Introduction
In the northeastern Mexico, small pelagic fish constitute one of fishery resources of most importance for industrial purse seine fleet, the main ports of fish landing are Guaymas and Mazatlán (SAGARPA, 2012).
In the littoral of Sinaloa, Nayarit and Jalisco, fishery of small pelagic fish of greater importance comprises three fish species of the Opisthonema genus (O. libertate, O. medirastre and O. bulleri) which are named thread herring (‘sardina crinuda’) (Jacob-Cervantes, 2010).
O. libertate is the most abundant species in fish catches, therefore most of studies on fisheries biology focused on this species (Gallardo-Cabello et al., 1993; Carmona & Alexandres, 1994; Jacob-Cervantes y Aguirre-Villaseñor, 2014; Ruiz-Domínguez & QuiñonezVelázquez, 2018), while fisheries biology for O. medirastre y O. bulleri is unknown.
Growth rate of organisms can be estimated by means of indirect and direct methods. In previous studies on O. medirastre, growth was tackled by means of an indirect method based on a length-frequency analysis (Soto-Rojas & Rodríguez-Murillo, 1999; Vega-Corrales, 2010), but it is unknown from age and growth studies of this species by direct methods. The most used bone structure for age determination in fish are otoliths (Campana, 2001).
On the other hand, the growth of O. medirastre has been described by selecting a priori Von Bertalanffy model, the most used in fisheries biology (Katsanevakis, 2006; Katsanevakis & Maravelias, 2008), since its parameters are used in models to estimate biomass (Sparre & Venema, 1997). A relatively new paradigm in biological sciences is the multi-model selection (Burnham & Anderson, 2002; Katsanevakis, 2006; Beninger et al., 2012), consisting in selecting from a set of candidate models, the model which best fits the data, by using the information theory and the parsimony concept, both integrated in Akaike Information Criterion (AIC). The best model is the one with lower AIC. Other models used to describe growth in fish are Gompertz (1825), Logístico de Ricker (1975), Schnute (1981) and Schnute & Richards (1990). Nevertheless, the generalized Schnute growth model is versatile and includes historic models as special cases (Schnute, 1981). At least eight different growth curves can be generated from the four special cases of this model. Therefore, the use of generalized Schnute growth model to describe growth increases the probability that the real model is included.
Currently, fishery of small pelagic fish in the southern Gulf of California is certified as sustainable by the Marine Stewardship Council, but performing evaluations of each species of the thread herring complex (‘sardina crinuda’) is included among the observations, comprising modeling growth for each species.
This study dealt with growth modeling of O. medirastre in the southern Gulf of California by means of reading otoliths and of a multi-model select approach, from cases of Schnute’s growth model as candidate models.
Material and Methods
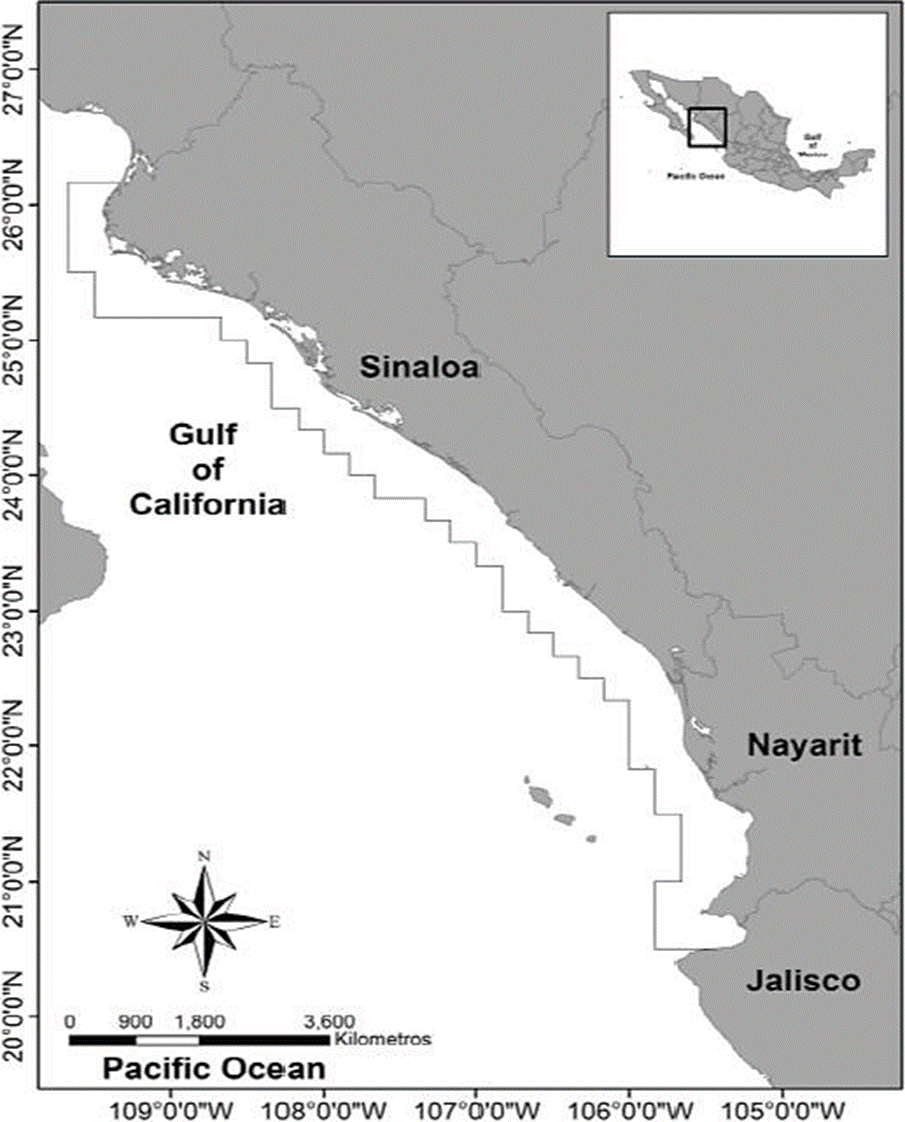
The fishing area of thread herring fleet including Punta Ahome, Sinaloa to Bahía Banderas, Jalisco, is delimited into a polygon included between Latitude 2027° N, la Longitude 105-110° W and at a maximum of 20 nautical miles offshore (Figure 1).
The analyzed information came from biological samples of commercial landings thread herring catches in the port of Mazatlán, Sinaloa, realized by the personnel of the “Small pelagic fish” (Pelágicos Menores) Program from CRIAP Mazatlán, INAPESCA, SADER.
Samples were randomly taken from 12 kg of the total fish catch of a vessel and ten individuals of each 5 mm-length interval were randomly selected, to which standard length (mm), weight (gr), sex were measured, sagittal otoliths were extracted to determine age and the first branchial arch for taxonomic identification according to criteria of Berry y Barrett (1963). Data of 2005 (n=163), 2008 (n=59) and 2015 (n=206) were selected because when gathering them, the annual cycle of continuous information was completed and data represent years with different environmental conditions (2005-Neutral, 2008-La Niña and 2015-El Niño).
Age was determined by counting opaque and hyaline bands of each otolith (Beckman & Howlett, 2013). For this, otoliths were cleaned and immersed in purified water for five minutes for its digitalization by means of a Carl Zeiss stereoscopic microscope (Stemi 508) equipped with an Axiocam ERc 5s camera. The precision of age determinations from otoliths was evaluated with an average percentage error (APE) according to Beamish & Fournier (1981) and Coefficients of Variation (CV).
Where: N is sample size, R number of readings per structure, Xij is the ith reading of the jth structure, Xj is the average number of growth marks for the jth structure.
Schnute’s growth model and its five cases was applied to describe the growth.
Where: Y(t) is standard length at age t, Y1 and Y2 are minimum and maximum standard length, α is a growth parameter with units of years, b is related to the inflexion point of a “S” shape growth curve and τ1, τ2 are minimum and maximum ages of the data set.
Parameters of each model were estimated by maximizing the of log-likelihood function (Hilborn & Mangel, 1997) with the Newton algorithm (Neter et al., 1996).
Where: LL is the log-likelihood function, n is sample size and σ is standard deviation, which was estimated with the following functions, considering additive error.
Additive error structure:
Where: Yti is observed standard length, Ŷti is estimated standard length.
In order to evaluate candidate models and select the best one, based on parsimony principle, a multi-model selection (MMS) approach was chosen, where various models compete and are simultaneously faced with data (Johnson & Omland, 2004; Hobbs & Hilborn, 2006).
A statistical method based on information theory was applied, commonly used in models select approach. Akaike information criterion (corrected Akaike Information Criterion, AICc) incorporates information on fitting, complexity and size of the sample (Johnson & Omland, 2004).
AIC is Akaike Information Criterion from model j (Akaike, 1981; Burnham & Anderson, 2002).
Where: n is the total number of observations, K is the total number of estimated parameters and LL is the maximum log-likelihood.
For each model, plausibility was estimated with Akaike weight (wi).
Results and Discussion
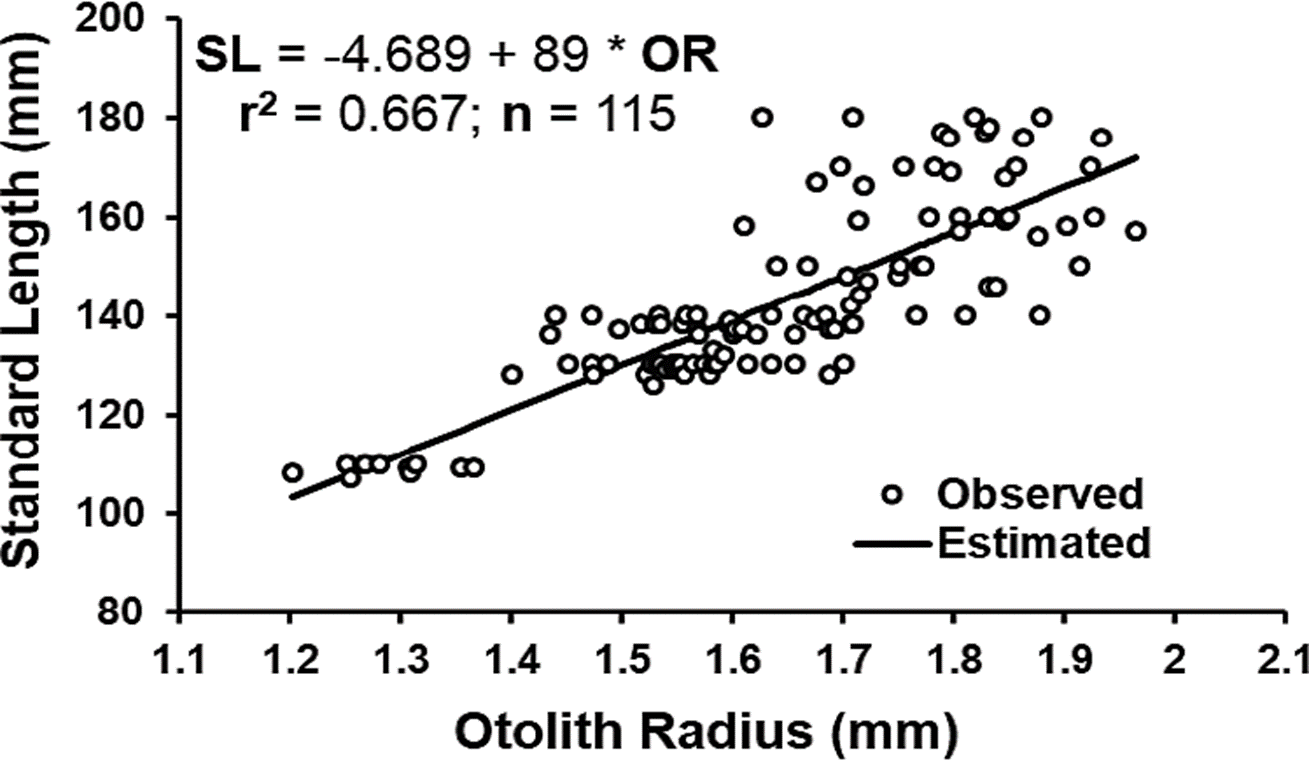
In Figure 2, the relationship between otolith radius and O. medirastre standard length was shown to be linear, with a significant coefficient of correlation (r = 0.817, t = 15.05 p < 0.001) and a coefficient of determination of 0.667. This demonstrated a proportional growth of the otolith with fish size, although the coefficient of determination indicated that 33.3 % of the variation in size was explained by another factor, independent from otolith size. In studies on fish growth, coefficients of determination for the relationship between otolith size and fish size have been observed to vary from 0.4 to 0.9 (Carmona & Alexandres, 1994; Quiñonez-Velázquez et al., 2002; Puentes-Ganada et al., 2004; Morales & González, 2010; Pajuelo & Lorenzo, 2011; Souza-Conceinao & Schwingel, 2011; Cerna et al., 2014). The proportionality of otolith size with fish size is very important when a retro-calculation of previous sizes has to be done, and the more precision you have, the better it is, especially because in some cases, a reabsorption of scale rings (Lee’s phenomenon) can be presented, however, in this study, the retro-calculation was not performed, since organisms of earlier ages were well represented. Otoliths were only examined to observe hyaline and opaque growth bands, which were present independently from the proportionality of otolith size with fish size. Otoliths had previously been used for age determination in Opisthonema species (Carmona & Alexandres, 1994; Ruiz-Domínguez & Quiñonez-Velázquez, 2018).
Monthly proportion of otoliths with opaque and hyaline edges showed an annual cycle; most of the proportion of hyaline edges was registered in September (0.75), while most of the proportion of opaque edges was registered in March (0.86) (Figure 3). Most of the proportion of opaque edges coincided with the period of maximum concentration of chlorophyll-a present from January to June in the coast located between Sinaloa and Cabo Corrientes, Jalisco (Cepeda-Morales et al., 2017), where the thread herring fleet operates. Phytoplankton or zooplankton availability during these months of high chlorophyll-a concentration represents a high food availability for O. medirastre and therefore, a fast growth associated with the formation of opaque zones of their otoliths. The reproductive maximum of this species occurs in August-Septembre (Cotero-Altamirano et al., 2014) and coincides with the period of low chlorophyll-a concentration (Cepeda-Morales et al., 2017). These two events represent a low food availability and an energy allocation towards gonads maturation, respectively, coinciding with the low growth phase and the formation of the hyaline zone of otoliths. The persistence of these two periods can show inter-annual variations, however Figure 3 represented the average of three years with different environmental conditions (El Niño, La Niña and Neutral), therefore it can be stated that in average, an opaque band and a hyaline band were formed annually.
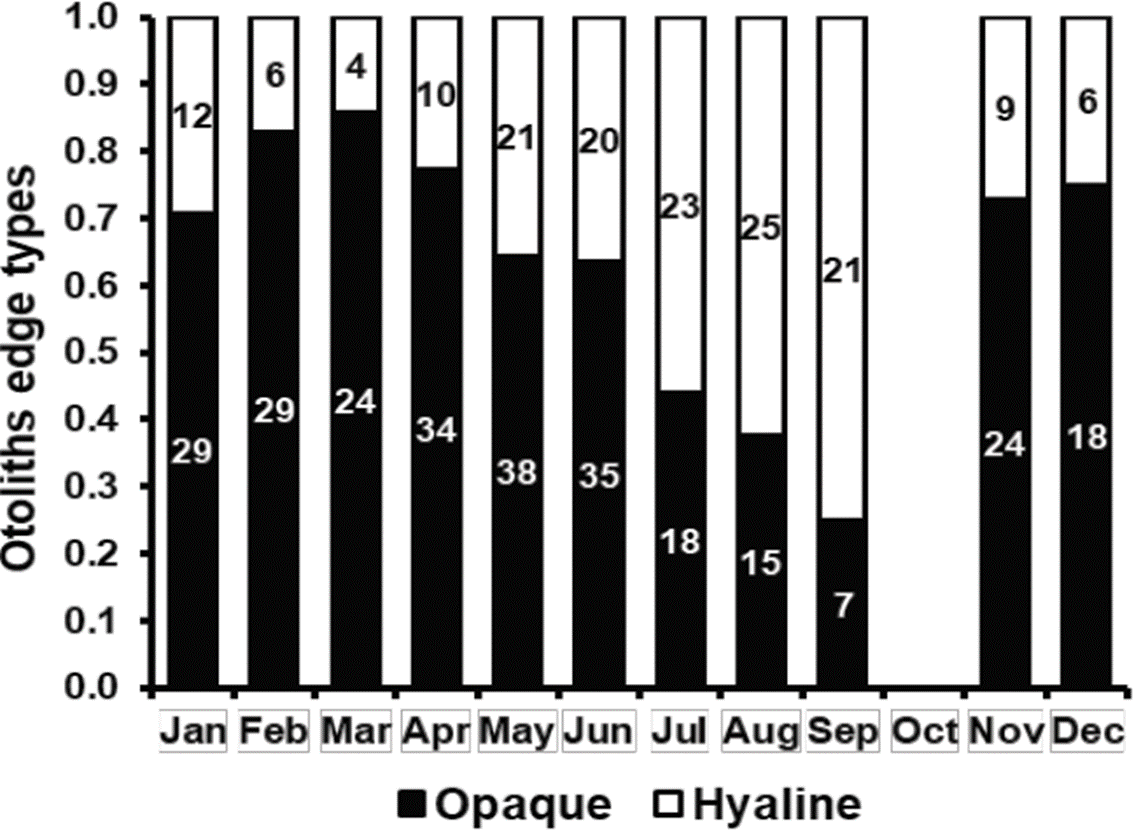
Figure 3 Monthly proportion of otolith edge types of O. medirastre in the southern Gulf of California.
In literature, no previous works on ages of O. medirastre have been found up to now to contrast the current results, but in O. libertate, an opaque band and a hyaline band have been observed to be formed annually (Ruiz-Domínguez & Quiñonez-Velázquez, 2018).
The count of growth bands allowed to identify five age groups (0-4 years); an age-length key for O. medirastre was shown in Table 1. The APE and the coefficient of variation (CV) of readings were 3.1 % and 2.4 % respectively. Although there were no reference points for the precision of age readings in fish, values of both indexes (APE = 3.1 %, CV = 2.4 %) used here were lesser than the median of studies on age of 117 fish (APE = 5.5 %, CV = 7.6 %), which were recommended as maximum limit for short lifespan organisms (Campana, 2001). A higher precision of age readings supports a greater determination of age (Cerna et al., 2014). Thus, the low values of APE and CV in this study indicated an acceptable determination of age in O. medirastre.
Table 1 Age-length key of middling thread herring O. medirastre.
| Length/Age | 0 | 1+ | 2+ | 3+ | 4+ | Total |
|---|---|---|---|---|---|---|
| 130-139 | 0.828 | 0.172 | 0 | 0 | 0 | 174 |
| 140-149 | 0.114 | 0.886 | 0 | 0 | 0 | 79 |
| 150-159 | 0.000 | 0.781 | 0.219 | 0 | 0 | 73 |
| 160-169 | 0 | 0 | 0.947 | 0.053 | 0 | 57 |
| 170-179 | 0 | 0 | 0.200 | 0.800 | 0 | 35 |
| 180-189 | 0 | 0 | 0 | 0.111 | 0.889 | 9 |
| 190-199 | 1 | 1 |
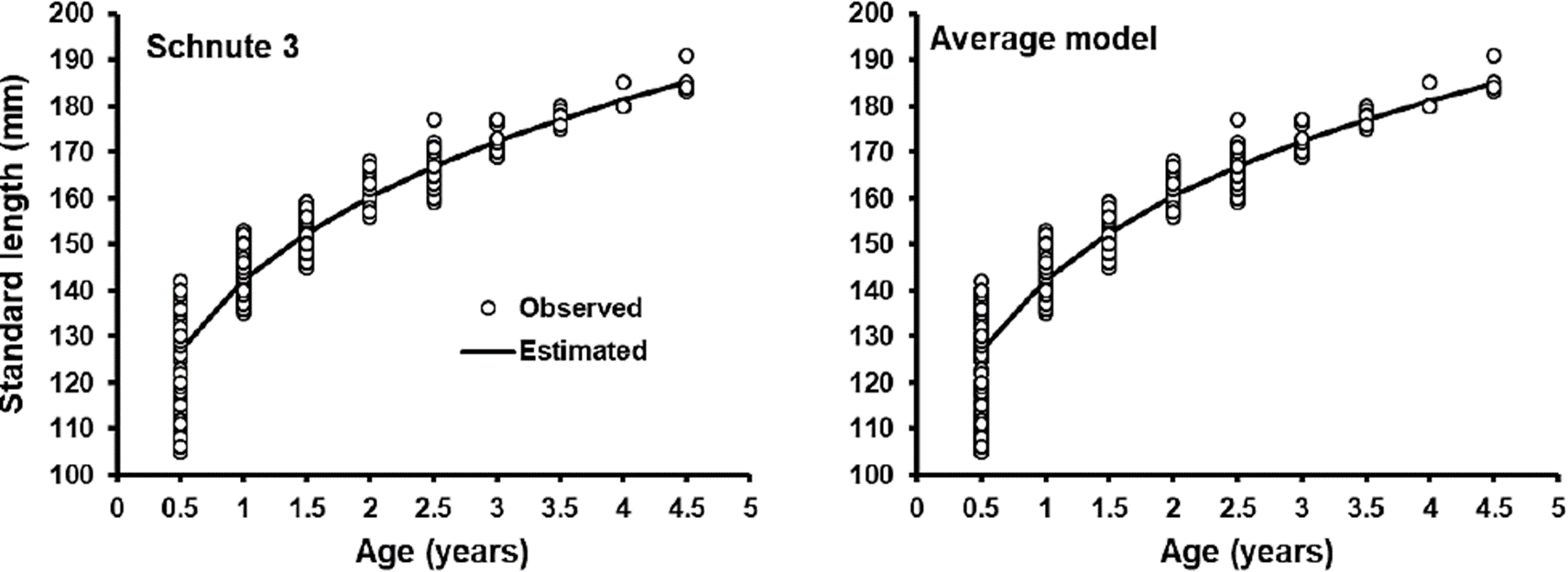
This is the first study on age and growth in O. medirastre using otoliths, previous works used length-frequency analysis to estimate growth (Soto-Rojas & RodríguezMurillo, 1999; Vega-Corrales, 2010). When comparing different cases of Schnute’s growth model in growth description of O. medirastre, Schnute´s case 3 was the best, as it obtained the lower AICc (2845) with WAICc = 63.7 %, however, in accordance with Katsanevakis (2006), this case is not a clear winner (WAICc > 90 %). Cases 1, 5 and 2 of Schnute’s growth model (23, 9.6 and 3.8 % respectively) had support in data as well to describe O. medirastre’s growth (Burnham & Anderson, 2002) (Table 2). When the best model is not a clear winner, a better procedure to infer size to age is by means of an average model resulting from the estimation of size to age of each candidate model, weighted by its respective WAICc (Burnham & Anderson, 2002). However, the difference of the estimate value of size between the best model and the average model was less than 1 mm (Figure 4). On the contrary, with case 5 (Von Bertalanffy), the difference was up to 3 mm in the most advanced ages. In this analysis, case 5, which is the equivalent of Von Bertalanffy growth model, was ranked in third place among candidate models and with little support in data, therefore the a priori use of this traditional model to describe growth in O. medirastre could lose information. Similar results have been reported by Katsanevakis & Maravelias (2008), who found that Von Bertalanffy model was not the best model in 87 of 133 fish species. Ruiz-Domínguez & Quiñónez-Velázquez (2018) modeled growth of a sympatric species, O. libertate and by means of MMS, they selected Von Bertalanffy model as the best one, which is contrary to results with O. medirastre in the present work. They included fry and juveniles specimens, which represent fast growth stage and Von Bertalanffy model fits very well in these phases, but in ages recruited for fishery (from age 1 and higher) a notable discrepancy can be observed between the model and observed data. In this study, although thread herrings aged less than one year were included, the fry were not included and the best model (case 3) crossed the central part of observed data.
Table 2 Growth model parameters, AICc, and WAICc of O. medirastre.
| Case | y1 | y2 | a | b | AICc | ΔAICc | WAICc |
|---|---|---|---|---|---|---|---|
| 1 | 127 | 185 | -0.001 | 5.49 | 2847 | 2.04 | 23 |
| 2 | 127 | 182 | 0.626 | 0 | 2851 | 5.64 | 3.8 |
| 3 | 127 | 185 | 0 | 5.48 | 2845* | 0 | 63.7 |
| 4 | 132 | 204 | 0 | 0 | 3028 | 183.3 | 0 |
| 5 | 127 | 182 | 0.508 | 1 | 2849 | 3.79 | 9.6 |
* best model τ1 = 0.5, τ2 = 4.5
In the graph of the growth curve of the best Schnute’s model case 3 (Figure 4), thread herring O. medirastre was observed to present an accelerated growth in the first year of life and reached 76.8 % of the value of maximum size (y2). In addition, a higher variation in size of 0.5 age organisms and a lower variation in size were observed while organisms increase age. This decrease in variance of the size with age is a characteristic of a compensatory-type growth (Dobson & Holmes, 1984; Aragón-Noriega et al., 2017), suggesting that organisms of O. medirastre went through a critical period of intraspecific competence; for instance a low food, space or environmental condition availability, affecting first life stages (Lasker et al., 1970; Lasker & Zweifel, 1978) and when conditions improved, the most stressed organisms, which remained in small sizes at their age, grew faster and compensate these differences in size with the other organisms of the cohort. Compensatory growth has been reported for marine fish (Ali et al., 2003) as well as in aquacultured fish and crustacean as rainbow trout and white shrimp (Dobson & Holmes, 1984; Aragón-Noriega et al., 2017)
Conclusion
The use of MMS, the integration of length data at the age of three different years representing different environmental conditions and compensatory growth are a guarantee that Schnute’s model case 3, selected as the best model, is useful to represent the average growth of O. medirastre for the southern Gulf of California.











 texto en
texto en