The interference of weeds with agricultural crops reduces their growth and causes significant losses of their yields and quality (Dukpa et al. 2020). These invasive plant species, often abundant and well adapted to adverse conditions present the first biotic limitation factor of the agronomic crops production. Weeds compete with agricultural crops for nutrients, light, water and space and release allelopathic substances into the soil which could support the development of pests and crop pathogens (Suksungworn et al. 2016).
The most commonly used method for weeds control is the application of chemical herbicides considered as more efficient and less time consuming (de Souza Barros et al. 2021). The excessive and often inappropriate use of chemical herbicides in agricultural systems engendered harmful effects on non-target species and caused serious damage to human and animal health and to the global environment (Böcker et al. 2019). In addition, chemical herbicides face the limits of losing their effectiveness due to the evolution of weeds resistance (de Souza Barros et al. 2021).
In order to reduce the dependence on chemical herbicides, the development of others alternative strategies is needed for weed control in modern sustainable agriculture (Sulistiani et al. 2021). Thus, there is an increasing necessity for the development of new methods for weeds control including the use of bioherbicides based on living organisms or their secondary metabolites (Kubiak et al. 2022). Recently, many investigations have been devoted to study the allelopathic effects of several plant species and their ability to control weeds. Many phytotoxic biomolecules of the class of secondary metabolites released by allelopathic plants present an alternative strategy to manage weeds (Hasan et al. 2021). Various phytochemicals of the groups of phenolic acids, flavonoids, tannins, terpenes and alkaloids have been reported as bioactive allelochemicals with high potential as botanical bioherbicides (Scavo & Mauromicale 2021).
The development of botanical bioherbicides has become one of the major challenges in the agrochemical research to replace or integrate the harmful chemical herbicides (Motmainna et al. 2021). Bioherbicides are reported as easily and rapidly biodegradable, less disruptive to ecosystems and considered as environmentally friendly (Kong et al. 2021). Moreover, plant phytotoxic compounds possess highly diverse molecular structures and then various sites of action which minimize the emergence of resistant weeds (Araniti et al. 2012).
Efficacy and selectivity are the two basic criteria for the development of new bioherbicides (Hasan et al. 2021). In addition to the investigation of the bioherbicidal efficacy of plant extracts, the evaluation of their selectivity is required. Based on the previous considerations, the present study presents as main objectives the evaluation of the bioherbicidal activity and the selectivity of the aqueous extracts of Artemisia arborescens L. based on five plant species of agronomic interest, namely lettuce used as model plant, radish, pea, durum wheat and maize. The phytotoxic efficacy and selectivity of A. arborescens (tree wormwood) water extracts were tested using both pre-emergence and post-emergence bioassays.
Material and methods
Plant material. Fresh leaves of two Artemisia arborescens genotypes were collected during the vegetative development stage of the species on March 2021 from the sites of Bousselem and Kef located in the North West of Tunisia. The plant material was dried in the shade at room temperature then ground with an electric grinder into a fine powder used for the preparation of the aqueous extracts. A voucher specimen of both Artemisia arborescens genotypes were deposited in the herbarium of the Higher School of Agriculture of Kef.
The phytotoxic effect of tree wormwood extracts on the germination and primary seedling growth was carried out against lettuce (Lactuca sativa L., Asteraceae) considered as model species for phytotoxic activities (Wu et al. 2001). Furthermore, this activity was tested against four others agronomic species representing two monocotyledonous species (Triticum turgidum L. and Zea mays L., Poaceae) and two dicotyledonous species (Raphanus sativus L., Brassicaceae and Pisum sativum L., Fabaceae). The in vivo phytotoxic activity of the extracts was tested on one-month-old seedlings of the five studied species based on pot tests under semi-controlled conditions.
Extracts preparation. The aqueous extracts were prepared by the maceration of 15 g of dried and finely ground leaves for each tree wormwood genotype in 200 mL of distilled water. The extracts were incubated in an orbital shaker and stirred continuously at 120 rpm for 24 hours at 30 °C in the dark. Then, the samples were filtered twice with Whatman No. 1 paper and stored at + 4 °C away from light until the various analyses were carried out.
Phytochemical characterization. All the used reagents in the chemical analyses were analytical grade and were purchased from Sigma-Aldrich (Steinheim, Germany). The total polyphenol contents of each extract (TPT) were estimated using the Folin-Ciocalteu method (Singleton & Rossi 1965). The calibration curve was carried out with gallic acid (0-200 µg/mL) and the results are expressed as mg of gallic acid equivalent per gram of dry weight (mg GAE/g DW). The flavonoids contents of the extracts were estimated following the aluminium chloride method (Djeridane et al. 2006). The concentrations of flavonoids in the studied extracts were deduced from a standard range established with rutin (0-200 µg/mL) under the same operating conditions as the extracts. The flavonoid contents are expressed as mg rutin equivalent per gram of dry weight (mg RE/g DW).
The condensed tannin contents of the extracts were determined using the vanillin method (Sun et al. 1998) with some modifications as described by Riahi et al. (2021). The contents of total condensed tannins were determined based on a calibration curve using catechin (0-200 µg/mL) as standard and results expressed as milligrams of catechin equivalent per gram of dry weight (mg CE/g DW). The terpenoids contents in the aqueous extracts of tree wormwood were determined following the method of Ghorai et al. (2012). The terpenoids contents were deduced from a calibration curve using linalool (0-120 µg/mL) as standard and results were expressed as milligrams of linalool equivalent per gram of dry weight (mg LE/g DW). All the phytochemical analyses were achieved in triplicate.
Germination and primary growth bioassays. For each tree wormwood genotype, the aqueous extracts were completely dried to determine their weigh. Then, the obtained residues were adjusted with distilled water to the concentrations 2 and 8 mg/mL. The seeds of each plant species were surface sterilized using 1 % sodium hypochlorite solution for 5 minutes then rinsed three times. Ten seeds of each species were sown in Petri dishes (6 cm Ø) covered with two layers of sterile Whatman No. 1 paper and watered with 4 mL of each tested extract. The control was watered with 4 mL of distilled water. All experiments were repeated three times.
Petri dishes were sealed with Parafilm to prevent desiccation and kept to germinate in the dark at a temperature of 25 °C. Seeds showing root extrusion at least 1 mm long were considered germinated. The number of germinated seeds is recorded every day until there is no more germination. Germination percentages were determined from cumulative germination data as GT (%) = N/NT × 100 where N is the number of germinated seeds and NT is the total number of seeds. The germination indexes were determined using the following formula (Chiapuso et al. 1997): GI = (N1) × 1 + (N2-N1) × 1/2 + (N3-N2) × 1/3 +.... + (Nn-Nn-1) × 1/n. Where, N1, N2, N3, …. Nn are the number of germinated seeds after 1, 2, 3,… n-1 and n days.
The effect of the extracts on the primary growth of the roots and shoots was assessed. Five pre-germinated seeds (48 hours) of each studied species were placed in sterile Petri dishes (6 cm Ø) and treated with 4 mL of each tested extract. After 72 hours of exposure to the different extracts, the length of the roots and shoots was determined. The experiments were realised in triplicates. The inhibition rates of germination, roots and shoots growth were calculated using the following equation: Inhibition rate (%) = [(Control - Treatment) / Control × 100].
The in vivo phytotoxic bioassays. One-month old seedlings of the five studied species were used as plant material to conduct the in vivo phytotoxic activity of tree wormwood water-soluble compounds at the concentration of 8 mg/mL. Seeds were sown in pots filled with agricultural soil and commercial peat Pindstrup (2v : 1v) and maintained in a greenhouse under semi-controlled conditions with a photoperiod of 16 hours of light and 8 hours of darkness, an average temperature of 25 ± 2 °C and an average relative humidity of 55 ± 5 %.
One-month old seedlings were sprayed with the aqueous extracts (8 mg/mL) of A. arborescens until dropping. The group of untreated plants (control) was sprayed with distilled water. The application of the extracts was repeated twice with an interval of three days. The experiments were arranged on a completely randomized block design with three replicates. Three days after the application of the second treatment, the treated and untreated seedlings were subjected to symptomatic evaluation and three samples of leaves were randomly harvested from each species to serve for the following analyses.
The method of Lichtenthaler & Wellburn (1983) was used to determine spectrophotometry the contents of Chlorophyll a (Chla), chlorophyll b (Chlb), total chlorophyll (ChlT) and carotenoids (Carot) in the leaves of the seedlings for each treatment. The contents of the photosynthetic pigments were calculated and expressed as milligrams per gram of fresh weight (mg/g FW). Proline contents were estimated according to the method of Bates et al. (1973) and the results are expressed as micrograms per gram of fresh weight (μg/g FW). Malondialdehyde (MDA) amounts were determined according to the method of Doblinski et al. (2003) and expressed as nanograms per gram of fresh weight (ng/g FW).
Data analysis. All experiments were performed in triplicate. The results are presented as mean values ± standard variations. The quantitative variations of the studied parameters were evaluated using the one-way variance analysis. The comparison of the means was carried out using Duncan's test at a significance level of 0.05. Statistical analysis was performed using IBM SPSS Statistics software version 28.0 for Windows.
Results
Phytochemical characterization of the extracts. The contents of total phenolics, flavonoids, condensed tannins and terpenoids compounds in the aqueous extracts of the two genotypes of A. arborescens were determined (Table 1). The obtained results reveal considerable levels of total polyphenols with a significant variation between the two genotypes. The genotype of Bousselem exhibited higher amounts (35.79 ± 2.04 mg GAE/g DW) of polyphenolic compounds than the genotype of Kef (27.89 ± 2.91 mg GAE/g DW). Non-significant variation was recorded for the flavonoid contents between Kef genotype (17.65 ± 0.43 mg RE/g DW) and Bousselem genotype (18.25 ± 0.84 mg RE/g DW).
Table 1 Variation of the contents of total polyphenols (TPC), total flavonoids (TFC), condensed tannins (CTC) and terpenoids (TTC) between tree wormwood genotypes.
| Genotype Bousselem | Genotype Kef | |
|---|---|---|
| TPC (mg GAE/g DW) | 35.79 ± 2.04 b | 27.89 ± 2.91 a |
| TFC (mg RE/g DW) | 18.25 ± 0.84 a | 17.65 ± 0.43 a |
| CTC (mg CE/g DW) | 7.26 ± 0.44 a | 8.01 ± 0.51 a |
| TTC (mg LE/g DW) | 19.88 ± 3.29 b | 12.42 ± 2.07 a |
Mean values in each line followed by different letters are significantly different.
Similarly, the obtained findings showed non-significant variation in the contents of condensed tannins between the aqueous extracts of the two genotypes. The concentrations range from 7.26 ± 0.44 mg CE/g DW (Bousselem genotype) to 8.01 ± 0.51 mg CE/g DW (Kef genotype). However, a significant variation in the contents of terpenoids between the extracts of kef and Bousselem was recorded. The genotype of Bousselem showed higher contents of terpenoids (19.88 ± 3.29 mg LE/g DW) than the extract of Kef genotype (12.42 ± 2.07 mg LE/g DW).
Effect of extracts on germination. The application of the aqueous extracts of the two genotypes of A. arborescens decreased significantly the germination rates of lettuce, radish, durum wheat and maize as compared to the control while no significant effect on the germination of pea was recorded (Figure 1).
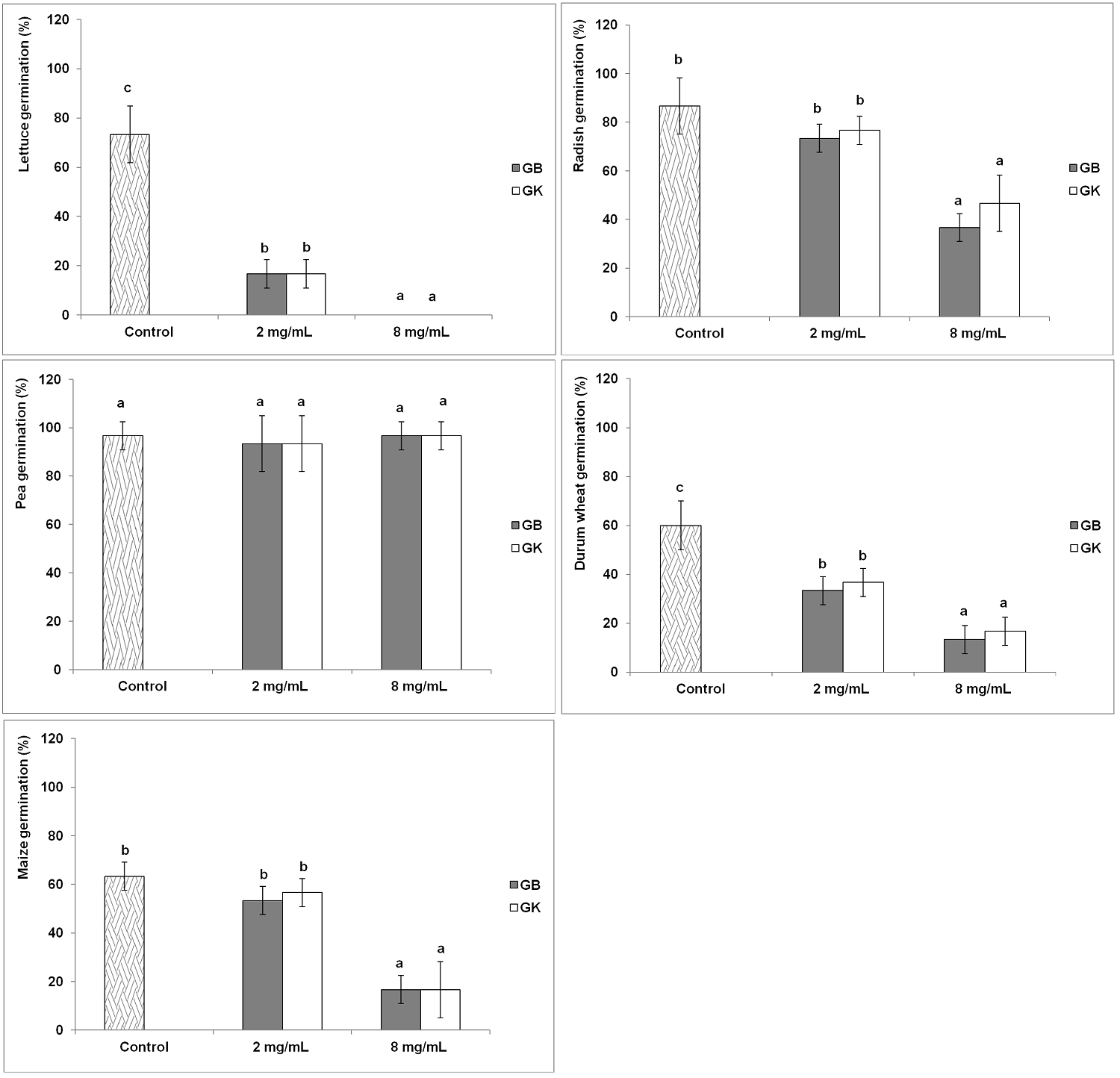
Figure 1 Variation of the germination rates among the tested agronomic species (GB: genotype Bousselem, GK: genotype Kef). Mean values followed by different letters are significantly different.
A significant variation of the responses according to the tested species and the applied concentrations was observed with no significant variation according to the genotype of tree wormwood. A total inhibition (100 %) of lettuce germination was observed with a concentration 8 mg/mL for both genotypes extracts. The concentration 2 mg/mL resulted in a significant reduction of the germination rates of treated lettuce seeds (16.66 %) compared to the control (73.33 %) giving a germination inhibition of 77 %. The treated seeds at this concentration showed a germination delay of two days compared to the control.
The two concentrations of tree wormwood water extract didn’t affect significantly the germination percentages of pea seeds compared to the control (96.66 %). For radish and maize, the application of the extracts at 2mg/mL to the seeds didn’t affect significantly the germination levels compared to control (radish: 86.66 %, maize: 63.33 %). However, the germination percentages decreased significantly at the concentration 8 mg/mL for radish (36.66-46.66 %) and maize (16.66 %) giving a reduction in the germination rates by 46-58% and 74 %, respectively. The two applied concentrations of extracts resulted in a significant reduction of the germination capacity of treated durum wheat seeds compared to the control (60 %). The lowest germination rates of durum wheat (13.33-16.66 %) were recorded with the concentration 8 mg/mL giving reduction rates of germination of 72-78 %.
Furthermore, the application of tree wormwood water extracts affected significantly the germination indexes which traduce the germination speed (Figure 2). A reduction of the germination indexes for lettuce and durum wheat with both 2 mg/mL and 8 mg/mL concentrations was recorded in a concentration-dependent manner. For radish, pea and maize a significant reduction of the germination indexes for treated seeds was recorded with the concentration 8 mg/mL.
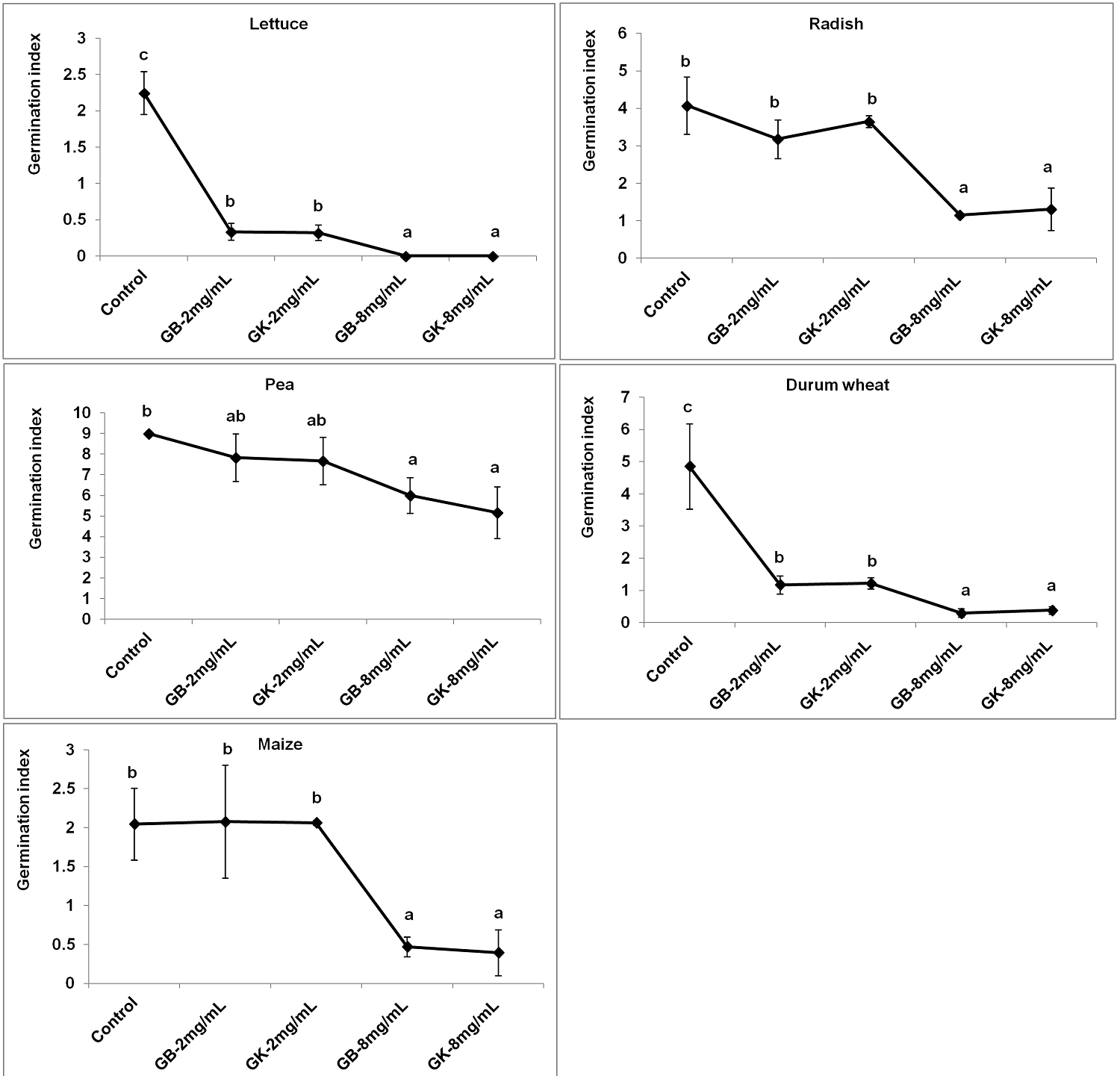
Figure 2 Variation of the germination indexes among the tested agronomic crops (GB: genotype Bousselem, GK: genotype Kef). Mean values followed by different letters are significantly different.
Effect of extracts on seedlings primary growth. The effect of A. arborescens extracts on the primary growth of roots and shoots of seedlings in the studied species was evaluated. The obtained results showed a significant variation in the effects depending on the applied concentrations and the tested species (Figure 3). No significant variation according to genotype was detected for the growth of roots and aerial parts for all the tested agronomic crops. A significant decrease in the root growth of treated lettuce seedlings compared to the control (1.14 cm) was observed with the two concentrations of the extracts. The same pattern was observed for lettuce shoots growth which were significantly reduced compared to the control (2.02 cm) for both concentrations. The highest inhibition effects on roots (0.52-0.56 cm) and shoots (0.84-0.86 cm) were recorded with the concentration 8 mg/mL.
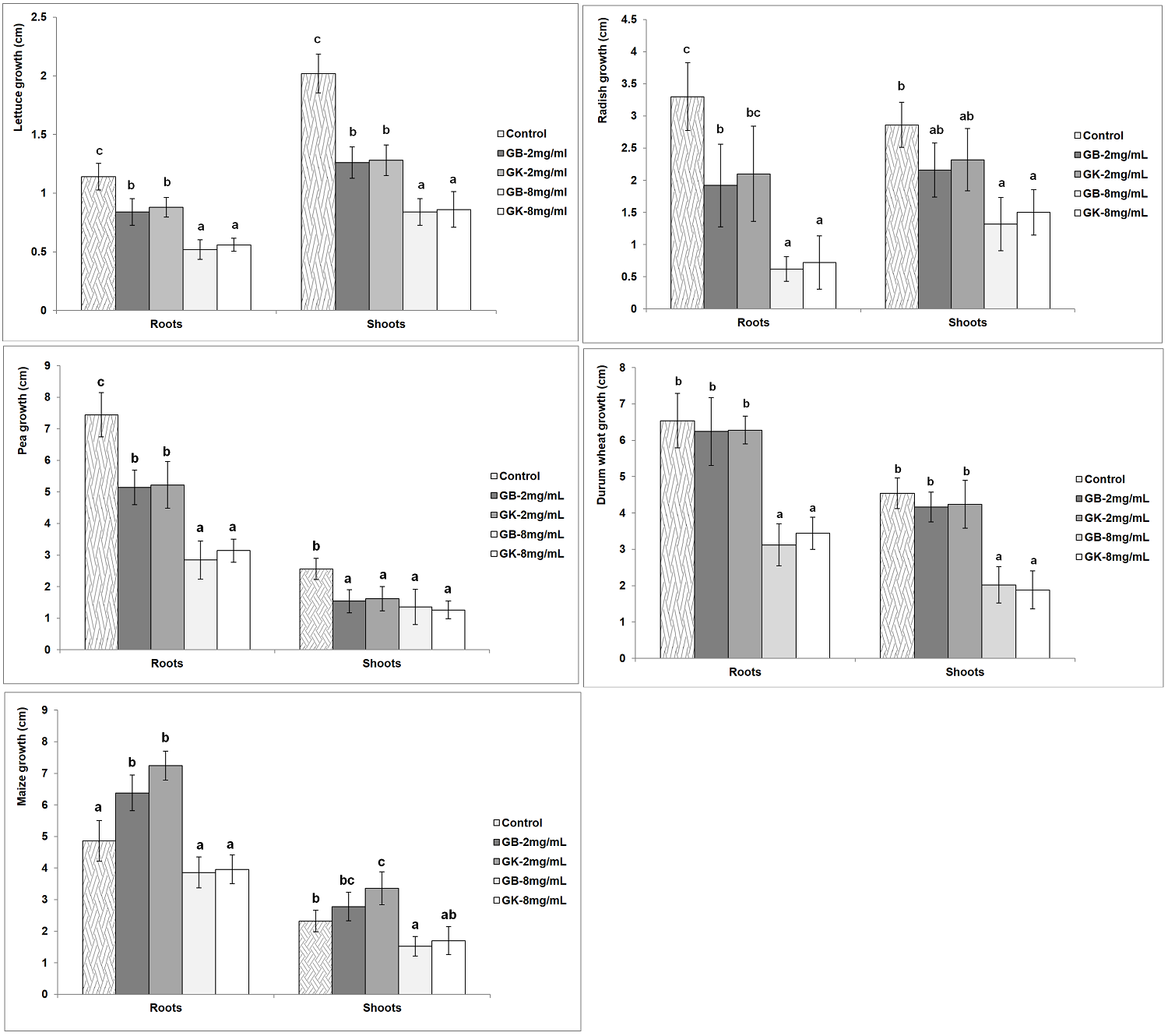
Figure 3 Variation of roots and shoots growth among the tested agronomic species (GB: genotype Bousselem, GK: genotype Kef). Mean values followed by different letters are significantly different.
With the exception of the treatment GK-2 mg/mL, all the others applied treatments caused a significant inhibition of radish root growth compared to the control (3.3 cm). The highest reduction of radish roots growth was recorded with the treatments 8 mg/mL (0.62-0.72 cm). On the other hand, the treatment 8 mg/mL induced a significant reduction in the growth of radish shoots (1.32-1.50 cm) as compared to the control (2.86 cm).
While no significant effect was recorded for tree wormwood extracts on the germination of pea, the application of these water-soluble compounds to pre-germinated seedlings gave a significant reduction in the growth of roots and shoots compared to the control with both concentrations. The two applied concentrations engendered a reduction in the growth of pea roots below that of control (7.44 cm). The highest inhibition of root growth of pea was recorded with the concentration 8 mg/mL (2.84-3.14 cm). The two applied concentrations reduce the shoot growth (1.26-1.62 cm) as compared to the control (2.56 cm) without showing a significant variation between the two concentrations.
A significant inhibition of root and shoot growth of durum wheat was observed with the concentration 8 mg/mL. This concentration engendered a reduction in the growth of durum wheat roots (3.12-3.44 cm) and shoots (1.88-2.02 cm) as compared to controls, 6.54 cm and 4.53 cm respectively. Interestingly, maize root growth was significantly improved over the control (4.86 cm) with the extract concentration 2 mg/mL (6.38-7.24 cm). No significant variation was recorded for root growth of treated maize seedlings at the concentration 8mg/mL compared to the control. The growth of maize shoots was significantly improved over the control (2.32 cm) with the treatment GK-2 mg/mL (3.36 cm). A significant inhibition of the shoot growth of maize with the treatment GB-8 mg/mL (1.52 cm) was obtained.
The in vivo phytotoxic effects. The one-month-old seedlings treated twice with the aqueous extracts of A. arborescens at the concentration 8 mg/mL were harvested. The treated lettuce, durum wheat and maize seedlings showed remarkable visual changes as compared to the controls sprayed with distilled water (Figure 4). Yellow and brown coloured zones in the leaves were clearly observed with a higher intensity for the seedlings sprayed with the extracts of Bousselem genotype. Morphological observations show a remarkable effect on the leaves of maize followed by wheat, whereas no apparent effect was detected for radish and pea.

Figure 4 Morphological appearance of the leaves of the tested agronomic crops after spraying with tree wormwood aqueous extracts (8 mg/mL). From the left to the right: control, genotype Bousselem, genotype Kef. Lettuce (A), Radish (B), Pea (C), Durum wheat (D) and Maize (E).
The in vivo effect of the extracts (8 mg/mL) on the tested species was evaluated based on various biochemical parameters, namely the contents of chlorophyll a, b and total, carotenoids, proline and malondialdehyde (MDA). For lettuce, the obtained results showed a significant decrease in the contents (mg/g FW) of chlorophyll a, b, T and carotenoids as compared to the control (Chla: 0.555 ± 0.015; Chlb: 0.186 ± 0.004; ChlT: 0.742 ± 0.020; Carot: 0.166 ± 0.004) with both tree wormwood extracts (Figure 5). The obtained results showed no significant variation for all the photosynthetic pigments between the control (Chla: 0.99; Chlb: 0.33; ChlT: 1.32; Carot: 0.29 mg/g FW) and the treated pea seedlings. Concerning radish seedlings, no significant variation in chlorophyll and carotenoid contents compared to the control (Chla: 1.04; Chlb: 0.35; ChlT: 1.40; Carot: 0.27 mg/g FW) was generated by the application of extracts of Bousselem genotype. However, a significant increase in the contents of chlorophyll b and total compared to the control was observed for the seedlings treated with the extracts of Kef genotype.
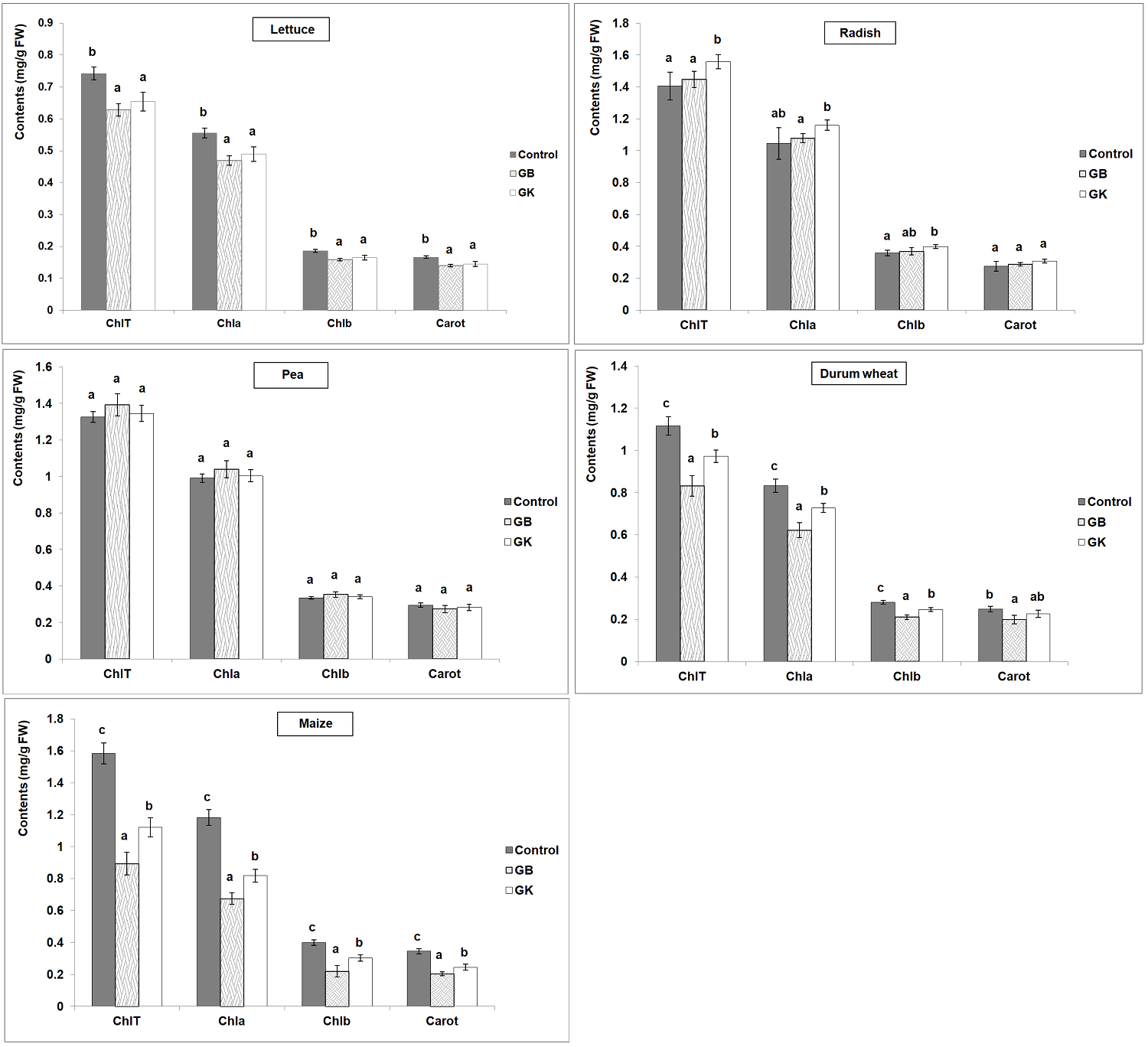
Figure 5 Variation of the contents of photosynthetic pigments (mg/g FW) among the five tested crops (GB: genotype Bousselem, GK: genotype Kef). Mean values followed by different letters are significantly different.
For durum wheat and maize seedlings, a reduction in the contents of chlorophyll a, b and total compared to the control (Durum wheat: Chla: 0.83, Chlb: 0.28, ChlT: 1.11 mg/g FW; Maize: Chla: 1.18, Chlb: 0.40, ChlT: 1.58 mg/g FW) was observed with the extracts of both genotypes. The highest decreases in chlorophyll a, b and total contents for durum wheat and maize were observed with extracts of Bousselem genotype. Concerning the carotenoids contents, a significant decrease in treated plants compared to the durum wheat control (0.24 mg/g FW) was observed under the effect of Bousselem extract. A significant decrease of carotenoids amounts in treated maize seedlings compared to the control (0.34 mg/g FW) was observed with the extracts of both genotypes. The highest decrease in carotenoids contents for maize was detected with Bousselem genotype.
The contents of proline (μg/g FW) in the leaves of the tested agronomic species were determined. The obtained results showed a significant variation among the studied treatments and the control for the four species lettuce, radish, durum wheat and maize while no significant variations were detected for pea (Figure 6). A significant increase in the proline contents in treated lettuce (67.82-69.11 μg/g FW) and radish (77.12-79.31 μg/g FW) seedlings over the untreated plants (lettuce: 49.34 μg/g FW, Radish: 70.45 μg/g FW) was observed. No significant variation of proline contents according to tree wormwood genotype was detected. The obtained results showed no significant variation in proline amounts between treated and untreated plants (92.32 μg/g FW) of pea. However, the concentration of proline increased significantly as compared to the control of durum wheat (120.38 μg/g FW) and maize (116.54 μg/g FW) in the treated seedlings with a significant variation according to the genotype of tree wormwood. For durum wheat and maize, the highest contents of proline were observed with the extracts of Bousselem genotype, 152.64 and 147.42 (μg/g FW), respectively.
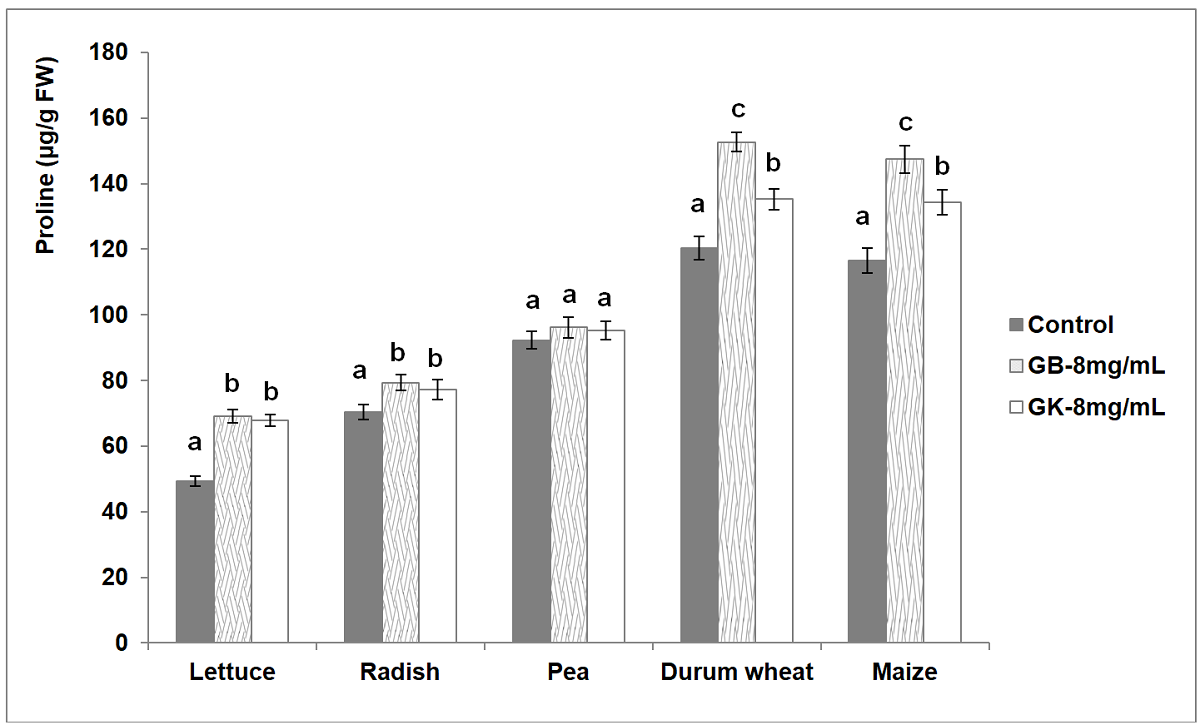
Figure 6 Variation of proline contents (μg/g FW) in the leaves of the five tested agronomic crops (GB: genotype Bousselem, GK: genotype Kef). Mean values followed by different letters are significantly different.
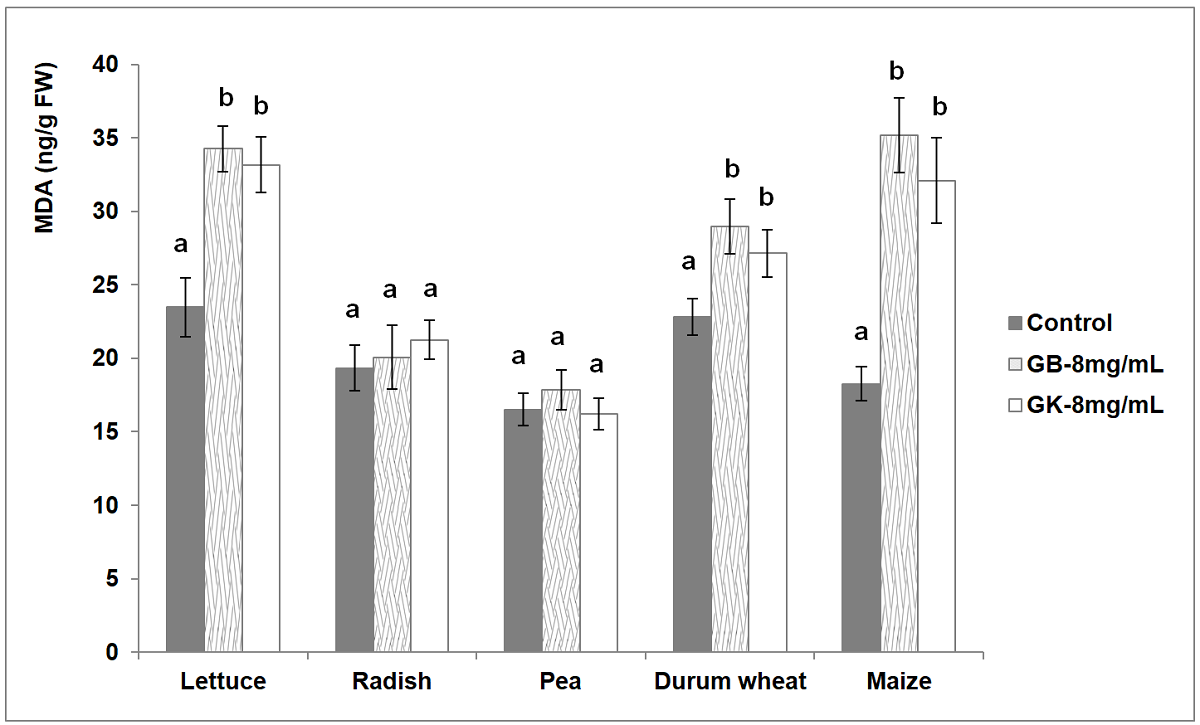
The variation in the accumulation of MDA in the leaf tissues of the five crops was evaluated (Figure 7). A significant increase in the concentration of MDA in treated lettuce (33.17-34.25 ng/g FW), durum wheat (27.14-28.96 ng/g FW) and maize (32.09-35.18 ng/g FW) over the untreated seedlings (lettuce: 23.48, durum wheat: 22.81, maize: 18.25 ng/g FW) was recorded with both tree wormwood genotypes. No significant variation of MDA contents was recorded according to tree wormwood genotype for the three-last species. The obtained results showed no significant fluctuation of MDA contents between the treated seedlings and the control of radish (19.34 ng/g FW) and pea (16.52 ng/g FW) for both tree wormwood genotypes.
Discussion
The application of plant allelopathic phytochemicals to control weeds is considered recently as an effective, low cost, eco-friendly and alternative strategy to the use of harmful chemical herbicides and time-consuming mechanical methods (Alsharekh et al. 2022). Artemisia species are considered among the candidate donor plants for the development of biopesticides including bioherbicides. This is due to their richness in phytotoxic secondary metabolites of the classes of phenolic acids, flavonoids, terpenoids and alkaloids (Araniti et al. 2012, Riahi et al. 2022). A considerable bioherbicidal activity against many weed species was reported for Artemisia absinthium (Kapoor et al. 2019), Artemisia argyi (Chen et al. 2022), Artemisia vulgaris (Pannacci et al. 2020), Artemisia fragrans (Pouresmaeil et al. 2020), among others.
The accumulation of secondary metabolites in plant species depends both on endogenous and exogenous factors (Thakur et al. 2019, Ben Rejeb et al. 2023). The genotype is reported to affect significantly the accumulation of these high-value bioactive compounds (Riahi et al. 2019). Moreover, the extraction process is the main step to recover and isolate the bioactive phytochemicals. Water is the solvent of choice for the extraction of plant phytotoxic secondary metabolites given its higher advantage of being the safest and most eco-friendly solvent used for extraction and separation processes (Bhat et al. 2020). The aqueous extracts are often used for testing the allelopathic activities of plant species (Vitalini et al. 2021). Actually, the use of plant aqueous extracts to inhibit weeds is gaining increasing interest in the environmentally friendly agriculture (Carrubba et al. 2020).
The in vitro bioassays are the first step to evaluate the allelopathic potential of plant extracts. Lettuce (Lactuca sativa) is a model species generally used for the screening of bioherbicidal activity due to its high inherited sensitivity to phytotoxic substances (Wu et al. 2001). The results showed that the aqueous extracts of A. arborescens exert an inhibitory effect on the germination of lettuce seeds with a total inhibition at the concentration 8 mg/mL. Moreover, the applied extracts inhibited the germination of radish, durum wheat and maize with various degrees. Interestingly, no significant effect was recorded on pea germination.
Generally, the inhibition of seed germination is attributed to the disruption of mitochondrial respiration (Abrahim et al. 2003) and the inhibition of the activity of certain metabolic enzymes involved in glycolysis and oxidative pathways (Omezzine et al. 2011). Germination inhibition is one of the most used parameters to assess the allelopathic potentials of plants extracts. The evaluation of the phytotoxic effect of plant extracts on the germination of weeds or model plants is of great importance. Indeed, the inhibition of the germination ability of the soil seed bank of these undesirable plant species is of great importance for their control. This could limit their emergence at least until the crop will develop a good competitiveness (Carrubba et al. 2020).
The results showed variation in the phytotoxic activity of the extracts between the five tested species (Figures 1-3). Different responses of plant species to phytotoxic substances have been reported by previous studies. According to some reports, the size of the seeds is a determining factor in their response to phytotoxic substances. Generally, a similar concentration of an extract can produce more inhibition on small-seeded species than on large-seeded ones (Kruidhof et al. 2011, Alvarez-Iglesias et al. 2014).
In addition, a significant inhibitory effect of tree wormwood water extracts on the growth of roots and shoots of the five agronomic crops was observed. The length reduction of roots and aerial parts of seedlings can be explained by the alteration of mitosis and cell elongation in the presence of certain allelochemicals in the applied extracts (Javaid & Anjum 2006). Higher effects on roots than on aerial parts are generally observed (Appiah et al. 2018, Kyaw & Kato-Noguchi 2020). The stronger harmful impacts of extracts on the roots reflect the fact that massive uptake of phytotoxic substances by the roots is higher than by the aerial parts (De Martino et al. 2012). Generally, the root tissues are more permeable to phytotoxic compounds than the aerial tissues (Nishida et al. 2005). Therefore, the results in this study confirm previous reports showing that root growth may be the best variable to assess the phytotoxic potential of plant extracts (Aslani et al. 2014).
It is noted that while Artemisia arborescens extracts did not reveal a significant effect on pea germination, a significant inhibition of root and shoot growth was recorded for this species. These findings corroborate previous results which reported higher effect of phytotoxic water extracts on root elongation than on seed germination (Araniti et al. 2012). Thus, A. arborescens water extracts could be a potential candidate for the development of bioherbicides with pre-emergence application on pea. At this stage, weeds are more susceptible to the phytotoxic compounds and their seeds bank will be reduced which avoid their competition with the agronomic crop. At lower concentrations of the extracts, a significant increase in the primary growth of maize was observed, corroborating previous findings which reported that allelopathic phytochemicals could exercise positive effects at lower concentrations. A positive effect of low concentrated allelopathic extracts on germination and seedling growth of wheat was previously reported (Perveen et al. 2019).
The no significant in vivo effect of Artemisia arborescens extracts on pea and radish, suggest that the allelopathic compounds of the studied species could be the subject of further investigations for the development of bioherbicides for post emergence application on pea and radish.
The phytotoxicity mechanisms of allelochemicals are reported to be complex. They affect almost all aspects of plant growth and development, including gene expression, phytohormone synthesis and activity, ion uptake and photosynthetic function (Yan et al. 2015). Photosynthetic pigments contents are considered as significant indicators of the effect of allelochemical extracts on plants. A decrease in their amounts after phytotoxic treatments has been recorded by previous investigations. Jali et al. (2021), reported that chlorophyll a, b and total contents were negatively affected upon the application of phytotoxic extracts on the species Macrotyloma uniflorum. Regarding carotenoids contents, many studies have reported that the accumulation of these pigments decreases significantly in treated plants (Ghimire et al. 2020). In some cases, an increase in carotenoids levels under the effect of allelopathic treatments has been observed, especially with low concentrations (Ben Jaballah et al. 2017). The reduction of photosynthetic pigments was considered responsible for the alteration of photosynthetic activity and thus the inhibition of the growth of treated plants (Siddiqui & Zaman 2005). The results corroborate others previous studies that have reported an increase in proline contents in plant species treated with allelopathic substances. Proline accumulation is a common response in many plants in response to a wide range of biotic and abiotic stresses (Verbruggen & Hermans 2008). Proline acts as electron acceptor and prevents membrane damage. It offers protection against ROS-induced disturbances and activates detoxification pathways which prevent programmed cell death (Szabados & Savouré 2010).
Polyunsaturated fatty acids present the main lipid components of the cell membrane sensitive to peroxidation and degradation. The increase in membrane permeability under the effect of allelochemical stress corresponds to an increase in the lipid peroxidation which results in the accumulation of malondialdehyde MDA (Omezzine et al. 2014). MDA accumulation, due to lipid peroxidation, has been reported as a response to various allelopathic plant extracts. The increase in lipid peroxidation levels can be considered as an index of oxidative damage in plants treated with phytotoxic extracts. Allelochemicals could damage the cell membrane by direct interaction with its constituents or may modify certain metabolic functions necessary to maintain its function (Goel & Sheoran 2003). The accumulation of MDA is toxic because it causes the alteration of membrane proteins and the decrease of cell membrane fluidity (Gill & Tuteja 2010).
Our findings highlighted that monocotyledonous species are more sensitive to the extracts than dicotyledonous ones. In others studies, dicots target plants were more severely affected than monocots when treated with Tinospora tuberculata leaf extract (Aslani et al. 2014). Differences in the sensitivity of various plant species to phyto-inhibitory compounds could be explained by several factors. Kobayashi (2004) suggested that this is due to the physiological and biochemical characteristics of each plant species. Different responses to the same allelopathic extract may also be due to seed structure and seed coat permeability (Hanley & Whiting 2005). Haq et al. (2010) suggested that a mixture of various compounds in the extract can act antagonistically or synergistically in different plant species. Some plant extracts such as Artemisia vulgaris L. exhibited significant herbicidal activity against weeds and biostimulant action on the crops (Pannacci et al. 2020). The selectivity of the phytotoxic effects of plant extracts makes them good candidates for further in-depth studies for the development of eco-friendly bioherbicides to control weeds (Vaccarini & Bonetto 2000). Moreover, the determination of the optimal concentrations range which engendered the inhibition of weeds germination and growth without having negative effects on crops is interesting for the development of herbal bioherbicides (Krumsri et al. 2019).
While the number of studies which focused on the screening of the in vitro and in vivo bioherbicidal activity of plants extracts is increasing, little is known about the side effects of the application of botanical bioherbicides. The results of Fuselier & Carreiro (2022) showed that Fern gametophyte establishment was negatively affected by glyphosate but not by two applied bioherbicides (cinnamon plus clove oil, and pelargonic acid). The development of bioherbicides of natural origins is highly encouraged for application in organic and conventional agriculture. Bioherbicides are reported to have no effects on non-target species and to present several advantages as compared to chemical herbicides including biodegradation ability, multiple modes of action and safety to human health and environment (Roberts et al. 2022). However, further investigations regarding the effects of their long-term and repeated application on non-target species and environment are required. Indeed, some natural phytotoxic products such as AAL-toxins are reported to be toxic to mammals which have reduced their interest for weed management (Hasan et al. 2021).
This study revealed a significant variation in the in vitro and in vivo phytotoxic activity of Artemisia arborescens water extracts against lettuce, radish, pea, durum wheat and maize. Pre-emergence bioassays showed that lettuce was the most sensitive to A. arborescens water-soluble phytochemicals while no significant effect was recorded on pea germination. The applied extracts inhibited the growth of the roots and aerial parts of the five agronomic crops with significant variation among the studied species. Higher in vivo phytotoxic effects were recorded against monocotyledonous than dicotyledonous crops. The extracts of Bousselem genotype exhibited relatively higher in vivo phytotoxic effect than Kef genotype. Interestingly, pea is the least affected crop based on both in vitro and in vivo bioassays. The obtained results highlighted a significant potential of tree wormwood extracts for the development of bioherbicides with pre-emergence or post-emergence applications according to the target agronomic crop.











 nueva página del texto (beta)
nueva página del texto (beta)