Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista mexicana de biodiversidad
versión On-line ISSN 2007-8706versión impresa ISSN 1870-3453
Rev. Mex. Biodiv. vol.80 no.3 México dic. 2009
Taxonomía y sistemática
Gracilaria, Subgenus Textoriella (Gracilariaceae, Rhodophyta) in the Gulf of Mexico and the Mexican Caribbean
Gracilaria, subgénero Textoriella (Gracilariaceae, Rhodophyta) en el golfo de México y el Caribe mexicano
Kurt M. Dreckmann* and Abel Sentíes
Departamento de Hidrobiología, División de Ciencias Biológicas y de la Salud, Universidad Autónoma Metropolitana – Iztapalapa. Apartado postal 55–535, 09340, México, D.F., México.
* Correspondent:
tuna@xanum.uam.mx
Recibido: 10 octubre 2007
Aceptado: 21 abril 2009
Abstract
Four species of Gracilaria (Gracilariaceae, Rhodophyta) with textorii–type spermatangial conceptacles (subgenus Textoriella Yamamoto) are recorded for the Gulf of Mexico and the Mexican Caribbean: Gracilaria blodgettii, G. cervicornis, G. mammillaris, and G. tikvahiae. The general distribution of the subgenus for Central America, both Pacific and Atlantic, displays a disjunct pattern explainable based on the geologic vicariant events that interrupted the connection between Pacific and Atlantic at the Isthmuses of Panama (closed 3.1–2.8 million years ago), and Tehuantepec (southern Mexico, closed 4–3.5 million years ago). Gracilaria cuneata/G. crispata, and G. mammillaris (G. hayi)/G. veleroae are 2 pairs of sibling species, or sister taxa, that diverged as a result of the final emergence of the Isthmus, and of the same age as the Central American Isthmus itself.
Key words: Gracilaria, Textoriella, floristics, biogeography.
Resumen
Se registran 4 especies de Gracilaria (Gracilariaceae, Rhodophyta) con conceptáculos espematangiales tipo textorii– (subgénero Textoriella Yamamoto) para el golfo de México y Caribe mexicano: Gracilaria blodgettii, G. cervicornis, G. mammillaris y G. tikvahiae. La distribución general del subgénero para Atlántico y Pacífico de Centroamérica despliega un patrón disyunto explicable por los eventos geológicos vicariantes que interrumpieron la conexión entre Pacífico y Atlántico en los istmos de Panamá (cerrado hace aprox. 3.1–2.8 millones de años) y Tehuantepec (sur de México, cerrado hace aprox. 4–3.5 millones de años). Gracilaria cuneata/G. crispata y G. mammillaris (= G. hayi)/ G. veleroae corresponden a 2 pares de especies hermanas que divergieron como resultado de la emersión del istmo, y con aproximadamente la misma edad del istmo centroamericano.
Palabras clave: Gracilaria, Textoriella, florística, biogeografía.
Introduction
The genus Gracilaria Greville sensu lato (incl. Gracilariopsis E.Y. Dawson, and Hydropuntia Montagne, Gracilariaceae, Rhodophyta) are distinguished from other gracilarioid algae in the structure and organization of male cells or spermatia. There are 4 recognized general arrangements (Homi 1958; Yamamoto 1975, 1978; Liao and Hommersand, 2003): in superficial or epicortical sori; in cortical concave depressions, in deep cortical oval depressions, single or confluents, and in subcortical multi–cavernous conceptacles. Based on the differential distribution of these anatomical patterns within the genus Gracilaria, Yamamoto (1975) proposed the taxonomic establishment of 3 subgenera. The first, Gracilariella Yamamoto, is characterized by superficial spermatangia distributed all over the algal surface. The type of this subgenus is Gracilaria chorda Holmes. The second subgenus is Textoriella Yamamoto, which is characterized by spermatangial conceptacles in shallow depressions, each primordium of spermatangial mother cell giving place to a branched system that covers the floor of conceptacle when mature, each spermatangial mother cell producing spermatia. The type of Textoriella is Gracilaria textorii Suringar De Toni. Finally, subgenus Gracilaria has spermatangia in deep oval conceptacles, with each primordium of spermatangial mother cells giving rise to a branched system that covers the inner surface of the whole conceptacle when mature; each spermatangial mother cell produces spermatia. The type of this subgenus is Gracilaria verrucosa (Hudson) Papenfuss. Recently, Tseng and Xia (1999) proposed a fourth subgenus, Hydropuntia, based on the ocurrence of spermatangia in multi–cavernous conceptacles. The type of Hydropuntia is Gracilaria edulis (S. G. Gmelin) P. C. Silva.
The only available monograph of Gracilariaceae on the Mexican shores of the Gulf of Mexico and the Caribbean Sea is Taylor (1960). Because the family has experienced a great deal of taxonomic changes during the last 45 years, a monographic project that incorporates those changes is needed. As a part of that project, we present here our results on the subgenus Textoriella.
Materials and methods
Algal material was collected along the Mexican shores of the Gulf of Mexico (Tamaulipas, Veracruz, Tabasco, Campeche), and the Mexican Caribbean (Yucatán and Quintana Roo) throughout the past 10 years, preserved in 3–4% formalin/seawater solution, and housed as herbarium specimens at UAMIZ. We also studied herbarium material from AHFH, CIQRO, ENCB, FCME, MEXU, UAT, UC, UNL, and US. Small segments were cut into cross or longitudinal sections with a razor blade, stained with aniline blue, and mounted in 100% glycerine for anatomical observations. When specimens came from dry material, segments were rehydrated in a 1–2% aqueous solution of children's shampoo and warmed for 1–2 min in a microwave oven before sectioning. Schematic drawings of the 4 gracilariacean spermatangial types mentioned above were drawn with pen and ink on bright–white 90 g paper from a LeicaTM DMLB compound microscope, and are presented below as figures 1–5 (Figures 1, 2, 3, 4 and 5). Maps were also drawn on bright–white 90 g paper, and composed with AdobeTM PhotoshopTM CS2. Photographs were taken with a NikonTM D60 digital camera equipped with a standard NikonTM 35–55 mm Zoom lens.

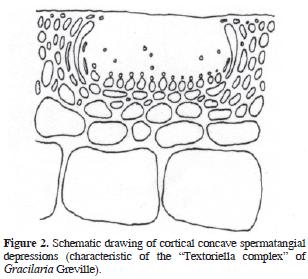
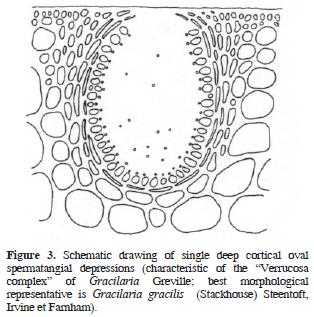
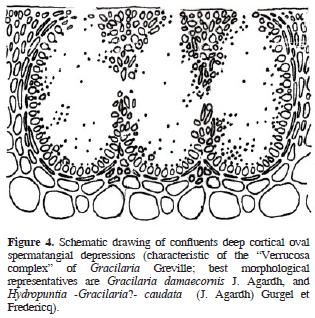
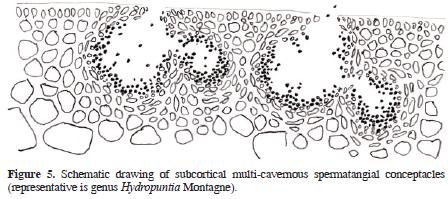
Descriptions
Gracilaria blodgettii Harvey, 1853: 111 (Type loc.: Key West, Florida, USA).
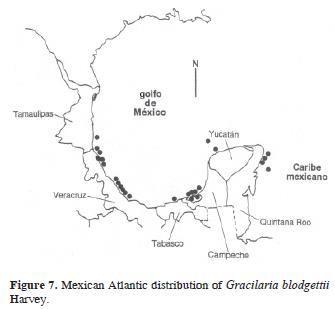
Synonym: Gracilaria cylindrica Børgesen, 1920: 375–377, figs. 364 and 365 (Syntype locs.: Virgin Islands; north of America Hill, St. John, and between St. John and St. Thomas). Figs. 6 and 7.
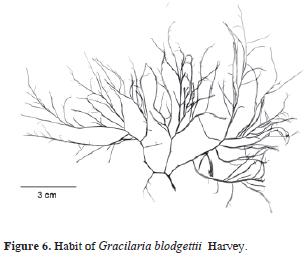
Thallus erect, up to 14 cm tall, texture soft, substance rubbery, dark red to pinkish in situ, pale brown once fixed. Adherence to herbarium sheets good. Holdfast a disc, small. Branching irregular, in more than one plane, secund or alternate in some parts. Axis and branches terete, (1.2)1.6–1.7(1.9) mm diameter, constant in width, branches strongly constricted at the bases, frequently arched. Segments between forks variable in length. Apex rounded. Stipe large. Transition between medulla and cortex abrupt. Medulla with 7–10 cells, subspherical to polygonal (240)340–600(740) pm diameter. Cortex formed by (1–3)8–12(15) cellular layers. Cortical cells (6)10–15(20) nm large, 7–10(20) pm diameter, anticlinal. Spermatangia 30–45 µm high, 30–50(50) µm wide. Cystocarps hemispherical, apiculate and sessile, 550–880 pm high, 750–1000(1200) µm diameter. Carpospores in clusters. Gonimoblast columnar. Tetrasporangia ovate, 28–50 µm large, 26–35 µm diameter, distributed all over the thallus except on main axis, apical region and stipe, rounded by 2–4 cortical cells, pyriform, and longer than ordinary.
Selected specimens: Tamaulipas: ENCB 4977, Escolleras de Altamira, Ma. E. Sánchez R., 28.09.1983. Veracruz: ENCB 10461, Sobre Malecón, calle Colón, C. Mendoza et al., 17.06.1993. Campeche: ENCB 2955, Playas cercanas a Ciudad del Carmen, Rzedowski #30048, 23.12.1972; ENCB 2969, 69 km al N.E. de Isla Aguada, sobre la carretera a Champotón, Rzedowski #30098, 23.12.1972; MEXU 1340, La Laguna de Términos, en la desembocadura de Ciudad del Carmen, M.M. Ortega #1027, 00.05.1964. Quintana Roo: ENCB 6680, Isla Mujeres, C. Mendoza, 01.03.1985; ENCB 8793, Puerto Morelos, C. Mendoza, 13.06.1987; ENCB 6614 Playa Lancheros, Isla Mujeres, L.E Mateo, 10.10.1983. Note: Synonym proposed by Norris and Fredericq (1990), and Fredericq and Norris (1992).
Gracilaria cervicornis (Turner) J. Agardh, 1852 [1851–1863]: 604.
= Fucus cervicornis Turner, 1808–1809: 131–132, pl. 121 (Type loc.: West Indies).
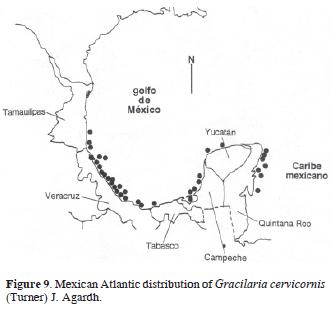
Synonym: Gracilaria ferox. J. Agardh, 1852 [1851–1863]: 592 (Lectotype loc.: Martinique, West Indies fide Oliveira et al., 1983:3003). Figs. 8 and 9.

Thallus erect, up to 10–25 cm tall, texture smooth, substance tough, brown red to yellowish green. Axes and branches complanate, 1–5 mm width. Branching dichotomous, in 2 planes, bases continuous (with no constrictions). Adherence to herbarium sheets regular to good. Margins with horn–like (cervicornis) branchlets. Segments between forks variable in length. Stipe very short or inconspicuous. Holdfast not distinct, disc–like. Apex, as margins, with horn–like processes or branchlets. Subcortex formed by 2 cellular layers, each 20–30 µm diameter, slightly pigmented. Transition is, however, abrupt. Cortex composed by 1–7 cellular layers, superficial or external ones 6–10 µm diameter, very pigmented, anticlinal. Medullar cells hemispherical, 170–300 µm diameter, thick cell walls with no pigmentation, less than 10 layers from one surface to the other (in cross section). Spermatangia 40–60 µm width, 35–40 µm depth. Cystocarps hemispherical, 0.5–1.1 mm diameter, mostly on horn–like branchlets, base–constricted and apiculate. Carpospores in clusters. Gonimoblast columnar. Nutritive tubular cells in contact with external pericarp. Tetrasporangia ovate, 30–40 µm large, 22–25 µm diameter, spread in wide groups (but not sori) below external cortical cells, specially on flattened branches at medium–superior thirds.
Selected specimens: Tamaulipas: ENCB 4785, Escolleras del río Pánuco, Ciudad Madero, Leg.: H. Alfaro García, 17.10.1982, Det.: J.M. López Bautista, Hab.: Sobre rocas a 2 m de profundidad. Veracruz: UAMIZ 487, Morro de La Mancha, K.M. Dreckmann, 02.12.1990; UAMIZ 752, Barra de Cazones, Laguna de Tamiahua, N. Hernández Soto M–26, 06.06.1994; US 34264 (Barcode: 00551339), Bajo Tuxpan, J.F. Longoria #102,17.07.1965. Campeche: ENCB 4519, Lerma, L. Aguirre J., 28.12.73; MEXU 1123, Lagune de Terminos; dans l'embouchure de Paso Real, LTS 11, Rodolfo Cruz #1269, 00.09.65. Yucatán: ENCB 8773, Yucaltepén, L. Huerta, 02.06.1987. Quintana Roo: ENCB 6649, Playa Maya, Cozumel, C. Mendoza, 27.02.1985. Note: Synonymy proposed by Oliveira et al. (1983).
Gracilaria mammillaris (Montagne) M. Howe, 1918: 515
= Rhodymenia mammillaris Montagne, 1842: 252 (Type locality: Martinique). Figs. 10 and 11.
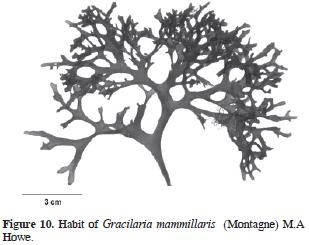
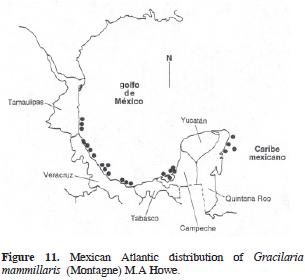
Thallus erect, (3)5–10(14) cm high, dark red, texture smooth, substance tough. Adherence to herbarium sheets minimal. Branching dichotomous, in 2 planes. Branches flattened, up to 1 cm width, no base–constricted. Margins free of proliferations or branchlets. Segments between forks constant in length up from the stipe. Apex rounded. Stipe short and stout. Holdfast not distinct, disc–like. Subcortex absent, ence, transition abrupt. Cortex formed by 1–3 cellular layers, external ones are slightly rectangular, 5–18 µm diameter, strongly pigmented, anticlinal. Medullary cells spherical to ovate, 50–150 µm diameter with no apparent pigmentation, less than 10 cellular layers from one surface to the other (in cross section). Spermatangia 18–20 µm depth, 40–45 µm width. Cystocarps hemispherical, 1.0–1.3 mm diameter, constricted and slightly to evidently apiculate. Carpospores in clusters, spherical to slightly pyriform, up to 30 µm large. Gonimoblast columnar. Nutritive tubular cells in contact with external pericarp. Tetrasporangia spherical to slightly ovate, 25–35 µm diameter, at external cortex, and rounded by enlarged cortical cells.
Selected specimens: Tamaulipas: ENCB 4071, Escolleras de Ciudad Madero, G. Galdi, 31.03.1978. Veracruz: ENCB 6644, Tuxpan, S. Pierdánt, 06.02.1986; UAMIZ 761, Mocambo–Boca del Río, N. Hernández Soto M2, 26.11.1994. Campeche: ENCB 4204 Ciudad del Carmen, A. Vargas #RHG 1543, 24.05.1965; US 36604 (Barcode: 00557649), Ciudad Del Carmen, Isla Carmen, A. Vargas #Rhg–1544, 24.05.1965; MEXU 1122, Lagune de Terminos; dans la lagune en face a Ensenada, LTS 12, Rodolfo Cruz #1026a, 00.04.1964. Quintana Roo: ENCB 6631, Muelle de Isla Mujeres, C. Mendoza, 13.09.1985; UAMIZ 762, Playa del Carmen, A. Sentíes G., 01.03.1994; US 14417, Xel–Ha, Quintana Roo, D.L. Ballantine #Dlb 1933, 12.04.1983.
Note: Schneider (1975), when comparing material of Gracilaria mammillaris from North Carolina (USA) with that of G. veleroae, collected by Dawson in the Gulf of California and housed in various herbaria, found no clear differences and proposed the heterotypic (or taxonomic) synonymy of G. veleroae with G. mammillaris. Norris (1985) suggested that, because Schneider never saw the type material belonging to either of the 2 binomials, both taxa should remain as distinct. Recently, Gurgel et al. (2004: 178) described Gracilaria hayi and included in it Gracilaria mammillaris sensu Schneider (1975) and Schneider and Searles (1991) not G. mammillaris, thus reducing the problem to nonexistent.
Gracilaria tikvahiae McLachlan, 1979: 19, Fig. 1 (Type loc.: Barrachois Harbour, Colchester County, Nova Scotia, Canada).
Holotype: Hoja #6919 in the Marine Algal Herbarium, National Research Council of Canada, Halifax (NRCC, !, in McLachlan [1979:21, fig. 1]). Figs. 12 and 13.
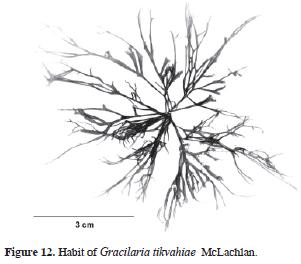
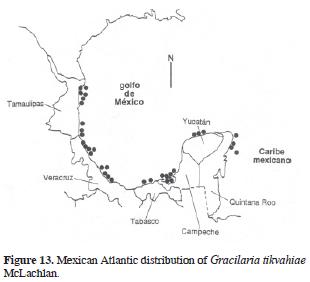
Thallus erect, (7)9–15(30) cm tall, texture smooth, substance loose, color highly variable, olive brown, reddish, dark purple or even, yellowish green. Adherence to herbarium sheets minimal. Branching dichotomous to irregular. Axes complanate, bases not constricted. Apex rounded to acute. Stipe large and slender. Cortical cells 8–9 µm diameter, in 1–2 layers, anticlinal. Medullary cells variable in size, around 100–120 µm diameter, more than 10 layers from one surface to the other (cross section). Spermatangia 18–20 µm depth 25–30(40) µm width. Cystocarps prominent, hemispherical, distributed all over the branch surface, 500–520 µm diameter, slightly apiculate, constricted. Carpospores in clusters. Gonimoblast columnar. Nutritive tubular cells in contact with external pericarp. Tetrasporangia ovate, distributed over the branches, variable in size, 25–30 µm width, 25–40 µm large.
Selected specimens: Tamaulipas: ENCB 5332, Punta Piedra, Municipio de San Fernando, J. Herrera, 20.09.1986. Veracruz: US 42509, Lagoon De Alvarado, G.B. Saunders #556, 31.01.1952. Campeche: MEXU 1056, Lagune de Terminos; dans la Lagune, en fase à Ensenada, LTS 12, Rodolfo Cruz #1026, 00.04.1964. Yucatán: US 45386 (Barcode: 00556795), Río San Miguel, Chicxulub, Puerto Progreso, M.A. Garza Hernandez#473, 12.03.67. Quintana Roo: ENCB 6678, Playa San Juan, Isla Cozumel, C. Mendoza, 04.06.1985.
Discussion
Phylogenetic relationships of subgenus Textoriella. Using 36 SSU rDNA sequences of Gracilariaceae from the tropical Atlantic and Pacific, Bellorin et al. (2002) found evidence that is relevant for subgenus Textoriella. For example, they found that the recognition of subgenera within Gracilaria based on spermatangial arrangement was not supported, and that most Pacific species with either "textorii" or "verrucosa" type spermatangia were deeply separated from Atlantic species (the exception being some sister taxa, as we will discuss below). Those Pacific taxa are G. chilensis Bird, McLachlan et Oliveira (central Chile), G. pacifica I.A. Abbott (California, subgenus Gracilaria Yamamoto), and Gracilaria "sp. Mexico" (California). Later, Gracilaria "sp. Mexico" proved to be G. vermiculophylla (Ohmi) Papenfuss (Bellorin et al., 2004). They also found that G. isabellana Gurgel, Fredericq et J.N. Norris (as G. lacinulata (Vahl) M. Howe; see Gurgel et al., 2004, and Wynne, 2005) from the Caribbean (Cumaná, Venezuela) and northern Brazil (Bahia), G. foliifera (Forsskål) Børgesen var. angustissima (Harvey) W.R. Taylor from the Caribbean, and G. tepocensis (E.Y. Dawson) E.Y. Dawson from Santa Catarina, Brazil, are closely related to G. tikvahiae from Canada, thus forming what they called the "tikvahiae" lineage. In this lineage, the temperate and subtropical isolates G. tikvahiae and G. tepocensis were closely related. It is significant, by the way, that the molecular study of Bellorin et al. (2002), supported the early proposal that G. foliifera var. angustissima is a taxonomic synonym of G. tikvahiae (Wynne, 1998). For the southern Pacific, Bellorin et al. (2002) found that G. chilensis and G. tenuistipitata C.F. Chang et B.M. Xia grouped in a single clade, although with very low bootstrap support (65%–68%).
Instead of SSU, Gurgel and Fredericq (2004) used the chloroplast–encoded rbc gene from 67 specimens worldwide, and found that selected taxa from subgenus Textoriella (G. textorii, G. cervicornis, G. curtissiae, G. tikvahiae, G. mammillaris, and G. chilensis) appear as belonging to sister groups such as verrucosa–type–bearing species (which is expected), as well as with Hydropuntia taxa (which they treat as a different genus); for example, G. tikvahiae is in the same clade of Gracilaria damaecornis, a multi–cavernous type bearing species that should belong to subgenus Hydropuntia sensu Tseng et Xia (1999). It is clear that not all textorii–type bearing taxa share a common ancestor (this, however, does not contradict the fact that G. cuneata/G. crispata, and G. mammillaris/G. veleroae are 2 pairs of sister taxa in the Central American Isthmus. See below). According to Gurgel and Fredericq's Fig. 1 (2004: 141), some of them even do share a common ancestor with Hydropuntia.
While authors like Tseng and Xia (1999), based on anatomy, support a subgeneric scheme for genus Gracilaria (subgenera Gracilariella, Textoriella, Gracilaria, and Hydropuntia), other researchers (Gurgel and Fredericq, 2004; Gargiulo et al., 2006; Kim et al. 2008), based on molecular evidence, suggest the taxonomic independence of genus Gracilariopsis, Gracilaria, and Hydropuntia (but see Liao and Hommersand, 2003, for a morphological study that also supports this view). Although well aware that the representation of the textorii–type in 2 distinct clusters of Gracilaria sensu stricto (Gugel and Fredericq, 2004) apparently represents homoplasy, we will recognize at least the subgenera Textoriella and Gracilaria, which are respectively close to the Bursa–pastoris and Gracilis morphological groups of Liao and Hommersand (2003), until more studies on textorii and verrucosa spermatangial arrangements are done, especially in the western Atlantic where, as Gurgel and Fredericq (2004) pointed out, the number of species is underestimated.
Distribution. The general distribution of subgenus Textoriella in the Central America, both Pacific and Atlantic (Lists 1 and 2), displays a regional pattern explainable with reference to geologic vicariant events that interrupted the connection between the Pacific and Atlantic at the Isthmus of Panama (closed 3.1–2.8 million years ago. Coates and Obando, 1996), and the Isthmus of Tehuantepec (southern Mexico, closed 4–3.5 million years ago. See below). If we look at the notes for lists 1 and 2, we will see an apparently more general pattern that would reflect an early pantropical Tethyan distribution, from the early–middle Eocene (60–50 million years ago) to the closure of the Tethys sea, about the middle Miocene (15–12 million years ago. Tomlinson, 1986; Brown and Lomolino, 1998).
List 1. Subgenus Textoriella in the tropical–subtropical western Atlantic (Oliveira, 1984; Schneider and Searles, 1991; Ganesan, 1994; Gurgel et al., 2004; Wynne, 2005).
Gracilaria armata (C. Agardh) J. Agardh1
G. blodgettii Harvey2
G. cervicornis (Turner) J. Agardh3
G. cuneata J.E. Areschoug4
G. curtissiae J. Agardh4
G. galetensis Gurgel, Fredericq et J. Norris
G. hayi Gurgel, Fredericq et J. Norris
G. intermedia J. Agardh subsp. ganesana Gurgel, Fredericq et J. Norris
G. isabellana Gurgel, Fredericq et J.N. Norris
G. mammillaris (Montagne) M. Howe
G. smithsonensis Gurgel, Fredericq et J. Norris
G. tepocensis (E.Y. Dawson) E.Y. Dawson
G. tikvahiae McLachlan5
Notes: 1Also recorded for the Mediterranean Sea (Gargiulo et al., 1992), and the Indian Ocean (Silva et al., 1996), 2Also recorded for Western Australia (Millar and Xia Bangmei, 1997), and the Indian Ocean (Silva et al., 1996). 3Also recorded for Canary Islands (Sosa et al., 1996), tropical West Africa (Lawson and John, 1982, as G. ferox), and the Indian Ocean (Silva et al., 1996). 4Not found in the subject area. 5 Recorded as a commercial introduction for the Hawaiian Islands (Abbott, 1999).
List 2. Subgenus Textoriella in the tropical–subtropical eastern Pacific (Taylor, 1945; Dawson, 1949; Norris, 1985; Dreckmann, 2002).
Gracilaria cerrosiana W.R. Taylor
G. crispata Setchell et Gardner
G. pachydermatica Setchell et Gardner
G. parvispora I.A. Abbott1
G. pinnata Setchell et Gardner2
G. ramisecunda E.Y. Dawson
G. subsecundata Setchell et Gardner3
G. tepocensis (E.Y. Dawson) E.Y. Dawson
G. textorii (Suringar) De Toni
G. turgida E.Y. Dawson
G. veleroae E.Y. Dawson4
Notes: 1Hawaiian Islands (Abbott, 1999). 2 Reported as endemic for Central, and 3Northern Gulf of California (Espinoza–Avalos, 1993). 4Also recorded for the Indian Ocean (Silva et al., 1996).
Taking into account the available distributional data, we found that there are 2 sibling pairs, or twin species (Collins, 1996), between the tropical eastern Pacific, and its western Atlantic counterpart: (1) G. cuneata/G. crispata, and (2) G. mammillaris (or G. hayi)/G. veleroae. Because these 2 pairs are very similar morphologically (Dreckmann, 2002. Also see note for G. mammillaris in this work), and the ranges of both are restricted to areas adjacent to the Central American Isthmus (they are not cosmopolitan), we take them as sister taxa that diverged as a result of the final emergence of the Isthmus, and to be of the same age as the Isthmus itself as the most parsimonious explanation for the pattern (Humphties and Parenti, 1999; Collins, 1996). Such patterns of morphologically similar sister taxa are common in most of the groups that have been studied in the area (Collins, 1996), including sea stars, sea urchins, brachyuran crabs, gastropods, bivalves, chitons, and polychaetes among others mentioned by Collins (1996. But also see Craw et al., 1999, for a worldwide review on vicariance).
Acknowledgments
This paper is a partial result of the first author's research in the Biological Sciences Doctorate Program at Universidad Autónoma Metropolitana. We wish to thank Dr. Michael J. Wynne (University of Michigan), and 2 anonymous referees for their very valuable suggestions and the improvement of the English version. Sincere thanks are due to the program's Academic Committee for helpful assistance and patience throughout the study. To the Universidad Autónoma Metropolitana Unidad Iztapalapa for financial support.
Literature cited
Abbott, I. A. 1999. Marine Red Algae of the Hawaiian Islands. Bishop Museum Press. Honolulu. 477 p. [ Links ]
Abbott, I. A., Zhang Junfu and Xia Bangmei. 1991. Gracilaria mixta, sp. nov. and other Western Pacific species of the genus (Rhodophyta: Gracilariaceae). Pacific Science 45:12–27. [ Links ]
Agardh, J. G. 1876. Species, genera et ordines algarum. Descriptiones succinctae specierum, generum et ordinum. Vol. III T. O. Weigel. Lipsiae. 724 p. [ Links ]
Bellorin, A. M., M. C. Oliveira and E. C. Oliveira. 2002. Phylogeny and systematics of the marine algal family Gracilariaceae (Gracilariales, Rhodophyta) based on small subunits rDNA and its sequences of Atlantic and Pacific species. Journal of Phycology 38:551–563. [ Links ]
Bellorin, A. M., M. C. Oliveira and E. C. Oliveira. 2004. Gracilaria vermiculophylla: A western Pacific species of Gracilariaceae (Rhodophyta) first recorded from the eastern Pacific. Phycological Research 52:69–79. [ Links ]
Børgesen, F. 1920. The marine algae of the Danish West Indies. Part 3. Rhodophyceae (6) with addenda to the Chlorophyceae, Phaeophyceae and Rhodophyceae. Dansk Botanisk Arkiv 4: 14–35. [ Links ]
Brown, J. H. and M. V. Lomolino. 1998. Biogeography. Second Edition. Sinauer. Sunderland. 691 p. [ Links ]
Coates, A. G. and J. A. Obando. 1996. The geologic evolution of the Central American Isthmus. In Evolution and environments in tropical America, J. B. C. Jackson, A. F. Budd and A. G. Coates (eds.). The University of Chicago Press. Chicago. p. 21–56. [ Links ]
Collins, T. 1996. Molecular comparition of transisthmian species pairs: rates and patterns of evolution. p. 303–334. In Evolution and environments in tropical America, J.B.C. Jackson, A.F. Budd, and A.G. Coates (eds.). The University of Chicago Press. Chicago. 415 p. [ Links ]
Chang, C. F. and B. Xia. 1963. Polycavernosa, a new genus of the Gracilariaceae. Studia Marina Sinica 3:119–126. [ Links ]
Craw, R. C., J. R. Grehan and M. J. Heads. 1999. Panbiogeography. Tracking the history of life. Oxford University Press. New York. 229 p. [ Links ]
Dawson, E. Y. 1949. Studies of northeast Pacific Gracilariaceae. Allan Hancock Foundation Pubs. Occasional Paper Number 7. The University of Southern California Press. Los Angeles. 105 p. [ Links ]
Dreckmann, K. M. 2002. El género Gracilaria (Gracilariaceae, Rhodophyta) en el Pacífico centro–sur mexicano. p. 77–118. In Monografías Ficológicas Vol. 1, A. Sentíes G., and K. M. Dreckmann (eds.). Universidad Autónoma Metropolitana Unidad Iztapalapa and Red Latinoamericana de Botánica. México, D.F. 192 p. [ Links ]
Espinoza–Avalos, J. 1993. Macroalgas marinas del Golfo de California. In Biodiversidad marina y costera de México, S. I. Salazar–Vallejo and N. E. González (eds.). Comisión Nacional para el Conocimiento y Uso de la Biodiversidad and Centro de Investigaciones de Quintana Roo. Chetumal. 865 p. [ Links ]
Fredericq, S. and M.H. Hommersand. 1989. Comparative morphology and taxonomic status of Gracilariopsis (Gracilariales, Rhodophyta). Journal of Phycology 25:228–241. [ Links ]
Fredericq, S. and J. N. Norris. 1992. Studies on cylindrical species of Western Atlantic Gracilaria (Gracilariales, Rhodophyta): G. cylindrica Børgesen and G. blodgettii Harvey. Børgesen. Taxonomy of Economic Seaweeds 3:211–231. [ Links ]
Ganesan, E. K. 1994. Morphology and taxonomy of the little–known species Gracilaria cuneata J.E. Areschoug (Gracilariales: Rhodophyta) from Venezuela. Carribbean Journal of Science 30:124–129. [ Links ]
Gargiulo, G. M., F. De Mazi and G. Tripodi. 1992. Morphology, reproduction and taxonomy of the Mediterranean species of Gracilaria (Gracilariales, Rhodophyta). Phycologia 31:53–80. [ Links ]
Gargiulo, G. M., M. Morabito, G. Genovese and F. De Masi. 2006. Molecular systematics and phylogenetics of Gracilariacean species from the Mediterranean Sea. Journal of Applied Phycology 18:497–504. [ Links ]
Gurgel, C. F. D. and S. Fredericq. 2004. Systematics of the Gracilariaceae (Gracilariales, Rhodophyta): A critical assessment based on rbc L sequence analyses. Journal of Phycology 40:138–159. [ Links ]
Gurgel, C. F. D., S. Fredericq and J. N. Norris. 2004. Molecular systematics and taxonomy of flattened species of Gracilaria Greville (Gracilariaceae, Gracilariales, Rhodophyta) from the Western Atlantic. Taxonomy of Economic Seaweeds 9:159–199. [ Links ]
Harvey, W. H. 1853. Nereis boreali–americana. Part I. Rhodospermae. Smithsonian Contributions to Knowledge 5(5), pls. XIII–XXXVI. 258 p. [ Links ]
Howe, M. A. 1918. Algae. p. 553–618 In The Flora of Bermuda, N. L. Briton (ed.). Charles Scribner's Sons. New York. 723 p. [ Links ]
Homi, H. 1958. The species of Gracilaria and Gracilariopsis from Japan and adjacent waters. Memoirs Faculty of Fisheries, Hokkaido University 6:1–66. [ Links ]
Humphries, C. J. and L. R. Parenti. 1999. Cladistic Biogeography. Interpreting patterns of plant and animal distribution. Second Edition. Oxford University Press. New York. 187 p. [ Links ]
Kim, M. S., M. Kim, R. Terada, E. C. Yang and S. M. Boo. 2008. Gracilaria parvispora is the correct name of the species known as G. bursa–pastoris in Korea and Japan. Taxon 57:231–237. [ Links ]
Lawson, G. W. and D. M. John. 1982. The marine algae and coastal environment of tropical West Africa. Nova Hedwigia 70:1–455. [ Links ]
Liao, L. M. and M. H. Hommersand. 2003. A morphological study and taxonomic reassessment of the generitype species in the Gracilariaceae. Journal of Phycology 39:1207–1232. [ Links ]
McLachlan, J. 1979. Gracilaria tikvahiae sp. nov. (Rhodophyta, Gigartinales, Gracilariaceae), from the northwestern Atlantic. Phycologia 18:19–23. [ Links ]
Millar, A. J. K. and Xia Bangmei. 1997. Studies on terete species of Australian Gracilaria. Taxonomy of Economic Seaweeds 6:103–109. [ Links ]
Montagne, J. F. C. 1842. Troisième centurie de plantes cellulares exotiques nouvelles. Annals Sciences Naturelle, Botanique II. 18:241–282. [ Links ]
Norris, J. N. 1985. Studies on Gracilaria Grev. (Gracilariaceae, Rhodophyta) from the Gulf of California, Mexico. Taxonomy of Economic Seaweeds 1:123–135. [ Links ]
Oliveira, E. C. 1984. Taxonomic criteria in the genus Gracilaria Grev. (Rhodophyta): An experience with the western Atlantic species. Hydrobiologia 116/117:55–58. [ Links ]
Oliveira, E. C. de, C. J. Bird and J. McLachlan. 1983. The genus Gracilaria (Rhodophyta, Gigartinales) in the western Atlantic. Gracilaria dominguensis, G. cervicornis, and G. ferox. Canadian Journal of Botany 61:2999–3008. [ Links ]
Tseng, C. K. and B. M. Xia. 1999. On the Gracilaria in the Western Pacific and the Southeastern Asia Region. Botanica Marina 42:209–217. [ Links ]
Schneider, C. W. 1975. Taxonomic notes on Gracilaria mammillaris (Mont.) Howe and Gracilaria veleroae Dawson. Taxon 24:643–646. [ Links ]
Schneider, C. W. and R. B. Searles. 1991. Seaweeds of the Southeastern United States. Duke University Press. Durham. 553 p. [ Links ]
Silva, P. C., P. W. Basson and R. L. Moe. 1996. Catalogue of the benthic marine algae of the Indian Ocean. University of California Publications in Botany 79:1–1259. [ Links ]
Sosa, P. A., M. A. Cabrera–Pérez and G. García–Reina. 1996. Genetic variation of Gracilaria cervicornis (Rhodophyta) gametophytes from the Canary Islands. European Journal of Phycology 31:143–147. [ Links ]
Taylor, W. R. 1945. Pacific marine algae of the Allan Hancock expedition to the Galapagos Islands. Allan Hancock Pacific Expeditions. Volume 12. The University of Southern California Press. Los Angeles. 528 p. [ Links ]
Tomlinson, P. B. 1986. The Botany of Mangroves. Cambridge University Press. Cambridge. 419 p. [ Links ]
Turner, D. 1808–1809. Fuci, sive plantarum fucorum generi botanicis ascriptarum, icones, descriptiones et historia. John & Arthur Arch, London. Vol. II. pls. 72–134. 162 (+ 2) p. [ Links ]
Wynne, J. M. 1989. The re–instatement of Hydropuntia Montagne (Gracilariaceae, Rhodophyta). Taxon 38:476–479. [ Links ]
Wynne, J. M. 2005. A checklist of benthic marine algae of the tropical and subtropical western Atlantic: Second revision. Nova Hedwigia 129:1–152. [ Links ]
Yamamoto, H. 1975. The relationship between Gracilariopsis and Gracilaria from Japan. Bulletin of the Faculty of Fisheries, Hokkaido University 26:217–222. [ Links ]
Yamamoto, H. 1978. Systematics and anatomical study of the genus Gracilaria in Japan. Memoirs Faculty of Fisheries, Hokkaido University 25: 97–152. [ Links ]














