INTRODUCTION
Animal agriculture is an important sector of the economy in Mexico, which ranks 7th globally in terms of the number of cattle produced for commercial purposes1. In 2010, bovines comprised the second highest livestock species raised in the country after poultry2. Despite increases in production forecasts due to improved genetics and feeding among other things3,4, parasitic diseases remain an important factor impacting the productivity of cattle in Mexico5. Furthermore, parasitic diseases can hinder the international trade of cattle raised in Mexico. For example, cattle intended for export are inspected at US ports of entry and are returned to Mexico if found infested with cattle fever ticks6.
The national herd is estimated to comprise 32,402,461 head of cattle (dairy= 2,420,289; beef= 29,992,172)7 distributed in almost 2 million square kilometers of land. Veracruz is the state with the largest herd that includes around 4 million head. Cattle graze in almost 2 million square kilometers of the territory, distributed in four pastoral regions according to agro-climatic conditions: a) Arid and semiarid, covering portions of nine states (Baja California, Baja California Sur, Sonora, Chihuahua, Coahuila, Nuevo León, Durango, Zacatecas and San Luis Potosí); b) Temperate, including nine states and the Federal District (Aguascalientes, Guanajuato, Hidalgo, Jalisco, México, Morelos, Puebla, Querétaro and Tlaxcala) and areas surrounding Mexico City; c) Dry tropical, comprising six states (Colima, Guerrero, Michoacán, Nayarit, Sinaloa, and Tamaulipas), and d) Humid tropical, extending across six states (Campeche, Chiapas, Quintana Roo, Veracruz, Tabasco, and Yucatán). Production systems and costs are largely dependent on the climatic conditions of the respective regions8.
Occurrence of internal and external parasites in cattle throughout Mexico is favored by the predominance of hot humid and sub-humid climates in tropical and subtropical latitudes. Cattle production provides an important economic activity for the large expanse of land in Mexico, and the United States (US) is the primary export market with an average of 1 million cattle imported by the US from Mexico between 1989 and 20099.
Economic loss due to parasites in some countries where the cattle industry is also an important economic activity has been estimated. Drummond et al10 calculated that annual losses due to cattle parasites in United States of America approximated US$ 2,260 million when the national herd comprised 124.7 million head11. This figure represented 10 % of the production value and sales of the cattle industry at that time. Estimates of annual economic loss for specific pests ranged from US$ 29.7 million for scabies and mange mites up to US$ 730.3 million for horn flies. Recently, Grisi et al12 reassessed the potential economic impact of internal and external cattle parasites in Brazil and estimated it to be at least US$ 13.96 billion annually.
A preliminary assessment revealed that the economic impact of parasitic diseases on cattle production in Mexico is uncertain5,13,14,15 and which recognized the challenges involving the estimation of economic loss on the productivity of cattle due to parasitic diseases related to multifactorial variables influenced by geographic area and climatic conditions, management practices, and herd size and density. This type of effort has been further complicated by the threat of parasiticide resistance in several populations of endo- and ectoparasites of cattle. Thus, the results from the study presented here represent a systematic approach to ascertain economic losses due to cattle parasitism in Mexico.
The present study aimed to calculate economic losses caused by cattle parasites in Mexico, including the main ecto- and endo-parasite species known to have a significant effect on the productivity of bovine populations in Mexico. Consideration was given to specific cases where parasiticide resistance increases the cost of efforts to control cattle parasites.
Methodology used
Endoparasites considered for the economic evaluation included: gastrointestinal nematodes (GIN), coccidia (Eimeria spp.), and liver fluke (Fasciola hepatica). The cattle tick (Rhipicephalus microplus), horn fly (Haematobia irritans), and stable fly (Stomoxys calcitrans) are the external parasites included in this analysis. Other parasites affecting cattle in Mexico were excluded because they: a) Were not reported to be of national economic importance, b) Are limited to small regions of the country, (c) Are associated with reports of enzootic instability, d) Are not associated with robust economic and epidemiological data in Mexico, e) Are missing economic and epidemiological information. Parasites falling on those categories include Dictyocaulus viviparus, Neospora caninum, Toxoplasma gondii, Cryptosporidium parvum, Tritrichomonas foetus, Dermatobia hominis, Tabanidae, and chewing lice.
The method to estimate the potential economic impact caused by each parasite species or group considered the number of animals at risk, data available on milk production loss, weight gain loss in beef cattle, or official reports on condemnation of livestock byproducts (i.e., livers)12. In most cases the economic analysis used data originated from local studies, generated primarily between 2000 and 2015. It must be clarified that a limitation of this approach is that estimates addressing data gaps were extrapolated from selected studies conducted in other countries on parasitic economic damage to cattle; whenever possible by making comparisons between studies conducted in Mexico under similar environmental conditions. Thus, the potential economic impact assessment for cattle parasites estimated here is dependent on a wide variety of climatic factors. Grisi et al12 validated this methodology to reassess the potential economic impact of cattle parasites in Brazil. Because information on production effects was obtained from control animals, i.e. parasitized animals kept untreated in the respective studies, or the estimated populations at risk of being affected by a particular parasitic disease, the resulting estimates of economic impact represent losses in untreated cattle or the estimated bovine population at risk. Thus, the estimates presented here do not necessarily represent the actual impact of cattle parasitism in Mexico but the potential losses expected in the absence of parasite control measures.
POTENTIAL IMPACT OF GASTROINTESTINAL NEMATODES
Helminthic infections are an important cause of disease and productivity loss in livestock production worldwide. Moreover, tropical and template climatic conditions favor the presence and incidence of GIN, potentiating the effect on animal health and reducing livestock production. This problem is not only caused by clinical parasitism; non-visible effects of the subclinical phase also provoke deficiencies in nutrient use, which can cause a major production loss16,17.
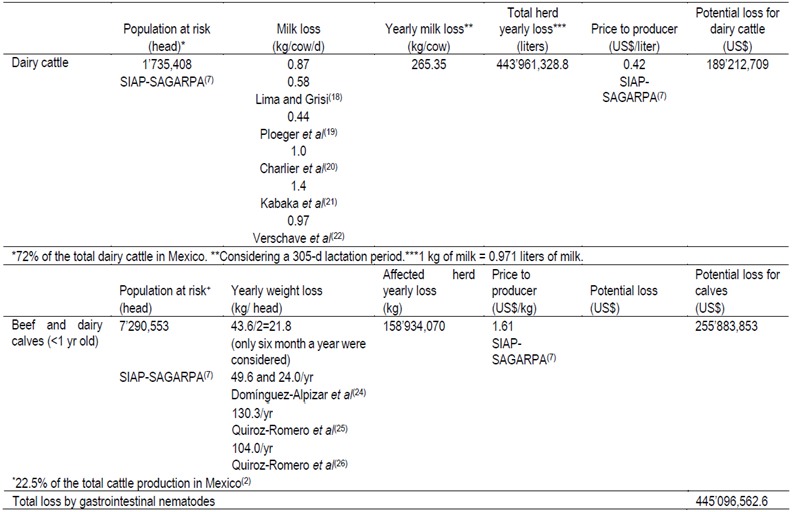
Impact of GIN on cattle production has been extensively studied worldwide. The effect of GIN on milk production was studied by Lima and Grisi18 in the state of Rio de Janeiro, Brazil where milk production of grazing cows medicated with albendazole at parturition was compared with untreated controls; an increase of 51.90 kg of milk per cow was reported in the medicated group during the 90-d study period, which corresponded to 0.58 kg of milk/cow/d. A similar study conducted in Holand19 reported an increase of 0.44 kg of milk/cow/d. Milk yield after anthelmintic treatment on pastured dairy cattle in the Netherlands was estimated to increase by 1.0 kg/cow/d20. When Kabaka et al21 evaluated the impact of GIN infection on cattle from two districts of Kenya, they found a daily loss of 1.4 kg/cow/d. Vercruysse et al22 evaluated 11 commercial dairy farms in Belgium to study the effect of eprinomectin treatment at calving on milk production; in this study eprinomectin treatment resulted in an average increase of 0.97 kg in daily milk yield. Considering that the average loss due to GIN in grazing cows is 0.87 kg of milk cow/d compared to animals with medication, the potential losses due to gastrointestinal worms for grazing dairy cattle population at risk in Mexico would be US$ 189,212,709 (Table 1). In spite of the evident economic benefit that could represent the control of GIN in grazing dairy cows, it is important to remember that most anthelmintic drugs cannot be used in those animals while they are producing milk for human consumption. Thus, other control methods, based on alternative control measures ought to be used to reduce the economic impact.
Table 1. Economic losses due to gastrointestinal nematodes among dairy and beef cattle in Mexico in 2013
Under the hot sub-humid conditions of Yucatan, Mexico, there was a superior average weight gain of 32.5 and 15.8 kg when the animals (Bos indicus) were treated with netobimin following two strategic programs (on August and October, or on August and January). In a period of 240 d, dewormed calves obtained an additional weight gain of 0.136 kg/d (49.6 kg/yr) or 0.07 kg/d (24.0 kg/yr) respectively, compared to non-treated calves23. The most common genera of GIN identified in this study area were Haemonchus, Cooperia and Trichostrongylus24.
In Chiapas, Mexico (hot and humid tropical region), an increase of 30.7 kg was obtained during a 90-d experiment (0.350 kg/d, 127.7 kg/yr) after deworming calves with ivermectin+abamectin25. Another study performed in Veracruz, Mexico also under hot and humid tropical conditions, the use of ivermectin+abamectin to control GIN in calves resulted in an additional weight gain of 20.0 kg (0.285 kg/d) and 25.03 kg (0.357 kg/yr) during a 70-d experiment (130.3 kg/yr and 104.0 kg/yr, respectively) compared with non-treated calves26.The most common genera of GIN identified in this study were Haemonchus, Cooperia, Trichostrongylus, Oesophagostomum and Ostertagia.
Studies have shown that the productivity of calves in tropical parts of Mexico is most affected by GIN during the rainy season24,27. Thus, for estimation purposes a rainy period of 6 mo was applied to derive potential losses caused by GIN in calves from Mexico. Considering an average loss of 87.1 kg of weight gain calves/yr without medication, the potential losses in the calf population caused by GIN in 6 mo would be US$ 255’883,853 (Table 1). Overall, annual losses in cattle due to GIN can potentially reach US$ 445,096,562.
POTENTIAL IMPACT OF EIMERIA SPP
Major weight loss associated with eimeriosis in cattle is likely to occur within the first year of life when animals are most susceptible to Eimeria infection28. Cattle eimeriosis reduces food consumption, body weight, feed efficiency and may cause 24 % mortality. An estimated 77 million young cattle in the United States are infected by coccidia during the first year of their life, 4 million will be treated for eimeriosis, and 80,000 cattle naturally infected with Eimeria, could die from the disease29. Annual economic losses due to eimeriosis in the US have been estimated at US$ 62 million30. Animals housed in large numbers with bad hygienic practices are more likely to contract the disease. Therefore, feedlot and dairy cattle are the most susceptible. Eimeriosis in feedlot cattle is associated with stress caused by shipping, changes in ration as well as weather conditions, and overcrowding31. Fitzgerald32 suggested that as a rule of thumb expenses associated with eimeriosis should be considered to be US$ 2.60 per calf/yr. Dairy farmers in Estonia were estimated to incur annual losses of generated income ranging from 8 to 9 % and assigned a loss of US$ 6.23 per calf/yr due to eimeriosis33.
Under hot subhumid tropical conditions, Rodríguez-Vivas et al34 studied the factors associated with bovine eimeriosis in calves and found Eimeria oocysts in 87.8 % of the samples. The most common species identified were E. bovis, E. zuernii, E. ellipsoidalis and E. auburnensis. These authors14 mentioned that these species are the most frequently found in cattle , and are reported among the most pathogenic35. A further study by Rodriguez-Vivas et al13 investigated the prevalence of gastrointestinal parasites in cattle of different ages in the south of Mexico. Prevalence of Eimeria spp represented 71.57 % of the fecal samples tested. Quezada et al36 estimated the prevalence of Eimeria spp in post-weaner calves in a dry region of Mexico and found 62 % of the animals to be positive. INEGI2 reported that in Mexico 22.5 % of the cattle population are bovines <12 mo of age. According to the total population of cattle in Mexico (32’402,461) reported by SIAP-SAGARPA7, and considering that 22.5 % of the national bovine herd is <12 mo of age, it was estimated that 7’290,553 calves are at risk of being affected by eimeriosis. Based on an average of 73.8 % prevalence of Eimeria spp13,34,36 among young cattle from different regions of Mexico, it was calculated that 5,380,428 young animals have been affected, each costing US$ 4.42/animal33 to treat eimeriosis33. Therefore, the estimated annual losses due to eimeriosis in Mexican cattle total US$ 23’781,491.
POTENTIAL IMPACT OF FASCIOLA HEPATICA
Liver fluke transmission depends on the presence of the snail intermediate host. In Mexico Lymnaea humilis, L. cubensis and L. bulimoides have been identified as intermediate host of F. hepatica37. Therefore, the distribution of the parasite is limited to those geographic areas where the appropriate snail species are present38. Liver flukes are enzootic in 29 of the 32 states of Mexico, especially where high annual rainfall and large areas of poorly drained pasture provide suitable habitats for the lymnaeid snails. Therefore, the economic impact of bovine fasciolosis caused by F. hepatica is relevant in areas where the parasite is enzootic or in non-enzootic areas where infected cattle are introduced39.
According to the four pastoral regions where cattle are raised in Mexico, the average (range) rates of liver condemnation due to fasciolosis from all cattle slaughtered are: Arid and semiarid region 2.89 %, (0.2 to 5.3 %)39,40; Temperate region 11.63 % (1.8 to 30.0 %)39,40; Tropical dry 6.9 % (2.0 to 11.63 %)39; Tropical humid (Yucatan and Quintana Roo States are considered free of F. hepatica) 12.5 % (0.2 to 35.8 %)39,41. The overall estimation of annual losses suffered by the cattle industry of Mexico due to liver condemnation as a result of fasciolosis caused by F. hepatica is US$ 4,239,667 (Table 2).
Table 2. Economic losses due to liver confiscation produced by Fasciola hepatica to cattle production in Mexico in 2013
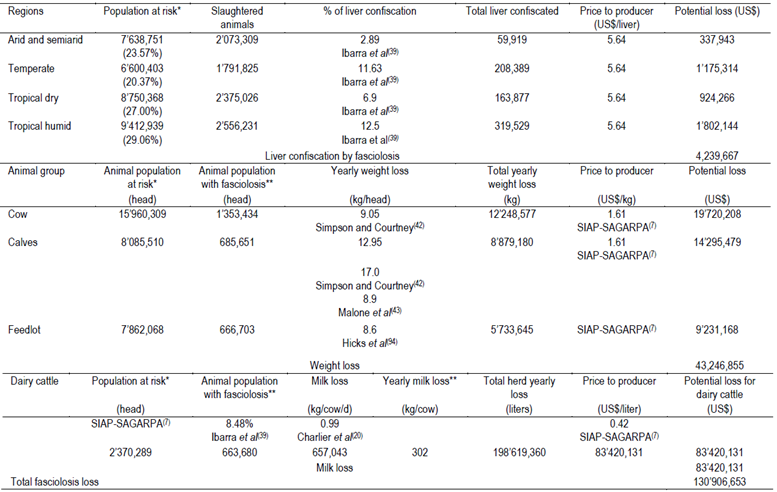
*Animals from three states considered free of F. hepatica were removed from the animal population at risk. **Only 8.48% of animal at risk were considered suffering fasciolosis39.
Simpson and Courtney42 conducted a survey in Florida, US, that showed the specific benefits of liver flukes control. Animals treated with the appropriate antiparasitic drug were 8.2 to 9.9 kg heavier when culled cows were considered, and 13.6 to 20.4 kg heavier when calves were treated at weaning. Another study conducted43 at a commercial cow-calf operation in Louisiana, showed that US calves from cows receiving treatments for both flukes and nematodes had an average weight gain advantage of 8.9 kg in 205-d adjusted wining weights compared to that of calves from cows receiving treatment for nematodes only. Because all treatment groups were maintained on the same conditions, the authors inferred that administration of flukicides to cows had a positive effect on productivity by increasing milk production, which resulted in heavier calves.
Recently, Charlier et al44 conducted a trial on 11 dairy herds exposed to F. hepatica to assess the effect of closantel treatment at dry-off (or 80-42 d before calving in first-calving heifers) on milk production parameters. The authors found that closantel treatment resulted in a noticeable increase of 303 kg in a 305-d milk production (0.99 kg of milk/d/animal). Based on this result it was estimated that the annual loss in milk production due to infection with F. hepatica is US$ 83’420,131 (Table 2). The estimated loss associated with the effects of F. hepatica infection in beef and dairy cattle that livestock producers in Mexico suffer totals US$ 130’906,653.
POTENTIAL IMPACT OF CATTLE TICKS
Ticks and the pathogens they transmit to domestic animal species, humans, and wildlife are widely distributed throughout the world, particularly in tropical and subtropical regions. It has been estimated that 80 % of the world’s cattle population is exposed to tick infestation45. Ticks are responsible for a variety of losses, caused by the direct effect of attachment, injection of toxins, or through the morbidity and mortality associated with tick-borne pathogens46, and secondary problems as the enhancement of transmission of Dermatophilosis, myiasis47, or udder damage by tick species belonging to the genus Amblyomma48. The estimated annual global costs associated with ticks and tick-transmitted pathogens range between US$ 13.9 billion and US$ 18.7 billion49. Recently, Grisi et al12 estimated the annual costs associated with the cattle tick Rhipicephalus (Boophilus) microplus (R. microplus) in the Brazilian cattle industry, to be up to US$ 3.24 billion. The national cattle fever tick control campaign in Mexico50 reported that 65 % of the national territory is infested with R. microplus15. Based on this information, the estimated number of cattle at risk of R. microplus infestation is 24’973,983 cattle.
Losses on milk production caused by the cattle ticks R. microplus, were recently estimated by Rodrigues and Leite51 for the state of Minas Gerais, Brazil. These authors estimated that ticks were responsible for a reduction of 90.24 L of milk per cow per lactation period. Based on this information, the estimated loss to the dairy herd of Mexico is US$ 68’878,694 (Table 3).
Table 3. Economic losses due to the cattle tick Rhipicephalus microplus, related to milk and beef production in Mexico in 2013
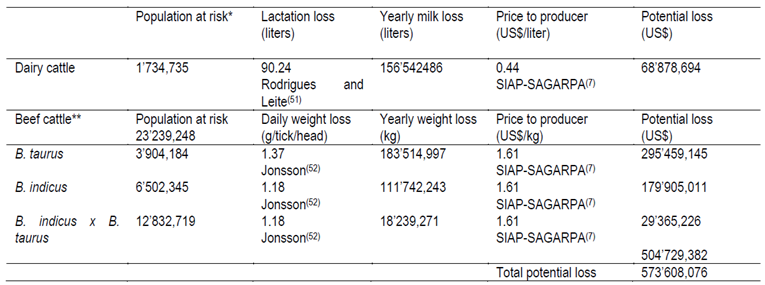
*Cattle at risk of tick infestation7;50. **16.80 % of Bos indicus, 27.98 % of Bos taurus, and 52.22 % of Bos indicus x Bos taurus cattle in Mexico. ***Considering the following mean daily tick burdens: B. taurus-94 ticks55, B. indicus x B. taurus-39.9 ticks5;53, and B. indicus-3.3 ticks56.
Estimates of the losses inflicted by cattle ticks on beef cattle were based on Jonsson52, who reported daily losses of 1.18 and 1.37 g per tick per animal for Bos indicus x Bos taurus cattle and B. taurus, respectively. An average of 70.3 adult ticks per cattle were reported during studies on population dynamics of R. microplus infesting B. taurus x B. indicus cattle in Yucatan5. Alonso-Diaz et al53 found an average of 14.0 and 35.4 R. microplus adult ticks per animal in two cattle crossbreed genotypes in Veracruz, Mexico. An average of 14.5 adult ticks per criollo cattle54 were found. Meanwhile, others55 found that B. taurus cattle had an average of 94 ticks/animal, while Gomes et al56 reported 3.3 ticks/per animal for B. indicus cattle. Considering that the beef cattle herd is composed of 16.80 % of B. indicus, 27.98 % of B. taurus, and 52.22 % of B. indicus x B. taurus crosses2, and the potential infestation rate for this bovine population, the estimated economic loss caused by this tick on beef cattle is US$ 504’729,382. Therefore, the total economic loss attributable to R. microplus in the Mexican cattle herd could approach US$ 573’608,076 (Table 3).
R. microplus is the vector of the hemoprotozoans Babesia bovis and B. bigemina, which cause bovine babesiosis, and the intraerythrocitic rickettsia Anaplasma marginale that causes anaplasmosis57,58. Babesiosis and anaplasmosis are responsible for important economic losses in the cattle industry of tropical and subtropical areas of the world. Annual losses (million) of US$ 500, 16.9, 5.1, 5.4, 6.8, 21.6, 19.4, 57.2, 3.1, and 0.6 caused by babesiosis and anaplasmosis have been reported for Brazil, Australia, Kenya, Zimbabwe, Tanzania, South Africa, China, India, Indonesia, and Philippines, respectively59,60. The economic impact is a consequence of direct losses like mortality, abortions, reduction in meat and milk yield, and indirect effects associated with the application of disease and tick control measures. In Argentina, 62 % and 38 % of the financial loss in dairy cattle affected by babesiosis were due to direct and indirect effects, respectively61. Babesiosis and anaplasmosis are prevalent wherever R. microplus and R. annulatus occur in Mexico62-66, and 75 % of the national cattle herd is considered to be at risk of becoming infected with B. bovis and B. bigemina67. However; we were unable to estimate the economic impact of babesiosis and anaplamosis on the cattle industry in Mexico because key epidemiological data, such as description of specific enzootic instability regions, mortality and incidence of babesiosis and anaplasmosis outbreaks, and number of abortions due to clinical cases and seasonal occurrence of outbreaks were not found during the search of the scientific literature.
POTENTIAL IMPACT OF HAEMATOBIA IRRITANS
The horn fly, Haematobia irritans, is one of the most economically important pests of cattle worldwide12,65. It is an obligate hematophagous ectoparasite that feeds almost exclusively on cattle. Because of H. irritans feeding behavior and infestation levels tend to be high, cattle expend a great degree of energy in defensive behavior, and reduce the grazing time, feeding efficiency and milk production in the case of dairy cattle67.
Losses due to horn fly infestation, based on the studies in Brazil68 and Argentina69, are estimated as an average yearly weight loss of 3.25 kg per cow and 28.0 g per day (305 d of lactation, 8.54 kg per cow), respectively. Estimated losses in calves also were based on Bianchin and Alves68 in Brazil and Steelman et al70 in the US, which estimated an average yearly weight loss of 2.0 and 8.1 kg per calf, respectively. Yearly weight losses of steer and heifers were estimated as 12.19 kg per animal71. Losses in milk production caused by H. irritans in dairy farms were estimated as 27.0 kg decrease in milk production per cow per year in the US72.
In general, Haematobia irritans in Mexico affects cattle during 7 mo of the year73. Cattle in the state of Veracruz (hot humid tropic) experienced high H. irritans infestation (70 to 121 flies/animal) between August and November73. Galindo-Velasco et al74 reported that cattle in the state of Colima, including sub-humid tropic and sub-tropic environments, had high horn fly infestations (120 to 236 flies per animal) during 6 mo of the year. However, in the state of Tamaulipas high horn fly infestation levels were observed in cattle throughout the year with population peaks higher than 200 flies/animal detected in September, April, May, and June75. Considering the beef cattle population at risk, losses due to horn fly parasitism in Mexico are estimated to be US$ 231’665,430 (Table 4). No estimates of horn-fly losses on dairy cattle or beef cattle pregnancy rates are provided here. These issues need further investigation.
Table 4. Economic losses due to the horn fly Haematobia irritans, related to beef cattle production in Mexico in 2013
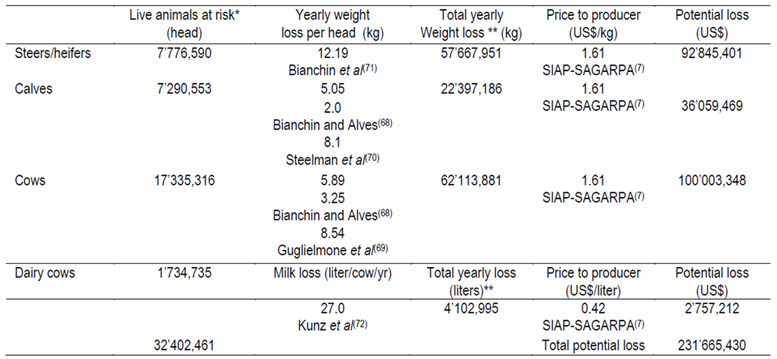
* From the total cattle production in Mexico7, 22.5 % was calves, 24.0 % steers/heifers, and 53.5 % adults (>3 yr)2. **7.3 mo per year were considered for H. irritans affect on cattle73,74,75.
POTENTIAL IMPACT OF STOMOXYS CALCITRANS
The stable fly, Stomoxys calcitrans, is another economically important pest of livestock worldwide. The stable fly problem can be exacerbated in areas where high organic content, like hay waste residues, are present at or nearby pastures where cattle graze76-78. Heavy stable fly infestations can results in a reduction of weight gain and feed efficiency of confined and grazing beef cattle79. Taylor et al76 and Grisi et al12 reported that the stable fly has an economic impact on US and Brazilian cattle production of US$ 2,211.0 and 335.5 million per year, respectively. Kunz et al72 estimated a potential loss due to stable flies in dairy and beef cattle of 0.1 kg per steer per day in feedlots and a 27.0 kg decrease in milk production per cow per year. Taylor et al76 estimated an annual per animal production loss of 139.0 kg milk for dairy cows, and 6.0, 26.0, and 9.0 kg body weight for preweaning calves, pastured stockers, and feeder cattle, respectively.
Cruz-Vázquez et al80 studied the annual variation of S. calcitrans adult stage infestation in Aguascalientes, Mexico and they found seasonal behavior of infestation with higher infestations between mid-summer and mid-fall period (2.5 mo) with abundance ranging from 4 to 40 flies per animal. Based on this information, economic losses due to the stable fly in Mexico were estimated to be US$ 6’786,792 (Table 5).
Table 5. Economic losses due to the stable fly Stomoxys calcitrans among dairy and feedlot cattle in Mexico in 2013
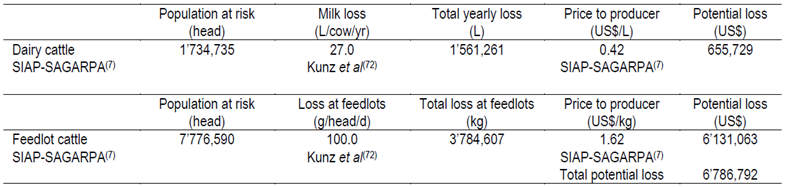
* 2.5 mo per year were considered to affect S. calcitrans to cattle80.
POTENTIAL ECONOMIC IMPACT OF ALL STUDIED PARASITES
Annual potential economic loss due to the six major endo- and ectoparasites of cattle in Mexico considered here was estimated to be US$ 1,411’845,004. Considering that the national cattle herd registered in 2013 included 32’402,461 head7, the estimated yearly loss per head caused by the selected cattle parasites was US$ 43.57. By comparison, the annual loss per head caused by a similar group of cattle parasites in Brazil, except the cattle grub (Dermatobia hominis) and New World screwworm fly (Cochliomyia hominivorax), was estimated to be US$ 65.4912.
The limitations of some of the baseline studies used to develop these estimates, particularly when extrapolated from local situations to a national scale12, are acknowledged. However, the general picture obtained from the present effort demonstrates the magnitude and importance of cattle parasitism in Mexico and the unfeasibility of a profitable livestock industry without adapting sustainable and integrated parasite control strategies. This approach is required given the favorable environmental conditions for livestock parasites in grazing areas where cattle are raised in Mexico.
PARASITICIDE RESISTANCE
The main strategy for cattle parasite control in Mexico is based on the use of chemicals such as anthelmintics, coccidiostats, flukicides, acaricides, insecticides and endectocides81. The indiscriminate use of these chemicals has resulted in the development of resistance in populations of internal and external parasites of cattle, which impacts the livestock industry in Mexico82-87.
Parasiticide resistance is a multifaceted problem that not only impacts the ability of producers to mitigate the impact of parasites on farm animal production, but it also poses risks to the environment and public health88,89. Resistance to conventional acaricides and macrocyclic lactones, including organophosphates (OPs), pyrethroids (SPs), amitraz, fipronil, and ivermectin among cattle fever tick populations has been identified as one of the most common problems in cattle from Mexico83,87. Resistance to OPs first developed in the 1980s in Mexico, and resistance to SPs emerged in the 1990s. Amitraz was introduced along with SPs to control OPs-resistant ticks in 1986. Amitraz was not widely used initially due to its higher cost, but its application became more prevalent and intensive after SPs resistance was discovered in 199385. The first case of amitraz resistance in R. microplus from Mexico was confirmed in 2001 in the state of Tabasco86. The first reports of R. microplus populations resistant to ivermectin and fipronil in Mexico were made in 2010 and 2013, respectively87,88. Rodríguez-Vivas et al89 studied 217 field populations of R. microplus and determined the prevalence of farms with resistance to SPs, OPs and amitraz in the southern part of the country. They found that resistance to SPs like cypermethrin, flumethrin, and deltamethrin was one of the most serious problems faced by livestock producers in the Mexican tropics where 66 to 96 % of the farms had cattle infested with R. microplus resistant to SPs. The presence of R. microplus populations resistant to ivermectin has been documented in the states of Yucatan and Veracruz where resistance prevalence ranges from 41 to 100 %, with most of the resistance level categorized as low. Resistance to multiple classes of antiparasitic drugs, including OPs, SPs, amitraz and ivermectin, appears to be an emerging problem among R. microplus populations in Mexico83.
Resistance to ivermectin in GIN has been identified in cattle from Mexico. Initial reports of ivermectin-resistant GIN in Mexico were obtained for infected cattle in the states of Campeche90, Yucatán84, and Veracruz91. Studies performed in Mexico suggest a high frequency of cattle herds with IVM-resistant GIN (>71 % of the surveyed herds), and they involved species in the genera Haemonchus and Cooperia.
Horn fly populations resistant to insecticides have been documented in Mexico. Insecticide resistance was reported for horn fly populations in the states of Tamaulipas, Veracruz, San Luis Potosi, Sinaloa, Aguascalientes, and Nuevo León92,93. The development of resistance to permethrin in horn flies from Veracruz and Nuevo León apparently was associated with strong selection pressure and the lack of integrated parasite management practices because in the farms SPs were used commonly to control R. microplus too92,93.
Antiparasitic resistance in endo- and ectoparasite populations affecting the health and productivity of cattle in Mexico increases the cost of parasite control and decreases the profitability of livestock producers. The use of commercial antiparasitic drugs to control endo- and ectoparasites in cattle is expected to continue increasing in Mexico due primarily to: availability and ease of use for endectocidal products, increasingly accessible prices especially for generic versions of macrocyclic lactones, and a dearth of products containing active ingredients with new modes of action83.
Studies assessing the actual economic impact of parasiticide resistance on the profitability of cattle producers in Mexico are needed. The establishment of integrated parasite control programs adapted to each of the four main agroecological regions could reduce the impact of endo- and ectoparasites on the health and productivity of the national cattle herd83. An adaptive and sustainable approach will help mitigate the burden of diseases caused by the six major endo- and ectoparasites considered here, which was estimated to cost the cattle industry of Mexico US$ 1.41 billion annually and a yearly loss per head of US$ 43.57.











 nueva página del texto (beta)
nueva página del texto (beta)


