INTRODUCTION
As a novel class of antidiabetic medication, incretin-based drugs, including glucagon-like peptide 1 receptor agonists (GLP-1 RAs) and dipeptidyl peptidase 4 inhibitors (DPP-4Is), are promising agents for the treatment of type 2 diabetes (T2DM)1,2. GLP-1 RAs are mainly known to stimulate glucose-dependent insulin secretion, suppress glucagon release, and inhibit gastrointestinal motility, whereas DPP-4Is prevent the breakdown and inactivation of GLP-1 and glucose-dependent insulinotropic polypeptide to improve blood glucose. Studies have shown that incretin-based drugs not only enhance blood glucose management without increased weight gain or hypoglycemia but also improve beta-cell function and insulin resistance in patients with T2DM2,3.
Recently, studies had shown that incretin-based therapies could reduce the levels of pro-inflammatory markers, which were thought to play a role in the development of insulin resistance4. Acute glucose excursion and post-prandial glucose excursion could exacerbate inflammation, which increased the risk of diabetic complications5,6. As one of the acute phase proteins, high-sensitive C-reactive protein (hs-CRP) was reported to be elevated in T2DM7. Although there have been two meta-analyses to explore the effect of GLP-1 RAs8 and DPP-4Is9 on CRP, the specific mechanism has not been explored. This paper further discussed the role of factors such as body mass index (BMI), blood glucose, and medication time on the effect of incretin-based therapies on CRP. Besides, this paper has added the data of the newly published literature so as to analyze the effects of incretin-based therapies on serum hs-CRP in T2DM more credibly. Hence, the aim of this study was to assess the reported effects of incretin-based therapies on serum hs-CRP by systematically reviewing the existing randomized control trials.
METHODS
Search strategy
All randomized clinical trials (RCTs) investigating the effects of incretin-based therapies (including GLP-1 RAs and DPP-4Is) on serum hs-CRP concentrations in adults with T2DM were identified by comprehensive computer-based searches of PubMed, EMBASE, Cochrane Library, and Web of Knowledge databases until August 2019 without restrictions of language. The search was performed using various combinations of keywords like (dipeptidyl peptidase-4 inhibitors OR dpp-4 inhibitor OR dutogliptin OR alogliptin OR linagliptin OR saxagliptin OR sitagliptin OR vildagliptin) OR (glucagon-like peptide 1 receptor agonists OR glp-1 agonists OR exenatide OR dulaglutide OR liraglutide OR taspoglutide OR incretin) AND (T2DM) AND (Controlled Clinical Trial[Publication Type]). The exact search was available on request from the authors. Additional studies were also identified by a hand search of all the references of retrieved articles.
Study selection
RCTs were eligible for this meta-analysis if they met the following inclusion criteria: (1) RCTs were human randomized, controlled trials, (2) adult patients aged more than 18 years with T2DM, and (3) hs-CRP change was reported for the intervention and control groups. We did not consider abstracts or unpublished reports. Case reports, editorials, review articles, and letters were excluded from the study. Articles were excluded if they did not include a control population. The present study was performed according to PRISMA guidelines.
Data extraction
Two researchers conducted an initial screening of studies independently. The next phase involved a review of abstracts and an examination of the full text in terms of the eligibility criteria. The final eligibility of the articles was determined through the agreement of the two reviewers. The following characteristics were extracted from each study: the first author, year of publication, study design, study duration, and baseline measures and changes from baseline of hs-CRP in intervention and control groups. Meanwhile, we did not define any minimum number of interventions or controls to be included in our meta-analysis.
Study quality assessment and risk of bias
The risk of bias of all included studies was assessed by the Cochrane Collaborations tool for assessing the risk of bias of randomized trials10. The risk of bias assessment of all included articles is shown in Table S1 (Supplementary Information).
Statistical analysis
The standard deviations of the changes of serum hs-CRP from baseline were estimated from the sample size and the standard error or the 95% confidence interval (CI) when the RCTs involved in this meta-analysis did not report them. Standard mean differences (SMDs) and 95% CIs were calculated to evaluate the influence of incretin-based therapies on serum hs-CRP levels due to different scales or units. The pooled SMD of each study was calculated by the random effects model (using the DerSimonian-Laird method) regardless of heterogeneity. Heterogeneity was quantitatively assessed using the I2 index.
Sensitivity analysis, subgroup analysis, and meta-regression were also to explore the source of heterogeneity. In addition, we assessed the probability of publication bias with funnel plots and Eggers test, with p < 0.1 considered representative of statistically significant publication bias. Statistical analysis was conducted using STATA 11.0 (Stata, College Station, TX, USA). The p < 0.05 was considered statistically significant.
RESULTS
A total of 2,164 records were identified, 1005 of which were excluded due to duplicates. Moreover, 1,101 articles were removed after reviewing titles and abstracts. An additional 33 articles were then removed for not satisfying the inclusion criteria (n = 7, not hs-CRP; n = 13, no data; n = 1, geometric data; n = 5, no control; n = 3, no baseline data; n = 4, not RCT). A flow chart on article selection for the meta-analysis is shown in figure 1.

Figure 1 The flow chart on article selection for the meta-analysis, a total of 2,164 records was identified, and 25 studies were included in this meta-analysis finally.
Characteristics of the included studies
Twenty-five studies with 28 trials, with 2,851 participants in total, were included in the final systematic review and meta-analysis. Twenty-five of the included studies were parallel randomized controlled trials, with eight studies reporting double-blind design, eight studies reporting open-label design, and one study reporting single-blind design. Another two included studies were cross-over randomized, controlled trials with open-label design. The characteristics of the 25 enrolled studies are shown in Table 1. The 25 studies were all published between 2010 and 2017. Of 25 studies, 12 used DPP-4Is as intervention11-25, 13 used GLP-1 RAs26-39. Among 12 studies concerning DPP-4Is, sitagliptin (dose range of 25-100 mg once daily), linagliptin (with 5 mg once daily), and vildagliptin (all with 50 mg twice a day) were studied with 8, 1, and 3 studies, respectively. In view of the 13 studies on GLP-1 RAs, exenatide (a dose of 5 µg twice daily for a period of 4 weeks, followed by a dose increase to 10 µg twice daily), liraglutide (range of 0.6-1.8 mg once daily), and taspoglutide (20 mg subcutaneously once weekly after 10 mg for the first 4 weeks) were studied in 6, 6, and 1 studies separately. The total number of individuals enrolled in each of the 25 studies ranged from 20 to 740. The duration of interventions ranged from 12 weeks to 104 weeks, with a median follow-up of 24 weeks. The mean age for the included studies was between 47.7 and 68.4 years. The mean disease duration of patients with diabetes for the included studies ranged from 5.4 months to 17.3 years.
Table 1 The characteristics of included trials
| Study (year) | Design | Background medications | Therapy duration | Intervention | Participants | Age (y) | Diabetes duration | CRP at baseline | BMI at baseline |
|---|---|---|---|---|---|---|---|---|---|
| Park KS (2017) | Open-label; Cross-over | Metformin | 12 w | I: Vildagliptin | 16 | 60.0 ± 9.6 | 7.4 ± 5.2 y | Log(hs-CRP): 0.42 ± 0.72 nmol/L | 25.5 ± 4.1 |
| C: Glimepiride | 16 | ||||||||
| Strozik A (2015) | Parallel | None | 12 w | I: Vildagliptin + Metformin (1.5 g) | 15 | 45.9 ± 4.6 | | hs-CRP: 2.8 ± 0.2 g/l | 28.2 ± 1.8 |
| C: Metformin (1.5 g) | 13 | 51.4 ± 7.2 | hs-CRP: 2.7 ± 0.4 g/l | 29.0 ± 3.5 | |||||
| I: Vildagliptin + Metformin (3.0 g) | 17 | 49.3 ± 4.4 | hs-CRP: 3.1 ± 0.1 g/l | 30.5 ± 1.5 | |||||
| C: Metformin (3.0 g) | 16 | 58.2 ± 2.7 | hs-CRP: 3.0 ± 0.2 g/l | 33.3 ± 2.2 | |||||
| Takihata M (2013) | Open-label; Parallel | None | 24 w | I: Sitagliptin | 58 | 60.3 ± 7.5 | | hs-CRP: 1388 ± 3095 ng/ml | 24.6 ± 3.3 |
| C: Pioglitazone | 57 | 60.7 ± 9.5 | hs-CRP: 1953 ± 3860 ng/ml | 25.8 ± 4.8 | |||||
| Bunck MC (2010) | Parallel | Metformin | 1 y | I: Exenatide | 30 | 58.4 ± 8.4 | 5.7 ± 4.8 y | hs-CRP: 1.81 ± 0.25 mg/l | 30.28 ± 1.44 |
| C: Glargine | 29 | 58.3 ± 7.5 | 4.0 ± 3.4 y | hs-CRP: 1.42 ± 0.27 mg/l | 30.34 ± 1.96 | ||||
| Derosa G (2012) | Open-label; Parallel | Metformin | 12 m | I: Exenatide | 81 | 57.3 ± 7.7 | 7.6 ± 2.8 y | hs-CRP: 2.0 ± 0.8 mg/l | 31.9 ± 1.7 |
| C: Placebo | 82 | 56.7 ± 7.3 | 7.8 ± 3.1 y | 31.7 ± 1.5 | |||||
| Shigiyama F (2017) | Parallel | None | 16 w | I: Linagliptin + Metformin | 29 | 60.4 ± 9.0 | | hs-CRP: 1372.8 ± 2489.4 ng/ml | 25.3 ± 4.4 |
| C: Metformin | 25 | 60.3 ± 12.3 | hs-CRP: 1743.3 ± 2586.2 ng/ml | 26.2 ± 4.0 | |||||
| Dutour A (2016) | Open-label; Parallel | Oral medicine | 26 w | I: Exenatide | 22 | 51 ± 2 | 4 (2-8) y | hs-CRP: 11.65 ± 10.36 mg/l | 37.2 ± 1.7 |
| C: Conventional treatment | 22 | 52 ± 2 | 4 (1-10) y | hs-CRP: 9.96 ± 11.01 mg/l | 31.7 ± 1.5 | ||||
| Wu JD (2011) | Double-blind; Parallel | None | 16 w | I: Exenatide | 12 | 54 ± 9.5 | 5.0 ± 2.5 y | hs-CRP: 0.4 ± 0.5 mg/l | 26.3 ± 1.9 |
| C: Placebo | 11 | 57 ± 10 | 7.3 ± 4.4 y | hs-CRP: 0.6 ± 0.4 mg/l | 26.3 ± 3.0 | ||||
| Nakamura K (2014) | Parallel | None | 12 w | I: Sitagliptin | 24 | 66.6 ± 11.9 | 57.6 ± 41.2 m | hs-CRP: 2194.4 ± 4079.1 | 27.8 ± 3.5 |
| C: Voglibose | 31 | 68.4 ± 9.2 | 41.9 ± 44.1 m | hs-CRP: 2052.2 ± 3816.2 | 25.7 ± 4.3 | ||||
| Mita T (2016) | Open-label; Parallel | None | 104 w | I: Sitagliptin | 122 | 63.8 ± 9.7 | 17.2 ± 8.5 y | hs-CRP: 506 (210-1310) ng/dl | 25.0 ± 4.3 |
| C: Conventional treatment | 121 | 63.6 ± 1.0 | 17.3 ± 8.7 y | hs-CRP: 487 (277-1004) ng/dl | 25.0 ± 3.8 | ||||
| Nomoto H (2015) | Open-label; Parallel | None | 14 w | I: Liraglutide | 16 | 61.1 ± 8.3 | | | 26.6 (23.6-29.9) |
| C: Glargine | 15 | 59.5 ± 12.3 | 25.8 (24.2-28.2) | ||||||
| Fan H (2013) | Parallel | None | 12 w | I: Exenatide | 49 | 51.02 ±10.10 | | hs-CRP: 3.14 ± 0.58 mg/l | 28.18 ± 1.86 |
| C: Metformin | 68 | 54.68 ± 12.14 | hs-CRP: 3.16 ± 0.68 mg/l | 27.61 ± 1.77 | |||||
| Oe H (2015) | Open-label; Parallel | None | 24 w | I: Sitagliptin | 40 | 67.8 ± 10.5 | 48 (6-240) m | hs-CRP: 3869 ± 9072 | 27.7 ± 4.1 |
| C: Voglibose | 40 | 66.7 ± 9.8 | 38.5 (3-28) m | hs-CRP: 1358 ± 2511 | 25.7 ± 4.3 | ||||
| Kato H (2015) | Open-label; Parallel | None | 24 w | I: Sitagliptin | 10 | 62 (56-70) | | hs-CRP: 0.18 (0.05-0.23) mmol/l | 25.6 (24.7-32.5) |
| C: Glimepiride | 10 | 55 (42-62) | hs-CRP: 0.19 (0.07-0.28) mmol/l | 26.6 (25.0-32.4) | |||||
| Derosa G (2010) | Single-blind; Parallel | None | 12 m | I: Exenatide | 59 | 57 ± 8 | | hs-CRP: 2.1± 0.8 mg/l | 28.7 ± 1.5 |
| C: Glimepiride | 57 | 56 ± 7 | hs-CRP: 2.0 ± 0.7 mg/l | 28.5 ± 1.4 | |||||
| Nomoto H (2016) | Open-label; Parallel | None | 26 w | I: Sitagliptin | 41 | 62 (35-80) | | | 25.7 ± 3.9 |
| C: Glimepiride | 49 | 60 (36-60) | 25.2 ± 3.5 | ||||||
| Faurschou A (2015) | Double-blind; Parallel | None | 8 w | I: Liraglutide | 11 | 54 ± 14 | | hs-CRP: 5.4 ± 4.4 mg/l | 37.0 ± 8.2 |
| C: Placebo | 9 | 48 ± 12 | hs-CRP: >3.8 ± 5.4 mg/l | 35.0 ± 11.5 | |||||
| Derosa G (2012) | Double-blind; Parallel | Metformin | 12 m | I: Vildagliptin | 84 | 54.2 ± 8.3 | 6.1 ± 3.7 m | hs-CRP: 1.9 ± 2.0 mg/l | 27.9 ± 1.5 |
| C: Placebo | 81 | 52.4 ± 7.1 | 6.3 ± 3.9 m | hs-CRP: 1.7 ± 0.8 mg/l | 27.8 ± 1.4 | ||||
| Derosa G (2012) | Double-blind; Parallel | Metformin | 12 m | I: Sitagliptin | 86 | 55.9 ± 8.8 | 5.8 ± 2.6 m | hs-CRP: 1.8 ± 0.7 mg/l | 28.1 ± 1.2 |
| C: Placebo | 83 | 54.8 ± 7.9 | 5.4 ± 2.3 m | hs-CRP: 2.0 ± 0.9 mg/l | 28.9 ± 2.0 | ||||
| Hollander P (2013) | Double-blind; Parallel | None | 24 w | I: Taspoglutide | 149 | 53.0 ± 10 | 5.2 ± 4.3 y | hs-CRP: 5.57 mg/l | 36.9 ± 5.0 |
| C: Placebo | 143 | 54 ± 10 | 4.9 ± 4.1 y | hs-CRP: 6.73 mg/l | 36.5 ± 4.8 | ||||
| Liang Z (2013) | Parallel | None | 12 m | I: Exenatide | 34 | 50.94 ± 5.89 | 7.17 ± 1.80 y | hs-CRP: 3.14 ± 1.14 mg/l | 30.9 ± 0.7 |
| C: Conventional treatment | 36 | 51.75 ± 6.70 | 7.24 ± 1.61 y | hs-CRP: 2.91± 1.55 mg/l | 30.1 ± 0.6 | ||||
| Seino Y (2012) | Double-blind; Parallel | None | 24 w | I: Liraglutide | 264 | 58.2 ±10.4 | 8.1 ± 6.7 y | hs-CRP: 0.1052 mg/dl | 24.5 ± 3.7 |
| C: Glimepiride | 129 | 58.5 ±10.4 | 8.5 ± 6.8 y | 24.4 ± 3.8 | |||||
| Koren S (2012) | Open-label; Cross-over | None | 3 m | I: Sitagliptin | 20 | 59 ± 10 | 7.8 ± 5.0 y | hs-CRP: 4.7 ± 6.0 mg/l | 31 ± 5 |
| C: Glimepiride | 20 | ||||||||
| Kaku K (2010) | Double-blind; Parallel | Sulfonylureas | 24 w | I: Liraglutide | 176 | 60.2 ± 10.6 | 10.5 ± 6.7 y | hs-CRP: 0.1145 ± 0.1312 mg/dl | 30.5 ± 1.5 |
| C: Placebo | 88 | 58.6 ± 9.7 | 10.1 ± 7.3 y | hs-CRP: 0.1478 ± 0.1523 mg/dl | 33.3 ± 2.2 | ||||
| Nandy D (2014) | Double-blind; Parallel | None | 12 w | I: Liraglutide | 16 | 57.7 ± 9.0 | 5.3 ± 4.1 y | - | 32.7 ± 4.5 |
| C: Placebo | 14 | 60.3 ± 7.3 | 8.4 ± 4.6 y | 31.6 ± 4.2 | |||||
| C: Glimepiride | 17 | 57.7 ± 5.3 | 6.8 ± 8.1 y | 31.1 ± 4.9 |
I: intervention group; C: control group; hs-CRP: high-sensitive C-reactive protein; w: weeks; m: months; y: years; Data are presented as the mean ± SD or median (range). SD: standard deviation.
Pooled estimate of incretin treatment on hs-CRP
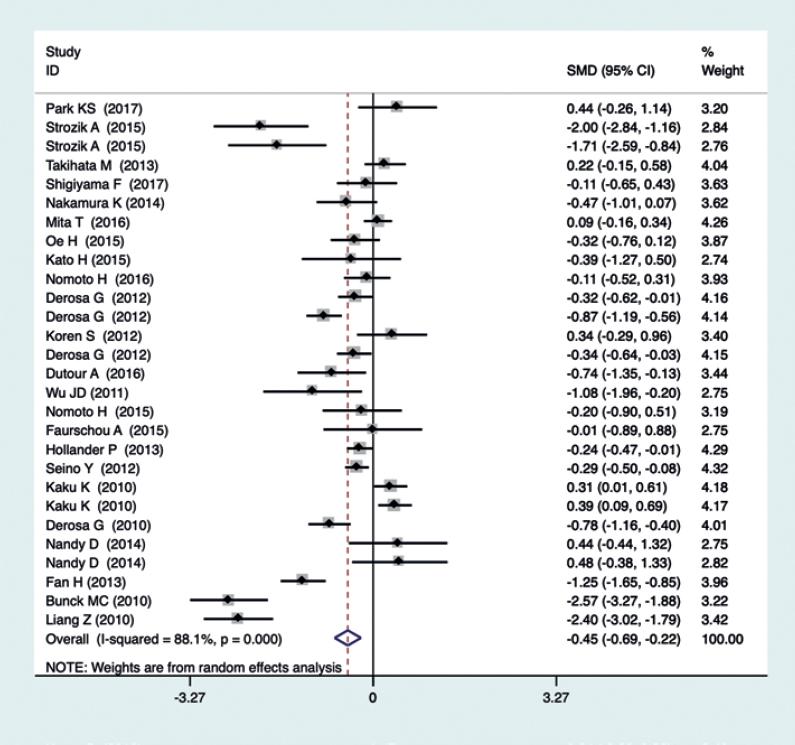
Of the 25 studies reporting changes in hs-CRP following treatment with incretin, hs-CRP decreased significantly in incretin group compared with the control group (SMD = −0.452, 95% CI = −0.688-−0.217, p < 0.001), with high heterogeneity (I2 = 88.1%, p < 0.001). The forest plot of the effect was presented in figure 2.
Evaluation of heterogeneity and sensitivity analysis
In exploratory analyses (Table S2, Supplementary Information), meta-analysis of trials excluding open and crossover studies also found a significant reduction of hs-CRP by 0.45 (95% CI = −0.95-−0.33, p < 0.001). The subgroup analysis showed that therapy with both DPP-4Is (SMD = −0.338, 95% CI = −0.635-−0.041, p = 0.026) and GLP-1 RAs (SMD = −0.544, 95% CI = −0.908-−0.181, p = 0.003) caused a significant reduction in hs-CRP versus all comparators. Subgroup analysis showed that there was no significant hs-CRP decrease in the subgroup with the intervention for a duration less than 24 weeks (SMD = −0.423, 95% CI = −0.889-0.043, p = 0.075), while hs-CRP decreased remarkably in the other subgroup (duration of intervention ≥24 weeks) (SMD = −0.465, 95% CI = −0.748-−0.182, p = 0.001) comparison with the placebo control group; the subgroup analysis based on mean baseline BMI showed that in the group with BMI more than 30 kg/m2, there was significant difference in the reduction in hs-CRP after incretin-based treatment (SMD = −0.680, 95% CI = −1.251-−0.110, p = 0.019). In the group with BMI <30 kg/m2, hs-CRP was also significantly decreased after incretin-based treatment (SMD = −0.330, 95% CI = −0.576-−0.082, p = 0.009).
Meta-regression analyses were performed to evaluate the extent to which different variables explained the heterogeneity. The results revealed that the heterogeneity could be explained by better glycemic control (linear regression coefficient = −0.993 [95% CI, −1.671-−0.316], p = 0.006) and more BMI decline (linear regression coefficient = −0.325 [95% CI, −0.605-−0.044], p = 0.027) in the effect of incretin on hs-CRP. However, age of subjects, year of publication, serum lipids, and sample size were not statistically correlated with heterogeneity (p > 0.05).
Publication bias diagnostics
We further identified the potential publication biases of literatures by Eggers test and funnel plot. In all trials, the shapes of the funnel plot indicated no obvious asymmetry (Fig. 3) and Eggers test provided statistical evidence for the funnel plot symmetry. No significant publication bias was found in the trials (p = 0.103).
DISCUSSION
In the present study, we used a complete search, integration, and analysis of data to perform a systematic assessment regarding the effects of incretin-based drugs treatment on hs-CRP compared to placebo or active drugs in patients with T2DM. Our meta-analysis, with 25 studies involving a total 2,851 participants, suggested that incretin-based regimens, both GLP-1 RAs and DPP-4Is, were associated with a significant reduction in hs-CRP compared with controls. These data strongly support the benefits of incretin-based therapy in improving inflammation in patients with T2DM.
The duration of incretin-based drugs intervention and mean baseline BMI varied between the trials included in this meta-analysis. Subgroup analysis indicated that a greater reduction in hs-CRP was observed following treatment when the duration of intervention was longer than 24 weeks. The previous meta-analysis by Mazidi et al. also showed that changes in serum CRP concentration following treatment with GLP-1 RAs were associated with the duration of treatment8. It is possible that intervention duration <24 weeks is not sufficient to observe an obvious change in hs-CRP. Subgroup analysis showed that incretin-based therapies significantly reduced serum hs-CRP levels regardless of the mean baseline BMI level greater than or <30 kg/m2. It is suggested that the effect of incretin-based drugs on hs-CRP is independent of baseline BMI, but the effect of incretin-based drugs on hs-CRP reduction seems to be higher in subgroup analysis with BMI greater than 30 kg/m2.
The impact of incretin-based drugs on serum hs-CRP concentration may work through several different reasons. First, it may be the result of reduced body weight. In this paper, it showed that more BMI decline was associated with hs-CRP reduction. It has been well-established that GLP-1 RAs can significantly reduce the body weight in T2DM, which improves chronic inflammation in visceral adipose40. CRP expression is influenced by pro-inflammatory cytokines secreted by visceral adipose tissue, such as tumor necrosis factor-a and interleukin-6. Second, it is due to the fact that incretin-based therapies improve glycemic excursion. In this paper, we found that better glycemic control was associated with hs-CRP reduction. Acute glucose migration following an oral glucose tolerance test increases the level of monocyte NF-κB, which is the main cellular signal of inflammation and induces the transcription of pro-inflammatory cytokines and enzymes that generate reactive oxygen species41. Previous studies have shown that CRP levels in diabetic patients increased significantly after an oral glucose tolerance test42. Moreover, incretin-based drugs might have direct effects on inflammation. In fundamental studies, GLP-1 RAs can directly inhibit NF-κB pathway and the secretion of inflammatory cytokines in macrophages43. Besides, GLP-1 RAs also improved inflammation in adipose tissue by improved angiogenesis and microcirculation in obesity44. Moreover, inhibiting DPP-4 with linagliptin can polarize macrophages and form an anti-inflammatory phenotype in adipose tissue, thereby reducing inflammation and insulin resistance caused by obesity45. Sitagliptin, an available DPP4 inhibitor drug, showed multidimensional anti-inflammatory effects among diabetic patients mostly by affecting on NF-κB signaling pathway46.
The following limitations, however, should be acknowledged. First, we cannot conduct a subgroup meta-analysis to explore the effect of baseline hs-CRP level on the outcome due to the inconsistency of detection methods and different reference ranges of normal values. Second, some trials included in the present meta-analysis with small sample sizes reduced the power of those trials. Third, most trials in this meta-analysis were not specially designed to assess the effects of incretin-based therapies on inflammatory biomarker hs-CRP. Finally, some data for changes in hs-CRP could not be extracted directly. Although we get data by calculation based on the Cochrane Handbook for Systematic Reviews of Interventions, it still leads to deviations.
This meta-analysis shows that incretin-based drugs, both GLP-1 RAs and DPP-4Is, have potentially beneficial effects on inflammatory biomarker hs-CRP regardless of baseline BMI. Moreover, the effect is related to long intervention duration, better glycemic control, and more BMI decline.











 text new page (beta)
text new page (beta)