oils, antioxidant activity, flavonoids, phenolics.
Resumen
El tomillo (Thymus vulgaris L.) es una planta aromática y medicinal con propiedades antioxidantes, antimicrobianas y antifúngicas de interés en las industrias farmacológica y alimentaria. Las sustancias húmicas (SH) son productos naturales que tienen efectos benéficos tanto en el suelo como en el crecimiento vegetal, efectos que han sido explicados por la interacción directa de las SH con los procesos fisiológicos. Pero se desconoce si estos factores pueden incrementar el rendimiento y la composición de los constituyentes bioactivos en tomillo. Para probar el efecto de estas sustancias, se obtuvieron plántulas de tomillo a partir de la germinación de semillas de T. vulgaris, cultivadas en diferentes concentraciones de SH (100, 200, 300 y 400 mg L–1) y tres niveles de humedad (20, 40 y 60 % de capacidad de campo). A la cosecha se midió el crecimiento de la planta, en altura, peso fresco y peso seco de la parte aérea. Sus constituyentes bioactivos se determinaron como rendimiento y componentes principales del aceite esencial (por destilación en aparato Clevenger y cromatografía de gases); además se midió actividad antioxidante y contenidos totales de fenoles y de flavonoides mediante técnicas espectrofotométricas. Las variables morfológicas mostraron diferencias significativas (P ≤ 0.05) entre tratamientos, excepto en el peso en fresco de la parte aérea. El rendimiento de aceite esencial y su composición, así como la actividad antioxidante y los contenidos totales de fenoles y flavonoides, fueron mayores con los máximos niveles de SH y de humedad del sustrato.
Palabras clave: Thymus vulgaris, aceites esenciales, actividad antioxidante, flavonoides, fenoles.
INTRODUCTION
The genus Thymus belongs to the family of Labiateae and includes many species. Thymus vulgaris L. is a small woody shrub 10–30 cm tall native to the Mediterranean region. It is an aromatic plant characterized by a broad chemical intraespecific variability (Bozin et al., 2006; Figueiredo et al., 2008). Thyme leaves can be used fresh or dried as a spice to add a distinctive aroma and flavor to food (Lee et al., 2004). The essential oils are rich in phenolic compounds and possess a wide range of biological and pharmacological properties (Bozin et al., 2006): antiseptic, carminative, antimicrobial and antioxidative (Baranauskiene et al., 2003; Burt 2004; Nejad et al., 2008). Because of this broad range of uses, this wild plant is gathered so extensively that it is in danger of extinction. Cultivation of thyme could help to prevent extinction. However, the bioactive properties of the secondary metabolites need to be measured in cultivated thyme plants. Also the trend toward healthy, safe and exotic food makes necessary to implement fertilization practices that can guarantee the consumer safeness and product quality.
The content and composition of thyme essential oils depend on several factor such as chemotype, biotype and cultivation conditions (Figueiredo et al., 2008). Several studies have shown that the humic substances (HS) used for plant nutrition, can enhance root, leaf and shoot growth as well as fruit yield (Chen et al., 2004; Siminis et al., 1998). These positive effects can be explained by the direct action of humic substances on physiological and metabolic processes (Eyheraguibel et al., 2008). Their effects appear to be exerted on cell membrane functions by promoting nutrient uptake and plant growth and development, because they are hormone–like substances (Nardi et al., 2002). Some studies have documented yield increases in vegetables, root crops, flowers and cereals by humic substances (Nikbaht et al., 2008). In addition, drought conditions can limit photosynthesis in plants and alter the nutrient uptake, thus decreasing the production of secondary metabolites (Figueiredo et al., 2008). Therefore, the purpose of this study was to determine biomass production and bioactive constituents in thyme in response to humic substances and soil moisture percent.
MATERIALS AND METHODS
Experimental conditions
This study was conducted in the winter 2009 in a greenhouse of the Medicinal and Aromatic Plants Department of the University of Massachusetts at Amherst, MA, located at 42.37° N and at 72.53° W. During the experiment the average temperature was 20 °C and daylight lasted 10 h.
Thyme plants were grown from seeds obtained from the company Botanical Interest* (USA). Plants were cultivated in different concentrations of humic substances (HS1:100; HS2: 200; HS3: 300; and HS4: 400 mg L–1), contained in the commercial products HumiSolve USA* (humic acids 57 %) and TM–7USA* (fulvic acids 10.5 %, derived from free water cretaceous humate deposits, soluble potash 3.7 %, sulfur 5 %, copper 0.31 %, iron 1.2 %, boron 0.37 %, manganese 1.2 %, cobalt 0.05 %, molybdenum 0.1 %, zinc 1 %). The HS were added at the beginning of each week during the experimental period. The substrate used for plant growth was Farfad 3B MIX* with a pH range of 5.5–6.5 after wetting, which has the following components: Canadian sphagnum 45 %, perlite 15 %, vermiculite 15 %, starter nutrients, wetting agents and dolomitic limestone. Thyme plants were grown also at three moisture levels (M1: 20, M2: 40, and M3: 60 percent of field capacity). The substrate moisture content was measured by the gravimetric method (Sanchez–Blanco et al., 2008). Water consumption was monitored throughout the experimental period and was determined every other day by subtracting the weight of pots without plant from that of pots with plant. A factorial experiment with completely randomized design and three replications was used.
Variables measured
Plant height. Plants were measured every week until harvest, 60 d after the transplant.
Fresh and dry weight. Fresh biomass was weighed in an analytical balance (Mettler Toledo* PL303). To determine dry weight the plant samples were placed in an oven at 70 C until a constant weight was reached. The rest of the plant material was harvested and stored at –20 °C until hydrodistillation.
Essential oils. Ten grams of shoot fresh material was steam distilled in a Clevenger type apparatus during 2 h, when water started to boil. Oil volume was measured directly in the extraction burette. Samples were stored in flasks and kept at 4 °C until gas chromatography analysis. For identification of major volatile components in the essential oils, extracts were analyzed in a Shimadzu GC–2014® gas chromatograph equipped with a flame ionization detector (FID). A capillary column (30 m x 0.25 mm inner diameter and 0.25 µm film thickness) was used to separate individual components. Helium was used as a carrier gas with a flow rate of 1 mL min–1. Temperature was programmed from 75 to 200 °C with a ramp rate of 4 °C for a final time of 5 min. Both injector and detector were maintained at 200 and 220 °C, respectively. The sample (1 µL) was injected at a five split ratio with an autoinjector Shimadzu AOC–20i®. The individual peaks were identified from their retention times, and compared to the following standards: carvacrol, linalool, thymol, 3–octanol, α–pinene, (+)–limonene and (–) thujone.
The antioxidant activity index, total phenolic content and total flavonoids were measured in fresh material, based on colorimetric reactions measured with a HACH DR/2000® spectrophotometer. All analytical analyses were performed three times. Samples were prepared according to Chizzola et al. (2008) and Nourhene et al. (2009), using ethanol 60 % (v/v). Two grams of fresh material was treated with 15 mL of solvent, and extraction was done at 5 °C for 24 h. The extract was filtered and stored at –20 °C until analysis.
Antioxidant activity by the DPPH method. This activity was determined according to the procedure described by Chizzola et al. (2008) and Scherer and Texeira (2009). This test is based on the ability of the antioxidant to give off hydrogen radicals. The ethanol extracts samples of 400 µL were adjusted to 1 mL with methanol 50 %, and thereafter 1 mL of DPPH (2'2–diphenyl 1–1picrylhydrazyl) (2.43 x 10–4 M) was added. After 30 min in the dark at room temperature, absorbance was measured against a blank, at 517 nm. A sample of 2.5 mM of Trolox (6–hidroxy–2,5,7,8–tetramethylchromane–2 carboxilic acid) in methanol was used as a reference substance. The blank was a solution containing 500 µL of trolox and 500 µL of methanol, which reacted with 1 mL of DPPH reagent to obtain a full decoloration of the radical. For the calibration curve, 0.1 to 2 mM trolox in 1 mL of methanol was used. The trolox standard solution was prepared under the same conditions. The results are expressed as percentage of DPPH inhibition, according to the following equation:
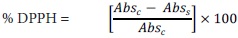
where Absc is the value of control absorbance, and Abss is the value of sample absorbance. A lower absorbance of the reaction mixture indicates a high free radical scavenging activity.
Total phenolic content was determined by the Folin–Ciocalteau method, according to Chizzola et al. (2008) with some modifications. The following reagents were used: caffeic acid, Folin–Ciocalteau reagent, sodium carbonate and ethanol. To 0.5 mL of the ethanolic extract, 1 mL of ethanol 95 % (v/v) and 5 mL of distilled water were added, plus 0.5 mL of Folin–Ciocalteau reagent diluted with distilled water 1:10. After 5 min, 1 mL of sodium carbonate solution in water (5 %, v/v) was added. The samples were mixed and kept 30 min in the dark, and then absorption was read at 725 nm. The blank was prepared by the same method using ethanol. The calibration curve was prepared using different concentrations of caffeic acid in ethanol. Total content of phenolic compounds in extract was expressed in |ig of caffeic acid equivalents (CAE) per 100 g of fresh plant material (FPM).
Content of flavonoids. It was measured according to Socha et al. (2009). To 1 mL of the extract solution, 5 mL of distilled water and 0.3 mL sodium nitrite solution (5 % v/v) were added. Then 0.3 mL of aluminium chloride solution (4 % v/v) was added, and after 5 min 2 mL NAOH (1 M) and distilled water were added to make up a total of 10 mL. The solution was mixed, and absorbance was measured immediately with a spectrophotometer against a the blank, at 510 nm. The blank was prepared with the same procedure using ethanol. The total flavonoid content was calculated on the basis of a standard curve of quercetin solutions. Results were expressed in |ig of quercetin equivalents (QE) per 100 g fresh plant material (FPM).
The experimental data were analyzed statistically by ANOVA (SAS Institute, 1999). Estimation of the significance of differences between means was based on a Tukey test (α = 0.05).
RESULTS AND DISCUSION
No visual differences in appearance or color were observed due to the use of humic substances on the crop. Nevertheless, differences (P ≤ 0.05) due to the effect of moisture were observed among morphological variables, except for shoot fresh weight and its interaction with HS (Table 1).
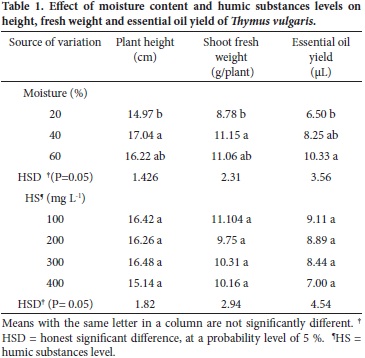
Plant height was affected (P ≤ 0.05) by moisture contents of 40 and 20 % (Table 1), suggesting that maintaining a high moisture level is important for plant growth. These results can be explained by the fact that differential provision of water to plants influences their morphology, physiology and dry matter partitioning between roots and shoot. Exposure to a water deficit has caused a significant decrease in plant height and weight (Sanchez–Blanco et al., 2008). In our study, plant height ranged between 14.9 and 17.0 cm.
Plant weight ranged from 8.78 to 11.15 g/plant, and it increased (P ≤ 0.05) as the irrigation level was increased also. The highest values were found with soil moisture contents of 40 % (M2) and 60 % (M3) of field capacity. A similar pattern was reported for thyme, hyssop (Hyssopus officinalis L.) (Khazaie et al., 2008) and oregano (Origanum vulgare L.) (Azizi et al., 2009). This response can be attributed to the availability of enough moisture for the root system.
There was a little effect of the HS level on the thyme yield, since very similar values of total plant fresh weight were observed (Table 1). There were no significant differences among HS levels regarding shoot dry weight and root fresh and dry weight, so their mean values are not shown here.
The essential oil yield (µL) varied significantly (P ≤ 0.05) with the soil moisture level, but not with the HS level nor with the interaction. The yield of essential oil was highest at 60 % of soil moisture (Table 1), effect that can be attributed to the favorable moisture conditions maintained during the crop growth. The oil yield was reduced by water stress which also affected crop growth adversely. In other studies it has been found that water supply significantly affects the yield and quality of essential oils in patchouli (Pogostemon cablin), as reported by Sing et al. (2002). Similarly, Azizi et al. (2009) indicated that an optimal water supply during the seedling development and stem elongation, followed by a restriction of water supply after flowering, may increase the content of essential oil and thus improve quality in oregano herbage. The lack of effect of HS on the accumulation of essential oils might have been modified by plant age, as reported by Khazaie et al. (2008); on this regard, the plants were 60 d old in our study.
The extracted oils were yellow and had a distinct sharp odor. The identified components were: β pinene (6.615), (+)–limonene (9.492), 3–octanol (15.215), (–)–thujone (16.399), linalool (20.087), thymol (36.310) and carvacrol (36.976), since their retention times (Rt) were the same as their external standards. The moisture percentages and levels of humic substances had a positive effect on essential oil components (Table 2).
The most abundant compounds in the extract were thymol and (+)–limonene, since both of them represented 72.3 % of the total volatiles quantified. Generally, thymol, a phenolic monoterpene, defines the essential oil quality because of its active pharmacological properties (Bozin et al., 2006; Loziene et al., 2007). Similar results have been reported by Nejad et al. (2008) who found the lowest content of carvacrol and high amounts of thymol during the vegetative stage of Thymus caramanicus. The essential oil composition varied (P ≤ 0.05) with the moisture percent and with the amount of humic substances supplied (Table 2). The soil moisture did not affect the production of thymol, but it did modify the concentration of the other components. With 300 and 400 mg L–1 of HS applied in the irrigation water, the concentrations of thymol (5.8 %) and (–)–thujone (11.4 %) were higlier (P ≤ 0.05) than at 100 or 200 mg L–1 of HS, while the opposite happened for carvacrol (33.8 %) and linalool (22.6 %).
The thyme antioxidant activity showed significant differences (P ≤ 0.05) among moisture and HS levels, being highest with 60 % of moisture and 400 mg L–1 of HS (Table 3). The total phenols and flavonoid contents varied also due to soil moisture and HS level (P ≤ 0.05).
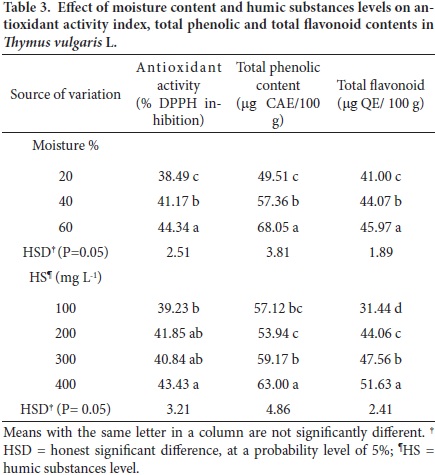
Water supply is one main factors in determining yield and composition of bioactive metabolites in several spices and herb crops (Azizi et al., 2009; Jordán et al., 2009; Sanchez–Blanco et al., 2008; Singh et al., 2002). In our study the highest inhibition of the free radical DPPH was attained with a moisture content of 60 % and 400 mg L–1 of humic substances. Conversely, the lowest values of this activity was found with 20 % of water content and 100 mg L–1 of humic substances. Nevertheless, the total antioxidant activity of DPPH determined in this study was lower (43.4 % DPPH inhibition) than the 55.9 % reported by Chizzola et al. (2008). The plant age may have affected this reaction.
The contents of phenolic compounds varied (P ≤ 0.05) among moisture levels, ranging from 49.5 to 68.0 µg CAE/ 100 g of fresh plant material (Table 3). The highest value was found at 60 % of soil moisture. This trend is in agreement with data reported by Jordán et al. (2009), whom indicated that an increase in watering favors the total phenolic content and the radical scavenging activity of DPPH. In this regard, Strail et al. (2006) claim that the antioxidant activity properties of plant extracts are associated to the presence of phenolic compounds with ability to donate hydrogen for reducing DPPH radicals. Our values are lower, however, than those reported by Chizzola et al. (2008) who found an average value of 65.2 µg g–1 caffeic acid in T. vulgaris plants. The humic levels also affected significantly the contents of total phenolics; the highest value (63 mg CAE/100 g) was found with 400 mg L–1, while the lowest (53.9 µg CA/100 g) was found for 100 mg L–1.
The total flavonoid content of thyme was higher (P ≤ 0.05) in the 60 % moisture level, with a value of 45.9 µg QE/100 g of fresh matter, compared to the moisture content of 20 % with 41 µg QE/100 g (Table 3). There was also a significant effect of the level of humic substances supplied. The lowest value was found for 100 mg L–1 (HS1), while the highest value was found for 400 mg L–1 (HS4). These results support previous observations (Scherer and Texeira, 2009; Socha et al., 2009; Strail et al., 2006) regarding the antioxidant activity of flavonoids.
CONCLUSIONS
Optimal water supply during the vegetative stage of thyme plants increases biomass yield and essential oil yield contents, since at 60 % of moisture level the antioxidant activity, total phenolic content and total flavonoid is increased. The level of humic substances supplied with watering also affects the biomass and the concentration of thymal, carvacrol, linalool and (–) thujone. At 400 mg L–1 of humic substances there were higher values for antioxidant activity, total flavonoid and total phenolic contents, compared to lower rates of these substances.
BIBLIOGRAPHY
Azizi A, F Yan, B Honormeier (2009) Herbage yield, essential oils content and composition of three oregano (Origanum vulgare L.) populations as affected by soil moisture regimes and nitrogen supply. Ind. Crops Prod. 29:554–561. [ Links ]
Baranauskiene R, P R Venskutonis, P Vikellis, E Dambrauskien (2003) Influence of nitrogen fertilizers on the yield and composition of thyme (Thymus vulgaris). J. Agric. Food Chem. 51: 7751–7758. [ Links ]
Bozin B, N Mimica–Dukic, N Simin, G Anackov (2006) Characterization of the volatile composition of essential oils of some Lamiaceae species on the antimicrobial and antioxidant activities of the entire oils. J. Agric. Food Chem. 54:1822–1828. [ Links ]
Burt S (2004) Essential oils: their antibacterial properties and potential applications in foods–a review. Internat. J. Food Microbiol. 94:223–253. [ Links ]
Chen Y, C E Clapp, H Magen (2004) Mechanisms of plant growth stimulation by humic substances: the role of organic iron complexes. Japan Soc. Soil Sci. Plant Nutr. 50:1089–1095. [ Links ]
Chizzola R, H Michitsch, C Franz (2008) Antioxidative properties of Thymus vulgaris leaves: Comparison of different extracts and essential oil chemotypes. J. Agric. Food Chem. 56:6897–6904. [ Links ]
Eyheraguibel B, J Silvestre, P Morard (2008) Effects of humic substances derived from organic waste enhancement on the growth and mineral nutrition of maize. Biores. Technol. 99:4206–4212. [ Links ]
Figueiredo A C, J G Barroso, L G Pedro, J J C Scheffer (2008) Factors affecting secondary metabolite production in plants: volatile components and essential oils. Flav. Frag. J. 23:213–226. [ Links ]
Jordán M J, R M Martínez, C Martínez, I Monino, J A Sotomayor (2009) Poliphenolic extract and essential oil quality of Thymus zygis spp. Gracilis shrubs cultivated under different watering levels. Ind. Crops Prod. 29:145–153. [ Links ]
Khazaie H R, F Nadjafi, M Bannayan (2008) Effect of irrigation frequency and planting density on herbage biomass and oil production of thyme (Thymus vulgaris) and hyssop (Hyssopues officinalis). Ind. Crops Prod. 27:315–321. [ Links ]
Lee S J, K Umano, T Shibamoto, K G Lee (2004) Identification of volatile components in basil (Ocinum basilicum L.) and thyme leaves (Thymus vulgaris L.) and their antioxidant properties. Food Chem. 91:131–137. [ Links ]
Loziene K, P R Venkutonis, A Sipailiene, J Labokas (2007) Radical Scavenging and antibacterial properties of extracts from different Thymus pulegioides L. chemotypes. Food Chem. 103:546–559. [ Links ]
Nardi S, D Pizzeghello, A Cuscolo, A Vianello (2002) Physiological effects of humic substances on higher plants. Soil Biol. Biochem. 34:1527–1536. [ Links ]
Nejad E. S, J Haddian, M H Mirjalilli, A Sonboli, M Yousefzadi (2008) Essential oil composition and antibacterial activity of Thymus caramanicus at different phenological stages. Food Chem. 110:927–931. [ Links ]
Nikbakht A, L Ancheng, E. Nemat–allah, X Yi Ping, K Mohsen, B Mesbah (2008) Effect of humic acid on plant growth, nutrient uptake and postharvest life of gerbera. J. Plant Nutr. 31:2155–2167. [ Links ]
Nourhene B, N Bahloul, I B Slimen, N Kechaou (2009) Comparison on the total phenol contents and the color of fresh and infrared dried olive leaves. Ind. Crops Prod. 29:412–419. [ Links ]
Sanchez–Blanco M J, S Alvarez, A Navarro, S Banon (2008) Changes in leaf water relations, gas exchange, growth and flowering quality in potted geranium plants irrigated with different water regimes. J. Plant Physiol. 166:467–476. [ Links ]
SAS Institute (1999) The SAS System for Windows. Release 8. Cary North Carolina. USA. 1643 p. [ Links ]
Scherer R, H Teixeira (2009) Antioxidant activity index (AAI) by the 2,2–diphenyl–1–picrylhydrazyl method. Food Chem. 112:654–658. [ Links ]
Siminis C, M Loulakis, M Kefakis, T Manios, V Manios (1998) Humic Substances from compost affects nutrient accumulation and fruit yield in tomato. Acta Hort. 469:353–358 [ Links ]
Singh M, S Sharma, S Ramesh (2002) Herbage, oil yield and oil quality of patchouli [Pogostemon cablin (Blanco) Benth.] influenced by irrigation, organic mulch and nitrogen application in semiarid tropical climate. Ind. Crops Prod. 16:101–107. [ Links ]
Socha R, L Juszczak, S Pietrzyk, T Fortuna (2009) Antioxidant activity and phenolic composition of herbhoneys. Food Chem. 113: 568–574. [ Links ]
Strail P B, Klejdus, V Kuban (2006) Determination of total content of phenolic compounds and their antioxidant activity in vegetables–evaluation of spectrophotometric methods. J. Agric. Food Chem. 54:607–617. [ Links ]














