INTRODUCTION
The measurement of arterial blood gases (ABG) is a standard invasive test to evaluate oxygenation, alveolar ventilation, and acid-base status. Therefore, it is irreplaceable by non-invasive tests such as pulse oximetry or capnography, which are able to follow the tendency in oxygenation or alveolar ventilation, respectively, but neither is able to assess acid-base status, nor the three issues simultaneously. The correct interpretation of ABG requires a comparison of these with reference values unless utilized longitudinally, comparing them with time-changes in the same person. The values of ABG change with altitude above sea level and, although expected changes can be estimated1-4, empirical verification is essential because ventilatory acclimatization varies with ethnicity and a population's adaptation to altitude. For example, at a high altitude (about 4000 m above sea level [masl]), alveolar ventilation is higher in the Tibetan population than in the Andean population5.
The Valley of Mexico has been inhabited for more than 10,000 years6 and is of mixed ethnic origin, although in general, inhabitants are considered Mexican mestizos, with a variable contribution of Amerindian genes, and could have developed mechanisms of adaptation to altitude. In Mexico City (at 2,240 masl), a summary of reference values for ABG was reported in 1985 and has been utilized since that time7. However, several technological advances have taken place, such as the routine direct measurement of SaO2, and now blood gas analyzers include modules to rapidly measure hemoglobin, lactate, and electrolytes that complement typical ABG, especially in emergencies. The degree of hyperventilation, therefore, PaCO2 is especially relevant, and in addition, we wonder whether a previously described postmenopausal increase in PaCO2 in women8, occurs in Mexico at moderate altitude.
It is commonly understood that acute hyperventilation occurs due to pain and stress during the measurement of ABG. This response modifies the gas exchange steady state, and in principle, the impact of stress could be reduced if the arterial blood sample can be taken after the puncture and placement of the arterial catheter.
Our main objective was to describe ABG in a healthy population residing at 2240 masl, to identify the mean level of alveolar ventilation (PaCO2), and to know whether a progressive increase in PaCO2 occurs with age, especially in women, and the impact of increasing body mass index (BMI).
METHODS
We conducted a cross-sectional study approved by the Ethics Committee of the National Institute of Respiratory Diseases (INER) of Mexico, a public referral hospital for respiratory diseases and research institution that cares for mainly uninsured patients. All participants signed informed consent and agreed to provide an arterial blood sample, taken at the INER through a radial intra-arterial catheter. Individuals had to be healthy, never smokers, older than 15 years of age, residing in the Valley of Mexico for at least 2 years, without a respiratory diagnosis or symptoms. Information was obtained through a standardized questionnaire; all participants had normal spirometry.
All blood samples were obtained with the study participants at rest. An intra-arterial line was placed in the radial artery, in the non-dominant wrist, after performing an Allen maneuver to demonstrate adequate collateral circulation. The puncture site was cleaned with chlorhexidine, and a catheter (18 G, BD insyte™ 1.3 × 30 mm or 20G 1.1 × 30 mm) was set in place after being anesthetized with Xylocaine. Blood samples were taken through an adaptor with a valve (Smartsite Adaptor MLL) with a syringe lubricated with 0.1 mL of 1000 u/mL heparin. An arterial blood sample was taken at least 5 min after catheter placement, without pain referred by the subjects, and with a stable respiratory pattern (mean BF 14 ± 3 min-1). The blood sample was processed at the INER Pulmonary Function Laboratory with a permanent quality-assurance program (INER Laboratory with ISO-9000 Certification) in a blood gas analyzer (ABL 800 FLEX, Radiometer, Denmark), including daily calibration checks and test samples. The device includes an environmental monitor and adjusts automatically for barometric pressure and humidity.
We report variables measured by the analyzer, including PaO2, SaO2, PaCO2, and pH, but also some variables calculated by the analyzer, including HCO3- from PaCO2, and pH (Henderson Hasselbach equation) also directly influencing the calculation of extracellular Base Excess (BE usually calculated by the Van-Slyke equation). The Alveolar-arterial PaO2 gradient (P(A-a)O2) was also reported by the gas meter, but in addition, was obtained by subtracting the PaO2 from the estimated Alveolar PO2 (PAO2) using the alveolar gas equation: PΑO2=0.21*(Pbar-47)-(PaCO2/R)+0.21*(PaCO2/R)*(1-R) , where R, the respiratory quotient, was considered 0.8, common with a mixed diet and, for the barometric pressure, we considered the mean value of Mexico City, that is, 585 mmHg.
Individuals with inadequate blood samples for processing (insufficient or clotted or when the gas analyzer displayed an error) were excluded from the analysis.
Statistical analysis
Descriptive statistics included indicators of central tendency and dispersion and frequencies or percentages for dichotomic variables. Group comparison was performed by Student's t test or Wilcoxon test for independent samples, and the χ2 test was employed for categorical variables. Associations among variables were explored with the Pearson or Spearman correlation coefficient and with linear regression models of PaO2, SaO2, and P(A-a)O2 as dependent variables as a function of age, BMI (weight in kg divided by height2 in meters), minute ventilation, or breathing frequency. Statistical analysis was performed by means of the Stata V.13.1 statistical software package.
RESULTS
We studied 217 healthy subjects (113 males) with a mean age of 40 ± 15 years and a mean BMI of 25 ± 3 kg/m2. Of the total, 6% were below 20 years of age, 48% (104) were between 20 and 40 years of age, and 46% were above 40 years of age; in table 1, we described the main characteristics of the studied population. The main results of ABG are depicted in table 2 separated by gender, mean PaO2 (71 ± 6 mmHg), SaO2 (94% ± 1.6%), PaCO2 (30.2 ± 3.4 mmHg), HCO3 (20 ± 2 mmol/L), and BE (−2.9 ± 1.9 mmol/L) were below normal values at sea level, but not the pH (7.43 ± 0.02) or the alveolar-arterial PaO2 gradient (7.5 ± 6.9 mmHg).
Table 1. Main characteristics of the studied population
| Variable | Males 113 (52%) | Females 104 (48%) | p-value |
|---|---|---|---|
| Age (years) | 36.7 ± 14.7 | 43.5 ± 14.4 | <0.001 |
| Height (m) | 1.7 ± 0.1 | 1.6 ± 0.1 | <0.001 |
| Weight (kg) | 74.2 ± 11.6 | 61.2 ± 9.1 | <0.001 |
| BMI (kg/m2) | 25.5 ± 3.4 | 25.0 ± 3.6 | 0.21 |
| Body surface area (m2) | 1.9 ± 0.2 | 1.6 ± 0.1 | <0.001 |
| FEV1/FVC (%) | 82.3 ± 4.5 | 82.0 ± 4.3 | 0.71 |
| FEV1 (L) | 4.0 ± 0.7 | 2.8 ± 0.5 | <0.001 |
| FEV1 (% P)* | 103.2 ± 9.7 | 104.1 ± 13.3 | 0.58 |
| FVC (L) | 4.9 ± 0.8 | 3.8 ± 0.6 | <0.001 |
| FVC (% P)* | 102.5 ± 9.2 | 104.0 ± 11.7 | 0.29 |
Values are represented with means and standard deviation. BMI: Body Mass Index (Weight/Height2). Body surface by the Haycok formula of BSA = 0.024265*height 0.3964*weight 0.5378.
*Spirometry Reference Values from Haankinson et al. for Mexican-Americans.
Table 2. Mean measurements of arterial blood gases according to gender
| Measurements | All | Men | Women | p-value |
|---|---|---|---|---|
| pH | 7.43 ± 0.02 | 7.43 ± 0.02 | 7.43 ± 0.02 | 0.32 |
| PaCO2 (mmHg) | 30.2 ± 3.4 | 30.8 ± 3.3 | 29.6 ± 3.4 | 0.01 |
| PaO2 (mmHg) | 71.2 ± 5.9 | 71.1 ± 5.6 | 71.4 ± 6.2 | 0.75 |
| HCO3 (mMol/L) | 20.0 ± 2.00 | 20.3 ± 1.8 | 19.6 ± 2.1 | 0.01 |
| Base excess*(mMol/L) | −2.9 ± 1.9 | −2.5 ± 1.7 | −3.2 ± 2.0 | <0.001 |
| SaO2 (%) | 94.0 ± 1.6 | 94.0 ± 1.4 | 94.0 ± 1.7 | 0.63 |
| Lactate (mMol/L) | 1.2 ± 0.7 | 1.3 ± 0.7 | 1.0 ± 0.6 | <0.001 |
| P(A-a)O2**(mmHg) | 7.5 (3.2-11.6) |
5.9 (2.9-11.6) |
7.9 (3.7 - 12.1) |
0.12 |
*Estimated by the gas meter: HCO3-from the Henderson-Hasselbach equation and Base Excess (BE) from the Van Slyke equation.
**P(A-a)O2 estimated from the alveolar gas equation; median and InterQuartile Range (IQR).
With an increase in age, PaO2 decreases at 0.16 mmHg per year (95% CI: 0.11-0.21 mmHg) and SaO2 0.04% per year (95% CI: 0.03-0.06%), without differences per gender. The linear regression equation was PaO2 = 77.5-0.16*age; p < 0.0001; with aging, P(A-a) O2 tended to increase (0.12 mmHg/year) (Fig. 1).

Figure 1. Effect of age on gasometric variables in a healthy population at an altitude of 2240 masl. (A) SaO2 (%); (B) PaO2 (mmHg); (C) PaCO2 (mmHg); (D) HCO3 (mmol/L); (E) Base Excess (mmol/L), and (F) P(A-a)O2.
Overall, PaCO2 in women increased with age by 0.075 mmHg/year (p = 0.0012, linear regression PaCO2 = 26.3 + 0.075*age), but the age coefficient was not different statistically from zero in men (0.02; p = 0.35). Consequently, in younger participants, the average PaCO2 was lower in women than in men but after 60 years of age, the average level of PaCO2 was higher in women than in men (Fig. 2).
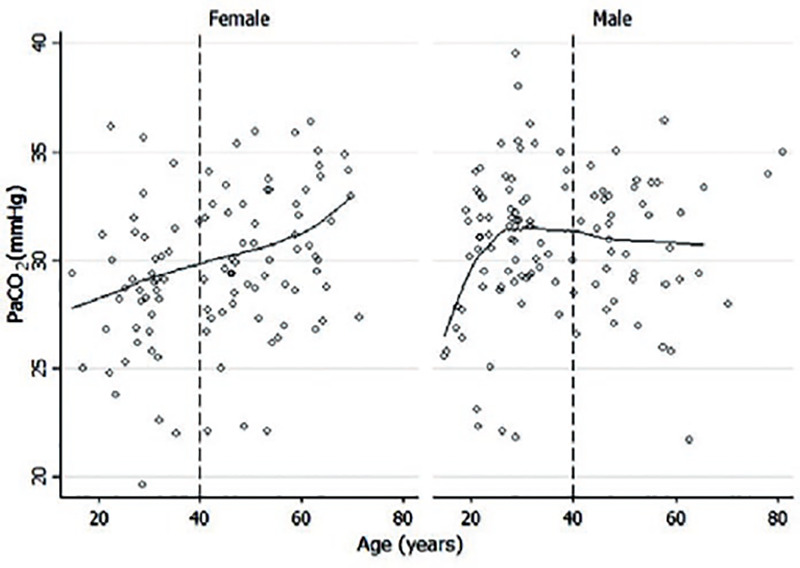
Figure 2. Arterial PaCO2 in female tends to increase with age at a rate of 0.10 mmHg/year after 40 years of age (95% CI, 0.003.0.2 mmHg; p = 0.04) in comparison with male, in whom that increase is no significant (0.02 mmHg/year) (95% CI, −0.08-0.12; p = 0.64).
SaO2 and PaO2 decreased significantly in women with higher BMI in a univariate analysis, (0.14% and 0.52 mmHg/kg/m2, respectively; p = 0.004 and 0.002). Both PaCO2 and the alveolar-arterial gradient tended to increase in women with a rising BMI (0.17 and 0.38 mmHg per kg/m2). However, after adjusting for age, changes in terms of BMI were not statistically significant (Fig. 3).
DISCUSSION
At an altitude of 2,240 masl, although PaO2 is considerably lower than at sea level, oxygen saturation remains on average above 90% (94%); thus, oxygenation at rest is preserved. Although SaO2 is above 90% on average, it is close to the knee of the hemoglobin-oxygen dissociation curve, and small decreases in PaO2 (expected, for example, with age, during sleep, or with heavy exercise) will lead to oxygen desaturation.
Mean PaCO2 was 30.7 mmHg, implying a mean alveolar ventilation of around 30%, above that at sea level (assuming a PaCO2 of 40 at sea level), with the result of a considerable increase in PaO2 in Mexico City compared with that expected in the absence of hyperventilation, but at the cost of greater work to breathe. Although Morris et al. reported an increasing PaCO2 with age8, we observed a rise in PaCO2 with age only in women as described previously in Bogota, Colombia, at 2600 masl9. Changes in PaCO2 were accompanied by expected adjustments in HCO3- and BE (both reduced or more negative) with a normal pH (7.43 on average). The level of PaCO2 observed in the report (30.2) is nearly identical to the 31 mmHg obtained by averaging the reference values of ABG in Mexico City in 19857 and below those reported in Bogota located at 2600 masl (reported at about 33 mmHg), those calculated from Morris for 2240 masl (PaCO2, 30.7 at 20 years of age and 34.7 at 60 years of age). [Equation: PaCO2 = 87.0423 + (Age*0.409901)- (Pbar*0.2264837) + (Pbar2*0.00021668) - (age*Pbar*0.0005306)]8 and below those predicted by a regression model on data from acclimatized lowlanders (PCO2, 32.3) [Equation: PaCO2 = (Pbar* 0.3300431) - (Pbar2*0.0004638)+(Pbar3*0.000000241) - 50.2436]4.
The increase in PaCO2 in older women may be due to the progressive lack of progesterone reported previously in a regression from several studies conducted in the U.S.8 and Bogota9 but has not been a consistent finding in other studies. Progestin receptors have been described both centrally and at the pulmonary level that participates in the lung development and function, as well as in control of ventilation by increasing minute volume10,11; therefore, a decreasing effect of progestin after menopause, could explain the presence of hypoventilation.
Although ethnic differences affecting ventilation explaining different mean PaCO2 found in Bogota compared to Mexico City cannot be ruled out, relevant methodological differences exist: in Bogota, the authors stabilized the participants (by reducing the respiratory quotient to < 1) before taking the blood sample, whereas in our study, the sample was taken by intra-arterial catheter 5 min after an arterial puncture in an attempt to reduce stress and pain at the moment of sampling; altitude must be taken into account for proper interpretation of oxygenation status and ventilation level (Table 3).
Table 3. PaCO2 reported at moderate altitudes in America
| Site | Altitude | Pbar (Torr) | pH | PaO2 (Torr) | PaCO2 (Torr) | SaO2 (%) | Comments | ||
|---|---|---|---|---|---|---|---|---|---|
| Overall | Male | Female | |||||||
| Salt Lake City. | 1,400 masl | 645 | 7.43 ± 0.02 |
74.7 ± 3.8 |
33.35 Approx. |
33.9 ± 2.2 |
32.8 ± 3.0 |
94.7 ± 0.7 |
Crapo et al.12 |
| Denver | 1,554 masl | 625 | 80.06 Approx. |
34.68 Approx. |
35.98 | 33.37 | 91 ± 0.6 |
Fitzgerald. Residents > 1 year14 | |
| Puebla | 2,158 masl | 585 | 7.39 ± 0.03 |
64.9 ± 5.9 |
35.8 ± 1.6 |
90.7 ± 3.2 |
Rojas et al. Residents persons, only men13 | ||
| Mexico City, Mex | 2,240 masl | 585 | 7.43 ± 0.02 |
71.2 ± 5.8 |
30.2 ± 3.4 |
30.8 ± 3.3 |
29.6 ± 3.5 |
94 ± 1.6 |
Cid-Pérez. (Actual study) |
| Ouray, USA | 2,371 masl | 573 | 28.5 Approx. |
30.65 | 27.18 | Fitzgerald. Residents >1 year14 | |||
| Matucana Peru | 2,390 masl | 572 | 7.37 ± 0.03 |
37.8 ± 1.8 |
91.7 ± 2.4 |
Hurtado et al. All subjects were men, American Indian15 | |||
| Bogota, Colombia | 2640 masl | 560 | 7.43 ± 0.02 |
33 Approx. |
33.5 ± 0.5 |
32.3 ± 2.1 |
Gonzalez-García M et al.9 | ||
| Cusco, Peru | 3350 masl | 505 | 7.43 | 61.08 | 30.62 | 31.6 | 29.9 | 91.13 | Pereira-Victorio et al. Native subjects16 |
masl: meters above sea level.
In contrast with other studies17,18, a drop in oxygen saturation with an increasing BMI was observed only in the univariate analysis and disappeared after adjusting for age. However, the variation in BMI was reduced in our participants (median= 25, ranging from 17 to 34 kg/m2), and lacked individuals with significant obesity and respiratory symptoms or diagnosis. On the other hand, we observed the expected adverse impact of age on oxygenation, with a decrease of 0.16 mmHg/year in PaO2, a drop of 0.04%/year in SaO2, and an increase in the alveolar-arterial gradient of 0.12 mmHg/year. Changes in terms of age were milder than those reported in other studies (0.27 and 0.42 mmHg/year)19-23. Aging of the lung gives rise to a variety of alterations in mechanics and gas exchange mechanisms, including decreases in ventilation/perfusion homogeneity, the mechanism explaining the increase in the alveolar-arterial PO2 gradient, and the drop in PaO2 with age (except for the increase in PaCO2 in women)24-27.
Our study's has limitations: it is difficult to know whether the ABG values measured reflect those present under habitual resting conditions, although these are unlikely to be maintained constant. Despite that, we separated the blood sample in time from the arterial puncture (carried out with local anesthesia) in an attempt to reduce the pain and stress often leading to hyperventilation, and although we reduced the likelihood of hyperventilation, we cannot rule out its presence when the sample was taken. We also did not measure the respiratory quotient during the procedure in order to observe whether the presence of steady-state could be supported. However, mouthpieces or facial masks are known to modify the breathing pattern and likely the respiratory steady-state, departing from habitual conditions. We do not know whether the mean pH 7.43, slightly on the alkalotic side, can reflect acute hyperventilation under resting conditions because of pain or stress. Furthermore, we have a limitation in the sample in subjects >60 years, mainly in the male sex; however, in this group, no significant changes were associated with age in PaO2 and PaCO2. We also lack hormonal measurements, especially in older women, to see whether a relationship exists between a higher PaCO2 and reduced progesterone levels. The population studied was not necessarily born in the Valley of Mexico but had resided for at least 2 years and was likely to become acclimatized to the altitude. We also lack the genetic ancestry of the participants, many of these typical Mexican mestizos with varied contributions of Amerindian genes.
On the other hand, we recruited a substantial number of normal subjects, with a wide age distribution, healthy from the respiratory point of view, and were studied with modern equipment and standardized techniques, including an intra-arterial line in an attempt to avoid pain and stress at the moment of the blood sample.
Although oxygenation status was maintained at 2240 masl at rest, the SaO2 was close to 90%, with more risk of hypoxemia on small decreases of PaO2. Mean hyperventilation is considerable (approximately 30% above sea level, and mean PaCO2 if 30.2 mmHg), slightly above that reported for acclimatized lowlanders and ameliorating progressively in post-menopausal women. The observed hyperventilation and the increased work of breathing reduce hypoxemia but may lead to more frequent dyspnea or fatigue.











 text new page (beta)
text new page (beta)



