1. INTRODUCTION
In 1856 W.H. Perkin discovered mauveine, the first synthetic organic dye (Pavlović, Racané, Čičak, & Tralić-Kulenović, 2009). In 1987 Zollinger reported that, over 7×105 tonnes of synthetic dyes are produced annually (Atkinson, 1952). Azo dyes are highly colored and have wide applications in leather industries (Jarad, 2012), textile dyeing (Raghavendra & Kumar, 2013) and agriculture (Hussain et al., 2016). They also found its applications in food stuff (Raj & Patel, 2015), cosmetics (Sanjay et al., 2012), paper printing (Shabir & Saeed, 2016), color photography & pharmaceutical (Sabnis & Rangnekar, 1989). They have been studied widely because of its excellent thermal stability (Rangnekar & Sonawane, 2000) and optical properties in recording medium (Christie, Standring, & Griffiths, 1988), toner, (Freeman et al., 1987) and printing (Ahmadi & Amani, 2012).
The azo compounds are applicable for biocidal treatment of textile materials (Hussain et al., 2016) because they exhibit biological activity (Hussain et al., 2016) (Ahmadi & Amani, 2012) (Loganathan, Sithick, Purushothaman, Silambarasan, & Jamal A, 2015). Azo compounds are well known for their medicinal importance (Raghavendra & Kumar, 2013) and are recognized for their applications as, antiseptics (Raj & Patel, 2015), antibacterial (Hallas & Towns, 1997) and antitumor (Rangnekar & Sonawane, 2000). They are involved in a many biological reactions such as inhibition of DNA, RNA (Rizk, Ibrahim, & El-Borai, 2015), protein synthesis and nitrogen fixation (Mahmoud, Mohamed, & Mohamedin, 2017). In recent years, azo metal dyes are used for DVD -R (digital versatile storage device), (Arun et al., 2009; Kurtoglu, 2009).
Azo dyes are synthesized in two steps i.e diazotization followed by coupling (Shabir & Saeed, 2016). About 60-70% of the synthetic dyes which are in industrial use are azo dyes and have high affinity for wool (Girish & Raksha, 2011), diversity in structure and easily availability of raw material (Arun et al., 2009; Crina & Ion, 2012; Hussain et al., 2016; Kurtoglu, 2009; Radulescu, Muntean, Todea, Verdes, & Andelescu, 2015; Shabir & Saeed, 2016). Due to its ecologically friendly effects, there has been a tremendous increase in the synthesis of large number of azo metal dyes. A metal complex dye shows great affinity towards proteins. In these dyes one or two dye molecules are coordinated with a metal ion (Szymczyk, El-Shafei, & Freeman, 2007). The dye molecule is typically a mono azo structure containing additional groups such as hydroxyl, carbonyls or amino which are capable forming a strong coordination complex with transition elements such as Cr, Ni, Co (Fazeli, Sohrabi, & Tehrani-Bagha, 2012). Metal complex dyes belong to numerous classes of dyes. For example, they are found among direct, acid, and reactive dyes (Chao & Yang, 1995; Ibrahim, El-Zairy, & Abo-Shosha, 1994; Venkatesh, Venkatesh, & Quaff, 2017). When applied in dyeing processes, metal complex dyes are used in pH condition that is regulated by user class. The pH level for wool typically ranges from strongly acidic ranging from 1.8 to 4 for 1:1 metal complex dye, moderately acidic neutral ranging from 4-7 for 1:2 metal complexes (Anitha, Reddy, & Vittala Rao, 2011; Hussain et al., 2016).
Textile is the largest manufacturing sector of Pakistan. It contributes 8.5% to GDP and employees 38% of the total manufacturing labor force which amounts to 15 million people. Pakistan has emerged as one of the major cotton textile product suppliers in the world market. Import of various dyes and pigments increased from 12.9 billion in 2009-2010 to Rs13.4 billion in 2012 (Hussain et al., 2016; Hussain et al., 2017).
The main objectives of this research study was to synthesize and characterize the acid dye and its metal complexes with transition elements such as copper, nickel, cobalt, Iron and chromium.
2. EXPERIMENTAL
2.1 MATERIALS AND METHODS
All commercial products were purchased from Sigma-Aldrich. Solvents used were of analytical grade and, when necessary, were purified and dried by the standard methods. Melting points were determined in open capillary tubes on a Stuart melting point apparatus. The IR spectra were run on the single beam Nicolet IR 100 (Fourier-Transform); while UV of all the samples were run in water using UV-Genesys spectrophotometer. Mass spectral data were obtained from Waters GCT premier spectrometer. The 1HNMR and 13C-NMR spectra were recorded in D2O using NMR Bruker DPX 300 spectrophotometer operating at 300 MHz. TMS was used as internal standard with the deuterium signal of the solvent as the lock and chemical shifts δ were recorded in ppm. The elemental analysis (C, H, N, S) of the compounds were performed using Flash EA 1112 elemental analyzer while the pH was monitored using Portable pH Meter Model PHB4. Compounds were routinely checked by TLC on silica gel G plates and paper chromatography using different eluting solvents. All crude products were isolated as solids and purified by a combination of column chromatography and recrystalli-zation. Fastness to light was assessed in accordance with BS 1006-1978. Rubbing fastness was checked with an Atlas Crock meter in accordance with AATCC TM 8-1961 and the wash fastness was determined according to ISO: 765-1979.
2.2 GENERAL PROCEDURE FOR THE SYNTHESIS OF 1-AMINO-2 NAPH THOL-6-NITRONAPHTHALENE-4-SULPHONIC ACID BASED ACID DYES
Acid dye based upon 1-Amino-2-naphthol-6-nitronaphthalene-4-sulphonic acid were accomplished in two step procedure which is as follows;
A-Diazotization and Coupling with Resorcinol
To the well stirred ice jacketed aqueous solution of 1-Amino-2-naphthol-6-nitronaphthalene-4-sulphonic acid (2.84g, 0.01mol) at 0-5 ̊C, was added 0.7g sodium nitrite and 3.5 ml conc. HCl. The reaction mixture was vigorously stirred for 1h at the above temperature to achieve the desired diazonium salt.
Freshly prepared diazonium salt of 1-Amino-2-naphthol-6-nitronaphthalene-4-sulphonic acid was coupled with resorcinol (1.10g, 0.01mol) at 15-20 oC. The coupling was facilitated using sodium carbonate as acid binding agent. The reaction mixture was stirred for 4hrs under the above conditions until completion of coupling was observed from paper chromatogram. pH of reaction mixture was reduced up to 4.5 with HCl which caused the precipitation of dye. Dye was filtered and dried in oven at 70-75o C till constant weight was obtained with percentage yield of 85%.
B. Metallization of Acid Dye
Metallization of the above synthesized acid dye 4 was achieved treating the dye (0.40g, 0.001mol) with FeSO4.7H2O 5a (0.13g, 0.0005mol) at pH 6.5. Then the reaction mixture was heated at 100oC. Stirring and heating at this temperature was continued for further 1.0 hour till the metallization was completed as shown by the comparative TLC. The dye was cooled to room temperature; its pH was reduced to 1.0 with conc. HCl. The dye was salted out with sodium chloride which was subsequently filtered and dried in oven at 80 º̊C till constant weight.
Similarly Nickel (II), Copper (II), Cobalt (II) and Chromium (III) complexes of dye 4 were prepared by reacting them respective metal salts (5b-e) at temperature 55-70 º̊C with mole ratio 1:1. In this way metal complex dyes 6a-g were synthesized from ligand dye 4. Physical and spectroscopic data of ligand acid dye and metal complex dyes are as under
4-((2, 4-dihydroxyphenyl)diazenyl)-3-hydroxy-7-nitronaphthalene-1-sulfonic acid (4)
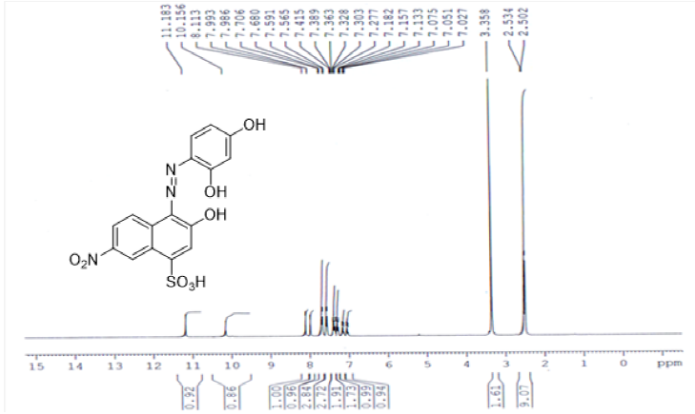
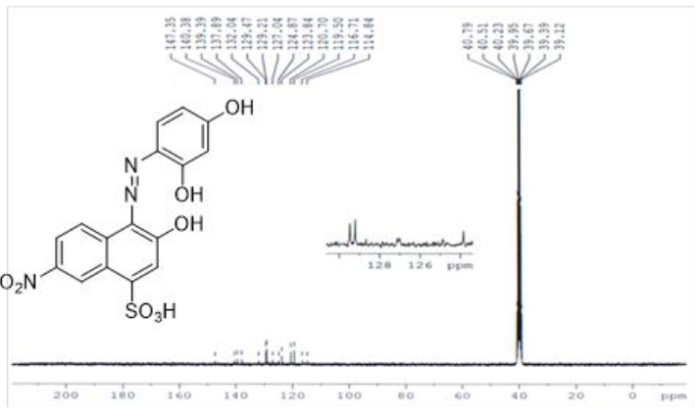
Reddish brown solid, (70%) λmax (nm):435; FT-IR (KBr, cm-1) νmax: 3478 br (OH), 3064 (C=C-H), 1643, 1590, 1518 (C=C aromatic), 1447 (N=N), 1220 (S=O), 1060 (C-O), 770 (Ar-H). 1H-NMR (300 MHz, DMSO-d6) δ (ppm): 7.02-7.41 (10H, m), 7.57 (2H, d, J=7.8Hz), 7.69 (2H, d, J=7.8Hz), 7.99 (2H, s), 8.11 (1H, br, SO3H ), 10.15 (1H, br, OH), 10.18 (1H, br, O-H). 13C-NMR (75 MHz, DMSO-d6) δ (ppm): 147.35, 140.38, 139.39, 137.89, 132.04, 129.47, 129.21, 127.98, 127.04, 124.87, 123.84, 120.70, 119.50, 116.71 and 114.84. Anal. Calcd. For C 16 H 11 N 3 O 8 S, C: 47.41; H, 2.74; N, 10.37; O, 31.58; S, 7.91; Found: C, 46.90; H, 2.65; N, 10.05; S, 8.10.
[C 32 H 18 FeN 6 O 16 S 2]SO 4 (6a)
Light greenish, λmax (nm): 485; FTIR (KBr, cm-1) νmax: 3021 (C=C-H), 2843 (C-H, str), 1590, 1532, 1492 (C=C aromatic), 1440 (N=N), 1374 (C-H, bend), 1225 (S=O), 1143 (C-O), 906 (C=C-H, bend), 590 (Fe-N, str). Anal. Calcd. [C32H18FeN6O16S2]SO4;C, 40.10; H, 1.89; N, 8.77; S, 10.03 Found:C, 39.90; H, 1.54; N, 8.32; S, 10.54.
[C32H18N6NiO16S2]SO4 (6b)
Greenish brown, λmax (nm): 510; FTIR (KBr, cm-1) νmax: 3045(C=C-H), 2928 (CH2), 2873 (C-H, str), 1620, 1584, 1541 (C=C aromatic), 1443 (N=N), 1365 (C-H, bend), 1232 (S=O), 1143 (C-O), 824 (C=C-H, bend), 745 (C=C-H, bend), 584 (Ni-O, str). Anal. Calcd. For [C 32 H 18 N 6 NiO 16 S 2]SO 4:C, 39.98; H, 1.89; N, 8.74; S, 10.00, Found: C, 39.31; H, 1.34; N, 8.20; S, 10.80.
[C 32 H 18 CuN 6 O 16 S 2]SO 4 (6c)
Greenish, λmax (nm): 500; FTIR (KBr, cm-1) νmax: 3048 (C=C-H), 1610, 1575 (C=C aromatic), 1436 (N=N), 1341 (C-H), 1222 (S=O), 1153 (C-O), 884, 725 (C=C-H, bend), 593 (Cu-N, str). Anal. Calcd. For [C 32 H 18 CuN 6 O 16 S 2]SO 4:C, 39.78; H, 1.88; N, 8.70; S, 9.95, Found: C, 39.14; H, 1.40; N, 8.10; S, 10.10.
[C32H18CoN8O22S2](NO3)2 (6d)
Olive brown, λmax (nm): 507; FTIR (KBr, cm-1) νmax:3053 (C=C-H), 1630, 1556, 1512 (C=C aromatic), 1440 (N=N), 1335 (C-H), 1218 (S=O), 1142 (C-O), 880, 715 (C=C-H, bend), 560 (Co-N, str). Anal. Calcd. For[C 32 H 18 CoN 8 O 22 S 2](NO 3 ) 2: C, 38.84; H, 1.83; N, 11.32; S, 6.48; Found:C, 38.10; H, 1.40; N, 11.04; S, 6.79.
[C 32 H 18 CrN 6 O 16 S 2](CH 3 COO)3 (6e)
Reddish brown, λmax (nm): 530; FTIR (KBr, cm-1) νmax: 3049 (C=C-H), 1610, 1578 (C=C aromatic), 1443 (N=N), 1339 (C-H), 1264 (S=O), 1160 (C-O), 865,730, 670 (C=C-H, bend), 594 (Cr-O, str). Anal. Calcd. For [C 32 H 18 CrN 6 O 16 S 2](CH 3 COO)3: C, 44.07; H, 2.63; N, 8.11; S, 6.19; Found:C, 43.78; H, 2.24; N, 7.75; S, 6.45.
2.3 DYEING METHOD
Dye solution (10 ml, 0.2 g dye on crust weight) was taken in a dyeing drum. The pH of the dye-bath was adjusted to 5.5 by adding acetic acid solution (1.0 ml. 10% w/v) solution. The total volume of the dye-bath was adjusted to 100 ml by adding required amount of water. The leather piece (crust leather) (10g) was introduced into the dye-bath with stirring. The contents of the dye-bath were stirred for 1h at room temperature and then temperature was gradually raised to 45-50oC over period of of 30 minutes and maintained for one hour. The dye-bath was kept rotating during the process of dyeing and was added formic acid 1.0 ml and pH adjusted to 2.0 and run the drum for half hour more till dye fixed on leather. After this, the dye liquor was taken in 250 ml volumetric flask. The fabric was washed with cold water and the combined solution of dye liquor and washings was then further diluted to 250 ml with water. From this diluted solution, 1ml was further diluted to 50 ml with water and the absorbance of this solution was measured to find out the exhaustion of dye on leather sheet. The dyed leather piece was dried and mounted on shade card. A weighed amount of leather piece was stirred in boiling acidified pyridine which dissolves the unfixed dye from fabric and from the absorbance of this solution percentage fixation was checked.
3. RESULTS AND DISCUSSION
3.1 SYNTHETIC PATHWAY TO ACID DYE AND METAL COMPLEXES
The reaction sequence employed for the synthesis of the targeted acid dye 4 and its metal complexes (6a-e) is illustrated in scheme 1. Accordingly, 1-Amino-2-naphthol-6-nitronaphthalene-4-sulphonic acid (1) was diazotized by treating with sodium nitrite in the presence of HCl at low temperature 0-5oC. Temperature and pH conditions were maintained to obtain dyes in maximum yields with high purity. The reaction of 1-Amino-2-naphthol-6-nitronaphthalene-4-sulphonic acid (1) with resorcinol (3) in alkaline medium resulted in the coupling at ortho to the hydroxyl group of resorcinol. The dye4 were precipitated by decreasing the pH of reaction mixture with HCl.Dyes were dried and purified by recrystallization in ethanol.
Metallization of above synthesized acid dyes 4 was done by treating the alkaline solution of dye with FeSO4.7H2O, NiSO4.6H2O, CuSO4.5H2O, Co(NO3)2.6H2O and Cr(OOCCH3)3with continuous stirring and heating the reaction mixture at 55-70oC for 4-5 h until the confirmation about completion of reaction was observed by taking the TLC of reaction mixture in 9:1 chloroform and methanol. Dyes were precipitated with addition of HCl, filtered and dried in oven at 70 oC. Dyes were again recrystallized from ethanol, dried, weighed and determined the percentage yield.
3.2 SPECTRAL PROPERTIES OF DYES
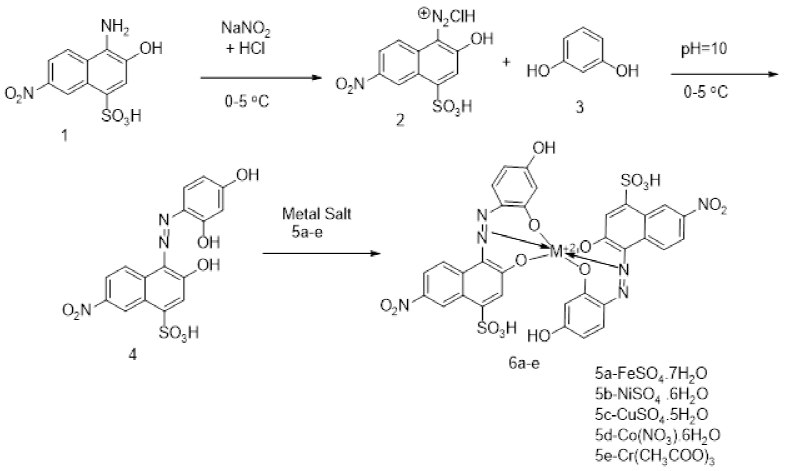
The absorption maxima (λmax) of the acid dye 4 and its metal complexes with Fe (II), Ni(II), Cu(II), Co(II) and Cr (II) 6a-e were recorded in ethanol and are shown in Fig. 1. These dyes showed two absorption maxima, one in the ultraviolet region in the range 300-350 nm and other in the visible region due to π-π* transition of azo linkage N=N of dyes. There is one ligand dye (dye 4) which is complexed with different metals. All the metal complexes exhibited bathochromic shifts as compared to the ligand dye which had λmax 435nm. Iron (II) complex (6a) produced light greenish color with λmax at 485 nm and there is shift of 50 nm in the wavelength of maximum absorption from their original ligand dyes (figure 1). Chromium (III, 6e) complex of dye 4 presented the bathochromic shift of 95nm which is the largest shift toward longer wavelength in this series of dyes 6a-e. Change in colour of dyes after metal complex formation depends upon electron accepting or donating ability of d-orbitals either they are in stable or unstable state of electronic configuration and also on the contribution of change in structure brought about after complexation, either it is large or small. This largest shift in Chromium complexes is attributed to five unpaired electrons in 3d-orbitals. Being highly paramagnetic complex, there is small energy gap between t2g and eg orbitals. Similarly other metals like nickel, cobalt and copper expressed different colors after complex formation.
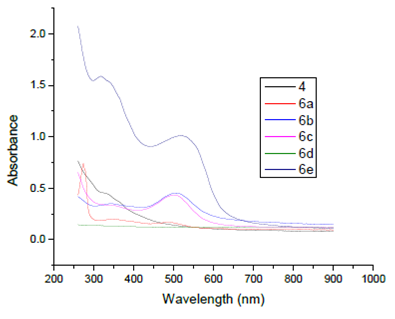
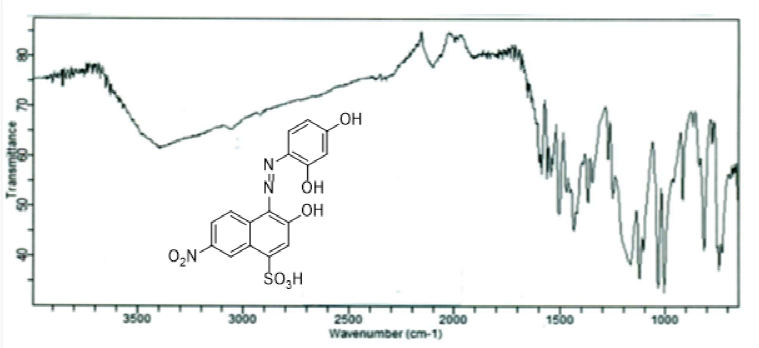
Infrared spectra of the synthesized acid dye 4 and its metal complexes exhibited absorption peaks due to O-H, Ar-H, C-H, C=C, N=N, SO3H, C-O and O-M stretching and bending vibrations at 3478, 3021-3053, 1610-1621, 1532-1578,1436-1443, 1218-1264, 1142-1160, 825-884 and 770-772 cm-1 as depicted from their FTIR spectra in Fig 2 and 3. Specifically speaking, using FTIR spectrum of dye 4, a broad band is observed in the range 3478 cm-1 which was due to H-bonding of O-H groups in close proximity to each other in dye molecules. Aromatic (benzene) rings are evidenced by presence of peaks in the range 3064 cm-1 due to C-H stretching of unsaturated carbon atoms which are further confirmed by their peaks at 1643, 1590 and 1518 cm-1. The absorption bands at 1447 cm-1 depicted the presence of N=N stretching vibrations of dye. Synthesis of dyes 6a-e has also been confirmed by their FTIR spectra. The metal complexes have been inveterated by the presence of peaks at low frequency region at 560-594cm-1 because of large masses of metal atoms and these peaks are absent in the ligand FTIR spectrum (Figure 2 and 3).
The 1H-NMR spectrum of acid dye4 showed signals down field at due to two OH groups present in the coupling and diazo components of dye at 10.15 and 10.18ppmand SO3H group showed signal at 8.113ppm. A singlet peak at 7.993 ppm is due to a single non coupled proton attached to naphthalene ring. Similarly a singlet peak at 7.986 ppm is due to one proton sandwiched between two hydroxyl groups at resorcinol coupler. Symmetrical doublet peaks at 7.40 and 7.57ppmwith same coupling constant is due to two mutually coupled protons attached to resorcinol ring. Similarly doublet signal at 7.31 and 7.69 ppm is due to mutually coupled set of protons attached naphthalene ring (Figure 4). A singlet signal at 7.36ppm because of non-coupled proton attached at naphthalene ring. In case of 13C-NMR of all dyes showed aromatic peaks in range 114.84-147.35 ppm are present (Figure 5). In this way dye was verified for its structure from respective NMR spectra.
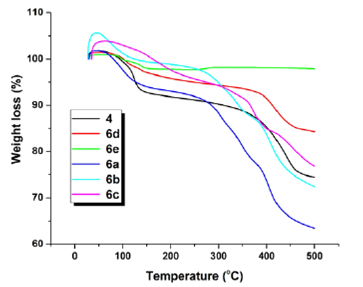
3.3 THERMOGRAVIMETRIC ANALYSIS OF ACID DYE AND ITS METAL COMPLEXES
Thermal gravimetric analysis plays an important role in the study of structure, stability, and its applicability in the field of dyeing, photo typing, printing, lasers and optical devices. The result obtained from thermal analysis can be used to describe its thermal stability and to determine the temperature range in which they can be used without changes in its properties, colour and composition. The TGA curve was obtained by using NETZSCH analyzer within temperature range of 20-500 oC in flowing Argon atmosphere at a flow rate of 50 using Al2 O3 as reference material. The TGA curve (Figure 6) display three stages of mass loss within temperature range of 130-340 oC. The weight loss occur at the first stage 2% for each of Co and Fe complex, 9% for original ligand 10 % for Ni complex, and almost 7% for Cr complex. In the second stage the weight loss is only 6% for Co complex, 11% for Fe complex 13% for Ni 12 % from original ligand and almost 12% for Cr complex (Table 1). The 3rd phase indicate that Fe complex decompose at 400 oC, Cr complex at 380oC,original ligand at 360 oC, Ni complex at 350 oC while Co complex shows higher temperature range.
3.4 DYEING PROPERTIES OF DYES
Acid dye 4 and its metal complexes 6a-e were applied at 2 % depth on course leather according to the standard procedure
3.4.1 Exhaustion and fixation study:
Exhaustion and fixation values are determined by the application of dyes at 2 and 5% dyeing on leather fabric. Exhaustion and fixation values are shown in Table 2.
Table 2 Exhaustion and fixation data of the Acid dye 4 and their metal complexes (6a-e).
| Dye | Shade on Leather | λ max (nm) in ethanol | Exhaustion &(oC) | Fixation %(oC) |
|---|---|---|---|---|
| 4 | Reddish brown | 435 | 75.75 | 87.43 |
| 6a | Light greenish | 485 | 81.43 | 91.87 |
| 6b | Greenish brown | 510 | 82.24 | 89.33 |
| 6c | Greenish | 500 | 86.27 | 95.46 |
| 6d | Olive brown | 507 | 79.37 | 85.54 |
| 6e | Reddish | 530 | 82.17 | 88.14 |
An exhaustion deals with the amount of dye transferred from dye bath to fabric and is articulated as percentage of the quantity of dye originally placed in the dye bath. Fixation of a dye is concerned with the amount of dye fixed with the fabric. Driving force for exhaustion is concentration of dye in two phases and for fixation is the physical as well as chemical interaction with fabric. In order to acquire high degree of exhaustion, assisting salt Na2SO4 was added in the dye bath to perk up exhaustion by increasing the electrostatic interactions of dyes with fabric.
The percentage exhaustion and fixation of 2 and 5% dyeing on cotton ranges from 80-90% and 94-97% respectively as it is represented in the Table 2. All the dyes have good exhaustion and fixation values which is expected due to the rapid diffusion of the dye molecule within the fabric under dyeing condition and physical as well as chemical interactions of polar groups present in diazo and coupling components. From the applications of dyes on leather it appears that metal complexes of the dyes have high exhaustion and fixation as compared to un-metallized dyes. Chromium (III) complexes of dyes have high fixation as compared with iron (II) and copper (II) complexes.
3.4.2 Fastness properties
Fastness properties of dyes were assessed after application of 2 and 5 % dye with respect to leather fabric as represented in Table 3. These were light fastness, wash fastness and rubbing fastness which provided the clear picture regarding quality of dye.
Table 3 Fastness properties data of the Acid dyes 4 and their metal complexes (6a-e).
| Dye | Wash Fastness | Light Fastness | Rubbing Fastness | Perspiration Fastness |
|---|---|---|---|---|
| 4 | 2-3 | 2-3 | 3-4 | 3-4 |
| 6a | 2-3 | 2-3 | 4-5 | 3-4 |
| 6b | 2-3 | 2-3 | 4-5 | 3-4 |
| 6c | 2-3 | 2-3 | 4-5 | 3-4 |
| 6d | 2-3 | 2-3 | 4-5 | 3-4 |
| 6e | 3-4 | 2-3 | 4-5 | 3-4 |
Light fastness is the property to which a dye resists fading due to light exposure. Different dyes have different degrees of resistance to fading by light. Light fastness of all dyes (4 and 6a-e) was high in range 2-3. These dyes have very little susceptibility to phtochromisim, due to involvement of electrons in complex formations. Wash fastness is the resistance offered by dyed fabric to retain color when washed by soaps and detergents. Wash fastness of all dyes was in range 2-3 except to those of chromium complex in which it was 3-4.
Rubbing fastness is the most important test which is always required for every colored fabric either it is printed or dyed. Rubbing fastness was planned to determine the degree of color which may transfer from the surface of a colored fabric to a particular test cloth for abrasion. Rubbing fastness of dye 4 and its metal complexes was very high 4-5. Rubbing fastness gives clue about other improved properties like wash fastness, substantively and sturdiness in use. It is obvious from rubbing fastness value that all these dyes have high fixation on leather fabric.
Perspiration fastness deals with ability of dyed fabric not to satin and not to fade when dyed fabric perspired. People’s sweat consists of complicated composition and its main composition is salt. Sweat is divided into acidity and basicity due to difference of people.
Perspiration fastness was found in the range 3-4 for ligand dye 4 and its metal complexes 6a-e. Dyed leather swatches are shown in Figure 7.
3.4.3 Antifungal activity
The synthesized azo ligand and its metal complexes were screened for possible antifungal activity in Sabouraud Dextrose Agar and Nutrient Agar against 72 hour old culture of fungi strain Candida albican using fluconazole as standard. Original azo dye has no antifungal inhibition against Candida albican (Table 4 and 5). Metal complex of copper, iron and chromium are inefficient against the antifungal strain. However, cobalt and nickel complexes show good inhibition effect against Candida albican. Nickel complex shows 65.2 % inhibition in 1000 μǥ/ml zone. Cobalt complex shows 43.5% in 500 μǥ/ml and 108.7% in 1000μǥ/ml respectively. Thus Nickel and cobalt complexes carry benefits of antifungal activity which can be beneficial in tanning industry to avoid fungal degradation of lather and textile.
Table 4 Antifungal Properties of Acid dye 4 and their metal complexes (6a-e).
| Concentration of samples (μg/ml) | Inhibition zone of Dyes (mm) | Standard | |||||
|---|---|---|---|---|---|---|---|
| Dye 4 | Dye 6a | Dye6b | Dye 6c | Dye 6d | Dye 6e | Fluconazole | |
| 200 | 0 | 0 | 0 | 0 | 0 | 0 | 100% |
| 300 | 0 | 0 | 0 | 0 | 0 | 0 | |
| 500 | 0 | 0 | 0 | 0 | 0 | 0 | |
| 1000 | 0 | 0 | 15 | 25 | 0 | ||
4. CONCLUSIONS
New acid dyes and its metal complexes have been synthesized and characterized well, and they expressed promising results in their application properties on leather in terms of their light fastness, wash fastness and rubbing fastness. High thermal stability was exhibited by acid decomposed at 350 oC. Cobalt and Nickel complex dyes showed valuable antifungal activity.











 nueva página del texto (beta)
nueva página del texto (beta)