Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Botanical Sciences
versão On-line ISSN 2007-4476versão impressa ISSN 2007-4298
Bot. sci vol.92 no.3 México Set. 2014
Ecología
Cacti community structure in a tropical Mexican dry forest under chronic disturbance
Estructura de la comunidad de cactáceas en una selva seca tropical mexicana bajo disturbio crónico
Luís Antonio Arias-Medellín1, Alejandro Flores-Palacios2 and Cristina Martínez-Garza2,3
1 Facultad de Ciencias Biológicas, Universidad Autónoma del Estado de Morelos, Cuernavaca, Morelos, México.
2 Centro de Investigación en Biodiversidad y Conservación, Universidad Autónoma del Estado de Morelos, Cuernavaca, Morelos, México.
3 Autor para la correspondencia: cristina.martinez@uaem.mx.
Received: April 13th, 2013
Accepted: September 4th, 2013
Abstract
When agricultural lands in formerly dry forest are abandoned, chronic disturbance by livestock grazing and wood extraction may set back successional processes precluding older dry forest structure from be reached. We compare cacti density, richness, diversity and evenness in twelve 50 × 50 m plots: six plots in secondary dry forest and six plots in older conserved dry forest at Sierra de Huautla, Central Mexico. Cacti were identified, the life-form of each species was recorded and size was measured to assign individuals into four life-stage classes. Nine cacti species were registered across both forest types. In this dry forest, richness, diversity and evenness of cacti community increased in sites under chronic disturbance, whereas density was not significantly affected. Size structure at community level in the conserved forest showed an increase of individuals with size, whereas in the secondary forest, size structure showed an uneven distribution of individuals, as in populations that regenerate by pulses. In the secondary forest, there was a lower proportion of adults and they were in average, smaller than those in the conserved forest. Chronic disturbance may eventually change cacti community structure, affecting animals that depend on large arboreal cacti for food, perches and/or refuge; therefore, maintenance of biodiversity will be at stake.
Key words: cattle ranching, Opuntia spp., Pachycereus grandis, restoration ecology, secondary dry forest, Sierra de Huautla.
Resumen
Cuando las tierras de cultivo son abandonadas, la ganadería extensiva y la extracción de productos naturales son perturbaciones crónicas que puede afectar el proceso de sucesión natural que llevaría a la selva estacional a su estructura original. Se comparó la densidad, riqueza, diversidad y equitatividad de la comunidad de cactáceas en 12 parcelas de 50 × 50 m: seis parcelas localizadas en la selva secundaria y seis parcelas en la selva conservada en Sierra de Huautla, centro de México. Todas las cactáceas fueron identificadas, se registró su forma de vida y todos los individuos se clasificaron en cuatro etapas de su ciclo de vida según el tamaño. Nueve especies de cactáceas fueron registradas en ambos tipos de selva. En esta selva estacional, la riqueza, diversidad y equitatividad de la comunidad de cactos aumentó en los sitios bajo perturbación crónica, mientras que la densidad no se vio afectada. En la selva conservada, la estructura de tamaños de la comunidad mostró un mayor número de individuos de tallas mayores, mientras que en la selva secundaria la estructura de tamaños mostró una distribución no uniforme de individuos, como sucede en poblaciones que muestran pulsos de regeneración. En la selva perturbada se registró una menor proporción de individuos adultos y estos fueron, en promedio, más pequeños que los registrados en la selva conservada. La perturbación crónica puede eventualmente cambiar la estructura de la comunidad de cactáceas con efectos negativos para los animales que dependen de la comida, perchas y refugios que brindan los grandes cactos arborescentes, lo que pondría en peligro el mantenimiento de la biodiversidad en este ecosistema.
Palabras clave: ecología de la restauración, ganadería, Opuntia spp., Pachycereus grandis, selva estacional secundaria, Sierra de Huautla.
The Cactaceae family includes more than 2,000 species of many life forms, from smaller prostrate species to large arboreal ones (Barthlott and Hunt, 1993). Mexico has around 63 genera and 913 taxa (669 species and 244 subspecies); a high percentage of those species (35%) are considered threatened by the destruction of their habitat (Hernández and Godínez, 1994; Hunt, 1999; Guzmán et al., 2003). Worldwide analysis has shown that Mexico is the most important country to preserve cacti diversity in different ecosystems as deserts, xerophitic shrub lands and tropical dry forests (Bravo-Hollis, 1978; Bravo-Hollis and Sánchez-Mejorada, 1991a,b; Anderson, 2001; Ortega-Baes and Godínez-Álvarez, 2006). In tropical dry forests, Cacta-ceae is the third most important family by number of species (Trejo-Vázquez, 1998); however, this family is seldom evaluated in this ecosystem, which is indeed the third one with the highest forest cover loss worldwide (Hansen et al., 2010). Given that tropical dry forests are dominated by wind-dispersed species, cacti represent an important source of fleshy fruits for birds and bats during the dry season (Naranjo et al., 2003; Reyes-Agüero et al., 2006) when most of the tree species do not produce fruits (De León-Ibarra, 2005; Martínez-Garza et al., 2011). Therefore, the conservation of this family is crucial to maintain ecosystem processes, as trophic chains.
Chronosequences studies in tropical dry forests have shown that a forest structure similar to that of mature forest may be reached after more than 30 years of abandonment of agricultural land (Kennard, 2002; Ruiz et al., 2005). However, young secondary forests originated from abandoned agricultural land where successional processes took place (Brown and Lugo, 1990; Finegan, 1996) may experience chronic disturbance; for example, in many areas with secondary forest, cattle graze and browse herbs and grasses at the understory and also at the lower foliage of trees (Stern et al., 2002). Additionally, disturbance due to extraction of wood for timber, firewood or fence posts is frequent (Linares-Palomino and Ponce, 2005). This kind of forest, with different ages and degrees of disturbance, now cover more area than conserved older forest in the tropics (Chazdon et al., 2007). After lands are no longer used for cultivation, chronic anthropogenic disturbance may continuously set back successional processes, precluding mature forest structure to be reached and threatening cactus species inhabiting those areas.
Disturbance may have positive or negative effects on populations of cacti. Some studies have shown that land use transformation and cattle ranching are the main factors affecting negatively the population dynamics of cactus species (e.g. del Castillo, 1987; Bowers, 1997; Martorell and Peters, 2009; Flores et al., 2010). Other studies have revealed that moderate levels of human activities may increase densities of some globose species (Martorell and Peters, 2005; Portilla-Alonso and Martorell, 2011). Demography of cactus species have been frequently studied (reviewed in Godínez-Álvarez et al., 2003) but its community structure and the effect of disturbance on it has not been evaluated. We may expect a differential effect of disturbance in the structure of a community depending from dominant life-form. For example, diversity, richness, and evenness of herbs and grasses increase under extensive cattle ranching in pasture-lands (Nai-Bregaglio et al., 2002; Floyd et al., 2003; Hayes and Holl, 2003), whereas livestock presence affects negatively tree richness and diversity in the dry forests (Roth, 1999; González-Iturbe et al., 2002; Stern et al., 2002; VanderWerf, 2004). On the other hand, selective logging may not affect tree evenness in the dry forest (Monroy-Ortiz, 1997), but livestock presence does (Sánchez-Velásquez et al., 2002). However, it is not known whether the effect of disturbance on cactus communities inhabiting the dry forest will follow patterns found in other ecosystems under chronic disturbance.
Population size structure is affected by recruitment and mortality rates. For cactus species, population size structure is frequently an uneven distribution of individuals in the different size classes (see Appendix 1 in Godínez-Álvarez et al., 2003). The uneven population size structure is associated to events of recruitment in pulses favoured by the occasional coincidence of some suitable biotic and abiotic conditions, as rain, temperature, low herbivory and/or presence of nurse plants (Godínez-Álvarez et al., 2003). A couple of studies in the dry forest have revealed that cacti show an uneven size distribution (Contreras and Valverde 2002; Méndez et al., 2004). Other studies, also in the dry forest, have shown a decreasing number of cactus individuals with size (Ferrer-Cervantes et al., 2012; Valencia-Díaz et al., 2012) which is related to constant regeneration and lower mortality of adults (Godínez-Álvarez et al., 2003). Irrespective of population size structure, neutral effect of disturbance on size structure of cactus species have been reported (e.g. ureta and Martorell, 2009). However, given that cattle may step on smaller life-stage classes (del Castillo, 1987), regeneration of cacti in arid environments may be reduced and thus, individuals reaching adult stage may be fewer compared to habitats experienced undisturbed succession. Therefore, chronic anthropogenic disturbance in older secondary forest may result in an overall size structure similar to the one in the conserved forest but with a lower proportion of individuals from older life-stages.
We investigated cacti community structure in two localities of the tropical dry forest of Sierra de Huautla, Morelos, central Mexico, in fragments of older conserved dry forest and in successional areas more than 30 years old which are frequently disturbed by livestock activities and selective extraction of wood (called from now on, secondary forest). We tested the following hypothesis: due to chronic disturbance, (1) density, diversity, richness, and evenness of cacti community differ in older conserved compared to secondary forest, and (2) the cacti community in the dry forest shows an uneven size structure with a lower proportion of adults in the secondary forest compared to the older forest.
Methods
Study area. This study was carried out in lands close to the localities of El Limón de Cuauchichinola (1,220 m of altitude, called from now on El Limón) and Quilamula (1,070 m of altitude), both located within the Sierra de Huautla Biosphere Reserve, Morelos, central Mexico. The Reserve (18° 20' 10" - 18° 34' 20" N and 98° 51' 20" - 98° 08' 15" W) comprises 59,030 ha where the main vegetation type is tropical dry forest. Mean annual temperature is 24.5 °C and average annual rainfall (1980-2001) amounts to 840 mm (CONAGUA, Gerencia Regional Balsas, unpublished data), ~90% falling between late May and October. During the dry season (November to April), most of the trees lose their leaves. Soils are shallow (< 30 cm in depth), with sandy-loam texture (CONANP, 2005). In the Reserve, the most important plant families by decreasing number of species are Fabaceae, Poaceae, Asteraceae, Burseraceae, and Cactaceae (CONANP, 2005). More than 70% of species and genera of cacti reported for Morelos has been recorded in the Reserve: 12 genera and 28 species (Martínez-Alvarado, 1985; CONANP, 2005). Most of the trees in this forest have compound leaves and some reach 8 to 12 m in high, with convex or flat canopies (Rzedowski, 1978).
The landscape in the Reserve is a mosaic of fragments of old and young tropical dry forest in different degrees of conservation, surrounded by agricultural lands and small towns. Two main forest types are recognized in the localities studied here: secondary forest and conserved forest. The secondary forest resulted from large parts of forest cleared more than 40 years ago (~1970), which were used for maize cultivation for ca. 6 years, and later abandoned. The secondary forest has an average tree density (> 5 cm diameter at breast height: 1.3 m) of 264 ind./ha from 14 species, from which Acacia cochliacantha (Fabaceae), Ipomoea pauciflora (Convolvulaceae), Acacia farnesiana (Fabaceae), and Mimosa benthamii (Fabaceae) are dominant (Martínez-Garza et al., 2011). These forests are chronically disturbed by anthropogenic activities: during the rainy season ca. 600 heads of livestock feed in these secondary forests (~7 heads/ha); these heads of livestock are brought from neighbouring towns. Accordingly to land owners and current research (B. Maldonado, unpublished data), cattle are maintained in farms during the dry season but goats, pigs and horses graze in the secondary forest throughout the year. Wood extraction from the secondary forest is a common activity and villagers extract between 74 and 124 kg of wood per trip (Monroy-Ortiz, 1997).
What we call conserved forest corresponds to older growth forest never deforested where cattle from neighbouring towns do not graze, although cattle from local owners occasionally do; nonetheless, cattle is never found in densities higher than 1 head/ha (G. Pacheco, local farmer, pers. comm.). In conserved forests, wood extraction is seldom practiced since the preferred tree species are not frequent (B. Maldonado, unpublished data). The most common canopy trees in the conserved forest are Conzattia multiflora, Lysiloma acapulcense, L. divaricata (Fabaceae), several species of Bursera (Burseraceae) and Ceiba (Bombacaceae) (CONANP, 2005). In this conserved forest, average tree density is 905 ind./ha (unpublished data). Native herbivores in both forest types are deer (Odocoileus virginianus) and rodents (Lyomis sp., Baiomys sp., Sigmodon sp.; D. Valenzuela, unpublished data).
Sampling protocol. In January 2006, 12 plots of 50 × 50 m were selected in both forest types (secondary and conserved forest) in El Limón (N = 6) and Quilamula (N = 6) for a total sampling area of 3 ha. Within each locality, we selected three plots in each forest type with similar slopes (< 30%) and soil characteristics (J. Campo, unpublished data). These plots are part of a large project of restoration ecology which aims to favour the coexistence of biodiversity and the economic activities of the region using enrichment plantings and exclusion of disturbance (Martínez-Garza et al., 2011). Distance between localities is 8.67 km. Distance among plots ranged from 0.08 to 1.59 km. All cacti found within the plots were counted and measured in January 2007. To analyze population size structure, each individual was assigned to a life-stage class (seedling, juvenile, pre-reproductive, and reproductive adult) based in size and presence of reproductive structures (Martínez-Alvarado, 1985; Table 1).
Statistical Analysis. To evaluate cacti density between forest types overall species, a nested Analysis of Variance (ANOVA) was used, where forest type (secondary and conserved) was nested in locality (El Limón and Quila-mula); forest type had three replicates in each locality. Data were transformed with the logarithm base 10 to fulfil ANOVA assumptions (Shapiro Wilk W = 0.95, P < 0.64). To describe the community structure by forest type, dominance/diversity curves were constructed (Wilson, 1991). The Shannon-Wiener diversity index (H') was calculated using the following formula:
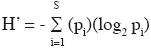 (Magurran, 2004)
(Magurran, 2004)
where H' is the species diversity index, S the number of species, and pi is the proportion of individuals from the total number of species that belongs to the species i. To estimate evenness, the Pielou index was used, but first we calculated the maximum evenness with the following formula:
Hmax = log2S (Magurran, 2004)
where Hmax is the maximum species richness and S is the species richness observed. When the maximum species richness was calculated, the following formula was used to estimate the Pielou evenness index (Krebs, 1985):
J = H/ H (Magurran, 2004)
where J is evenness, Hmax is the observed species richness and H is the maximum species richness. H' was used because it is clearly linked to the dominance/diversity curves (Wilson, 1991). H' and J were transformed with natural logarithms prior to analysis (Shapiro Wilk W = 0.94, P < 0.51 and W = 0.93, P < 0.35 respectively). H' values between forest types were compared using ?-test for each locality (Zar, 1996).
The Simpson diversity index expresses the probability that two individuals taken randomly from a sample belong to the same species and is calculated as:
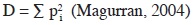
where pi is the proportion of the individual of the i species. As D increases, diversity decreases, so it is more commonly used as its inverse (Dinv = 1/D). It takes values from 1 (when there is only one species) to the total species richness of the community (Magurran, 2004).
The Berger-Parker index is a dominance measure that expresses the proportional abundance of the most abundant species:
B = Nmax/N (Magurran, 2004)
where Nmax is the number of individuals of the most abundant species and N is the total number of individuals in the community. An increase of B indicates a decrease in evenness and diversity, so its reciprocal is used (Brec = 1-B); therefore, an increase of Brec results in an increase in diversity and evenness (Magurran, 2004).
Community size structure (proportion of individuals overall species in each life-stage class) between forest types was compared with Kolmogorov-Smirnov tests and also individually for those species with enough sample size to run statistical analyses. Back-transformed means and standard deviations are shown throughout results. The diversity indexes and species rank curves were calculated using Excel v12.0 and the statistical analyses were carried out using the program R version 2.14.1 (R Development Core Team, 2011).
Results
In total, 943 individuals from nine species were registered, measured and identified (Table 2). Two large arboreal m height), three short arboreal (> 3 m height), one large shrub (~ 2 m height), and three small size life-forms (shrubs, prostrate and globose < 60 cm height) were recorded. All species were registered in both forest types except for Opuntia atropes and Pachycereus weberi, which were found exclusively in the secondary forest (Table 2). The large arboreal species Pachycereus grandis was the most abundant in conserved (70.65% of individuals) and secondary forest (31.75%; Table 2, 3). After P. grandis, the short arboreal O. velutina (27%) and the prostrate O. puberula (22.59%) were the species with the highest abundance in the secondary forest, whereas the small shrub O. pumila (19%) was the most abundant in the conserved forest (Table 2, 3). In average and taking into account all species and life-stage classes, there was a higher cacti density in the secondary (309.14 ± 2.62 ind./ha) than in the conserved forest (119.69 ± 3.85 ind./ha). However, density was statistically similar in both forest types (F(2,8) = 1.94, P > 0.10).
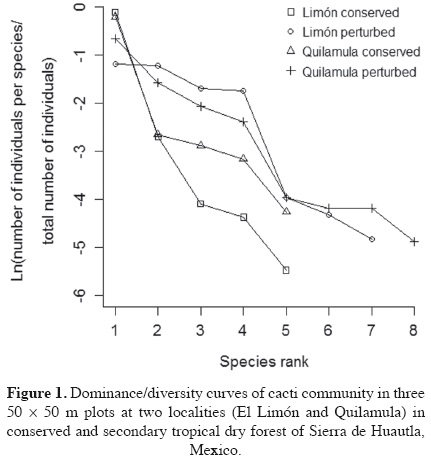
The dominance/diversity curve showed that there was higher richness in the secondary than in the conserved forest. The dominance/diversity curve was steeper in the conserved forest (Figure 1, Table 4) suggesting higher richness and evenness in the secondary forests of both localities. The Shannon-Wiener, inverse Simpson and the reciprocal of Berger-Parker diversity index confirmed the result of the curves: a significantly higher indexes were found in the secondary forest (H' > 1.39, Dinv > 2.88, Brec > 0.53) than in the conserved forest (H' > 0.42, Dinv > 1.23 Brec > 0.82) from two localities (t(2,8) = 4.8, P < 0.005) due to both, a higher richness and a higher evenness in the secondary forest (Figure 1, Table 4).
Size structure. At community level, the proportion of individuals in smaller life-stage classes (seedlings and juveniles) was two times higher in the secondary forest compared to the conserved forest, whereas the proportion of reproductive adults was two times higher in the conserved forest (Table 5). At community level, size structure was significantly different by forest type (Dmax = 310, P < 0.001; Table 5).
Two species, the large arboreal Pachycereus grandis and the small shrub Opuntia pumila had enough individuals at all life-stage classes and in both forest types to run statistical analyses at species level. The population size structure of P. grandis showed that the proportion of individuals in the seedlings and juveniles life-stage classes were 18 and three times higher, respectively in the secondary forest than in the conserved forest; whereas the proportion of individuals in pre-reproductive and reproductive adults classes were almost three times lower in the secondary forest compared to the conserved forest (n = 66 in the secondary forest and n = 219 in the conserved forest, Figure 2A). The population size structure of P. grandis showed significant differences between forest types (Dmax = 114, P < 0.001).
The population size structure of Opuntia pumila showed that the proportion of seedlings and adults was higher in the conserved forest compared to the secondary forest, whereas the proportion of juveniles and pre-reproductive adults were higher in the secondary forest (Figure 2B; Table 5). However, the population size structure of O. pumila was statistically similar between forest types (Dmax = 6, P > 0.1).
Discussion
Richness, diversity and evenness of the community of cacti in the dry forest of Sierra de Huautla increased in sites under chronic disturbance whereas density was not significantly affected. Size structure at community level in the conserved forest showed an increase of individuals with size, whereas in the secondary forest size structure showed an uneven distribution of individuals, as in populations that regenerate by pulses. Further, in the perturbed forest, there was a lower proportion of adults and they were, in average, smaller than those recorded in the conserved forest.
Community structure. Studies at the population level have shown positive, negative or neutral effects of disturbance in cacti density. Some species (called ruderal cacti sensu Martorell and Peters, 2005) may benefit from moderate levels of human activities as the globose species Coryphantha werdermannii in Coahuila, North Mexico (Portilla-Alonso and Martorell, 2011) and the cylindrical cactus Echinocereus lindsayi in Baja California, Mexico (Martorell et al., 2012). In the present study, further analysis at species level revealed that Opuntia velutina and O.puberula were significantly more abundant in the secondary forest under chronic disturbance compared to the conserved forest (t(1,10) = 2.6, P < 0.05 and t(1,10) = 6.4, P < 0.0001, respectively). This is in agreement with a study in the dry forest of Yucatan, Mexico, where it was found that the globose cactus Mammillaria gaumeri benefits from an increase of light after moderate disturbance of the forest canopy (Cervera et al., 2007). Further, four species evaluated in the present study showed similar abundance in secondary and conserved forest: Coryphantha elephantidens (t(1,10) = 1.8, P > 0.1), Pachycereus grandis (t(1,10) = 0.9, P > 0.4), O. pumila (t(1,10)= 0.6, P > 0.5) and Stenoce-reus beneckei (t(1,10) = 1.8, P > 0.1). This was in agreement to the neutral effect of disturbance found for Mammillaria crucigera and M. huitzilopochtli in the Tehuacan Valley, Puebla, Mexico (Martorell and Peters, 2009). More than 60 % of the cactus species occurring in this dry forest showed neutral or positive effect of disturbance; this seems to be reflected in the neutral effect of disturbance in the density of the entire cacti community considering all life-stage classes.
Cacti diversity, richness, and evenness increased in the secondary forest under chronic disturbance. These results are in agreement with what is known for smaller life-forms, as grasses and herbs experiencing faster succession after disturbance. For example, in mountain grasslands of Argentina (Nai-Bregaglio et al., 2002) and in the tropical dry forest of Sierra de Huautla, Mexico (De la O-Toris et al., 2012), a higher diversity and richness of herbs and grasses was found in disturbed areas compared to conserved ones (but see Floyd et al., 2003). In this study, seven out of nine cactus species and more than 55% of individuals registered in both forest types were < 3 m in height. Therefore, the higher richness, diversity and evenness of cacti in the secondary forest may reveal the response to disturbance of the smaller life-forms, similar to what happen to herbs and grasses in natural grasslands under chronic disturbance by livestock.
It is expected that cactus species inhabiting the secondary forest be a subset of those occurring at the conserved old forest. However, in this study, Opuntia atropes and Pachycereus weberi were only found in the secondary forest from one of the localities evaluated (Quilamula). These two species are used as food and also for feeding cattle (Luna-Morales and Aguirre, 2001; López-Acosta et al., 2008; Arias and Terrazas, 2009). Opuntia atropes (commonly named White nopal) is reported as native in various states in central Mexico (Guanajuato, Guerrero, Jalisco, Mexico, Michoacan and Morelos; Instituto de Biología, 2013) and it is also used in traditional medicine and to build living fences (López-Acosta et al., 2008). Further, O. atropes is one of the two species more frequently used as vegetable and therefore it has been cultivated probably since prehistoric times (Callen, 1967). P. weberi is also used as food for cattle and their seeds used to elaborate flatbread (tortillas), sauces (mole), and also for construction (Luna-Morales and Aguirre, 2001; Arias and Terrazas, 2009). The presence of P. weberi has been registered in ancient homegardens and is usually tolerated in agricultural lands (Luna-Morales and Aguirre, 2001). Many plant species widely used by people in the dry forest grow in sites under chronic disturbance (De la O-Toris et al., 2012; Maldonado et al., 2013). The multiples uses of these two species and their presence in low density only in the secondary forest suggest that they could be heavily extracted at both forest and their populations have not recovered. In this study, the presence of two more cactus species in the secondary forest and a similar density of individuals between forest types increased community evenness; this forest structure seems to be the result of some cactus species been favoured by microenvironmental conditions whereas others were heavily extracted.
Size structure. In the conserved forest, the proportion of individuals increased with size in all the species, which is a structure seldom registered for cactus species (reviewed in Godínez-Álvarez et al., 2003). In the arid region of Cuatro Cienegas, Coahuila, North Mexico, an increment of individuals with size was registered for the globose species Coryphantha werdermannii, at sites with very low disturbance (Portilla-Alonso and Martorell, 2011). A higher proportion of individuals in the larger than the smaller size-classes may be related to recruitment limitation (Nathan and Muller-Landau, 2000); recruitment limitation may occur at saturated undisturbed communities with a continuous canopy due to unsuitable microenvironmental conditions for seed germination and recruitment (see e.g. Clark et al., 1998). On the other hand, changes in size structure of cactus species due to disturbance has been registered in forest ecosystems. In the dry forest of the state of Veracruz, southeast Mexico, the cylindrical cactus Mammillaria eriacantha showed a decrease of individuals with size in most of the fragments evaluated (Valencia-Díaz et al., 2012); that size structure is related to continuous regeneration (Godínez-Álvarez et al., 2003). However, at the most disturbed fragment, the population of M. eriacantha showed an uneven size structure (Valencia-Díaz et al., 2012). In this study, the large arboreal cactus Pachycereus grandis showed a change in size structure with disturbance similar to that registered for the entire cacti community, whereas the shrub-like cactus Opuntia pumila showed a similar size structure in both forest types: the size structure related to constant regeneration. Therefore, a change in the size structure in the entire community and in the population of P. grandis due to disturbance may be the result of better recruitment conditions in the secondary forest.
In the conserved forest, the proportion of adults overall species was two times higher than the one recorded in the secondary forest, according to our hypothesis. These results are in line with others studies at population level; for example for Ferocactus histrix (del Castillo, 1987) and Coryphantha wer-dermannii (Portilla-Alonso and Martorell, 2011), a higher proportion of adults were found at the sites with less chronic disturbance. The proportion of adults under conditions of chronic disturbance may decrease due to high mortality of small size-classes and slow growth rates (Jiménez-Sierra et al., 2007; Alba-García, 2011; Martorell et al., 2012). Also, some studies have shown that even when recruitment is higher in grasslands, cacti growth faster in sites denuded of grasses (Mandujano et al., 2001) because belowground competition (Briones et al., 1996). This seems to be supported by the average height of adults of Pachycereus grandis in this study which was three times higher in the conserved forest (468 ± 43) compared to the secondary one (148 ± 81; Table 3). In the conserved forest, the higher proportion of larger adults was probably due to higher growth rates and lower recruitment. Few studies have explored the regeneration mechanisms of dry forest trees (reviewed in Quesada et al., 2009), whereas cactus species, with a variety of life-forms, are seldom included in the studies of woody species or totally excluded from the ones evaluating understory vegetation. Therefore, the mechanisms that explain the size structure of cactus species in the dry forest and its changes with disturbance needs to be further explored.
Implications for conservation and restoration of biodiversity in dry forests. Secondary forests that resulted from land abandonment after anthropogenic activities now cover more areas than conserved forest (Chazdon et al., 2007). These forests are important reservoir of biodiversity and because cactus populations may have different responses to disturbance, additional studies should be carry out to support management projects for their conservation (Martorell and Peters, 2009; Ureta and Martorell, 2009; González et al., 2013). In this study, 18 cactus species previously reported for Sierra de Huautla Biosphere Reserve (Martínez-Alvarado, 1985; CONANP, 2005) were not recorded in our plots probably because some of them are specialists of habitats not covered (e.g. rupicolous species). In these dry forests, some cactus species are specialists of uncommon microhabitats; conservation in those areas where many microhabitats are represented is crucial for cacti conservation (see e.g. Gibson et al., 2011).
Studies of succession have shown that vegetation that tolerates very dry conditions may successfully invade degraded landscapes (Ewel, 1980). We believe this may be the case for Opuntia puberula and O. velutina which have densities of one order of magnitude higher in the secondary forest compared to the conserved forest. However, the higher densities in the secondary forest may also be the result of asexual reproduction. For example, in the Southern Chihuahuan Desert, sexual reproduction of O. rastrera is lower than asexual reproduction, because the latter is enough to maintain these populations (Mandujano et al., 1996, 2001). Therefore, further studies should be conducted in the dry forest to evaluate if densities of Opuntia spp. are higher due to microclimatic conditions of secondary forests or to their asexual reproductive potential.
Conclusions
Chronic disturbance in this secondary forest during more than 30 years changed size structure of cactus species resulting in less adult individuals of reduced size. Under chronic disturbance, animals that depend on large arboreal cacti for food (i.e., bats, ants, bees, and butterflies) and perches or refuge (i.e., woodpecker) may move out affecting negatively many plant-animals interactions and the maintenance of diversity derived from them (Noy-Meir, 1974). For example, disturbed dry forests are usually dominated by trees with dry fruits dispersed by wind (Martínez-Garza et al., 2011) and therefore, these areas are not attractive to frugivores. One option to favour the coexistence of economic activities and biodiversity is to exclude fragments or medium size (e.g., ca. 0.25 to 1 ha) from chronic disturbance so younger cacti already recruiting may grow and reach older life-stage classes and full size. Adult cacti offering fleshy fruits during the harsh dry season will attract many frugivore animals and favour the dispersal of forest species they consume in older conserved areas (Wolf and Martínez del Rio, 2003). Sites far away from seed sources may be protected from disturbance and enriched by planting arboreal cacti and trees with fleshy fruits. The maintenance of fragment of undisturbed forest within large areas under chronic anthropogenic disturbance may increase the quality of the landscape matrix (Vandermeer and Carvajal 2001; Martínez-Garza and Howe, 2003), allowing plants and animals to survive as metapopulations in permanent agricultural landscapes.
Acknowledgments
We are grateful to Gerardo Pacheco and Rene Gadea for expert field assistance and to Carlos Martorell, Teresa Terrazas and one anonymous reviewer who provided constructive criticism of the manuscript. This work was supported by the Universidad Autónoma del Estado de Morelos, the Mexican Program for the Improvement of Professors (PROMEP), granted to CM-G (103.5/05/1901), the Cuerpo Académico de Ecología Evolutiva (PROMEP), and the National Council of Science and Technology of Mexico (CONACyT, Grant # 80027) granted to CM-G. LAA received a scholarship from PROMEP (103.5/05/1901).
References
Alba-García L. 2011. Reclutamiento de especies leñosas en la selva seca de Sierra de Huautla, Morelos, Mexico. Bs. Thesis. Universidad Autónoma del Estado de Morelos, Cuernavaca, Morelos, Mexico. 57 pp. <http://www.eco-index.org/search/results. cfm?ProjectID=1350> (accessed August 2, 2013). [ Links ]
Anderson E.F. 2001. The Cactus Family. Timber Press, Portland. [ Links ]
Arias S. and Terrazas T. 2009. Taxonomic revision of Pachycereus (Cactaceae). Systematic Botany 34:68-83. [ Links ]
Barthlott W. and Hunt D.R. 1993. Cactaceae. In: Kubitski K., Rohwer J.G., Bittrich V. Eds., The Families and Genera of Vascular Plants. Vol. II, pp. 161-197. Springer, Berlin. [ Links ]
Bowers J.E. 1997. Demographic patterns of Ferocactus cylindraceus in relation to substrate age and grazing history. Plant Ecology 133:37-48. [ Links ]
Bravo-Hollis H. 1978. Las Cactáceas de México. Vol. I. 2a ed. Universidad Nacional Autónoma de México, Mexico City. [ Links ]
Bravo-Hollis H. and Sánchez-Mejorada H. 1991a. Las Cactáceas de México. Vol II. 2a ed. Universidad Nacional Autónoma de México, Mexico City. [ Links ]
Bravo-Hollis H. and Sánchez-Mejorada H. 1991b. Las Cactáceas de México. Vol III. 2a ed. Universidad Nacional Autónoma de México, Mexico City. [ Links ]
Briones O., Montaña C. and Ezcurra E. 1996. Competition between three Chihuahuan desert species: Evidence from plant size-distance relations and root distribution. Journal of Vegetation Science 7:453-460 [ Links ]
Brown S. and Lugo A.E. 1990. Tropical secondary forests. Journal of Tropical Ecology 6:1-32. [ Links ]
Callen E.O. 1967. Analysis of the Tehuacán coprolites. In: Byers D.S. Ed. Environment and Subsistence, pp. 261-289, University of Texas Press, Austin. [ Links ]
Cervera J.C., Andrade J.L., Graham E.A., Durán R., Jackson P.C. and Simá J.L. 2007. Photosynthesis and optimal light microhabitats for a rare cactus, Mammillaria gaumeri, in two tropical ecosystems. Biotropica 39:620-627. [ Links ]
Chazdon R.L., Letcher S.G., van Breugel M., Martínez-Ramos M., Bongers F. and Finegan B. 2007. Rates of change in tree communities of secondary Neotropical forests following major disturbances. Philosophical Transactions of the Royal Society B Biological Sciences 362:273-289. [ Links ]
Clark J.S., Macklin E. and Wood L. 1998. Stages and spatial scales of recruitment limitation in Southern Appalachian forests. Ecological Monographs 68:213-235. [ Links ]
CONANP. Comisión Nacional de Áreas Naturales Protegidas. 2005. Programa de Conservación y Manejo Reserva de la Biosfera Sierra de Huautla México. Comisión Nacional de Áreas Naturales Protegidas, Mexico, D.F. [ Links ]
Contreras C. and Valverde T. 2002. Evaluation of the conservation status of a rare cactus (Mammillaria crucigera) through the analysis of its population dynamics. Journal of Arid Environments 51:89-102 [ Links ]
De la O-Toris J., Maldonado B. and Martínez-Garza C. 2012. Efecto de la perturbación en la comunidad de herbáceas nativas y ruderales de una selva estacional mexicana. Botanical Sciences 90:469-480. [ Links ]
De León-Ibarra M.A. 2005. Fenología de especies de plantas con frutos carnosos y disponibilidad espacial y temporal de este recurso en la Reserva de la Biosfera Sierra de Huautla: Implicaciones para los vertebrados. Bc. thesis, Universidad Autónoma del Estado de Morelos, Cuernavaca; Mexico. 76 pp. [ Links ]
del Castillo R.F. 1987. Efectos del disturbio y la orientación de la ladera en Ferocactus histrix. Cactaceas y Suculentas Mexicanas 32:8-16. [ Links ]
Ewel J. 1980. Tropical succession: manifold routes to maturity. Biotropica 12:2-7. [ Links ]
Ferrer-Cervantes M.E., Méndez-González M.E., Quintana-Ascencio P.F., Dorantes A., Dzib G. and Durán R. 2012. Population dynamics of the cactus Mammillaria gaumeri: an integral projection model approach. Population Ecology 54:321-334. [ Links ]
Finegan B. 1996. Pattern and process in Neotropical secondary rain forests: The first 100 years of succession. Trends in Ecology and Evolution 11:119-124. [ Links ]
Flores M.A., Manzanero M.G.I., Golubov J., Montaña C. and Mandujano M. C. 2010. Demography of an endangered endemic rupicolous cactus. Plant Ecology 210:53-66. [ Links ]
Floyd M.L., Fleischner T.L., Hanna D. and Whitefield P. 2003. Effects of historic livestock grazing on vegetation at Chaco Culture National Historic Park, New Mexico. Conservation Biology 17:1703-1711. [ Links ]
Gibson L., Lee T.M., Koh L.P., Brook B.W., Gardner T.A., Barlow J., Peres C.A., Bradshaw C.J.A., Laurance W.F., Lovejoy T.E. and Sodhi N.S. 2011. Primary forests are irreplaceable for sustaining tropical biodiversity. Nature 478:378-381. [ Links ]
Godínez-Álvarez H., Valverde T. and Ortega-Baes P. 2003. Demographic trends in the Cactaceae. The Botanical Review 69:173-203. [ Links ]
González-Iturbe J.A., Olmsted I. and Tun-Dzul F. 2002. Tropical dry forest recovery after long term Henequen (sisal, Agave fourcroydes Lem.) plantation in northern Yucatan, Mexico. Forest Ecology and Management 167:67-82. [ Links ]
González E.J., Rees M. and Martorell C. 2013. Identifying the demographic processes relevant for species conservation in human-impacted areas: does the model matter? Oecologia 171:347-356. [ Links ]
Guzmán U., Arias S. and Dávila P. 2003. Catálogo de Cactáceas Mexicanas. Universidad Nacional Autónoma de México, Comisión Nacional para el Conocimiento y Uso de la Biodiversidad. Mexico, D.F. [ Links ]
Hansen M.C., Stehman S.V. and Potapov PV. 2010. Quantification of global gross forest cover loss. Proceedings of the National Academy of Sciences of the United States of America 107:8650-8655. [ Links ]
Hayes G.F. and Holl K.D. 2003. Cattle grazing impacts on annual forbs and vegetation composition of mesic grasslands in California. Conservation Biology 17:1694-1702. [ Links ]
Hernández H.M. and Godínez A.H. 1994. Contribución al conocimiento de las cactáceas mexicanas amenazadas. Acta Botanica Mexicana 26:33-52. [ Links ]
Hunt D. 1999. CITES Cactaceae Checklist. Royal Botanic Gardens, Kew London. [ Links ]
Instituto de Biología. 2013. Opuntia atropes Rose - IBUNAM:-MEXU:OPUSsn243. UNIBIO: Colecciones Biológicas. 2010-07-04. Universidad Nacional Autónoma de México. <http://unibio.unam.mx/collections/specimens/urn/IBUNAM:MEXU:OPUSsn243> (accessed August 2, 2013). [ Links ]
Jiménez-Sierra C., Mandujano M.C. and Eguiarte L.E. 2007. Are populations of the candy barrel cactus (Echinocactus platyacanthus) in the desert of Tehuacán, Mexico at risk? Population projection matrix and life table response analysis. Biological Conservation 135:278-292. [ Links ]
Kennard D.K. 2002. Secondary forest succession in a tropical dry forest: patterns of development across a 50-year chronosequence in lowland Bolivia. Journal of Tropical Ecology 18:53-66. [ Links ]
Krebs C.J. 1985. Ecology: The Experimental Analysis of Distribution and Abundance. 3rd ed. Harper and Row, New York. [ Links ]
Linares-Palomino R. and Ponce A.S.I. 2005. Tree community patterns in seasonally dry tropical forests in the Cerros de Amotape Cordillera, Tumbes, Peru. Forest Ecology and Management 209:261-272. [ Links ]
López-Acosta S., Nuño-Romero R. and Torres-Morán M.I. 2008. Prueba de significancia de tres métodos para calcular el área de cladodios en plantas de nopal para verdura. In: Carvajal S. and Pimienta-Barrios E. Eds. Avances en la Investigación en el CUCBA. Memorias de la XIX Semana de la Investigación Científica, pp. 49-53. Centro Universitario de Ciencias Biológicas y Agropecuarias, Guadalajara. [ Links ]
Luna-Morales C.C. and Aguirre J.R. 2001. Traditional classification, utilization and ecological distribution of the Pachycereeae in Mixteca Baja, Mexico. Interciencia 26:18-24. [ Links ]
Magurran A.E. 2004. Measuring Biological Diversity. Blackwell Publishing. Oxford. [ Links ]
Maldonado B., Caballero J., Delgado-Salinas A. and Lira R. 2013. Relationship between use value and ecological importance of floristic resources of seasonally dry tropical forest in the Balsas River Basin, Mexico. Economic Botany 67:17-29. [ Links ]
Mandujano M.C., Montaña C. and Eguiarte L.E. 1996. Reproductive ecology and inbreeding depression in Opuntia rastrera (Cactaceae) in the Chihuahuan Desert: Why are sexually derived recruitments so rare? American Journal of Botany 83:63-70. [ Links ]
Mandujano M.C., Montaña C., Franco M., Golubov J. and Flores-Martínez A. 2001. Integration of demographic annual variability in a clonal desert cactus. Ecology 82:344-359. [ Links ]
Martínez-Alvarado D. 1985. Las cactáceas del estado de Morelos. Bch. Thesis, Universidad Autónoma del Estado de Morelos, Cuernavaca, Mexico. 176 pp. [ Links ]
Martínez-Garza C. and Howe H.F. 2003. Restoring tropical diversity: beating the time tax on species loss. Journal of Applied Ecology 40:423-429. [ Links ]
Martínez-Garza C., Osorio-Beristain M., Valenzuela-Galván D. and Nicolás-Medina A. 2011. Intra and inter-annual variation in seed rain in a secondary dry tropical forest excluded from chronic disturbance. Forest Ecology and Management 262:2207-2218. [ Links ]
Martorell C. and Peters E.M. 2005. The measurement of chronic disturbance and its effects on the threatened cactus Mammillaria pectinifera. Biological Conservation 124:199-207. [ Links ]
Martorell C. and Peters E.M. 2009. Disturbance-response analysis: A method for rapid assessment of the threat to species in disturbed areas. Conservation Biology 23:377-387. [ Links ]
Martorell C., Garcillán P.P. and Casillas F. 2012. Ruderality in extreme-desert cacti? Population effects of chronic anthropogenic disturbance on Echinocereus lindsayi. Population Ecology 54:335-346. [ Links ]
Méndez M., Durán R., Olmsted I. and Oyama K. 2004. Population dynamics of Pterocereus gaumeri, a rare and endemic columnar cactus of Mexico. Biotropica 36:492-504. [ Links ]
Monroy-Ortiz C. 1997. La leña como recurso energético, implicaciones ecológicas y etnobotánicas. M. Sc. Thesis. Facultad de Ciencias, Universidad Nacional Autónoma de México, Mexico, D.F. 112 pp. [ Links ]
Nai-Bregaglio M., Pucheta E. and Cabido M. 2002. El efecto del pastoreo sobre la diversidad florística y estructural en pastizales de montaña del centro de Argentina. Revista Chilena de Historia Natural 75:613-623. [ Links ]
Naranjo M.E., Rengifo C. and Soriano P.J. 2003. Effect of ingestion by bats and birds on seed germination of Stenocereus griseus and Subpilocereus repandus (Cactaceae). Journal Tropical Ecology 19:19-25. [ Links ]
Nathan R. and Muller-Landau H.C. 2000. Spatial patterns of seed dispersal, their determinants and consequences for recruitment. Trends in Ecology and Evolution 15:278-285. [ Links ]
Noy-Meir I. 1974. Desert ecosystems: Higher trophic levels. Annual Review of Ecology and Systematics 5:195-214. [ Links ]
Ortega-Baes P. and Godínez-Álvarez H. 2006. Global diversity and conservation priorities in the Cactaceae. Biodiversity and Conservation 15:817-827. [ Links ]
Portilla-Alonso R.M. and Martorell C. 2011. Demographic consequences of chronic anthropogenic disturbance on three populations of the endangered globose cactus Coryphantha werdermannii. Journal of Arid Environments 75:509-515. [ Links ]
Quesada M., Sanchez-Azofeifa G.A., Alvarez-Añorve M., Stoner K.E., Avila-Cabadilla L., Calvo-Alvarado J., Castillo A., Espírito-Santo M.M., Fagundes M., Fernandes G.W., Gamon J., Lopezaraiza-Mikel M., Lawrence D., Cerdeira M.L.P., Powers J.S., Neves F.D., Rosas-Guerrero V., Sayago R. and Sanchez-Montoya G. 2009. Succession and management of tropical dry forests in the Americas: Review and new perspectives. Forest Ecology and Management 258:1014-1024. [ Links ]
R Development Core Team. 2011. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna. [ Links ]
Reyes-Agüero J.A., Aguirre J.R. and Valiente-Banuet A. 2006. Reproductive biology of Opuntia: A review. Journal of Arid Environments 64:549-585. [ Links ]
Roth L.C. 1999. Anthropogenic change in subtropical dry forest during a century of settlement in Jaiqui Picado, Santiago Province, Dominican Republic. Journal of Biogeography 26:739-759. [ Links ]
Ruiz J., Fandiño M.C., Chazdon R.L. 2005. Vegetation structure, composition, and species richness across a 56-year chronose-quence of dry tropical forest on Providencia island, Colombia. Biotropica 37:520-530. [ Links ]
Rzedowski J. 1978. Vegetación de México. Limusa, Mexico, D.F. [ Links ]
Sánchez-Velásquez L.R., Hernández-Vargas G., Carranza-M. M.A., Pineda-López M.R., Cuevas-G. R. and Aragón-C. F. 2002. Estructura arbórea del bosque tropical caducifolio usado para la ganadería extensiva en el norte de la Sierra de Manantlán, México. Antagonismos de usos. Polibotánica 13:25-46. [ Links ]
Stern M., Quesada M. and Stoner K.E. 2002. Changes in composition and structure of a tropical dry forest following intermittent cattle grazing. Revista de Biología Tropical 50:1021-1034. [ Links ]
Trejo-Vázquez R.I. 1998. Distribución y diversidad de selvas bajas de México: relaciones con el clima y el suelo. Ph. D. Thesis. Facultad de Ciencias, Universidad Nacional Autónoma de México, México, D.F. 210 pp. [ Links ]
Ureta C. and Martorell C. 2009. Identifying the impacts of chronic anthropogenic disturbance on two threatened cacti to provide guidelines for population-dynamics restoration. Biological Conservation 142:1992-2001 [ Links ]
Valencia-Díaz S., Flores-Palacios A. and Castillo-Campos G. 2012. Tamaño poblacional y características del hábitat de Mammillaria eriacantha, una cactácea endémica del centro de Veracruz, México. Botanical Sciences 90:195-202. [ Links ]
Vandermeer J. and Carvajal R. 2001. Metapopulation dynamics and the quality of the matrix. The American Naturalist 158:211-220. [ Links ]
VanderWerf E.A. 2004. Demography of Hawai'i 'Elepaio: Variation with habitat disturbance and population density. Ecology 85:770-783. [ Links ]
Wilson J.B. 1991. Methods for fitting dominance/diversity curves. Journal of Vegetation Science 2:35-46. [ Links ]
Wolf B.O. and Martínez del Rio C. 2003. How important are columnar cacti as sources of water and nutrients for desert consumers? A review. Isotopes in Environmental Health Studies 39:53-67. [ Links ]
Zar J.H. 1996. Biostatistical Analysis. 3rd ed. Prentice-Hall, Inc., Englewood Cliffs. [ Links ]














