Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista odontológica mexicana
versión impresa ISSN 1870-199X
Rev. Odont. Mex vol.18 no.2 Ciudad de México abr./jun. 2014
Original research
Chemotherapy-induced oral stomatotoxicity
Blanca Leticia Martínez Goyri,* María Elena Cruz Ramos,§ Enrique Echevarría y PérezII
* Former resident Maxillofacial Prosthesis Department, Graduate and Research School. National School of Dentistry, National University of Mexico (UNAM).
§ DDS attached to the Oncology Service. General Hospital, Mexico City.
II Professor, Maxillofacial Prosthesis Department, Graduate and Research School. National School of Dentistry, National University of Mexico, (UNAM).
ABSTRACT
The principal aim of the present study was to determine the type of oral lesions most frequently found in breast cancer patients treated with chemotherapy at the General Hospital in Mexico City and establish under which circumstances these manifestations occur. A file review was undertaken, spanning from 1994 to 1999. Inclusion and exclusion criteria were established. Out of 405 cases under consideration, 327 cases were included in the sample. In these selected cases, stomatotoxicity was present in 129 cases: 89 cases exhibited only a single oral manifestation, 19 cases showed two and 14 cases exhibited more than two (three or more). Relationship between histological lineage was reflected; it was present in the infiltrating ducts, lobular and others (p < .005). Treatment schemes with chemotherapy were integrated with 5-fluorouracil, adriamycin, cyclophosphamide and methrotrexate (p < .005) dosage and stomatotoxicity were related (p < .005).
Key words: Stomatotoxicity, mucositis, xerostomy, sialorrhea.
INTRODUCTION
Cancer is a chronic-degenerative disease which presently is on the rise due to the increase in life expectancy of the population. In our country, it represents the second cause for mortality. It is characterized by an anarchic growth of cells, which gives rise to the onset of tumors with the potential to disseminate through lymphatic and haematogenic routes. Cancer treatment is multidisciplinary; it can be local, at the affected area, or systemic, by means of chemotherapy.1
Breast cancer is very frequent in female population. Approximately 6% of all deaths are caused by this disease. Breast cancer is considered a systemic disease; it can be locally treated with surgery and radiotherapy; nevertheless, chemotherapy represents its systemic treatment, which represents an important advance since it favorably impacts in 25 to 50% of the afflicted population.2-4
Prognosis and treatment protocol mainly depend on two elements: histological lineage and clinical stage.
These two factors must be taken into consideration when establishing a scheme, therapeutic dosage and number of programmed cycles.
DOSAGE
The specialist is the professional who determines, calculates and administers the chemotherapy dosage to cancer-afflicted patients. He establishes schemes which refer to selected medication, and cycles which correspond to the number of treatments.
In general terms, it can be established that dosage is based upon weight or body surface of the patient (milligrams/m2 surface or milligrams/weight in kilograms). At advanced clinical stages, high dosages and increasing number of cycles are applied, which generally will induce more toxic manifestations.5
According to consulted scientific literature, among 40 to 60% of cancer-ridden patients are treated systemically. Of the aforementioned, 40% present oral toxicity, even in cases where the cervico-facial region is not involved in the primary lesion.6,7
Mucositis appears due to a decrease in the renovation rate of the basal epithelium. It becomes thinned down and presents ulcers in the soft tissues, mainly in non-keratinized tissues such as oral and labial mucosae, as well as the lateral and ventral surfaces of the tongue. Severity varies according to the scheme employed, dosage and general state of the mouth before the therapy.
Almost all chemotherapeutic agents elicit stomatotoxicity to a certain degree. Clinical experiences of some authors reveal the following substances as the most frequent causal agents: anti-metabolites, methotrexate, 5-fluorouracil, doxorubicin, bleomycin, dactinomycin, daunorubicin and cyclophosphamide. If to this we add disrupted renal and hepatic functions, the risk increases as a result of reduced metabolism, which decreases drug secretion. When these circumstances are present, stomatitis cases are more severe and lasting. Histaminic and blood concentrations of cytotoxic agents delay the re-epithelialization process, therefore, as the varied proposed cycles are administered, manifestation probabilities increase.8,9
Breast cancer cases are very frequent in Mexico; therefore, there is a need to administer the aforementioned treatments; most patients seek treatment when the disease is at advanced stages, where stomatological manifestations are already present. These manifestations can be mitigated and sometimes avoided whenever cases are dentally treated before, during and after the chemotherapy treatment.10-12
METHODS
A retrospective and descriptive study was undertaken. Clinical files from breast cancer patients from years 1994 through 1999 were reviewed. All files belonged to the Oncology Service of the GeneralHospital, Mexico. All studies were conducted in consultation with services pertaining to breast tumors, chemotherapy and maxillofacial prosthesis
EXCLUSION CRITERIA
The following were excluded:
1) Patients afflicted with diseases pertaining to the oral cavity exhibiting manifestations before the chemotherapy treatment.
2) Patients afflicted with systemic disease and stomatological manifestations such as diabetes mellitus or acquired immunodeficiency syndrome.
3) Patients previously subjected to head and neck radiotherapy.
INCLUSION CRITERIA
All files included were of diagnosed breast cancer patients, who had been treated, from the beginning of treatment, at the chemotherapy and maxillofacial services of the General Hospital, Mexico.
The following variables were included:
1) Type of lesion through specific search.
2) Onset of oral lesions related to the chemotherapy scheme.
Files were examined with the help of a data collection sheet which included: age, gender, histopathological diagnosis, clinical stage, chemotherapy scheme, stomatological group, as well as lesion-specific search.
Results analysis were undertaken according to descriptive statistics methods.
• Student t test for continuous variables.
• χ2 for non-continuous variables (statistics included).
RESULTS
A review of files dating 1994 to 1999 was undertaken. We selected 405 cases of breast cancer patients, out of which only 327 fulfilled the inclusion criteria for the present study.
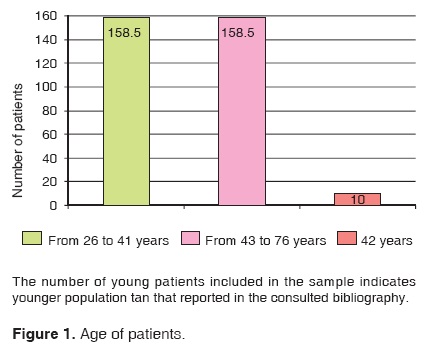
Breast cancer patients' age ranked from 26 to 76 years, the average was 42 years and the rank 50 years (Figure 1).
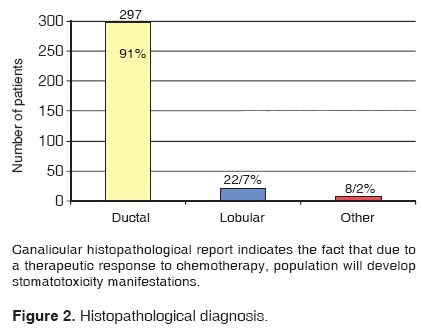
The histopathological analysis revealed the following: 297 patients presented diagnosis of infiltrating ductal carcinoma, 22 presented diagnosis of lobular carcinoma, and 8 cases were included in the diverse group (papillary, medullar) (Figure 2).
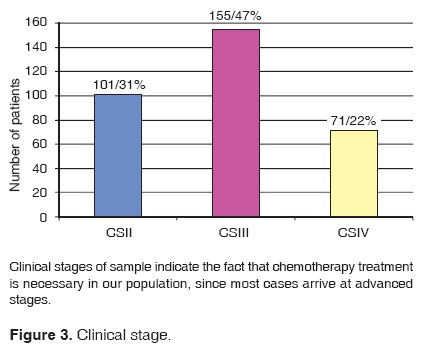
Assessed files revealed that 101 patients were at clinical stage II, 155 patients were at clinical stage III and 71 patients were at clinical stage IV (Figure 3).
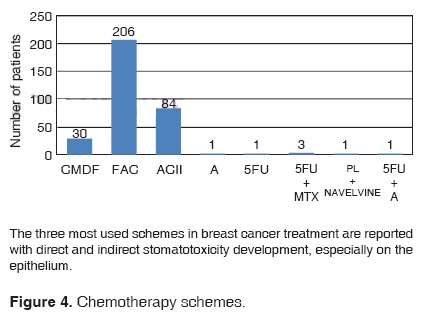
According to the scheme administered to these patients during treatment, the following was obtained: 201 patients received 5-fluorouracil, cyclophosphamide and doxorubicin (FAC), 84 patients were treated with adriamycin and cyclophosphamide (ACII), 30 patients were treated with cyclophosphamide, methotrexate and 5-fluorouacil (CMF), and only three were treated with 5-fluorouracil plus methrotrexate (5FU + MTX). The following schemes were each used in one case only: Adriamycin (A), 5-fluorouracil (5FU), A + 5FU, platinum and navelvine (OL + NV) (Figure 4).
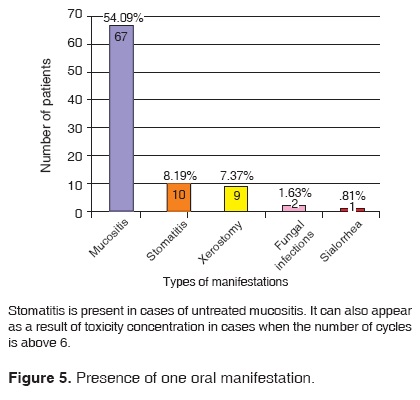
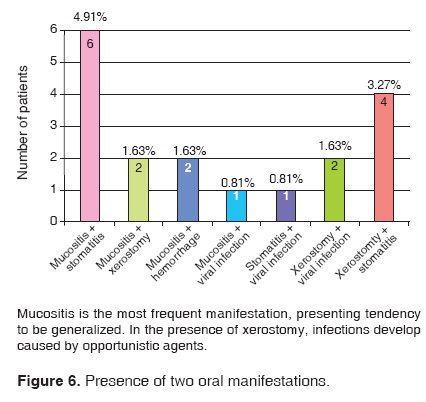
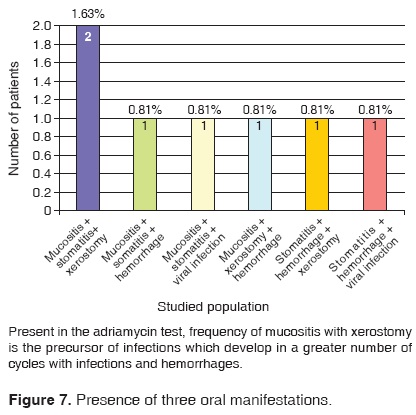
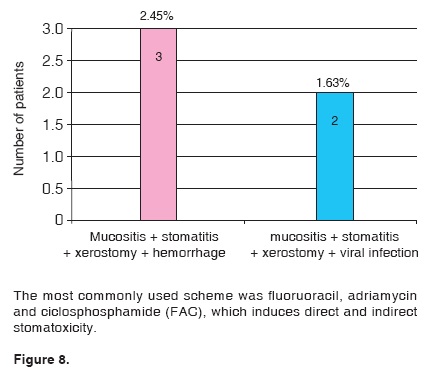
122 patients out of the 327 files included in this study presented stomatological manifestations. This represents a 37.31% of the studied sample. Out of these 122 patients, 89 exhibited a single oral manifestation during the chemotherapy treatment; 19 exhibited two different manifestations and 14 patients exhibited three or more stomatological manifestations (Figures 5, 6, 7, 8).
Statistical analysis was developed using the package designed for this purpose. In these analyses, the following variables were included from the data base:
1. Within the scope of the histopathological diagnosis, the following relationships were studied:
a) Infiltrating ductal versus lobular.
b) Infiltrating ductal versus others.
In both cases, statistical comparison resulted significant with p < .005.
Another studied factor was:
2. Different chemotherapy schemes; in this case, the following were compared:
a) FAC versus ACII
b) FAV versus CMF.
In both cases p < .005 (statistically significant).
Finally, the following factors were assessed:
3. Stomatological manifestations:
a) To identify the amount of present manifestations by comparing:
- 1 versus 2 (manifestations).
- 1 versus 3 (manifestations).
b) To determine the type of manifestations present by comparing:
- Mucositis versus sialorrhea.
- Mucositis versus xerostomy.
- Mucositis versus fungal infections.
Study of these cases revealed values of p < .005, indicating statistically significant results.
DISCUSSION
According to consulted literature, the average age of breast cancer patients ranks from 43 to 53 years. In our review we obtained an average of 42 years. This shows a tendency for disease onset in younger women. In our study group, this age range represented 90.82%.13,14
After conducting a histopathological examination of our patients, our review revealed that the most frequent carcinoma was the infiltrative duct carcinoma (91% of cases). Most examined cases (69.12%) were at advanced stages (III and IV). This data was supported by the First National Consensus on Breast Cancer Treatment. This report revealed that in Mexico, 50% of patients were diagnosed at these advances stages. This fact then renders chemotherapy indispensable in order to increase the life expectancy of patients.15,16
Chemotherapy systemic treatments mainly target cancer cells, preventing their growth and multiplication. Nevertheless, they also act upon rapid-dividing body cells, such as bone marrow cells, digestive tract cells, reproductive system cells, hair follicles, as well as skin cells.17
Our findings confirm studies reporting chemotherapy schemes, including adriamycin, cyclophosphamide and 5-fluorouacil as the most effective schemes for breast cancer treatment. Due to their moderate cost, these are the most commonly used drugs in thirdworld countries.11,15
Data found in scientific literature confirm the fact that stomatological manifestations are secondary reactions elicited by chemotherapy. In the present study 100% of all patients received poly-chemotherapy, or at least, some drug reported as associated to stomatological manifestations.18
Our patients received the following schemes: CMF,FAC, ACII, 5FU, 5FU + MTX, PL + NAVELVINE and 5FU + A)
Our results confirmed those obtained by previous studies, which mention mucositis as a common and potentially serious manifestation caused by chemotherapy.
Out of 122 patients, 91 presented isolated mucositis, or mucositis associated to other manifestations. This represented 54% of our population according to the Oral Mucositis Assessment Scale (OMAS), developed in 1996 by a team of oncology specialized physicians and experts in pain assessment and statistics.
Mucositis can be classified into the following degrees:
a) Grade 0. No affected sites.
b) Grade 1. Sore mouth and erythema.
c) Grade 2. Presence of erythema and ulcers; nevertheless, the patient can ingest solid foods.
d) Grade 3. Sore and ulcerated mouth; the patient can only achieve liquid diet.
e) Grade 4. Oral feeding is impossible.10,19-26
In concordance with other authors, we suggest that the chemotherapy drugs which mainly cause mucositis are: 5FU, methotrexate, doxorubicin, bleomycin, dactinomycin, daunomycin, cyclophosphamide, vincristin, cytosine-arabinoside, cisplatin and leocovorin. 10,23,26,27
Stomatitis was the manifestationwhich appeared in second place, since it appeared either by itself or associated with other manifestations in 33 out of the122 patients (26.95%).
Dreizen and his team indicated that methotrexate (folic acid antagonist) was one of the main causing agents of this manifestation, which is due to a folic acid deficiency that can interfere with epithelial cells production and maturation, thus eliciting atrophy or extinction of lingual papillae as well as ulcer formation, which in turn causes nutritional deficiency in the patient due to the pain these ulcers elicit. 19,27,36-39
Other authors have shown that 5FU (furacil metabolic antagonist) is another-stomatitis causing agent, since it produces thymine deficiency, which in turn interferes with nucleic acid synthesis and reflects into a rapid cellular division elicited by a cellular death and growth disequilibrium, provoking then ulceration and epithelial descamation.19,26,28. Based on the present study, we might also suggest the fact that gingival haemorrhage is another constant manifestation in these patients. It was observed in nine patients, although it was always associated with some other manifestation. Its main cause was vincristin cytosine-arabinside and prednisone. It is generated by drug-induced thrombocytopenia and by disseminated intravascular coagulation, especially in patients exhibiting platelet levels of 20,000 to 50,000 per mm3.
These drugs alter blood cell maturation and synthesis causing leukopenia, thrombocytopenia and anemia, depriving the immune system of protection. Myelosuppression can precipitate spontaneous periodontal bleeding and exacerbate chronic periodontal disease, as well as pulp problems.19,29,30
Xerosthomia was another commonly found manifestation in our patients. It was found in 27 out of the 122 patients (22.05%). This proportion was concurrent with data previously found in scientific literature.
Xerostomia appears as a mucositis aggregated or secondary effect. From the second day onwards, there is qualitative and quantitative reduction of salivary flow. Acini atrophy, cell necrosis, degeneration and salivary gland fibrosis are also present. All these factors elicit decrease29 of salivary flow and its pH, as well as saliva's buffering capacity. All the aforementioned factors accelerate periodontal deterioration and enhance the onset of dental caries.
This decrease of salivary proteins, minerals and enzymes directly affects tooth remineralization. Other affected functions are the patient's diction, mastication an deglutition. 14,30-35
There are reports indicating that cyclophosphamide, doxorubicin and vincristin are causing agents for viral infections. Our study seems to concur with this assertion: 8 patients (7.32% of the sample) presented viral infections during treatment, in all cases associated to other manifestations.
According to William Carl and his group28 this manifestation takes place because drugs interfere with the growth and maturation of oral mucosa cells, by reducing the tissue that functions as a barrier for this type of infections. Dreizen and his team19 supported this same theory. They further suggested that, in addition to viral infections, fungal infections might occur. Nevertheless, in the present study, only two patients were found to exhibit fungal infection; this represented a minimal percentage (1.63%). These infections were caused by oral flora and saliva alterations, and were mainly triggered by the use of prednisone, vincristin, doxorubicin and methotrexate. 10,21,22
Some studies mention the presence of sialorrhea as another frequent manifestation. Nevertheless, in the present study we only found this condition in one patient (0.81%). Authors report that sialorrhea has its onset before xerostomia, which then would explainthat it is common to report the latter and not suitably assess the presence of sialorrhea in these patients.29
The present study on stomatoxicitity manifestations induced by chemotherapy-treated breast cancer clearly establishes the need to formalize simultaneous treatment between the oncologist and a specialized stomatologist.
Appropriate oral care is critical during chemotherapy treatments. Manifestations will thus be prevented or reduced, and the patient will be able to eat properly and, thus, not delay her chemotherapy treatment. Prevention of stomatological manifestations reported in the present study shows the importance of conducting a meticulous oral examination in patients who are chemotherapy candidates.
CONCLUSIONS
1. Chemotherapy-induced stomatological manifestations in breast cancer patients mainly appear when schemes based on 5FU, adriamycin and cyclophosphatase are used.
2. The most frequent stomatological manifestations in patients included in this study were: mucositis, stomatitis, xerostomia and viral and fungal infections.
3. Based on the results obtained in the present study, it can be suggested that all patients treated with chemotherapy must receive dental treatment before, during and after chemotherapy treatment. A radiographic study of the patient must be obtained so as to devise a treatment plan which must include preventive measures, within the scope of the programmed conventional treatments. Special attention must be given to population with the same characteristics as the one studied in this sample, since they are normally afflicted by periodontal problems. Special care must be given to plaque control as well as topical fluoride application.
PROPOSAL
Elimination of dental and periodontal disease (caries, dental plaque and dental calculi) is of paramount importance. Poor fillings must also be redressed before the patient initiates her chemotherapy treatment.
Due to the aforementioned reasons, it is paramount to emit early diagnosis and devise appropriate treatment for fungal and viral infections in order to avoid generalized infections in the patient.
We will additionally require a comprehensive study for this chemotherapy-treated population, as well as for others. This study will enable us to know alterations that might appear in the saliva's enzymatic quality, as well as alterations appearing on the enamel structure, which can affect the quality of life of a population which every day presents increasing survival rates to breast cancer.
REFERENCES
1. López-Ríos O, Tovar-Guzmán V, Lazcano-Ponce E, Hernández-Ávila M. La epidemia de cáncer de mama en México ¿Consecuencias de la transición demográfica? Salud Pública. 1997; 39: 259-265. [ Links ]
2. Boring C, Squires T, Tong T. Cancer around the world, 19861988. Cancer statistics. Cancer J. Clin. 1992; 42: 19-38. [ Links ]
3. Atlanta American Cancer Society. Cancer facts and figures. 1991: 9. [ Links ]
4. Dirección General de Estadística y Evaluación. Secretaría de Salud. Daños a la salud. Boletín de información estadística. 1992; 12. [ Links ]
5. Peterson D, Sonis S. Oral complications of cancer chemotherapy. The Hague: Ed. Martinus Nijhoff Publishers; 1983. pp. 11-12. [ Links ]
6. Wahlin YB, Matsson L. Oral mucosal lesions in patients with acute leukemia and related disorders during cytotoxic therapy. Scand J. Dent Res. 1988; 96 (2): 128-136. [ Links ]
7. Hou GL, Tsai CC. Oral manifestations of agranulocytosis associated with methimazole therapy. J Periodontol. 1998; 59 (4): 244-248. [ Links ]
8. Dreizen M. Oral complications of cancer therapies. Description and incidence of oral complication. NCI Monog. 1990; 9: 11-15. [ Links ]
9. Simard-Savoie S. Oral manifestations in the patient taking anti-neoplastic medication. The role of the dentist. J. Dent Que. 1989; 26: 51-53. [ Links ]
10. Carl W. Oral complications of local and systemic cancer treatment. Curr Opin Oncol. 1995; 7: 320-324. [ Links ]
11. De Vita SH, Helman SA, Rosenberg S. Principles and practice of oncology. 5th edition. Philadelphia: Lippincott Williams & Wilkins; 1997. pp. 2705-2725. [ Links ]
12. National Cancer Institute monographs. Consensus development conference on oral complications of cancer therapies: diagnosis, prevention and treatment. Bethesda: National Institutes of Health; 1990. [ Links ]
13. Hernández-Muñoz GA. Avances en mastología. 2a. ed. Caracas Venezuela: Editorial Cromotip; 1990. pp. 147-155. [ Links ]
14. Frykberg. Management of in situ and minimally invasive breast carcinoma. J. Surgery. 1994; 18: 45-57. [ Links ]
15. Fernández-Ci et al. Anomalías del desarrollo mamario. En: Patología Mamaria. Barcelona: Salvat Editores; 1982. pp. 475-477. [ Links ]
16. Cárdenas J. Primer Consenso Nacional sobre Tratamiento del Cáncer Mamario. Colima, México; Septiembre, 1994. Disponible en: http://www.consensocancermamario.com/documentos/Folleto-Consenso-Nacional-1-Revision.pdf [ Links ]
17. Simon AR, Roberts MW. Management of oral complications associated with cancer therapy in pediatric patients. Journal of Dentistry for Children. 1991; 58: 384-388. [ Links ]
18. Peterson D, Sonis S. Oral complications of cancer chemotherapy. The Hague: Martins Nijhoff Publishing; 1983. pp. 113-149. [ Links ]
19. Dreizen S. Oral complications of cancer therapies. Description and incidence of oral complications. NCI Monogr. 1990; 9: 11-15. [ Links ]
20. Gordon B, Spadinger A, Hodges E, Ruby E, Stanley R, Coccia P. Effect of granulocyte macrophage colony stimulating factor on oral mucositis after hematopoietic stem cell transplantation. Journal of Clinical Oncology. 1994; 12 (9): 1917-1922. [ Links ]
21. O'Sullivan E, Duggal MS, Bailey CC, Curzon MEJ. Changes in the oral microflora during cytotoxic chemotherapy in children being treated for acute leukemia. Oral Surgery, Oral Medicine, Oral Pathology. 1993; 76 (sppl. 2): 161-168. [ Links ]
22. Bunetel L, Martine B, Rennes F. Oral pathoses caused by candida albicans during chemotherapy update on development mechanisms. Oral Surgery, Oral Medicine, Oral Pathology. 1996; 82 (2): 161-165. [ Links ]
23. Chi KH, Chen CH, Chan WK, Chow KC, Chen SY, Yen SH et al. Effect of granulocyte-macrophage colony stimulating factor on oral mucositis in head and neck cancer patients after cisplatin, fluoracil and leucovorin chemotherapy. Journal of Clinical Oncology. 1995; 13 (supl. 10): 2620-2628. [ Links ]
24. Barash A, Peterson D, Tanzer J, D'ambrosio J, Nuk K, Schubet M et al. Helium-neon lasser affects on conditioning induced oral mucositis in bone marrow transplantation patients. Cancer. 1995; 76 (12): 2550-2556. [ Links ]
25. Epstein J, Frances LWW. The efficacy of oral sucralfate suspention in prevention of oral mucositis due to radiation therapy. Int J Radiat Oncol Biol Phys. 1994; 28 (3): 693-698. [ Links ]
26. Osaki T, Elisaku V, Kazunori Y, Josui H, Yetsuya Y. Prophyla-sis of oral mucositis associated with chemoradiotherapy of oral carcinoma by azelastine hydrochloride with other antioxidants. Head and neck. 1994; 16: 331-339. [ Links ]
27. Fidler P, Loprizi Ch, Fallon J, Leitch J, Lee J, Hayes D et al. Prospective evaluation of a chamomile mouthwash for prevention of 5-FU induced oral mucositis. Cancer. 1996; 77 (3): 522-525. [ Links ]
28. Grandisher W. Docetaxel como tratamiento nedyuvante en pacientes con estadio III de cáncer de mama. Oncology. 1997; 8: 15-18. [ Links ]
29. William CW Lawrence S. Management of oral mucositis during local radiation and systemic chemotherapy: A study of 98 patients. J. Prosthetic Dent. 1991; 66 (sppl 3): 361-369. [ Links ]
30. Rosenberg S. Atención bucal de los pacientes bajo quimioterapia. "Care of the mouth". Manual de Oncología. 1989. pp. 227-237. [ Links ]
31. Peterson D. Oral toxicity of chemotherapeutic agents. Semin Oncol. 1992; 19 (5): 478-491. [ Links ]
32. Overhoster CD, Peterson DE. Infection in acute nolynphocytic leukemia prevalence of acute exacervations. Arch Inter Med.1982; 142: 51-54. [ Links ]
33. Ramos de la RC. Manifestaciones orales en pacientes con carcinoma mamario bajo tratamiento de quimioterapia con esquemas a base de adriamicina [Tesis]. México: Hospital General de México; 1999. p. 20. [ Links ]
34. Díaz-Rubio E. Monografías clínicas en oncología: efectos secundarias de la quimioterapia antineoplásica. Vol. II. Barcelona: S. A. Ediciones; 1990. [ Links ]
35. Lockhart PB, Soriss T. Alterations in the oral mucosa caused by chemotherapeutic agents. J. Dental Surgery Oncology. 1981; 7: 15. [ Links ]
36. Lynch M et al. Medicina bucal de Burkcet. la. ed. México: Ed. Mc Graw-Hill Interamericana; 1996. [ Links ]
37. Scully C, Macfarlane TW. Orofacial manifestations of childhood malignancy:clinical and microbiological findings during remission. ASDJ Dent Child. 1983; 50: 121-125. [ Links ]
38. Williams MC, Martin MV. A longitudinal study of the effects on the oral mucosa of treatment for acute childhood leukemia. Inter J Paed Dent.1992; 2: 73-79. [ Links ]
39. Wimenga ANM, Vandergraff WTA, Spijkervet FLK, Times W, Tinner-Bosscha H, Slviter MJ et al. A new in vitro assay for cuan-titation of chemotherapy induced mucositis. British Journal of Cancer. 1997; 76: 1062-1066. [ Links ]
Note This article can be read in its full version in the following page: http://www.medigraphic.com/facultadodontologiaunam Mailing address:
Mailing address:
Enrique Echevarría y Pérez
E-mail: eechevarriayperez@hotmail.com











 texto en
texto en 


