Introduction
Having fully healthy ecosystems is unfeasible. Zoonotic diseases are becoming increasingly common and more widespread in time and space due to the destruction and fragmentation of natural ecosystems caused by anthropogenic activities and climate change (Daszak et al. 2001; Stokholm et al. 2021). Understanding and identifying diseases that induce high wildlife mortality and morbidity is key for ecosystem management and conservation (Loots et al. 2017).
Ecological studies of diseases affecting carnivores emerged in the 1990s (McCallum and Dobson 1995). Populations infected with the canine distemper virus (CDV) were studied, finding that CDV was the primary cause of the local extinction of the lycaon (Lycaon pictus) in Serengeti, Tanzania (Creel and Creel 1998) and Masai Mara, Kenya (Alexander and Appel 1994), and of the black-footed ferret (Mustela nigripes) in Wyoming, United States (Williams et al. 1988). In 1994, Young concluded that the highest mortality in the tropics was due to disease in carnivores and food scarcity in herbivores; today, we know that intra- and inter-specific relationships are far more complex.
CDV belongs to the genus Morbillivirus, family Paramyxoviridae (Murphy et al. 2012). It is highly important for wildlife, particularly carnivores. It enters the body through the mucosae and spreads in the environment through urine and feces. Its rapid transmission among carnivores is related to their behavior, i. e., communicating through smell, coupled with the ability of the virus to occur in all fluids of the infected organism, including aerosols from respiratory secretions (Barrett 1999; Ávila et al. 2015; Loots et al. 2017); besides, CDV can also spread from the consumption of infected organisms (Tahara et al. 2008).
Similar to any virus, CDV needs a host to replicate itself. As it is associated with cellular tropism, CDV-infected organisms suffer a range of systemic alterations affecting the respiratory system (breathing disorders, as the virus enters directly by air and replicates in the respiratory tract), digestive system (diarrhea), urinary system (kidney disorders), lymphatic system (lymphopenia and persistent immunosuppression), skeletal system (multiple sclerosis), skin disorders, and the central nervous system (leukoencephaltis; Lempp et al. 2014). Infected individuals usually die from the disease (Rendon-Marin et al. 2019). CDV does not only affect canids, as the name suggests, but has been recognized as a multi-host pathogen that has caused massive deaths, mainly in carnivorous species (Loots et al. 2017; Rendon-Marin et al. 2019). There are records of the adverse effects of this virus in felids such as lion (Panthera leo; Weckworth et al. 2020), tiger (Panthera tigris; Myers et al. 1997; Seimon et al. 2013), Iberian lynx (Lynx pardinus; Meli et al. 2010); hyenids: spotted hyena (Crocuta crocuta); ursids: giant panda (Ailuropoda melanoleuca; Feng et al. 2016); and phocids: crabeater seal (Lobodon carcinophagus; Bengston et al. 1991), Caspian seal (Pusa caspica; Forsyth et al. 1998) and nerpa (Pusa sibirica; Grachev et al. 1989). Martínez-Gutiérrez and Ruiz-Sáenz (2016) produced a literature compendium on the records of CDV-infected species, either naturally or experimentally, in all continents except Australasia. They found 217 articles that mention 12 families of the order Carnivora (Ailuridae, Canidae excluding the domestic dog, Felidae, Hyaenidae, Mephitidae, Mustelidae, Odobenidae, Otariidae, Phocidae, Procyonidae, Ursidae, and Viverridae), four of Rodentia (Caviidae, Cricetidae, Muridae, including Mus musculus; and Sciuridae), two of primates (Cebidae and Cercopithecidae), three of Artiodactyla (Cervidae, Suidae and Tayassuidae), and one of Proboscidea (Elephantidae).
The main factors impacting disease dynamics in carnivores are population size, density, and distribution (Dye et al. 1995). A suitable case study may be the coyote (Canis latrans) because it is a common species with a large amount of data and records, widely distributed in America, from Alaska to Central America. Some studies contend that its distribution may also include Colombia in South America (Monroy-Vilchis et al. 2020). The wide distribution of this species is due to its plasticity to adapt to different habitats and its varied diet (Gompper and Gittleman 1991). The broad geographic range of the coyote makes it hugely important as a model of disease dispersal in wild species. This species has shown a high prevalence of canine parvovirus, canine distemper, canine infectious hepatitis, and many other infectious pathogens (Gese et al. 1991; Gese et al. 1997; Bischof and Rogers 2005). The presence of these diseases was significant due to the transmission of many of these diseases to the populations of wolves in Yellowstone National Park, making their recovery slower than estimated (Gese et al. 1997).
The ability to monitor the spread of CDV quickly and sparing the need to capture individuals to collect blood samples - which involves the use of traps, chemicals, or containment instruments - may improve disease control and reduce time and costs. As an alternative to conventional sampling and detection methods, the use of biosensors based on advanced materials allows for indirect, non-invasive, and effective identification of the presence of the virus (Vidic et al. 2017). These biosensors are made from nanoparticles (NPs); in most cases, the operation principle is related to the intrinsic features of this nanomaterial, e.g., its optical properties (Maddali et al. 2020).
Biosensors can recognize, either qualitatively or quantitatively, a specific pathogen for which they are designed, thus avoiding interactions with other microorganisms or molecules. It functions through a bioreceptor with chemical qualities, i.e., a cell, an enzyme, a tissue, DNA, or RNA (Vidic et al. 2017). Biosensors are extensively used in biomedicine to detect and diagnose viral diseases, so their application in the detection of CDV in wild mammals seems feasible (Saylan et al. 2019). The objective of this paper is to establish an updated overview of the use of biosensors for monitoring and controlling viruses in wildlife, with special emphasis on the canine distemper virus.
Materials and methods
A comprehensive literature review was conducted in search of references addressing the use of biosensors for diagnosing viral diseases, including the canine distemper virus (CDV). A search and analysis of nanovaccines designed and used to control canine distemper was carried out. We used the search engines PubMed, Google Scholar, and Scopus, with the keywords "Virus", "Canine Distemper Virus", "nanoparticles", "NPs", "AuNPs", "QD", "Biosensor", and "Sensor".
Results
We found a total of 50 studies published in scientific journals in domestic animals. None of them refers to the use of biosensors for viruses in wild mammals because most are in the experimental and testing phase (Supplementary Material 1). Publications that had not been peer-reviewed, such as blogs, personal web pages, and social media, were discarded. The studies analyzed were sorted into three groups: use of biosensors to detect viruses, use of biosensors to detect the canine distemper virus (CDV), and use of NPs to develop vaccines.
Discussion
The publications analyzed were grouped as follows:
Use of nanoparticles in virus detection. The use of nanomaterials for the development of biosensors is a subject of extensive research in the biomedical area. For example, there is a wide variety of systems used for the detection and diagnosis of the human papillomavirus (Cheng et al. 2022), human immunodeficiency virus (Nandi et al. 2020), and SARS-CoV-2 (Deepshikha et al. 2022). A biosensor is a system made up of three main components: a transducer that can be an NP, a biological recognition element that must be selective to interact with the analyte of interest (enzymes, DNA, antibodies, cells, and others), and a detector (Saylan et al. 2019; Figure 1).
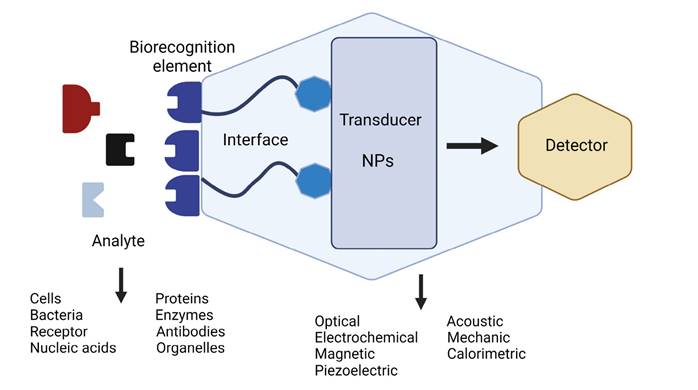
Figure 1 Schematic representation of a biosensor capable of selecting a specific analyte as its main characteristic.
These systems stand out from other sensors because of their high selectivity, sensitivity, and direct real-time detection capability. For the manufacture of biosensors, several NP systems have been used as biological receivers and transducers, most importantly NPs with high optical, magnetic, electrochemical, or electroacoustic properties. Their morphological (shape and size) and functional properties confer quick and easy identification, even for people unfamiliar with biosensors (Ortega-Berlanga et al. 2021). It should be mentioned that there is no universal biosensor, i. e., specific bionsensors should be designed and synthesized for the detection of a particular virus or analyte. Depending on the technique and NP used, biosensors can be highly selective and sensitive. However, there will always be limitations; for example, degradation of the biosensor by temperature or light, or saturation by interference with other biological agents that may induce false positives or affect detection limits and time (Wu et al. 2018). Our search found that systems with high optical properties have been the simplest and most effective devices for detecting viral diseases, which is why these systems are discussed in detail in this work.
Optical nanoparticles used as biosensors. The basic principle of an optical biosensor consists of inducing a change in the optical properties of the transducer associated with the analyte to be recognized, either through changes in fluorescence, light polarization, dispersion, or refractive index (Damborsky et al. 2016). Quantum dot-based (QD) optical biosensors and metallic NPs are the most suitable options due to several characteristics that make them superior to other types of NPs, including low manufacturing cost, high portability, high sensitivity and selectivity, short response time, and high spatial resolution (de La Rosa et al. 2020). Materials based on cadmium-selenium (CdSe), zinc sulfide (ZnS), selenium sulfide (SES2), or gold nanoparticles (AuNPs) have boosted the development of biosensors for the detection of human and zoonotic diseases, making the detection and diagnosis of diseases more efficient and affordable, and with less impact on individuals (Bollella and Katz 2020).
We found a large number of studies on the use of biosensors in the detection of viral diseases, their main characteristics, and hosts (Supplementary Material 1); only the most recent results, corresponding to the period 2017 to 2022, are shown in Table 1. The information found showed that, to date, the methodology has only been applied to domestic animals, with potential for use in wild mammals. The biosensors described can detect, in real time, diseases caused by a large number of viruses, such as the sheep-and-goat plague, hepatitis B, E, norovirus, Zika, or gumboro, among many others. Therefore, its replication in wildlife species seems feasible.
Diagnosis of CDV using biosensors. CDV has been diagnosed in different species of domestic and laboratory mammals using biosensors (Table 2). The results indicate that the virus can be detected either directly or through the antibodies produced by the host. The systems used most frequently include gold nanoparticles (AuNPs) as transducers, given their great versatility, allowing them to shift color in real time upon coming into contact with antibodies or virus particles. The color shift in AuNPs derives from the properties conferred by Localized Surface Plasmon Resonance (LSPR; Hidayah and Herbany 2020). LSPR is a physical phenomenon that consists of making free electrons on the NP surface oscillate coherently with the electrical field of incident light to form a plasma. This collective oscillation of electrons depends on electron density, effective medium, refraction index, NP shape and size, and separation between NPs. The maximum lambda spectral position (λmax) indicates the position of the LSPR, and this can be tuned by modulating the size and shape of nanoparticles and changing the physicochemical properties of the surface and the medium where NPs are found (Aguilera-Juárez et al. 2022). The LSPR is composed of the extinction coefficient, which is the sum of the absorption coefficient and the scattering coefficient (Cext = Cabs + Cscat), i. e., of the total light photons interacting with the NP, one part is absorbed and the other is scattered mostly without apparent energy loss (elastic dispersion). In spherical nanoparticles between 40 and 80 nm, the LSPR is located within the visible spectrum. However, as nanoparticle size increases, the shape of NPs changes, or their surface chemistry changes, Cext is dominated by the Cscat determined by a red shift of λmax. This shift in the LSPR position induces the color shift in the biosensor and indicates the presence of the analyte anchored on the NP surface (Hernández-Adame et al. 2019).
Silver nanoparticles (AgNPs) have been studied as antivirals, e. g., in the case of SARS-CoV-2 (Jeremiah et al. 2020). They also have optical properties via the LSPR; hence, they could be used to detect CDV. Unlike AuNPs, AgNPs are chemically unstable and sensitive to pH and temperature, which may cause nanoparticle degradation and release metal ions (Ag2+), leading to interference and toxicity. However, stabilizing the surface of AgNPs with more stable ligands, e. g., thiol terminal groups, would make their use safer.
These biosensors are convenient because the color shift is a qualitative indicator of the presence of viruses, thus sparing the need for additional equipment or techniques. Alternatively, by determining the wavelength at which the sample has the highest absorption (λmax), its intensity in the spectrum may be used as a quantitative indicator of the viral load (Damborsky et al. 2016). These devices are efficient, portable, and highly efficient for diagnosing the presence of the disease in real-time using blood, feces, urine, or saliva samples from a potential host (carnivores, in this case).
Table 1 Development of common biosensors used for the detection of viral diseases.
| System | Main characteristics | Virus detected | Host | Highlights | Reference |
|---|---|---|---|---|---|
| QD-LFIAS | QD-Lateral Flow Immunoassay Band Particle size: 20 nm | Sheep-and-goat plague (PPR) | Sheep and cattle | Diagnostic results are obtained within 15 minutes in a convenient and simple way, so the method shows great potential for detecting PPR in the field. | Cheng et al. 2017 |
| QD@MHS NPs | QD + silica oxide Particle size: 6-14 nm | Hepatitis E virus, Norovirus | Crab-eating macaque (Macaca fascicularis) | Detection and diagnosis of hepatitis E virus equivalent to the RT-qPCR test. | Ganganboina et al. 2020 |
| CdSe/ZnS QDs | QD Particle size: 157 nm | H1N1 bird flu | Human / zoonosis | Result equivalent to the PCR method; detection range 3.2 hemagglutination units/mL. | Jo et al. 2020 |
| GO-QD | QD Particle size: 12 nm | H1N1 bird flu | Human / zoonosis | The virus was isolated and detected without interruption and impurities and can be used in complex biological samples. | Lee et al. 2018 |
| AuNPs - PFAB | AuNPs - Optic fiber | SARS-CoV-2 coronavirus | Human | The portable biosensor can easily detect SARS-CoV-2 directly from saliva samples. | Murugan et al. 2020 |
| CdZnSeS/ZnSeS + AuNPs | QD +AuNPs Particle size: 20-35 nm | Influenza virus | Human | Biosensors can be a suitable alternative for detecting a wide range of infectious viruses. | Nasrin et al. 2020 |
| AuNPs | AuNPs + PCR Particle size: 1-100 nm | Canine coronavirus I and II | Canids | PCR using AuNPs was effective for the rapid detection of CCoV I and CCoV II. | Qin et al. 2021 |
| CdTe QDs | QD Particle size: 3.1 nm | Zika virus | Human | The sensor is 100-fold more sensitive than traditional tests measuring the immune response of protein E. The immunofluorescent platform with QD can be considered convenient and simple for detecting Zika or monitoring the immune response in vaccine testing. | Ribeiro et al. 2019 |
| CdSe/CdS/ZnS QD | QD Particle size: 7.8 nm | H1N1 and H3N2 influenza viruses | Human | Soluble QDs can be used to develop fluorescent sensors for the rapid diagnosis of influenza A in humans. | Nguyen et al. 2020 |
| GO-AuNPs | AuNPs-Graphene Hybrid Particle size: 10-28 nm | Foot-and-mouth disease virus (HFMD) | Human / zoonosis | PCR detection is improved 1000-fold with AuNPs. High specificity and detection can be applied to diagnose the disease even in the early stages, so it can be a valuable tool in biomedical and clinical applications. | Kim et al. 2020 |
| GO-AuNPs | AuNPs-Graphene Hybrid Particle size: 14 nm | Respiratory syncytial virus | Human | The graphene surface significantly increased the area of exposure of AuNPs. The immunoassay successfully detected the virus in artificial samples, showing great potential for commercial applications. | Zhan et al. 2021 |
| AuNPs-ASOs | AuNPs-antisense oligonucleotides Particle size: 30-60 nm | SARS-CoV-2 coronavirus | Human | Sensors with modified AuNPs can diagnose positive COVID19 cases in less than 10 minutes from isolated RNA samples. The results are read at a glance with no need for specialized equipment. | Moitra et al. 2020 |
| MNPs/AuNPs | Magnetic Nanoparticles-AuNPs Particle size: 15 nm | H1N1 influenza virus | Human | Sensor sensitivity better than the ELISA test. The technique is convenient, low-cost, and detects the H1N1 virus quickly and easily. The biosensor has great potential in clinical and pharmaceutical diagnosis. | Ye et al. 2021 |
Treatment of CDV with nanovaccines. The nanovaccines topic is beyond the scope of the present document. Mentioning it is nonetheless important because this area shows that the use of nanotechnology may contribute significantly to the diagnosis and control of diseases in mammals. Table 3 shows the CDV nanovaccine prototypes evaluated in various mammal species with encouraging results. Examples include the use of silver nanoparticles (AgNPs) for the treatment of CDV in the early stages of the disease (Bogdanchikova et al. 2016) or the use of proteins plus nanoparticles to enhance the immunological response of organisms against CDV (Dong et al. 2021; Wang et al. 2022).
The use of biosensors for the detection of diseases affecting ecosystem health is feasible through the NP methodology. A case of global importance because it affects several species is the CDV, which affects different groups of wild mammals and may be easily detectable. Carnivores susceptible to CDV include coyote (Canis latrans), a species for which this methodological technology can be developed. Monitoring coyote populations would be a valuable early indicator of the potential presence of CDV at some sites, which may affect various species and be transmitted to domestic ones, even to humans. Coyote can become a sentinel species for CDV because its populations are abundant and it not listed under a risk category on national and international lists (Kays 2018; DOF 2019). This species has a broad distribution, including all of North and Central America, probably reaching South America (Monroy-Vilchis et al. 2020). Besides, it inhabits different ecosystems, including urban landscapes, so it is a "bridge" species susceptible to infection of various diseases, including CDV, and capable of passing them from natural to peri-urban environments and vice versa (Gese et al. 1991; Bischof and Rogers 2005). Additionally, it has great vagility (Gompper and Glitterman 1991) and can rapidly transmit viruses to other mammals with populations under a risk category, as is the case of the Mexican wolf (Canis lupus baileyi; Boitani et al. 2018), different species of fox, skunk, raccoon, or even felines, including jaguar (Panthera onca; Quigley et al. 2017).
Future perspectives. The health of ecosystems largely depends on the wellness of the wildlife populations in them. In increasingly fragmented ecosystems, the spread of diseases among organisms can occur more rapidly, so timely disease identification, management, and control are essential (Mathews 2009). As predators, carnivores are hosts for many pathogens that directly impact populations of other wild and domestic species, occasionally affecting humans (Bischof and Rogers 2005). The ecological importance of some species is worth highlighting, particularly the coyote. Besides being omnivorous and in contact with species of different trophic niches, this species inhabits a broad range of ecosystems, from pristine to the most disturbed ones. This is why it can act as a carrier of diseases across isolated environments.
| Type | Main characteristics | Host | Application | Lower detection limit | Hightlights | Reference |
|---|---|---|---|---|---|---|
| AuNPs | AuNPs + antibody - Antigen | Not specified | Diagnostic | 0.7 ng CDV antigen/mL | Urine samples were analyzed and the results were visible (colorimetric) and obtained in an easy, simple and viable way at a low cost. | Basso et al. 2015 |
| AuNPs | AuNPs - Antigen | Not specified | Diagnostic | 107.5 TCID50 CDV antigen/mL | Flow bands with AuNPs can serve as in-situ diagnostic tools for the detection of distemper virus antibodies in dogs and felids. | Karki et al. 2022 |
| AuNPs | AuNPs-antibody + Antigen | Rabbits | Diagnostic | 635 µg CDV antigen/mL | The biosensor is very simple in showing CDV presence/absence results colorimetrically; therefore, it has potential for use in clinical tests. | Mazzu-Nascimento et al. 2017 |
| AuNPs | AuNPs + PCR | Canids | Diagnostic | 5.4 x 102 CDV primers/25 µL | The PCR using AuNPs was 100-fold more sensitive than conventional PCR tests. The technique allowed rapid and efficient CDV detection, with potential for use in the diagnosis of animal epidemics. | Wang et al. 2021 |
The canine distemper virus (CDV) affects several species and families (Martínez-Gutiérrez and Ruiz-Sáenz 2016); it has been recorded in the orders Artiodactyla, Primates, Proboscidea, Rodentia, besides Carnivora (Martínez-Gutiérrez and Ruiz-Sáenz 2016). The extinction of local populations of carnivores such as the wolf has been recorded as a result of this virus (Gese et al. 1997), which highlights the importance of CDV for the health of ecosystems.
CDV is detected by blood antibody tests several days after the individual has been infected (Rendon-Marin et al. 2019). Carrying out these tests requires capturing the individuals, which implies the use of different trapping methods and having basic equipment and staff trained in carnivore handling. Blood samples should be processed in the laboratory, with results that are not readily available and can be expensive. These aspects restrain the implementation of this approach as a baseline for monitoring CDV in ecosystems.
The biosensors analyzed in domestic animals in this study have yielded highly promising results for the diagnosis of CDV, so these biosensors are likely suitable for use in wild mammals such as coyote. The current methodological progress allows for detecting CDV with a device containing an AuNP-based biosensor specific for this virus, which would work optically: when placing the sample in the device, the biosensor would shift to a reddish color if the virus is detected. Our review did not demonstrate the use of AuNPs for CDV detection by the Surface-Enhanced Raman Spectroscopy (SERS). We consider this option should be explored because star-shaped AuNPs can amplify the detection signal to diagnose CDV, even under very low viral loads.
Table 3 Use of nanovaccines in disease biocontrol.
| Type | Main characteristics | Host | Application | Hightlights | Reference |
|---|---|---|---|---|---|
| AgNPs | AgNPs | Canids | Treatment | Canids infected with CDV that had not yet developed symptoms of neurological damage recovered favorably from the disease after the administration of AgNPs. | Bogdanchikova, et al. 2016 |
| NPs-CDV H | NPS with recombinant CDV H protein | Rodent | Biocontrol | Mice inoculated with the vaccine showed better immune responses versus the control group, so the H protein can be used for the development of vaccines against canine distemper virus. | Dong et al. 2021 |
| NPs-HA-F | NPs + hemagglutinin CDV sequences + ferritin | Rodents | Biocontrol | All organisms inoculated with the nanovaccine showed an increased production of antibodies against CDV and were able to recognize and eliminate cells with antigens on their membrane. The nanovaccine has great potential for CDV biocontrol in domestic and wild organisms. | Wang et al. 2022 |
The advantage of using this type of device with biosensors for CDV detection is that it works not only with blood samples but with any body fluids. This avoids the need to capture potential individuals to collect samples for testing and perform monitoring. One of the easiest indirect samples for testing is feces. CDV is a highly unstable virus outside its host, so its life expectancy in the environment is just a few hours, depending on the ambient temperature (Gilbert et al. 2015). The ideal scenario would be to track targeted locations assisted with predictive distribution models. Once the sampling site is selected, it should be searched for fresh feces. First, a fragment of feces should be dissolved in distilled water because the device would not accept solid samples; then, a single drop of the dissolved sample would be placed in the device. The biosensor would then detect the presence of the virus through a color shift. The system would allow immediate real-time detection of the health status of coyotes in the target area, which would likely also reflect the condition of several coexisting species. Also, the signal produced by the biosensor could be sent to a cell phone so that the information would be stored in a virtual space.
There are very few products commercially available; most are still in the development and research stages (Bollella and Katz 2020), so a market price for the device has not been established. One way to reduce costs could be the development of biosensors capable of detecting several analytes at a time (Haleem et al. 2021).
Biosensors have potential for use to assess the health status of ecosystems. Early detection of viruses in infected wild carnivores will allow for timely and effective CDV containment plans (Funk et al. 2001), avoiding high costs related to the long-term conservation, management, and protection of species and ecosystems. Above all, this strategy would avoid the spread of viruses between different species of mammals, including humans.











 nova página do texto(beta)
nova página do texto(beta)


