Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista mexicana de biodiversidad
versión On-line ISSN 2007-8706versión impresa ISSN 1870-3453
Rev. Mex. Biodiv. vol.83 no.4 México dic. 2012
https://doi.org/10.7550/rmb.26165
Anatomía
Threatened Neotropical mollusks: analysis of shape differences in three endemic snails from High Paraná River by geometric morphometrics
Moluscos neotropicales amenazados: análisis de diferencias de forma en tres caracoles endémicos del río Alto Paraná mediante morfometría geométrica
Roberto E. Vogler1,2*, Ariel A. Beltramino1,3, Diego E. Gutiérrez-Gregoric1,2, Juana G. Peso4, Miguel Griffin2,4 and Alejandra Rumi1,2
1 División Zoología Invertebrados, Facultad de Ciencias Naturales y Museo, Universidad Nacional de La Plata. Paseo del Bosque s/n (B1900FWA), La Plata, Argentina. *robertovogler@fcnym.unlp.edu.ar; robertovogler@yahoo.com.ar
2 Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), Argentina.
3 Agencia Nacional de Promoción Científica y Tecnológica (ANPCyT), Argentina.
4 Laboratorio de Plancton y Bentos, Facultad de Ciencias Exactas, Químicas y Naturales. Universidad Nacional de Misiones, Rivadavia 2370 (N3300LDX), Posadas, Argentina.
5 División Paleozoología Invertebrados, Facultad de Ciencias Naturales y Museo, Universidad Nacional de La Plata. Paseo del Bosque s/n (B1900FWA), La Plata, Argentina.
Recibido: 17 julio 2011
Aceptado: 11 mayo 2012
Abstract
Variation in shape among a living and 2 extinct aquatic snails of the genus Aylacostoma, using a geometric morphometric method of thin plate splines and multivariate analysis was investigated. The analysis was performed to evaluate the diagnostic capability of this method and to explore shell shape differences, due to the lack of other data, in an attempt to answer why only 1 of the species persisted in the wild. Sixteen landmarks in a bi-dimensional space for 32 shells of type, paratype and reference specimens deposited in museums of Argentina were defined. Analysis was successful in assigning individual specimens to particular species. Statistically significant differences in last whorl, aperture, and spire were found for the first 4 non-uniform components explaining an 85% of local variation observed. Differences could be related to a differential use of habitat and/or to the degree of exposure to water current. More globose shell found in the extinct species could be associated to habitats and substrata with the highest water currents, whereas the more stylized shell in the third species could be related to a preference for more protected habitats, like those where it presently occurs.
Key words: Argentina, Aylacostoma, freshwater, native species, Paraguay, TPS.
Resumen
La variación de forma entre una especie viviente y dos extintas de caracoles acuáticos del género Aylacostoma, fue investigada mediante el método de morfometría geométrica de "thin plate splines" y análisis multivariado. El análisis se realizó para evaluar la capacidad diagnóstica del método y explorar las diferencias de forma de conchilla, debido a la falta de otros datos, en un intento por responder por qué sólo una de las especies persistió en la naturaleza. Dieciséis "landmarks" fueron definidos en un espacio bi-dimensional para 32 conchillas de ejemplares tipo, paratipo y de referencia depositados en museos de Argentina. El análisis fue exitoso en la asignación de los individuos a especies particulares. Se encontraron diferencias significativas en el último anfracto, apertura y espira respecto de los 4 primeros componentes no uniformes, que explicaron el 85% de la variación observada. Estas diferencias podrían vincularse a un uso diferencial del hábitat y/o al grado de exposición a la corriente. La conchilla más globosa en las especies extintas pudo estar asociada a hábitats y sustratos con mayores corrientes, mientras que la conchilla más estilizada en la tercera especie podría estar relacionada con la preferencia por hábitats más protegidos, como los que actualmente habita.
Palabras clave: agua dulce, Argentina, Aylacostoma, especies nativas, Paraguay, TPS.
Introduction
Freshwater gastropods are found on every continent and in nearly all aquatic habitats (Strong et al., 2008). South America still lacks global estimates of species richness of mollusks (Leveque et al., 2005), although local diversity estimates for large South American rivers (i.e. Uruguay, Paraná and Río de la Plata) and their tributaries, indicate that diversity of freshwater gastropods in this continent might be very high, including faunas that are sometimes extremely speciose and frequently do not occur in other continents (Lévêque et al., 2005; Gutiérrez-Gregoric et al., 2006; Rumi et al., 2006, 2008; Strong et al., 2008; Núñez et al., 2010). Nonetheless, many native snail populations are declining in numbers as a consequence of continuous degradation and destruction of their natural ecosystems because of unabated human activity (Rumi et al., 2006; Strong et al., 2008; Darrigran and Damborenea, 2011).
The genus Aylacostoma Spix, 1827 is included in the gastropod family Thiaridae Gill, 1871. The understanding of Thiaridae has notably increased in recent times (e.g. Glaubrecht, 1996, 1999, 2006; Michel, 2004; Gomez et al., 2011; Strong et al., 2011). However, although more than 30 species of Aylacostoma from Central and South America are reported in the literature (e.g. Simone, 2006), the genus has never been comprehensively revised. Excepting some anatomical data (i.e. Hylton-Scott, 1953; Morrison, 1954; Simone, 2001) and 2 phylogenetic studies based on comparative morphology (Simone, 2001, 2011), current knowledge of species of Aylacostoma is based solely on original descriptions and records of their occurrence as published in inventories of molluscan or benthic faunas (e.g. Souza et al., 2008; Jorcin et al., 2009).
The occurrence of species of this genus in Argentina and Paraguay was first reported by Hylton-Scott (1953, 1954). She described A. guaraniticum (Hylton-Scott, 1953), A. chloroticum Hylton-Scott, 1954 and A. stigmaticum Hylton-Scott, 1954 (Fig. 1). These species are considered viviparous, they reproduce by parthenogenesis and no males have been reported (Quintana and Mercado-Laczkó, 1997; Ostrowski de Núñez and Quintana, 2008). Of the 3 species, the only available anatomical data is for A. guaraniticum (Hylton-Scott, 1953), whereas for the other species only shells have been described (Hylton-Scott, 1954).
The 3 species were recorded in highly oxygenated freshwater habitats near the Yacyretá-Apipé rapids in the Paraná River (Argentina-Paraguay), between the Argentine cities of Ituzaingó (27°37' S, 56°40' W) and Posadas (27°20' S, 55°55' W) (Ostrowski de Núñez and Quintana, 2008). Fifty years later, these endemic species were threatened by extinction as the result of major alteration in the flow regime along the rapids. Such alteration was caused by the construction of the Yacyretá Binational Hydroelectric power plant (Seddon, 2000; Ostrowski de Nuñez and Quintana, 2008). Of the 3 species described until 1993 (before impoundment), only A. chloroticum still persists currently in 2 relictual populations at the upstream section of the reservoir (Ostrowski de Nuñez and Quintana, 2008) while A. guaraniticum and A. stigmaticum are categorized as extinct in the wild (Mansur, 2000a, b).
The 3 species from Argentina-Paraguay constitute the southernmost record of the entire South and Central American range of the genus and are supposed to represent closely related species (Castellanos, 1981). However, the lack of previous studies and the absence of soft parts deposited in museum collections, jointly with the extinction in the wild of A. guaraniticum and A. stigmaticum, have hampered further insight into their anatomy, biology, ecology and evolution.
Shape analysis is a fundamental part of much biological research and an indispensable technique in the identification of species (Adams et al., 2004), and may be a valuable tool for understanding species in which no other information is available. Information about the shape of an organism can be quickly and precisely captured by processing digital images with the "landmark methods" of geometric morphometries that quantifies deformations of morphometric points in coordinate space and separate size and shape variation as a standard part of the analysis. Landmark methods have demonstrated to be very effective, particularly when combined with multivariate statistical techniques (Rohlf et al., 1996; Cadrin, 2000; Zelditch et al., 2004; Conde-Padín et al., 2007).
Considering that morphology could reflect special adaptations to some environmental features, we investigated if the geometric morphometric analysis will allow characterizing shell shape in these Aylacostoma species, in an attempt to answer why only 1 of these species persisted in the wild. Also we evaluate the diagnostic capability of this method for referring individual specimens to particular species; especially given that the anatomy is unknown.
Material and methods
Samples examined include 32 shells of Aylacostoma from Argentina and Paraguay hosted in the Argentine Museum of Natural Sciences "Bernardino Rivadavia" -MACN- (Buenos Aires, Argentina); La Plata Museum -MLP- (La Plata, Argentina) and the National University of Misiones -UNaM- (Posadas, Argentina). This study was based only on museum material, given the lack of living populations of A. stigmaticum and A. guaraniticum (Mansur, 2000a, b). Neither fixed specimens, soft parts, radulae nor previous studies of them (except for original descriptions) were found in museum collections and published reports.
All the material include type, paratype and reference adult specimens of A. guaraniticum (MACN-In N° 29251; MLP N° 11213; UNaM CR-1; total length range [last 3 whorls]: 24.70 - 34.76 mm); A. stigmaticum (MACN-In N° 488-2; MLP N° 10963/64/65; UNaM CR-1; total length range [last 3 whorls]: 21.89 - 27.88 mm) and A. chloroticum (MLP N° 10958 and 11596; UNaM CR-1; total length range [last 3 whorls]: 23.17 - 33.76 mm).
All specimens were photographed with a Samsung SL-76 camera (8 mega pixels definition). The same shell orientation was used for all specimens, with coiling axis of shell on the y-axis and aperture on the same plane as the camera objective (Carvajal-Rodríguez et al., 2005, 2006). All images included a 5 cm graded scale. Sixteen landmarks were selected along the shell perimeter in order to capture differences in all regions of the shell (Fig. 2), following a criterion similar to that of Conde-Padín et al. (2007) (Table 1).

As stated by Carvajal-Rodríguez et al. (2005) in a study of Littorina snails, these points do not necessarily represent homologous landmarks -from a developmental point of view- in different specimens, although they allow to capture and analyze objectively and repeatably, shell shape in species of Aylacostoma. Six landmarks (LM4, LM5, LM8, LM9, LM11, LM12) were treated as sliding semi-landmarks by using TPSUTIL version 1.44 (Rohlf, 2009), thus relaxing the homology criterion.
The raw coordinate configurations of all specimens were aligned (i.e. translated, rotated and scaled to match one another) using the Generalized Procrustes Analysis (GPA) procedure to eliminate variation due to differences in scale and orientation, which establishes an average configuration by minimizing the sum of squared distances between homologous landmarks from different specimens (Rohlf and Slice, 1990; Rohlf, 1999; Rufino et al., 2006). This average configuration of landmarks resulting from GPA or tangent configuration (Cavalcanti et al., 1999) served as the "reference configuration" in subsequent calculations. For each specimen, a variable for size (centroid size) and a set of variables for shape (uniform and non-uniform components) of shell shape were also obtained. Centroid size is obtained as a scaling factor during GPA (Bookstein, 1991; Conde-Padin et al., 2007). The 2 uniform components describe differences that affect equally all parts of the shell (global differences). In contrast, non-uniform components account for local shape deformations of the reference configuration at different spatial scales (Conde-Padin et al., 2007).
The coordinates of all aligned specimens were used for the thin plate splines -TPS- and relative warp analysis -RWA- (Bookstein, 1989, 1991; Rohlf, 1993), in order to analyze and display the direction of shape differences among species. In the TPS method, a hypothetical infinitely thin metal plate (grid) is fitted over the reference configuration, which is then deformed until it matches exactly the target shape (Bookstein, 1989, 1991; Rohlf et al., 1996). Relative warps (RWs) are the principal components of any kind of shape variables and reflect the major trends in shape variation (Rufino et al., 2006).
Landmark data were obtained by digitizing images of the shells as in Fig. 2, using software TPSDIG version 2.12 (Rohlf, 2008) to generate coordinates. Relative warp analysis and computation of partial warp scores were computed using TPSRELW version 1.49 (Rohlf, 2010) with the scaling option α = 0 that weighs all landmarks equally and is considered to be more appropriate for systematic studies (Loy et al., 1993; Rohlf, 1993; Rohlf et al., 1996; Cavalcanti et al., 1999).
The first 4 derived morphometric variables studied (RWs1-4) were included in a non-parametric MANOVA (NP-MANOVA) (Anderson, 2001) based on Mahalanobis distance (9 999 permutations), in order to test the significance of the mean shell shape of the species. After NP-MANOVA, a canonical variates analysis (CVA) was conducted on the same first 4 RWs, in order to maximize the separation between groups (Zelditch et al., 2004), to estimate misclassification rates, and to evaluate the shape differences that best distinguish among the 3 species (Márquez et al., 2010). TPS deformation grids along the canonical axes were generated in TPSREGR version 1.38 (Rohlf, 2011).
Statistical analyses were performed with PAST, version 2.14 (Hammer et al., 2001). Most of the programs used in this study are available at http://life.bio.sunysb.edu/morph/
Results
Relative warp analysis showed that approximately 85% of the local shell variation is explained by the first 4 RWs (i.e. RW1= 57.41%; RW2= 13.15%; RW3= 8.78%; RW4= 5.48%). The NP-MANOVA conducted in order to test the significance of the mean shell shape of the 3 species was higly significant (F= 7.164, p= 0.0001). Shape differences among species were maximized using CVA, and species were successfully discriminated (Fig. 3, Table 2). The CV1 (explaining 79.08% of the observed variation) can discriminate among the 3 species at the same time, while CV2 (explaining 20.92% of the observed variation) only discriminates between A. guaraniticum and the others (Fig. 3).
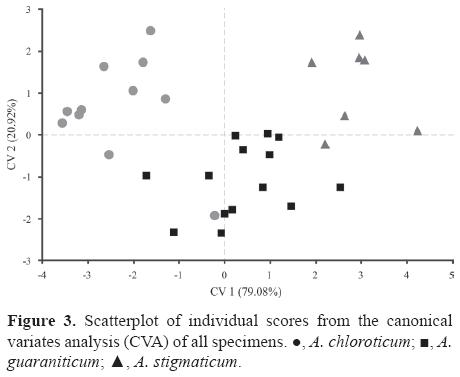
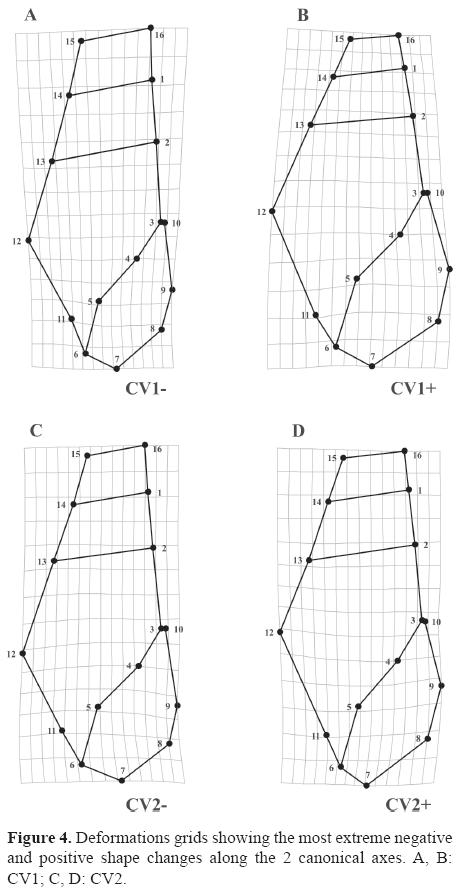
External landmarks and those that represent the aperture were connected by lines for an easier visualization of the meaning of deformations (Fig. 4). When analyzed from the lowest to the highest scores, first canonical axis can be mainly described as an expansion of the last whorl and aperture, involving almost all landmarks in the area. Also an antero-posterior contraction of spire was observed, showing an upward displacement of landmark pair defining the lower suture of penultimate whorl (LM13-LM2), as well as a downward and inwards displacement of landmark pair located on the upper suture of antepenultimate whorl (LM15-LM16) (Figs. 4A, B). In turn, when analyzed from the lowest to highest scores, the second canonical axis reveals no noticeable differences in spire shape, and variation was mostly associated to last whorl including an upward displacement of LM12 and a shrinking of LM6 and LM7 (Figs. 4C, D).
Discussion
Thin plate splines methods are increasingly used for morphometric research because they: i) allow complete separation of size and shape into distinct variables; ii) permit segregation of shape into uniform and non-uniform components; iii) have powerful means to visualize morphologic differences; iv) enable the incorporation of shape variables derived from geometric morphometric analyses into commonly used multivariate analyses; v) are cheap, and vi) employ user-friendly software (Anderson and Roopnarine, 2005; Carvajal-Rodríguez et al., 2005).
Application of geometric morphometric methods has demonstrated to be very useful in determining both intra (e.g. Palmer et al., 2004; Krapivka et al., 2007; Márquez et al., 2010; Valladares et al., 2010) and interspecific groups in mollusks (e.g. Ferson et al., 1985; Innes and Bates, 1999; Dommergues et al., 2003; Aguirre et al., 2006; Rufino et al., 2006). Like bivalves, gastropods have hard shells which make them excellent candidates for shape analysis by means of geometric morphometrics (e.g. Carvajal-Rodríguez et al., 2005, 2006), as no deformation occurs during manipulation.
In this study, significant differences in shell shape were found in the available museum material for A. guaraniticum, A. chloroticum and A. stigmaticum and almost 95% of the individual specimens were correctly assigned to species. We believe that the success of the method in revealing these differences seems to provide a robust basis to examine the morphological variation in these mollusks. This geometric sensivity has been noted by others in revealing shape differences in populations and specimens of sibling species of Littorina or British rough periwinkles (Carvajal-Rodríguez et al., 2005; Conde-Padín et al., 2007) and sibling species of Nassarius (Carvajal-Rodríguez et al., 2006). In the species studied here, the main shape differences evidenced along canonical axes can be summarized as changes in spire, last whorl and aperture. The most notorious discriminating differences were concentrated on CV1, where the species shape varies in a range from more stylized shells in A. chloroticum to more globose shells in A. stigmaticum.
The detection of 2 statistically significant variables, which summarize the main differences in shape among the 3 species, is interesting because previous geometric morphometric studies in gastropods have suggested that an interpretation of shape in simple biological terms is possible (Carvajal-Rodríguez et al., 2005; Conde-Padín et al., 2007). As an example Conde-Padín et al. (2007) proposed that the more globose shell found in an ecotype of Littorina saxatilis (Olivi, 1792) is possibly related with the need for resisting crab attacks, which are common where this ecotype lives. Unfortunately, no interpretations of this nature -as could be resistance to molluscivorous fish- can be inferred for the Aylacostoma species from the Paraná River because of: i) the disappearance in 1993 of the rapids where the species lived (Quintana and Mercado-Laczkó, 1997); ii) the extinction in the wild of A. guaraniticum and A. stigmaticum (Quintana and Mercado-Laczkó, 1997; Mansur, 2000a, b), and iii) the lack of previous studies. Nevertheless, considering that the 3 species inhabited highly oxygenated shallow freshwater habitats in the rapids (Quintana and Mercado-Laczkó, 1997), it seems probable that the differences between the 3 species regarding last whorl, aperture and spire shapes could possibly be explained by a differential use of the habitat and/or perhaps by different degrees of exposure to water currents.
As previously demonstrated for other freshwater snails (Greenwood and Thorp, 2001), a larger foot could be related to the ability to avoid being dislodged by waves. Thus, we hypothesized that the more globose shell and more oval aperture found in A. stigmaticum, followed to a lesser degree by A. guaraniticum, may be associated to habitats and substrata with the highest water currents in the rapids. Contrarily, the stylized shell seen in A. chloroticum may be related to the preference for more protected habitats as those found at the upstream section of the Yacyretá Reservoir, where 2 relictual populations of A. chloroticum still persist (Ostrowski de Nuñez and Quintana, 2008). Further studies should be made on these populations to test our hypothesis.
Finally, we believe that geometric approaches should not be limited only to the species of Aylacostoma from the Argentina and Paraguay as we consider that they are valuable analytical tools for quantifying and exploring the shape, that together with other data (e.g. anatomical, morphological, ecological and genetical) could contribute to the revision of this particular group of mollusks.
Acknowledgements
We thank Dra. Verónica Núñez, Dr. Rolando González-José and two anonymous reviewers for their helpful comments and suggestions.
Literature cited
Adams, D., F. Rohlfand D. Slice. 2004. Geometric morphometrics: ten years of progress following the 'revolution'. The Italian Journal of Zoology 71:5-16. [ Links ]
Aguirre, M. L., S. I. Perez and Y. N. Sirch. 2006. Morphological variability of Brachidontes Swainson (Bivalvia, Mytilidae) in the marine Quaternary of Argentina (SW Atlantic). Palaeogeography, Palaeoclimatology, Palaeoecology 239:100-125. [ Links ]
Anderson, M. J. 2001. A new method for non-parametric multivariate analysis of variance. Austral Ecology 26:32-46. [ Links ]
Anderson, L. C. and P. D. Roopnarine. 2005. Role of constraint and selection in the morphologic evolution of Caryocorbula (Mollusca: Corbulidae) from the Caribbean Neogene. Palaeontologia Electronica 8:32A:18p. [ Links ]
Bookstein, F. L. 1989. Principal warps: thin-plate splines and the decomposition of deformations. IEEE Transactions on Pattern Analysis and Machine Intelligence 11:567-585. [ Links ]
Bookstein, F. L. 1991. Morphometric tools for landmark data. Cambridge University Press, New York. 435 p. [ Links ]
Cadrin, S. X. 2000. Advances in morphometric identification of fishery stocks. Reviews in Fish Biology and Fisheries 10:91-112. [ Links ]
Carvajal-Rodríguez, A., P. Conde-Padín and E. Rolán-Álvarez. 2005. Decomposing shell form into size and shape by geometric-morphometric methods in two sympatric ecotypes of Littorina saxatilis. Journal of Molluscan Studies 71:313-318. [ Links ]
Carvajal-Rodríguez, A., J. Guerra-Varela, B. Fernández, E. Rolán and E. Rolán-Álvarez. 2006. An example of the application of geometric morphometric tools to the morphological diagnosis of two sibling species in Nassarius (Mollusca, Prosobranchia). Iberus 24:81-88. [ Links ]
Castellanos, Z. A. 1981. La familia Thiaridae, Morrison 1952 en la Argentina. In Fauna de agua dulce de la República Argentina. Moluscos gasterópodos, R. A. Ringuelet (ed.). Vol. 15, fas 3. FECIC, Buenos Aires. p. 7-18. [ Links ]
Cavalcanti, M. J., L. R. Monteiro and P. R. D. Lopes.1999. Landmark-based morphometric analysis in selected species of serranid fishes (Perciformes: Teleostei). Zoological Studies 38:287-294. [ Links ]
Conde-Padín, P., J. W. Grahame and E. Rolán-Álvarez. 2007. Detecting shape differences in species of the Littorina saxatilis complex by morphometric analysis. Journal of Molluscan Studies 73:147-154. [ Links ]
Darrigran, G. and C. Damborenea. 2011. Ecosystem engineering impact of Limnoperna fortunei in South America. Zoological Science 28:1-7. [ Links ]
Dommergues, E., J. L. Dommergues, F. Magniez, P. Neige and E. P. Verrecchia. 2003. Geometric measurements analysis versus Fourier series analysis for shape characterization using the gastropod shell (Trivia) as an example. Mathematical Geology 35:887-894. [ Links ]
Ferson, S., F. J. Rohlf and R. K. Koehn. 1985. Measuring shape variation of two dimensional outlines. Systematic Zoology 34:59-68. [ Links ]
Glaubrecht, M. 1996. Evolutionsökologie und Systematik am Beispiel von Süb- und Brackwasserschnecken (Mollusca: Caenogastropoda: Cerithioidea): Ontogenese-Strategien, paläontologische Befunde und Historische Zoogeographie. Backhuys Publishers, Leiden. 544 p. [ Links ]
Glaubrecht, M. 1999. Systematics and the evolution of viviparity in tropical freshwater gastropods (Cerithioidea: Thiaridae sensu lato) - an overview. Courier Forschungs-Institut Senckenberg 215:91-96. [ Links ]
Glaubrecht, M. 2006. Independent evolution of reproductive modes in viviparous freshwater Cerithioidea (Gastropoda, Sorbeoconcha): a brief review. Basteria 69 (Suppl. 3):28-32. [ Links ]
Gomez, M. I., E. E. Strong and M. Glaubrecht. 2011. Redescription and anatomy of the viviparous freshwater gastropod Hemisinus lineolatus (W. Wood, 1828) from Jamaica (Cerithioidea, Thiaridae). Malacologia 53:229-250. [ Links ]
Greenwood, K. S. and J. H. Thorp. 2001. Aspects of ecology and conservation of sympatric, prosobranch snails in a large river. Hydrobiologia 455:229-236. [ Links ]
Gutiérrez-Gregoric, D. E., V. Núñez, A. Rumi and M. A. Roche. 2006. Freshwater gastropods from Del Plata Basin, Argentina checklist and new locality records. Comunicaciones de la Sociedad Malacológica del Uruguay 9:51-60. [ Links ]
Hammer, Ø., D. A. T. Harper and P. D. Ryan. 2001. PAST: Paleontological Statistics Software Package for Education and Data Analysis. Palaeontologia Electronica 4:9. http://palaeo-electronica.org/2001_1/past/issue1_01.htm; 4:1:4:9 p. [ Links ]
Hylton-Scott, M. I. 1953. El género Hemisinus (Melaniidae) en la costa fluvial argentina (Mol. Prosobr.). Physis 20:438-443. [ Links ]
Hylton-Scott, M. I. 1954. Dos nuevos melanidos del Alto Paraná (Mol. Prosobr.). Neotropica 1:45-48. [ Links ]
Innes, D. J. and J. A. Bates. 1999. Morphological variation of Mytilus edulis and Mytilus trossulus in eastern Newfoundland. Marine Biology 133:691-699. [ Links ]
Jorcin, A., M. G. Nogueira and R. Belmont. 2009. Spatial and temporal distribution of the zoobenthos community during the filling up period of Porto Primavera Reservoir (Paraná River, Brazil). Brazilian Journal of Biology 69:19-29. [ Links ]
Krapivka, S., J. E. Toro, A. C. Alcapán, M. Astorga, P. Presa, M. Pérez and R. Guiñez. 2007. Shell-shape variation along the latitudinal range of the Chilean blue mussel Mytilus chilensis (Hupe 1854). Aquaculture Research 38:1770-1777. [ Links ]
Lévêque, C., E. V. Balian and K. Martens. 2005. An assessment of animal species diversity in continental waters. Hydrobiologia 542:39-67. [ Links ]
Loy, A., M. Corti and L. F. Marcus. 1993. Landmark data: size and shape analysis in systematics. A case study on Old World Talpidae (Mammalia, Insectivora). In Contributions to morphometrics, L. F. Marcus, E. Bello and A. García-Valdecasas (eds.). Museo Nacional de Ciencias Naturales, Madrid. p. 215-240. [ Links ]
Mansur, M. C. D. 2000a. Aylacostoma guaraniticum. IUCN 2011. IUCN Red List of Threatened Species. Version 2011.1. http://www.iucnredlist.org; last consult: 18.VII.2011. [ Links ]
Mansur, M. C. D. 2000b. Aylacostoma stigmaticum. IUCN 2011. IUCN Red List of Threatened Species. Version 2011.1. http://www.iucnredlist.org; last access: 18.VII.2011. [ Links ]
Márquez, F., J. Robledo, G. Escati-Peñaloza and S. Van der Molen. 2010. Use of different geometric morphometrics tools for the discrimination of phenotypic stocks of the striped clam Ameghinomya antiqua (Veneridae) in north Patagonia, Argentina. Fisheries Research 101:127-131. [ Links ]
Michel, E. 2004. Vinundu, a new genus ofgastropod (Cerithioidea: 'Thiaridae') with two species from Lake Tanganyika, East Africa, and its molecular phylogenetic relationships. Journal of Molluscan Studies 70:1-19. [ Links ]
Morrison, J. P. E. 1954. The relationship of Old and New World Melanians. Proceedings of the United States National Museum 103:357-394. [ Links ]
Núñez, V., D. E. Gutiérrez-Gregoric and A. Rumi. 2010. Freshwater gastropods provinces from Argentina. Malacologia 53:47-60. [ Links ]
Ostrowski de Núñez, M. and M. G. Quintana. 2008. The life cycle of Stephanoprora aylacostoma n.sp. (Digenea: Echinostomatidae), parasite of the threatened snail Aylacostoma chloroticum (Prosobranchia, Thiaridae), in Argentina. Parasitology Research 102:647-655. [ Links ]
Palmer, M., G. X. Pons and M. Linde. 2004. Discriminating between geographical groups of a Mediterranean commercial clam (Chamelea gallina (L.): Veneridae) by shape analysis. Fisheries Research 67:93-98. [ Links ]
Quintana, M. G. and A. C. Mercado-Laczkó. 1997. Caracoles de los rápidos en Yacyretá. Ciencia Hoy 7:22-31. [ Links ]
Rohlf, F. J. and D. Slice. 1990. Extensions of the Procrustes method for the optimal superimposition of landmarks. Systematic Zoology 39:40-59. [ Links ]
Rohlf, F. J. 1993. Relative warp analysis and an example of its application to mosquito wings. In Contributions to morphometrics, L. F. Marcus, E. Bello and A. García-Valdecasas (eds.). Museo Nacional de Ciencias Naturales, Madrid. p. 131-159. [ Links ]
Rohlf, F. J., A. Loy and M. Corti. 1996. Morphometric analysis of old world Talpidae (Mammalia, Insectivora) using partial-warp scores. Systematic Biology 45:344-362. [ Links ]
Rohlf, F. J. 1999. Shape Statistics: Procrustes superimpositions and tangent spaces. Journal of Classification 16:197-223. [ Links ]
Rohlf, F. J. 2008. TPSDIG: Version 2.12. New York State University at Stony Brook. [ Links ]
Rohlf, F. J. 2009. TPSUTIL: Version 1.44. New York State University at Stony Brook. [ Links ]
Rohlf, F. J. 2010. TPSRELW: Version 1.49. New York State University at Stony Brook. [ Links ]
Rohlf, F. J. 2011. TPSREGR: Version 1.38. New York State University at Stony Brook. [ Links ]
Rufino, M. M., M. B. Gaspar, A. M. Pereira and P. Vasconcelos. 2006. Use of shape to distinguish Chamelea gallina and Chamellea striatula (Bivalvia: Veneridae): linear and geometric morphometrics methods. Journal of Morphology 267:1433-1440. [ Links ]
Rumi, A., D. E. Gutiérrez-Gregoric, V. Núñez, I. I. César, M. A. Roche, M. P. Tassara, S. M. Martín and M. F. López-Armengol. 2006. Freshwater Gastropoda from Argentina: species richness, distribution patterns, and an evaluation of endangered species. Malacologia 49:189-208. [ Links ]
Rumi, A., D. E. Gutiérrez-Gregoric, V. Núñez and G. A. Darrigran. 2008. Latin American malacology. Freshwater mollusks from Argentina. International Journal of Tropical Biology 56:77-111. [ Links ]
Seddon, M. B. 2000. Molluscan biodiversity and the impact of large dams. IUCN, Switzerland. 42 p. [ Links ]
Simone, L. R. L. 2001. Phylogenetic analyses of Cerithioidea (Mollusca: Caenogastropoda) based on comparative morphology. Arquivos de Zoologia 36:147-263. [ Links ]
Simone, L. R. L. 2006. Land and freshwater molluscs of Brazil. EGB Fapesp, Sao Paulo. 390 p. [ Links ]
Simone, L. R. L. 2011. Phylogeny of the Caenogastropoda (Mollusca) based on comparative morphology. Arquivos de Zoologia 42:161-323. [ Links ]
Souza, G. T. R., M. H. Machado, M. L. G. G. Dias, F. H. Yamada, J. P. A. Pagotto and G. C. Pavanelli. 2008. Composifao e sazonalidade dos moluscos do Alto Rio Paraná, Brasil, e sua potencialidade como hospedeiros intermediários de digenéticos. Acta Scientiarum - Biological Sciences 30:309-314. [ Links ]
Strong, E. E., O. Gargominy, W. F. Ponder and P. Bouchet. 2008. Global diversity of gastropods (Gastropoda; Mollusca) in freshwater. Hydrobiologia 595:149-166. [ Links ]
Strong, E. E., D. J. Colgan, J. M. Healy, C. Lydeard, W. F. Ponder and M. Glaubrecht. 2011. Phylogeny of the gastropod superfamily Cerithioidea using morphology and molecules. Zoological Journal of the Linnean Society 162:43-89. [ Links ]
Valladares, A., G. Manríquez and B. Suárez-Isla. 2010. Shell shape variation in populations of Mytilus chilensis (Hupe 1854) from southern Chile: a geometric morphometric approach. Marine Biology 157:2731-2738. [ Links ]
Zelditch, M. L., D. L. Swiderski, H. D. Sheets and W. L. Fink. 2004. Geometric morphometrics for biologists. Elsevier Academic Press, New York. 443 p. [ Links ]














