Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Revista mexicana de ingeniería química
versão impressa ISSN 1665-2738
Rev. Mex. Ing. Quím vol.10 no.2 Ciudad de México Ago. 2011
Termodinámica
Purification and characterization of a thermostable alkaline protease produced by Yarrowia lipolytica
Purificación y caracterización de una proteasa termoestable alcalina producida por Yarrowia lipolytica
R. Hernández–Martínez1, A. Sancho–Solano2, O. Loera–Corral1, A. Rojo–Domínguez3, C. Regalado–González4, S. Huerta–Ochoa1 and L.A. Prado–Barragán1*
1 Departamento de Biotecnología, División de Ciencias Biológicas y de la Salud. Universidad Autónoma Metropolitana. Unidad Iztapalapa, 09340, México, D.F. *Corresponding author. E–mail: lapb@xanum.uam.mx Fax: 5804–4999
2 Centro Nacional de Innovaciones Biotecnológicas (CENIBiot), laboratorio de Bioprocesos. San José, Costa Rica 1174–1200.
3 Departamento de Ciencias Naturales. Universidad Autónoma Metropolitana. Unidad Cuajimalpa, 11850, México D. F.
4 Facultad de Química, Universidad Autónoma de Querétaro, Centro Universitario s/n. Querétaro, 76010 Qro. México.
Received 15 of November 2010.
Accepted 30 of January 2011.
Abstract
Purification of the extracellular prote ase produced by Yarrowia lipolytica was realized in fo ur steps: ammonium sulfate precipitation, anionic exchange (2X) and gel filtration. The enzyme showed m olecular weight of 61.5 kDa (SDS–PAGE) and optimum activity at 52.4°C at pH 10–11. The thermal stability was modified in presence of Ca2+ (10 mM) providing an in crease of 73, 6 and 11% at 40, 50 and 60°C respectively. The thermodynamic parameters (enthalpy and entropy) indicate that the stability of the enzyme is not provided by non–covalent linkages. Furthermore the ion Ca2+ is important for thermodynamic stabilization of the enzymatic structure. The proteolytic activity was inhibited by PMSF; suggesting that the enzyme can be classify in the serine protease family. The results of thermodynamic stability allow classifying the protease studied as thermostable. The importance of the Ca2+ on the thermostability was corroborated; this is the first report on thermal stability and thermodynamic properties of proteases produced by Y. lipolytica.
Keywords: Yarrowia lipolytica, protease, thermostable, thermodynamic stability.
Resumen
La purificación de la proteasa extracelular producida por Yarrowia lipolytica se realizó en cuatro etapas: precipitación con sulfato de amonio, intercambio amónico (2X) y filtración en gel. La enzima presentó un peso molecular de 61.5 kDa (SDS–PAGE) y actividad óptima a 52.4°C en el rango de pH 10–11. La estabilidad térmica se ve modificada en presencia de Ca 2+ (10 mM) observándose un aumento de 73, 6 y 11% a 40, 50 y 60°C respectivamente. Los parámetros termodinámicos (entropía y entalpia) indican que la estabilidad de la enzima no es debida a enlaces no covalentes. El ion Ca2+ estabiliza termodinámicamente la estructura enzimática. La actividad proteolítica fue inhibida por PMSF, sugiriendo que la enzima puede clasificarse como serán proteasa. Este es el primer trabajo que reporta estudios de estabilidad térmica y propiedades termodinámicas de proteasas producidas por Y. lipolytica.
Palabras clave: Yarrowia lipolytica, proteasa, termoestable, estabilidad termodinámica.
1 Introduction
Proteases are enzymes widely distributed in nature; they can be extracted from plants and animals however microorganisms represent an interesting source of proteases with clear industrial projection due to the rapid and profitable growth and high enzyme titers produced. Furthermore, the relatively easy genetic modification allows building of high productive strains (Peñalva and Gerbert, 2002). Yeast extracellular proteases are of particular interest for their high commercial applications and potential use in expression systems of heterologous proteins (Glover et al., 1997).
Among the yeast reported for protease production are included Candida lipolytica, Aureobasidium pullulans and Yarrowia lipolytica. In particular, higher titers of extracellular protease have been attributed to Y. lipolytica under optimized conditions (Mazdak et al., 2004). Y. lipolytica is an obligate aerobic dimorphic ascomycete considered as non–pathogen strain and it's approved as GRAS (Generally Recognized as Safe). Y. lipolytica secrets a large variety of metabolites such organic acids, lipases, phosphatases and proteases (Aloulou et al., 2007; González–López et al., 2006). Mazdak et al. (2004) reported that the XPR2 gene from Y. lipolytica encodes the inducible alkaline extracellular protease (AEP), a major protein secreted by this strain. González–López et al. (2006) reported that the type of protease secreted depends on the culture initial pH; then at initial acid pH, an acidic protease is produced (AXP), whereas at neutral or alkaline initial pH, production of an AEP is induced. Poza et al. (2007) characterized one AEP serine proteases from the subtilisin subfamily with molecular weight of 30 kDa and optimum activity pH of 9–10 when they used a neutral initial pH.
Protein inestability at high temperature is one of the main setbacks in extending the enzymes industrial application, and then protein thermostability has been vigorously studied in the biophysical and biotechnological areas. With the understanding of the factors that regulate protein thermostability, the enzymes thermostability could be modeled by rational design (Pack and Yoo, 2003). The objective of this work was to characterize the thermostable extracellular protease produced by Y. lipolytica in solid state culture (SSC) in order to provide information on the responsible factors for enzyme thermostability.
2 Materials and methods
2.1 Microorganism and inoculum
Y. lipolytica from coconut copra was provided by the Laboratory of Bioprocess from the University of Guadalajara (UdeG) Mexico. The strain was maintained on potato dextrose agar (PDA) at 4°C. The yeast was propagated in conical flasks (125 mL) with 30 mL of PDA at 45°C for 7 days. The cells were harvested with 25 mL of tween–80 (0.01%) and used as inoculum.
2.2 Protease production by solid state culture
The protease production was performed in tubular columns (25 × 200 mm) as previously described by Saucedo et al. (1992). Polyurethane foam and fish flour were used as inert support and substrate respectively at 70/30 (w/w) ratio. The culture medium was (g/L): KH2PO4, 1; MgSO4 7H2O, 0.5; KCl, 0.5 in phosphate buffer 0.1 M, pH 7 (Sandhya et al., 2005). The inoculum was adjusted at 2×107 cells per g of dry matter and moister content at 50%. The culture was maintained at 45°C and aeration rate of 40 mL/min for 72 h.
2.2.1 Enzyme extraction
The enzymatic extract was obtained by adding 10 mL of TRIS–HCl buffer (20 mM, pH 8) per gram of fermented matter; the slurry was gently stirred for 30 min at 4°C. The suspension was filtered and centrifuged at 15 000g for 15 min at 4 °C (Beckman Coulter 25R) to remove cells and other debris. The supernatant was stored at 4°C and used as protease extract.
2.3 Protein assay
Protein content was measured following the method reported by Bradford using bovine serum albumin (BSA) as standard (Bradford, 1976).
2.4 Assay of protease activity
Protease activity was determined by the modified method of Johnvesly and Naik (2001). The enzyme (50 μL) was added to 950 μL of 1% of casein Hammerstein (w/v) (dissolved in 50 mM phosphate buffer pH 7) pre–incubated at 50°C for 5 min. The enzymatic reaction was performed during 15 min and stopped by addition of 1.5 ml of 5% (w/v) trichloroacetic acid (TCA). The mixture was centrifuged at 15,000g at 4°C for 15 min and filtered through Whatman No. 1. The soluble peptides in TCA were estimated spectrophotometrically at 280 nm. A standard curve of tyrosine was used as reference. One unit of protease activity was defined as the amount of enzyme required to liberate 1 μg of tyrosine per minute under the assay conditions.
2.5 Enzyme purification
The enzyme extract was precipitated by ammonium sulphate at 80% saturation and the suspension was kept overnight with mild stirring at 4°C. The precipitated protein was separated by centrifugation at 15 000g, 30 min at 4oC and re–dissolved in 20 mM phosphate buffer pH 6 and dialyzed for 24 h against the same buffer with three changes of buffer. The dialyzed extract was loaded onto anion exchange chromatography column (AEC) (5 mL, HiTrapTM Q FF GE Healthcare®) previously equilibrated with the buffer above and eluted with a linear gradient from 0 to 2 M of NaCl in the same buffer. Fractions with protease activity were pulled and re–chromatographed in the same column but this time the enzymatic extract was loaded with Tris–HCl buffer 20 mM pH 9 and eluted with a linear gradient from 0 to 1 M NaCl in the same buffer. The flow rate was 1mL/min in both times of AEC. The major protease activity fractions from the second AEC were pulled and subjected to gel filtration (GF) in a 50 mL column packed with Ultrogel® AcA 34 previusly equilibrated with Tris–HCl (20 mM ,pH 9) added with NaCl (200 mM). Load and elution were performed at 0.5 mL/min. In all cases, separation was performed in FPLC Biologic LP (Bio–Rad®).
2.6 Characterization of the pure enzyme
2.6.1 Molecular weight determination
The molecular weight and purification progress were followed by electrophoresis (silver stain, SDS–PAGE) (Laemmli, 1970).
2.6.2 Effect of pH on activity and stability
The effect of pH on the activity of the pure protease was evaluated at 50oC at different pH values. The pH of the substrate (casein 1 %) was adjusted by the following buffers: 0.1 M sodium phosphate (pH 6 and 7); 0.1M Tris–HCl (pH 8 and 9); 0.1M glycine–NaOH (pH 10 and 11); sodium carbonate (pH 12). For pH stability the enzyme was diluted in the pH buffer to be tested and incubated at 25°C for 2 h. The residual protease activity (%) was subsequently determined as described above.
2.6.3 Effect of temperature on activity and stability
The effect of temperature on enzyme activity was evaluated from 40–80°C in presence and absence of Ca2+ (10 mM). Optima temperature activity was pointed out by means of Arrhenius correlation (Iqbal et al., 2003; Siddiqui et al., 1997).
Thermal stability (thermal inactivation) was studied from 40 to 70°C for 60 min both, in presence and absence of calcium (10 mM) (Adinarayana et al., 2003). Aliquots were withdrawn at different intervals and residual activity was measured as described above.
2.6.4 Thermodynamic studies
Thermal stability data were fitted and first order rate constant (kd) was determined by linear regression of ln V versus time (t). The thermodynamic data were calculated by rearranging the Eyring's absolute rate equation:
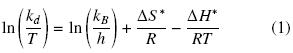
where kd, T, kb, h, ΔS*, ΔH* and R stand for specific reaction velocity, absolute temperature, Boltzmann's constant, Planck's constant, activation entropy, activation enthalpy and gas constant respectively (Iqbal et al., 2003).
2.6.5 Inhibition studies
The effect of protease inhibitors (5mM): phenylmethylsulphonyl fluoride (PMSF), β–mercaptoethanol and ethylene diamine tetra acetic acid (EDTA) on the activity of the pure enzyme was individually determinate by pre–incubation of the enzyme on the inhibitor solution at 25°C for 30 min and residual activity was measured. Residual activities in presence of the enzyme inhibitors were compared to the activity obtained without inhibitors (Charles et al., 2008).
3 Results and discussion
3.1 Protease production
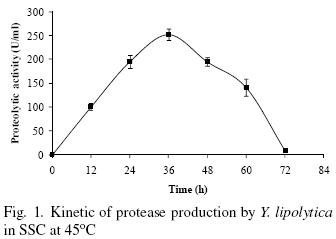
Production of bioactive compounds by SSC is taking a renewed importance due to its high productivity in short time fermentations (Ruíz–Leza et al., 2007). Figure 1 shows that protease production by Y. lipolytica in SSC increases with time and the maximum protease activity (252 U/mL) is observed at 36 h culture, after this time the enzyme production decreases. The reduction in enzyme production through time might be due to nutrients limitation and hydrolysis of the protease itself (Sandhya et al., 2005). The time of highest protease production observed in this work (36 h) is shorter than protease production by Candida humicola (96 h) and Candida buinensis (48 h) regardless of the previously optimized processes in both cases (Ray et al., 1992; De Araújo et al., 2010). The time production reported in this work represents a time reduction of 62 and 25% respectively. Nevertheless, Aerobasidium pullulans and Kluyveromyces marxianus presented maximum protease production at 30 h (Chi et al., 2007; Ramírez–Zavala et al., 2004), however, the authors do no mention the activity levels.
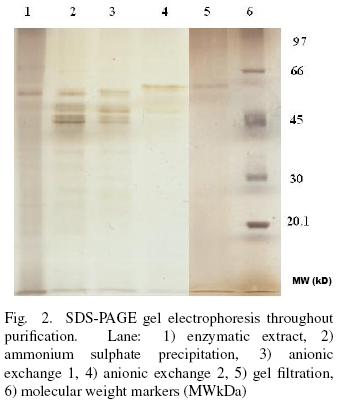
3.2 Enzyme purification
Purification of extracellular protease from Y. lipolytica is summarized in Table 1. The enzyme was purified 9.3– fold with 13.5% final yield. SDS–PAGE (silver stained) analysis of the purification progress shows a single band at 61.5 kDa after GF (Fig. 2). The molecular weight of proteases reported for Y. lipolytica, Debaryomyces hansenii and Candida humicola are from 30 to 36 kDa (Poza et al., 2007; Bolumar et al., 2005; Ray et al., 1992). Hernández–Montañez et al. (2007) reported a neutral intracellular protease (aminopeptidase) produced by Y. lipolytica with molecular weight of 97 kDa, highest that the extracellular protease studied in this work but similar to proteases from Debayromyces hansenii (101 and 370 kDa) (Bolumar et al., 20013 a; Bolumar et al., 2003b). The molecular weight of the pure protease is comparable with proteases produced from Debaryomyces hansenii (42 and 68 kDa), Kluyveromyces marxianus (46 kDa) and Saccharomyce cerevisiae (62 kDa) (Bolumar et al., 2008; Ramírez–Zavala et al., 2004; Kuhn et al., 1974). The molecular weight estimated for the protease studied is higher that the characteristic molecular weight presented by monomeric proteases produced by other yeast strains, probably due to some post–transcriptional modification (Spiro, 2002).
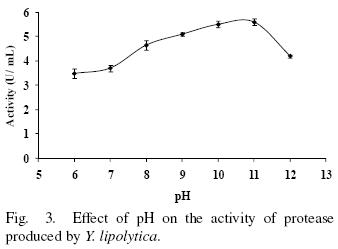
3.3 Effect of pH on activity and stability
The protease activity at different pH values is shown in Fig. 3. The protease is most active in a pH range from 6 to 12 showing at least 62% of maximum activity. However, optimal activity was observed at pH from 10–11. The pH values for optimal activity are in accordance with the extracellular protease produced by Yarrowia lipolytica CX161–1B and Aerobasidium pullulans that showed optimum activity at pH values of 9–10 and 9.5–10.5 respectively (Ogrydziak and Scharf, 1982; Donaghy and McKay, 1993). A large number of alkaline proteases produced by Aerobasidium pullulans, Candida humicola and Debaryomyces hansenii showed highest activity at alkaline pH from 8 to 9 (Chi et al., 2007; Ray et al., 1992; Bolumar et al., 2005; Bolumar et al., 2008).
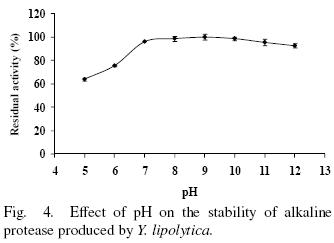
The enzyme was stable over a broad range of pH from 7 to 12 retaining more than 92% of residual activity. However at pH 5 and 6 retained 74 and 65% of activity respectively, after incubation for 2 h (Fig. 4). These results differ the ones reported by Ramírez–Zavala et al. (2004) whom reported that a protease from Y. lipolytica is stable at pH from 4 to 8, then the strain studied provide a wide pH range of high activity and gives a major advantage in industrial processes.
3.4 Effect of temperature on activity and stability
Enzyme activity in the temperature range from 35 to 70°C was evaluated in absence and presence of 10 mM Ca2+ (Figs. 5 and 6). The enzyme showed an optimal activity at 52.7 (5 U/mL) and 52.4°C (5.6 U/mL) in presence and absence of Ca2+ respetively. The results shown that the optimal temperature in presence or absence of Ca2+ are equal; nevertheless the enzyme activity was 12% increased in presence of Ca2+ suggesting that Ca2+ had an assisting effect on enzyme activity by providing stability. The optima temperature showed for the p1r3otease studied in this work is higher than the proteases produced by Y. lipolytica reported by Hernández–Montañez et al. (2007), Ogrydziak and Scharf (1982) and Jing et al. (2009) which display their optimum temperature at 37, 40, 45°C. Furthermore, the protease studied showed optimum temperature value higher than proteases produced by Candida humicola (37 °C), Debaryomyces hansenii (37 °C), Kluyveromyces marxianus (45 °C), Aerobasillum pullulans (45 °C) and Debaryomyces hanseni (Ray et ai, 1992; Bolumar et ai, 2003b; Ramíres–Zavala et ai, 2004; Chi et ai, 2007; Bolumar et al., 2003a).
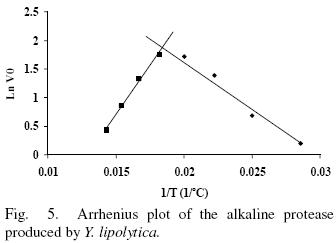
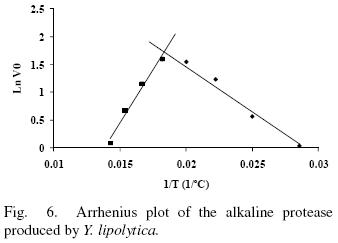
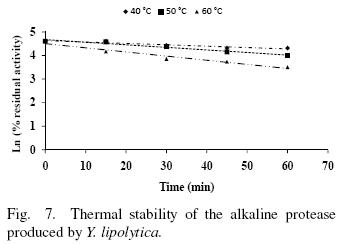
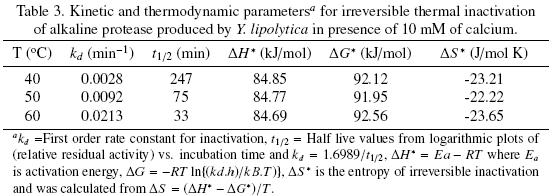
The thermal stability of the pure protease in presence and absence of Ca2+ (10 mM) is shown in Figs. 7 and 8. With the experimental date at different temperature values and the use of reverse lineal model (Azeredo et al., 2004), the half live (t1/2) was calculated (Table 2 and 3). The thermal stability profile of the protease showed that Ca2+ is required for enzyme stability because t1/2 is increased by 61, 6 and 11% at 40, 50 and 60°C respectively in presence of Ca2+; whereas at 70°C no activity was detected in thermostability at higher temperatures. This is the first report of thermal stability of proteases produced by Y. lipolytica.
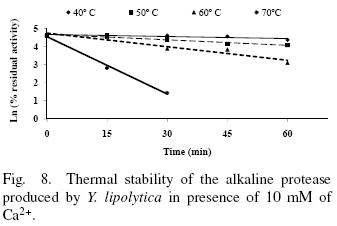
3.5 Thermodynamic studies
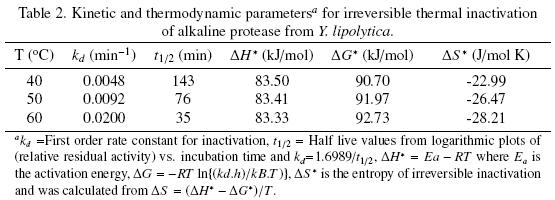
Tables 2 and 3 list the inactivation parameters of thermal inactivation at temperatures from 40 to 70°C. The results shows that in both cases (absence and presence of Ca2+) the enthalpy (ΔH*) is independent of temperature, thus there is no change in enzyme heat capacity, indicating that the enzyme stability is not provided by non–covalent linkages, since ΔH* value can be correlated with the number of non–covalent broken bonds during the process of protein denaturation (Ortega et al., 2004).The results showed that ΔG* is slightly higher in presence of Ca2+ indicating additional stability and in consequence the importance of Ca2+ to increase stability on the protease structure (Volquen et al., 2009). All entropy values (ΔS*) for thermal inactivation of the protease produced by Y. lipolytica from 40 to 60 °C were negative, however when Ca2+ was added, a slightly increase of ΔS* indicate that the structure is better organized than in absence of Ca2+ (Table 2 and 3). The most common cause of the heat inactivation on most enzymes is the loss of the native conformation (unfolding of the active tertiary protein structure to a disordered polypeptide), a process defined as thermodenaturation, which takes place as result of the increased molecular mobility at elevated temperature (Volquen et al., 2009; Iqbal et al., 2003).
The activation parameters related to Eyring's model supposed as irreversible first order inactivation shows that activation enthalpy exhibited a positive values in absence of Ca2+ (ΔH* =58.8 kJ/mol) and in presence of Ca2+ (ΔH* =85.3 kJ/mol). The difference on the value of this parameter indicates major thermostability when Ca2+ is present, suggesting that the protein structure presents higher rigidity (De Araújo et al., 2010). On the other hand, the activation entropy was negative in both, absence (–102.2 J/mol K) and presence of Ca2+ (–21.6 J/mol K), when Ca2+ is present, the increase entropy value indicates reorganization of the molecular structure of the enzyme.
3.6 Inhibition studies
The protease activity was inhibited 88% in presence of PMSF (5 mM) suggesting that the extracellular protease can be classify in the serine protease family. PMSF is known to sulphonate the essential serine residue in the active site of the protease resulting in a loss of enzyme activity (Kumar, 2002). Minimal or no inhibitory effect on the protease activity was observed with the other protease inhibitors tested (data not show).
Conclusions
Although many reports on production and purification of proteases by Y. lipolytica have been published, little information about thermal characteristics of this enzyme is available. In this study, we demonstrated that Y. lipolytica produce a thermostable serine protease in SSC at 45°C. The serine protease exhibited important properties such as broad range of pH stability and temperature. In presence of Ca2+ the stability was improved up to 73% at 40°C. The results of thermodynamic stability allows to class the protease studied as thermostable, additionally the importance of the Ca2+ on the thermostability was corroborated; this is the first report of thermal stability and thermodynamic properties of proteases produced by Y. lipolytica.
References
Adinarayana, K., Ellaia, P. and Davuluri, P. (2003). Purification and partial characterization of thermostable serine alkaline protease from a newly isolated Bacillus subtilis PE–11. AAPS pHARMSciTech 4 (4), 1–9. [ Links ]
Aloulou, A., Rodriguez, J., Puccinelli, D., Mouz, N., Leclaire, J., Leblond, Y. and Carriere, F. (2007). Purification and biochemical characterization of the LIP2 lipase from Yarrowia lipolytica. Biochimica et Biophysica Acta 1771, 228–237. [ Links ]
Azeredo, L., Freire, D., Soares, R., Leite, S., Coelho, R. (2004). Production and partial characterization of thermophilic proteases from Streptomyces sp. isolated from Brazilian cerrado soil. Enzyme and Microbial Technology 34, 354–358. [ Links ]
Bolumar, T., Sanz, Y., Aristoy, M. and Toldrá, F. (2003a). Puri?cation and characterization of a prolyl aminopeptidase from Debaryomyces hansenii. Applied and Environmental Microbiology 69(1), 227–232. [ Links ]
Bolumar, T., Sanz, Y., Aristoy, M. and Toldrá, F. (2003b). Purification and properties of an arginyl aminopeptidase from Debaryomyces hansenii. International Journal of Food Microbiology 86, 141– 151. [ Links ]
Bolumar, T., Sanz, Y., Aristoy, M. and Toldrá, F. (2005). Protease B from Debaryomyces hansenii: Purification and biochemical properties. International Journal of Food Microbiology 98, 167–177. [ Links ]
Bolumar, T., Sanz, Y., Aristoy, M. and Toldrá, F. (2008). Purification and characterization of proteases A and D from Debaryomyces hansenii. International Journal of Food Microbiology 124, 135–141. [ Links ]
Bradford, M. (1976). A rapid and sensitive method for the quanti?cation of microgram quantities of protein utilizing the principle of protein dye binding. Analytical Biochemistry 72, 248–54. [ Links ]
Charles, P., Devanathan, V., Anbu, P., Ponnuswamy, M., Kalaichelvan, P., Hur, B. (2008). Purification, characterization and crystallization of an extracellular alkaline protease from Aspergillus nidulans HA–10. Journal of Basic Microbiology 48, 347–352. [ Links ]
Chi, Z., Ma, C., Wang, P., Li, F. (2007). Optimization of medium and cultivation conditions for alkaline protease production by the marine yeast Aureobasidium pullulans. Bioresource Technology 98, 534–538. [ Links ]
De Araújo, D., De Albuquerme, C., Pereira, R., Souza, C., Aparecida, K., De Lima–Filho, J., Holanda, M., Converti, A., Figueiredo, A. (2010). Production and stability of protease from Candida buinensis. Applications in Biochemistry and Biotechnology 162, 830–842. [ Links ]
Donaghy, J. and MacKay, A. (1993). Production and properties of an alkaline protease by Aerobasidium pullulans. Journal of Applied Microbiology 74(6), 662–666. [ Links ]
Hernández–Montañez, Z., Araujo–Osorio, J., Noriega–Reyes, Y., Chávez–Camarillo, G. and Villa–Tanaca, L. (2007). The intracelular proteolytic system of Yarrowia lipolytica and characterization of an aminopeptidase. FEMS Microbiology Letters 268, 178–186. [ Links ]
Glover, D., MacEwen, R., Thomas, C. and Young T. (1997). pH–regulated expression of the acid and alkaline extracellular proteases of Yarrowia lipolytica. Microbiology 143, 3045–3054. [ Links ]
González–López, C., Ortiz–Castellanos, L., Ruiz–Herrera . (2006). The ambient pH response Rim pathway in Yarrowia lipolytica: Identification of YlRIM9 and characterization of its role in dimorphism. Current Microbiology 53, 8–12. [ Links ]
Iqbal Z., Rashid, M., Jabbar, A., Malana, N., Khalid, A., Rajoka, M. (2003). Kinetics of enhanced thermostability of an extracellular glucoamylase from Aranchniotus sp. Biotechnology Letters 25, 1667–1670. [ Links ]
Jing, L., Zhenming, C., Xianghong, W., Ying, P., Zhe, C. (2009). The selection of alkaline protease producing yeasts from marine environments and evaluation of their bioactive peptide production. Chinese Journal of Oceanology and Limnology 27, 753–761. [ Links ]
Johnvesly, B. and Naik, G. (2001). Studies on production of thermostable alkaline protease from thermophilic and alkaliphilic Bacillus sp. JB–99 in a chemically defined medium. Process Biochemistry 37, 139–144. [ Links ]
Kumar, G. (2002). Puri?cation and characterization of a thermostable alkaline protease from alkalophilic Bacillus pumilus. Letters in Applied Microbiology 34, 13–17. [ Links ]
Kuhn, R., Walsh, K.and Neurath, H. (1974). Isolation and partial characterization of an acid carboxypeptidase from yeast. Biochemistry 13(19), 3871–3877. [ Links ]
Laemmli, U. (1970). Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680–685. [ Links ]
Ma, C., Ni, X., Chi, Z., Ma, L., Gao, L. (2007). Purification and characterization of an alkaline protease from the marine yeast Aureobasidium pullulans for bioactive peptide production from different sources. Marine Biotechnology 9, 343–351. [ Links ]
Mazdak C., Gaillardin C. and Beckerich J. 2004. Heterologous protein expression and secretion in the non–conventional yeast Yarrowia lipolytica: a review. Journal of Biotechnology 109, 63–81. [ Links ]
Ogrydziak, D. and Scharf, S. (1982). Alkaline extracellular protease produced by Saccharomycopsis lipolytica CX 161– IB. Journal of General Microbiology 128, 1225–1234. [ Links ]
Ortega, N., Diego, S., Rodríguez, J., Perez, M. and Busto M. (2004). Kinetic behaviour and thermal inactivation of pectinlyase used in food processing. International Journal of Food Science and Technology 39, 631–639. [ Links ]
Pack, S. and Yoo, Y. (2003). Protein thermostability: structure–based difference of residual properties between thermophilic and mesophilic proteins. Journal of Molecular Catalysis 26, 257–264. [ Links ]
Peñalva, M. and Gerbert N. (2002). Regulation of gene Expression by ambient pH in filamentous fungi and yeasts. Microbiolgy and Mollecular Biology Reviews 66(3), 426–446. [ Links ]
Poza, M., Sestelo, A., Ageitos, J., Vallejo, J., Veiga–Crespo, G. and Villa, T. (2007). Cloning and expression of the XPR2 eene from Yarrowia lipolytica in Pichia pastoris. Journal of Agricultural and Food Chemistry 55, 3944–3948. [ Links ]
Ramírez–Zavala, B., Mercado–Flores, Y., Hernández–Rodríguez, C., and Villa–Tanaca, L. (2004). Purification and characterization of a lysine aminopeptidase from Kluyveromyces marxianus. FEMS Microbiology Letters 235, 369–375. [ Links ]
Ray, M., Devi, K., Kumar, S. and Shivaji, S. (1992). Extracellular protease from the Antarctic yeast Candida humicola. Applied and Environmental Microbiology 58 (6), 1918–1923. [ Links ]
Ruíz–Leza H.A., Rodríguez–Jasso R., Rodríguez–Herrera R., Contreras–Esquivel J.C. y Aguilar C.N. (2007). Diseño de Biorreactores para Fermentación en Medio Sólido. Revista Mexicana de Ingeniería Química 6(1), 33–40. [ Links ]
Sandhya, C., Sumantha, A., Szakacs, G. and Pandey, A. (2005). Comparative evaluation of neutral protease production by Aspergilus oryzae in submerged and solid state fermentation. Process Biochemistry 40, 2689–2694. [ Links ]
Saucedo, G., Lonsane, B., Navarro, J., Rusos, S. and Raimbault, L. (1992). Potential of using fermenter for biomass build–up, starch hydrolysis and ethanol production. Applied Biochemistry and Biotechnology 36, 47–61. [ Links ]
Sidiqqui, K., Rashid, M. and Rajoka, M. (1997). Kinetic analysis of the active site of an intracellular β–glucosidase from cellulomonas biazotea. Folia Microbiology 42 (1), 53–58. [ Links ]
Spiro, R. (2002). Protein glycosylation: nature, distribution, enzymatic information, and disease implications of glycopeptide bond. Glycobiology 12, 43–53. [ Links ]
Volquen, C., Faccin, D., Mertins, O., Heck, J., Silveira, N., Secchi, A. and Ayub, M. (2009). Kinetics of thermal inactivation of transglutaminase froma newly isolated Bacillus circulans BL32. Journal of Chemical Technology and Biotechnology 84, 1567–1575. [ Links ]














