Mexico is losing an average of 318,000 hectares of forest annually and is one of the countries with the highest deforestation rates, resulting in rapid ecosystem loss and decreasing native biodiversity (including plants and mushrooms) (Pérez-Moreno et al. 2020). Mexico is also the center of diversity of Quercus and Pinus species (Perry 1991, Valencia & Flores-Franco 2006) which are both widely known as ectomycorrhizal (ECM) trees. As Mexico harbors a great diversity of ECM trees, a high number of ECM mushrooms are expected. These ectomycorrhizal associations occur in all forest ecosystems from the tropics to the subpolar zones (Nara 2015). The ECM trees depend on the ECM fungi for obtaining greater access to mineral nutrients and water, while the trees provide a supply of carbohydrates to the symbiotic fungi (Smith & Read 2008).
Laccaria species associate with many ECM trees (Trappe 1962, Mueller 1992, Kropp & Mueller 1999, Wilson et al. 2017, DEEMY 2021). It is estimated that 116 species of Laccaria are distributed across temperate and tropical regions of the world (Wilson et al. 2017). The diversity and taxonomy of the genus Laccaria have been well documented (Mueller 1984) including with phylogenetic and molecular studies (Osmundson et al. 2005, Vincenot et al. 2012, Sheedy et al. 2013, Wilson et al. 2013, Popa et al. 2014, 2016, Montoya et al. 2015, Cho et al. 2018). More recently, biogeographic (Wilson et al. 2017) and genomic studies have used some Laccaria taxa (e.g.,Martin et al. 2008, Kang et al. 2020, Li et al. 2020) as model species in the understanding of the physiology, ecology, and evolution of ectomycorrhizal symbiosis.
In general, Laccaria species are considered early-stage colonizers (Nara et al. 2003, Reverchon et al. 2012) and therefore play a paramount ecological role in the establishment of ECM host plants (Kropp & Mueller 1999, Nara et al. 2003, Ashkannejhad & Horton 2005). They can grow vegetatively and are relatively easy to manipulate under experimental conditions. Several species of the genus, including for example Laccaria bicolor (Maire) P.D. Orton, L. laccata (Scop.) Cooke, and L. proxima (Boud.) Pat., have successfully been used in the production of ECM inoculants for forest management purposes (Molina 1980, Molina & Chamard 1983, Chakravarty & Hwang 1991, Duponnois & Garbaye 1991, Werner & Zadworny 2003, Pérez-Moreno et al. 2020). Laccaria species such as Laccaria amethystina Cooke. and L. laccata (Scop.) Cooke also have great cultural, social, and economic value and are harvested for income and as food in different parts of the world (McKnight et al. 1998, Pérez-Moreno et al. 2008, Kalita et al. 62016, Wu et al. 2019). Laccaria squarrosa Bandala, Montoya & A. Ramos, however, is a recently described species. It is only known from its type locality, where it associates with endangered Fagus grandifolia var. mexicana (Martínez) E. Murray trees in eastern Mexico (Ramos et al. 2017), and from discovery at a market in western Mexico, where it was identified as a rare species by Farfán-Heredia et al. (2018) without stating the vegetation type or location from where the specimens were gathered.
Pinus pseudostrobus Lindl. has a wide geographical distribution in Mexico and Central America with a broad altitudinal range between 1,600-3,300 m above sea level (Delgado et al. 2007). The species usually establishes pure stands or occurs with some other species of Pinus, Abies, Arbutus, Juniperus, or Quercus (Perry 1991). In Mexico, P. pseudostrobus has been used in reforestation programs especially on degraded soils, and its resins and wood are highly commercialized (Gómez-Romero et al. 2013).
During some expeditions to forest located in Piedra Canteada, Nanacamilpa, Tlaxcala, in Central Mexico, to document the diversity of wild edible mushrooms consumed by the local people, we found and collected specimens of L. squarrosa (a legitimate species included in the Mycobank database with the number 823034). The study site is in a natural reserve called the “Santuario de las Luciernagas” (The Sanctuary of the Fireflies) famous for the displays of fireflies that inhabit its Pinus-Quercus forest. The local people make an income from this activity during the summer, motivating them to conserve the forest and manage its resources. Basidiomata of L. squarrosa are also harvested as a source of food by members of the Nahua ethnic groups in this area.
In this study, we aim to determine for the first time whether Laccaria squarrosa establishes ectomycorrhizal symbiosis with the gymnosperm Pinus pseudostrobus in natural conditions, confirming the taxonomic identity of both symbionts with molecular analyses. The confirmation of this association would provide evidence to promote the use of L. squarrosa as a potential native inoculum in the production of ectomycorrhized P. pseudostrobus seedlings in successful reforestation programs in the studied area.
Materials and methods
Study area. The study area is in the state of Tlaxcala, Mexico, on the Trans-Mexican Volcanic Belt (TMVB; Figure 1). This ecoregion connects the Sierra Madre Oriental and the Sierra Madre Occidental, and serves as a center of biodiversity. The sampling site is in Piedra Canteada, Nanacamilpa municipality (Figure 1). The dominant climate is temperate subhumid with rains in summer and annual temperatures of 12-18 °C. Mean annual precipitation is < 500 mm (INEGI 1997). The type of vegetation in the study area is dominated by Pinus-Quercus (pine-oak) forest and Abies (fir) forest. The most representative species in the Pinus-Quercus Forest include Pinus montezumae Lamb, P. pseudostrobus, P. teocote Schiede ex Schltdl. & Cham., which usually co-occur with Quercus spp. such as Quercus crassipes Bonlp., Q. laurina Bonpl., and Q. rugosa Née. The Abies forest is composed of Abies religiosa (Kunth) Schltdl. & Cham., Alnus jorullensis Kunth., Salix paradoxa Kunth., and Arbustus xalepensis Kunth., all coexisting in the same area.
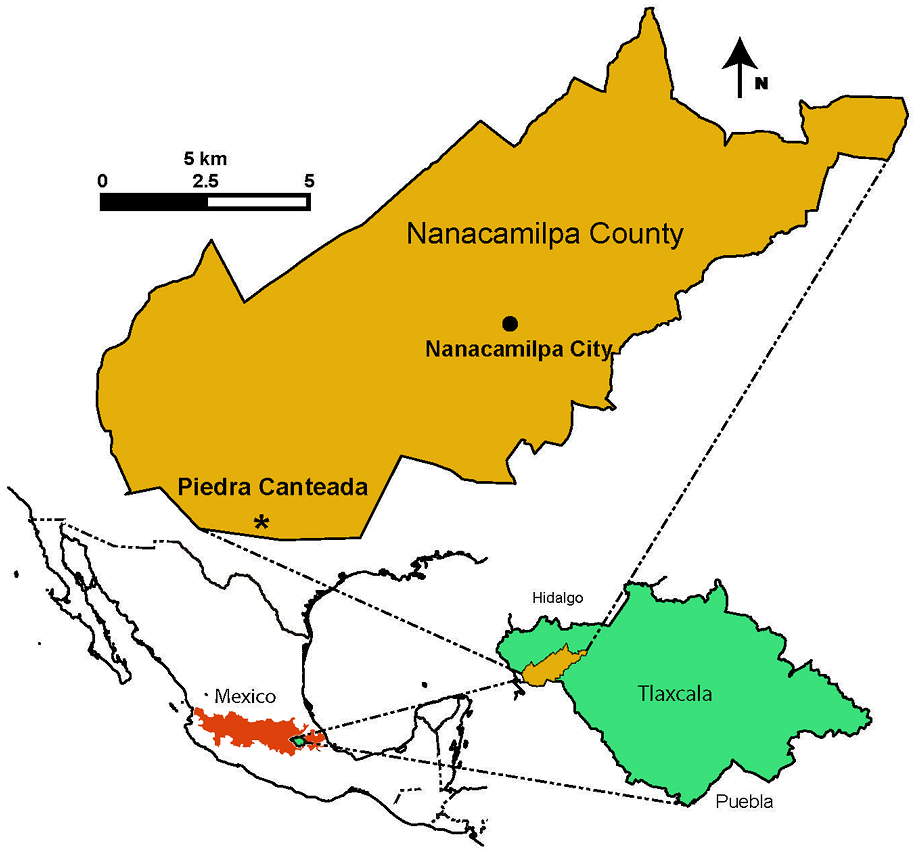
Figure 1 Map of the sampling site in Piedra Canteada, Nanacamilpa (brown), Tlaxcala (green), Mexico. Range of the Trans-Mexican Volcanic Belt (red).
Sampling and description. L. squarrosa basidiomata were collected in a Pinus-Quercus forest area which is known by local mushrooms pickers as a fruiting area for this species of Laccaria. The collection was made in a small area where Pinus pseudostrobus dominates (Figure 2). The basidiomata were identified in the field as L. squarrosa due to the squarrose surfaces on the stipe which is a diagnostic macromorphological feature of this species. We compared the macroscopic and microscopic characters of our samples with those described for the L. squarrosa type specimen (Ramos et al. 2017), and as additional evidence we extracted DNA and amplified the internal transcribed spacer (ITS) region (see the following section) from the basidiomata collected in the study area to confirm their identity. Soil samples were collected below the basidiomata following Agerer (1991) and Gardes & Bruns (1996). A stereomicroscope (Leica S8APO) was used to separate fine roots from the soil samples to select, photograph, and describe the morphology of the ectomycorrhizae that were found (Agerer 1987-2002). From each ectomycorrhiza, hand-made sections were made (cross and longitudinal), mounted in 3 % KOH and observed under a compound light microscope (Leica DM2500) for microscopic characterization (Agerer 1987-2002, DEEMY 2021). The ectomycorrhizae were preserved in 75 % ethanol and tips were stored at -20 °C for DNA extraction to identify the symbionts.
DNA extraction, PCR, and phylogenetic analyses. Total genomic DNA from both basidiomata and ectomycorrhizae was obtained using a modified cetyltrimethylammonium bromide (CTAB) procedure of Gardes & Bruns (1993). The polymerase chain reaction was performed targeting the nuclear ribosomal ITS regions using the primers ITS1F and ITS4 (White et al. 1990). PCR conditions were performed as follows: 94 °C for 5 min; then 35 cycles of 94 °C for 30 s, 55 °C for 30 s, and 72 °C for 30 s; followed by a final elongation step of 72 °C for 7 min. We constructed a dataset of aligned sequences obtained from root tips and basidiomata using the Phylogenetic Data Editor (PhyDE, v.0.997, Muenster, Germany, www.phyde.de). Genbank sequences that showed highest similarity scores (> 97) using the BLAST tool (Altschul et al. 1997) and sequences used in previous studies (Osmundson et al. 2005, Wilson et al. 2013) were included in the analyses. Cortinarius violaceus (L.) Gray (DQ486695) and Psathyrella rhodospora G. Weaver & A.H. Sm. (DQ486695) were included as outgroups (Table 1). To identify the host tree species at the nucleotide level, the plastid/chloroplastic single-locus trnL region was amplified from DNA isolated from the root tip. We amplified the trnL region using trnC/trnD primer pairs (Tedersoo et al. 2008). A second dataset was constructed including the new trnL sequences and 10 sequences of Pinus species retrieved from BLASTN results in GenBank (Altschul et al.1997) and one of Pseudolarix amabilis (NC030631) included as an outgroup. Both datasets (ITS-trnL) were separately aligned using the MUSCLE program (Edgar 2004). The best DNA model was selected using Mega 6.06 (Tamura et al. 2013). For the ITS dataset, Maximum likelihood (ML) and Bayesian inference (BI) were used. ML analysis was performed using MEGA 6.06 (Tamura et al. 2013) with 500 bootstrap replicates, while the BI analysis was estimated using MrBayes on XSEDE (3.2.6) using the CIPRES portal (Miller et al. 2010), employing two independent runs of 1,000,000 generations. For each run, four chains were employed, and one tree was sampled every 1,000 generations. respectively. Only Bootstrap values (BS) higher than 75 and Bayesian posterior probabilities (BPPs) above 0.85 were considered significant. As with the ITS dataset, ML analysis of the trnL dataset was performed using MEGA 6.06, and only BS values higher than 65 were considered significant.
Table 1 Laccaria species included in this study: voucher specimen, location, and sequence accession numbers. Black asterisk (*) indicates new sequences generated in this study.
| Taxon | Voucher Specimen | Location | Genbank ITS |
|---|---|---|---|
| Laccaria sp. | ALB183 | Tibet, China | JX504092 |
| Laccaria sp. | GMM6019 | Costa Rica | KU685757 |
| Laccaria sp. | GMM6012 | Costa Rica | KU685758 |
| L. alba Zhu L. Yang & Lan Wang | AWW438 | Yunnan, China | JX504094 |
| L. alba | F1120750 | China | JX504126 |
| L. amethystea (Bull.) Murrill | FP-98556 | Vorpommern, Germany | DQ499640 |
| L. amethystea | TUB 011464 | Germany | AF539737 |
| L. amethysteo-occidentalis G.M. Muell. | AWW556 | California, USA | JX504107 |
| L. amethysteo-occidentalis | AWW590 | Oregon, USA | JX504112 |
| L. amethystina Cooke | GMM7041 | Caucasus, Russia | KU685654 |
| L. amethystina | GMM7621 | France | JX504150 |
| L. angustilamella Zhu L. Yang & L. Wang | BAP226 | Yunnan, China | JX504118 |
| L. angustilamella | HKAS58714 | Yunnan, China | JX504168 |
| L. aurantia Popa, Rexer, Donges, Zhu L. Yang & G. Kos | KUN-F78557 Type | Yunnan, China | JQ670895 |
| L. aurantia | MB-FB-101109 | Yunnan, China | JQ681209 |
| L. bicolor (Maire) P.D. Orton | AWW539 | Illinois, USA | KM067817 |
| L. bicolor | AWW537 | Illinois, USA | JX504105 |
| L. nobilis A.H. Sm. | F1091206 | Michigan, USA | KU685636 |
| L. ochropurpurea (Berk.) Peck | JMP0038 | Wisconsin, USA | EU819479 |
| L. ochropurpurea | PRL4777 | Illinois, USA | KU685733 |
| L. ochropurpurea | KH_LA06_016 | Louisiana, USA | KU685721 |
| L. ochropurpurea | PRL3777 | Illinois, USA | KU685732 |
| L. proxima (Boud.) Pat. | F1133825 | Mississippi USA | KU685642 |
| L. roseoalbescens T.J. Baroni, Montoya & Bandala | LM5042 | Veracruz, Mexico | KJ874327 |
| L. roseoalbescens | LM5099 Type | Veracruz, Mexico | KJ874328 |
| L. salmonicolor A.W. Wilson & G.M. Muell. | GMM7596 Type | Tibet, China | JX504143 |
| L. squarrosa Bandala, Montoya & A. Ramos | DM63 Type | Veracruz, Mexico | MF669958 |
| L. squarrosa | DM93 | Veracruz, Mexico | MF669959 |
| L. squarrosa | DM121 | Veracruz, Mexico | MF669960 |
| L. squarrosa* | Hernández-Santiago 67 | Tlaxcala, Mexico | MT026967 |
| L. squarrosa* | Hernández-Santiago 65 | Tlaxcala, Mexico | MT026968 |
| L. squarrosa* | Martínez-Reyes 273 | Tlaxcala, Mexico | MT026969 |
| L. squarrosa* | Martínez-Reyes 274 | Tlaxcala, Mexico | MT026970 |
| L. trichodermophora G.M. Muell. | GMM7697 | Texas, USA | KM067863 |
| L. trichodermophora | GMM7698 | Texas, USA | KM067864 |
| L. trichodermophora | GMM7703 | Texas, USA | KM067865 |
| L. trichodermophora | GMM7712 | Texas, USA | KM067866 |
| L. trichodermophora | GMM7714 | Texas, USA | KM067867 |
| L. trichodermophora | GMM7716 | Texas, USA | KM067869 |
| L. trichodermophora | TENN42523 Type | Texas, USA | DQ149868 |
| L. trichodermophora | Montoya 4394 | Veracruz, Mexico | MF669962 |
| L. trichodermophora | Bandala 4282 | Veracruz, Mexico | MF669963 |
| L. trichodermophora | F1111951 | Costa Rica | KU685640 |
| L. trichodermophora | KH_LA06_013 | Lousiana, USA | KM067881 |
| L. trichodermophora | GMM7733 | Texas, USA | JX504157 |
| Psathyrella rhodospora M.G.Weaver & A.H. Sm. | MP133 MN | ------- | DQ267129 |
| Cortinarius violaceus (L.) Gray | MTS 4854 (WTU) | Washington, USA | DQ486695 |
Results
Four soil samples were collected beneath basidiomata of Laccaria squarrosa (MT026967 and MT026968) (Figure 3) from different sites in a pure Pinus pseudostrobus stand in Nanacamilpa, Tlaxcala, Mexico. A total of eight morphotypes were found in the soil samples and photographed, described, and sequenced. After BLAST analysis (Genbank), the samples labeled as Martínez-Reyes 273 ECM (MT026969) and Martínez-Reyes 274 ECM (MT026970), were confirmed as L. squarrosa. In our phylogenetic analysis, these samples clustered together (with high bootstrap and posterior probabilities values, BS = 99, BPPs = 1) with the sequence from the basidiomata of L. squarrosa collected in the study area and the sequence of the type specimen (Figure 4). In the case of the host tree the ML analyses clustered together our samples (Martínez Reyes 273, MW082603; Martínez Reyes 274, MW082604) with Pinus pseudostrobus sequences with a moderate but significant support value (BS = 65; Figure 5).

Figure 3 Laccaria squarrosa. A-B Mature basidiomata in their natural environment in Piedra Canteada, Nanacamilpa, Tlaxcala, Mexico (Hernández-Santiago 65); C-D Scanning electron micrograph of echinulate spores (Hernández-Santiago 65).
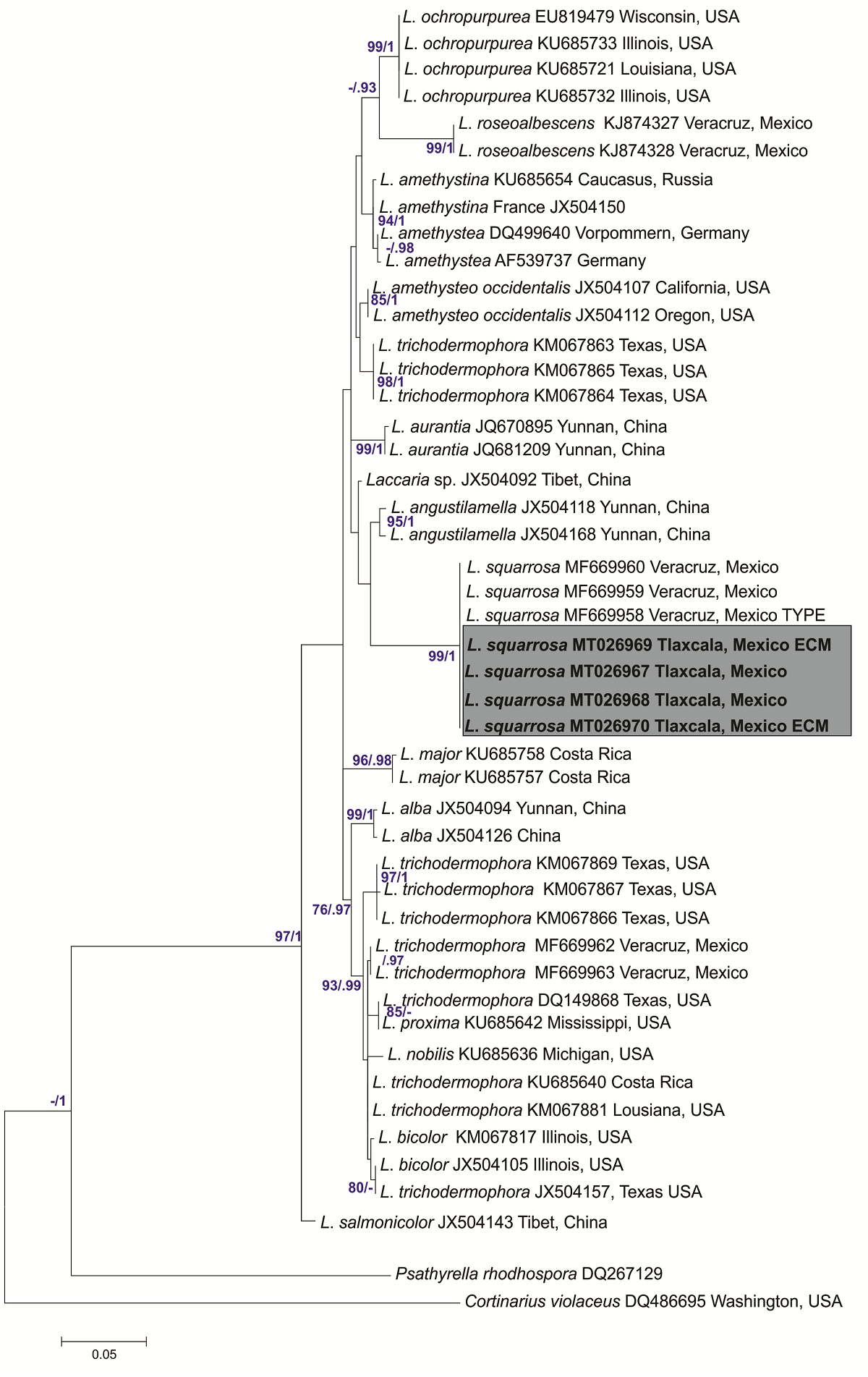
Figure 4 Phylogenetic relationships of Laccaria squarrosa based on ITS sequences from basidiomata and ECM root tips and inferred from the maximum likelihood tree, including bootstrap values (≥ 70) and Bayesian posterior probabilities (≥ 0.95). New sequences obtained here are indicated in bold. Specimen vouchers, accession numbers of ITS sequences, and country of origin are indicated for each branch.
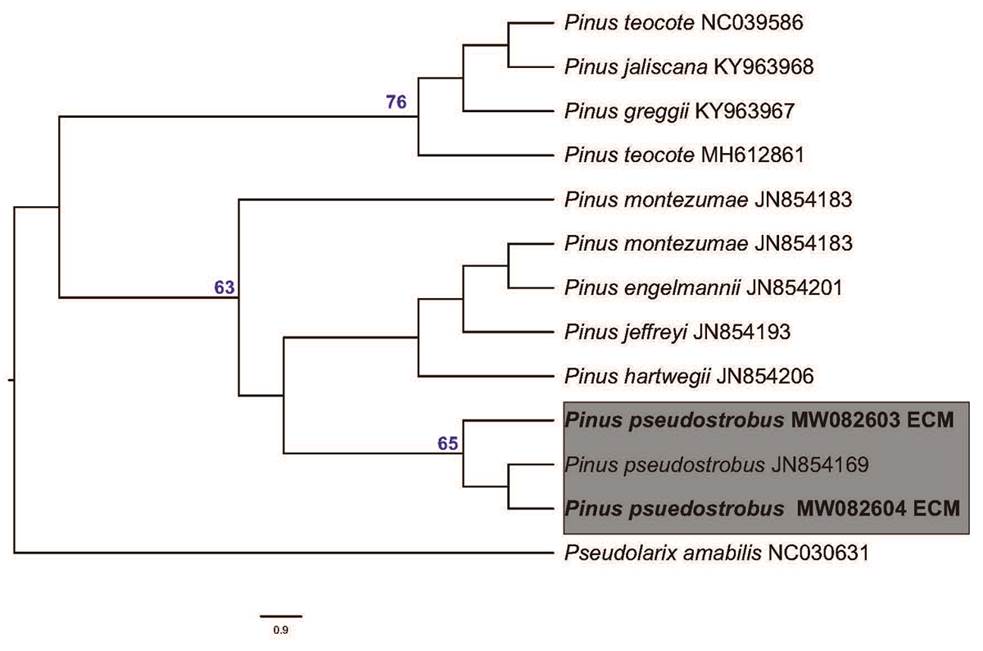
Figure 5 Phylogenetic relationships of Pinus species based on trnL sequences and inferred from the maximum likelihood tree, including bootstrap values (≥ 65). New sequences obtained here are indicated in bold.
Description of the ectomycorrhiza. Laccaria squarrosa (Bandala, Montoya & A. Ramos) + Pinus pseudostrobus Linl. (Figure 6). Ectomycorrhizal systems 1.83 mm in length and 3.15 mm wide, dichotomous, with short branches in two levels of ramification, branches 0.54-0.80 mm in length × 0.38-0.41 mm wide, straight to sinuous, brownish to pale brownish, dark-brownish to the ends, pale at the tip; smooth with some white appressed fibrils that give a glistening aspect, emanating hyphae scattered and loose; tips cylindrical to rounded; mantle opaque at distal end, hydrophilic, hyaline; rhizomorphs and sclerotia absent (Figure 6A-B). Longitudinal section: Mantle 15-35 μm thick, two layered (Figure 6C). Outer mantle 14 μm wide, loose, plectenchymatous, not gelatinized, with cylindrical cells up 2.5-3.5 μm, thin-walled, smooth. Inner mantle 18 μm broad, plectenchymatous, compact, hyphae up to 5 μm wide, with cylindrical cells 2.5-4 µm hyaline, thin-walled, tannin cells present, parallel to root axis. Hartig net palmetti, paraepidermal, penetrating the second layer of cortical root cells (Figure 6C-D), the hyphae between the root cells up to 3.5 μm wide, rounded; cortical cells rectangular, cylindrical 55-75 × 45-60 μm (in longitudinal section) and 27-65 × 27-51 μm (in cross-section) (Figure 6C-D). Clamp connections absent. Cross section: Mantle disposed in a plectenchymatous arrangement (Figure 6D), two layers discernible, hypha disposed in parallel and prostrate, some loose hyphae in the external mantle and compact in the inner mantle, tannin cells cylindrical, cortical cells 3.5 μm wide rectangular to rounded, hyphae between cortical cells cylindrical. Plan view: Outer mantle in a plectenchymatous layer, hyphae in an irregular arrangement composed of cylindrical cells 3-4 μm wide, sinuous, compact layer thin-walled, smooth hyphae (Figure 6E). Inner mantle in a plectenchymatous layer, in an irregular arrangement composed of cylindrical 2.5-5 (8) μm wide sinuous, tortuous; compact layer, thin-walled, smooth hyphae (Figure 6F).
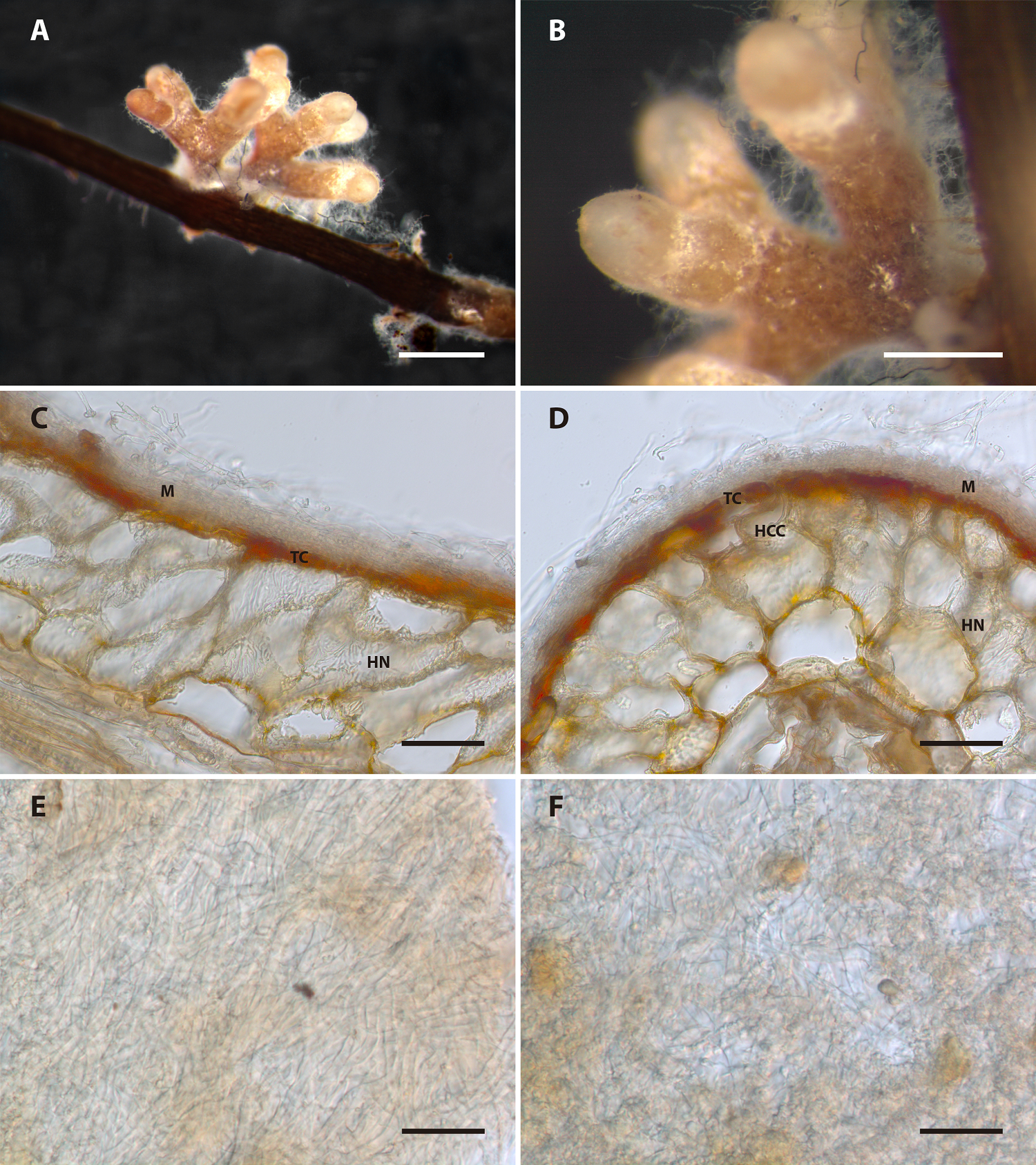
Figure 6 Laccaria squarrosa + Pinus pseudostrobus ectomycorrhiza. A) Ectomycorrhizal (ECM) system; B) Details of the pruinose surface and tips of ECM roots; C) Tangential section showing mantle (M), Tannin cells (TC), and Hartig net (HN); D) Ectomycorrhiza in cross-section showing Mantle (M), Hartig net (HN), Tannin cells (TC), and hyphae between cortical cells (HCC); E) Plan view of the outer mantle; F) Plan view of the inner mantle. Bars 1 mm in A; 0.5 mm in B; 50 μm in C) and D); and 20 μm in E) and F).
Discussion
For the first time, we describe the ectomycorrhiza of Laccaria squarrosa + Pinus pseudostrobus in natural conditions and extend the distribution and host associations of L. squarrosa. L. squarrosa was previously known only from the type locality (in Veracruz state located in eastern Mexico) and associated only with Fagus grandifolia var. mexicana, and from a traditional Purépecha market (in Michoacán, located in western Mexico) without stating the associated host trees. Our new data show that the species is distributed along the Trans-Mexican volcanic belt (TCMV; Figure 1). Detecting the ECM association in the field (through the morphological and molecular characterization of the mycobiont and host tree) is the first step toward the identification of suitable hosts for forest management and reforestation programs. This is the second record of L. squarrosa in Mexico and confirms that this species is distributed along the Trans-Mexican-Volcanic-Belt in association with two different ECM host tree species. Laccaria squarrosa can therefore be used not only as native inoculum for reforestation programs in the study area (Nanacamilpa, Tlaxcala), but also in the type locality of the species (Acatlán, Veracruz, Mexico), where the Fagus grandifolia var. mexicana forest is endangered. Further sampling should be performed in Mexico to identify additional ECM host tree species associated with this ECM fungus.
The ectomycorrhiza here described displays some similarities with the previously documented ectomycorrhizae of other Laccaria species associated with different host trees (Table 2). They display similar colors (except those formed by L. amethystina Cooke + Quercus sp. and L. trichodemorphora G.M. Muell. + Pinus montezumae), present emanating hyphae (however, abundance differs among the species), lack of rhizomorphs (except those formed by L. amethystina Cooke + Quercus sp. and L. amethystina Cooke + Betula sp.) and has a plectenchymatous outer mantle. Features such as the ramification type, mycorrhizal system length, and mantle thickness vary among all the species. However, more detailed descriptions are needed for further discussion about Laccaria ectomycorrhizae morphotypes.
Table 2 Diagnostic morphological characteristics of ectomycorrhizas established between species of Laccaria associated with different host trees. RT: Ramification type; MSL: Mycorrhizal system length; MC: Mycorrhiza color; MS: mantle surface; EHP: Emanating hyphae presence; Rhiz: Rhizomorphs; MT: Mantle thickness; MO: Mantle organization; D: Dichotomous; M-pin: Monopodial-pinnate; M-pyr: monopodial pyramidal; C: Coralloid; Pl: Plectenchymatous; Ps: pseudoparenchymatous; Nd: No data; N: natural conditions, A: artificial conditions.
| Species + host | RT |
MSL (mm) |
MC | MS | EHP | Rhiz | MT (μm) |
MO | Reference | Origin |
|---|---|---|---|---|---|---|---|---|---|---|
| Laccaria squarrosa + Pinus pseudostrobus | D | 1.83 | Brownish to pale brownish, dark-brownish | White fibrils glistening aspect | Scattered and loose | Absent | 15-35 | Pl | *This study | N |
| L. amethystina + Betula pendula | M-pin | 1-2 | Ochre, yellowish brown, or orange or red | Loosely woolly | Concentrated distally | Present | 36-93 | Pl | Cuvelier 1991 | N |
| L. amethystina + Fagus sp. | M-pin M-pyr | 1- 6 | Brown, ochre, yellowish brown of grey | Loosely woolly | Concentrated distally | Absent | 25-70 | Pl | DEEMY 2021 | N |
| L. amethystina + Quercus robur. | M-pyr | Nd | Violet, dark reddish blue or white | Loosely cottony | Abundant | Present | Nd | Pl | DEEMY 2021 | N |
| L. bicolor + Pinus montezumae | D | 2.5-3.66 | Brown to pale brownish and orange brownish | Metallic aspect | Abundant | Absent | 23.5 -31.4 | Pl | Santiago-Martínez et al. 2003 | A |
| L. bicolor + Pinus montezumae | D | Nd | Reddish-brown | Cottony | Scattered | Nd | Nd | Ps | Rodríguez-Gutiérrez et al. 2019 | A |
| L. bicolor + P. patula | D | 1-4.5 | Brown to orange, reddish brown | Glistening aspect | Nd | Absent | Nd | Pl | Carrasco-Hernández et al. 2010 | A |
| L. laccata + Pinus patula | D | 5.5 | Whitish orange to greyish brown | Pubescent | Present | Absent | 10-50 | Pl | Mohan et al. 1993 | A |
| L. laccata var. laccata + P. montezumae | D | Nd | Golden yellow | Cottony | Scattered | Nd | Pl | Rodríguez-Gutiérrez et al. 2019 | A | |
| L. proxima. + Betula spp. + Picea sitchensis +Pinus spp. | M-pin M-pyr | 1-8 | Brown to white | Shiny | Present | Absent | 15-40 | Pl | Ingleby et al. 1990 | A |
| L. proxima + P. pseudostrobus | D | 1-8 | Pale Brown to dark brown | Nd | Nd | Nd | Nd | Pl | Carrasco-Hernández et al. 2010 | A |
| L. tortilis + Betula pendula and Picea sitchensis | M-pin M-pyr | 7 | Brown to white | Loosely or cottony | Present | Absent | 10-30 | Pl | Ingleby et al. 1990 | A |
| L. trichodemorphora + Pinus montezumae | D C | 0.4-2.9 | White to pale white | Loosely cottony | Abundant | Absent | 14.4 -81.5 | Pl | Galindo-Flores et al. 2015 | A |
| L. trichodermophora + P. montezumae | D | Nd | Orange, yellowish brown, bright orange | Cottony | Nd | Nd | Nd | Pl | Rodríguez-Gutiérrez et al. 2019 | A |
| L. vinaceobrunnea + P. montezumae | D | Nd | Orange to brownish yellow | Cottony | Nd | Nd | Nd | Ps | Rodríguez-Gutiérrez et al. 2019 | A |
Both the previous record of the holotype of Laccaria squarrosa under a pure stand of Fagus grandifolia var. mexicana and our results suggest that L. squarrosa is a generalist species that can be associated either with Angiosperms or Gymnosperms. This record of L. squarrosa with both Fagus and Pinus supports the hypothesis of Wilson et al. (2017) who suggest that Laccaria´s association with Fagaceae aided their dispersal into the northern hemisphere and its eventual association with Pinus. Although in our sampling area there were other ECM host trees (Abies religiosa, Pinus teocote and P. montezumae, and Quercus spp.) we only found basidiomata of L. squarrosa associated with P. pseudostrobus. This result was confirmed by our sequencing and phylogenetic analyses with a good support (BS = 65; Figure 5). Further fieldwork sampling of root tips in the study area would provide valuable ecological information of its possible symbiotic associations with other host species.
Despite the great diversity of Laccaria species and their associated host trees in both northern and southern hemispheres, only thirteen descriptions of ectomycorrhizae of six species (L. amethystina, L. bicolor, L. proxima, L. tortillis, L. squarrosa, and L. trichodermophora) have been described (Table 2). Most of the records are ectomycorrhizae synthesized under artificial conditions and only four of them have described the morphoanatomical features of Laccaria ectomycorrhizae collected in natural conditions (Ingleby et al. 1990, Cuvelier 1991, DEEMY 2021). Therefore, the ectomycorrhiza here described represent one of the few morphotype descriptions of the ecologically and economically relevant Laccaria genus in natural conditions in the world.
Laccaria squarrosa was recorded as an edible wild mushroom representing an important food source for local gatherers in the study area, who have traditionally considered the forests as a source of ecosystem services and food. Our robust confirmation of the establishment of ectomycorrhizal symbiosis between L. squarrosa and P. pseudostrobus, with morphoanatomical, molecular, and phylogenetic evidence, open the way to future biotechnological research to enhance sustainable forest management in the study area. Production of bioinoculants using this pioneer fungal mycobiont in P. pseudostrobus plant production (either as mycelial inoculum, dry pilei, or spore slurries) is a real prospect, as it has been previously demonstrated for other phylogenetically-related Neotropical Laccaria species (Pérez-Moreno et al. 2020). In doing so, it is hoped that we can protect endangered Mexican forest species and ecosystems by promoting efficient plant production and successful reforestation.











 nova página do texto(beta)
nova página do texto(beta)




