Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista mexicana de ingeniería química
versión impresa ISSN 1665-2738
Rev. Mex. Ing. Quím vol.10 no.1 Ciudad de México abr. 2011
Biotecnología
Effect of glucose on 1,3–propanediol production by Lactobacillus reuteri
Efecto de la glucosa sobre la producción de 1,3–propanodiol por Lactobacillus reuteri
R. Baeza–Jiménez, L.X. Lopez–Martinez, J. De la Cruz–Medina, J.J. Espinosa–de–los–Monteros and H.S. García–Galindo*
UNIDA, Instituto Tecnológico de Veracruz. M.A. de Quevedo 2779, Col. Formando Hogar. Veracruz, Ver. 91897, México. *Corresponding author. E–mail: hsgarcia@itver.edu.mx; hugosgg@gmail.com
Received 24 of November 2010.
Accepted 8 of February 2011.
Abstract
Given the increasing demand for biofuels and biopolymers, there is an existing need to investigate new and more efficient alternatives for their production. 1,3–propanediol (PDO) is a typical product of glycerol fermentation by bacteria such as Klebsiella; however, Lactobacillus reuteri, a recognized probiotic, offers an alternative for its production in glucose/glycerol co–fermentations. The aim of the present work was to study the effect of glucose on PDO production using different initial concentrations (20–200 mM) under anaerobic conditions by L. reuteri ATCC 23272. Biomass was monitored by optical density at 600 nm and substrates and products profiles were analyzed by HPLC. The highest PDO concentration attained was 16.81 g/L in flasks (37°C, 100 mM glucose/200 mM glycerol for 16 h at pH 5.5, under anaerobic conditions). At fermenter level, a production of 28.69 g/L was reached under the same conditions, except glucose (200 mM) and glycerol (400 M). Stoichiometric and kinetic parameters showed that the 0.5 glucose/glycerol ratio allowed the highest PDO production. The flux calculated for glucose (Fglu) and glycerol (Fgly) in the co–fermentations with the highest PDO productions, had almost a 0.5 ratio.
Keywords: Lactobacillus reuteri, 1,3–propanediol, glucose, glycerol, co–fermentation.
Resumen
Dada la creciente demanda de biocombustibles y biopolímeros, existe la necesidad de investigar nuevas y más eficientes alternativas para producirlos. El 1,3–propanodiol (PDO) es un producto característico de la fermentación del glicerol por bacterias como Klebsiella; sin embargo, Lactobacillus reuteri, un probiótico reconocido, ofrece una alternativa para su producción en cofermentaciones glucosa/glicerol. El objetivo del presente trabajo fue estudiar el efecto de la glucosa en la producción de PDO usando diferentes concentraciones iniciales (20–200 mM) bajo condiciones anaeróbicas por L. reuteri ATCC 23272. La biomasa fue seguida por densidad óptica a 600 nm y los sustratos y el perfil de productos fueron analizados por HPLC. La concentración más alta de PDO obtenida fue de 16.81 g/L en matraces (37°C, 100 mM glucosa/200 mM glicerol, 16 h, a pH 5.5, bajo condiciones anaeróbicas). A nivel fermentador, se alcanzó una producción de 28.69 g/L en las mismas condiciones, excepto glucosa (200 mM) y glicerol (400 mM). Los parámetros estequiométricos y cinéticos muestran que la relación 0.5 glucosa/glicerol permiten la producción más alta de PDO. El flux calculado para glucosa (Fglu) y glicerol (Fgly) en las cofermentaciones con la producción más alta de PDO, casi tuvieron una relación de 0.5.
Palabras clave: Lactobacillus reuteri, 1,3–propanodiol, glucosa, glicerol, cofermentación.
1 Introduction
1,3–Propanediol (PDO) is one of the oldest known fermentation products and it is a promising bulk chemical which has attracted worldwide attention due to its enormous applications in polymers, cosmetics, foods, adhesives, lubricants, laminates, solvents, antifreeze and medicines (Colin et al., 2000; Zhu et al., 2002; Cheng et al., 2007; Willke and Vorlop, 2008). The majority of commercial synthesis of PDO are from acrolein by Degussa (now owned by DuPont) and from ethylene oxide by Shell (Haas et al., 2005). Problems in these conventional processes are the high pressure applied in the hydroformylation and hydrogenation steps along with high temperatures, the use of expensive catalysts and release of toxic intermediates.
Considering the yield, product recovery and environmental protection, much attention has been paid to its microbial production. Microbial production of PDO is carried out by very few organisms, all of them bacteria. These include enterobacteria of the genera Klebsiella (K. pneumoniae), Enterobacter (E. agglomerans), and Citrobacter (C. freundii), Lactobacilli (L. brevis and L. buchneri) and Clostridia of the C. butyricum and the C. pasteurianum group (Biebl et al., 1999; Saxena et al., 2009). Production data reported in the literature are 58.8 g/L by K. pneumoniae M 5a1 (Cheng et al., 2007), 64.9 g/L by C. butyricum VPI 3266 (González et al., 2005) and 135 g/L by a modified E. coli strain (Sanford et al., 2004).
In glycerol fermentation, part of the polyol is converted to the same products as in sugar fermentation, providing the necessary energy for growth but, for many of the products, reducing equivalents are released, which are oxidized in a reductive conversion of glycerol leading to the formation of PDO (Biebl et al., 1999). Lactobacilli have only this reductive conversion and need an additional fermentation substrate for growth and generation of the reduction equivalents (Veiga da Cunha and Foster, 1992).
L. reuteri, a recognized probiotic strain, can convert glycerol to PDO in co–fermentation with glucose. The first stage is catalyzed by glycerol dehydratase (GDHt, EC 4.2.1.30) that converts glycerol to 3–HPA (3–hydroxypropionaldehyde, reuterin), while the second stage involves an aldehydic dismutation of 3–HPA by PDO oxidoreductase (1,3–PDOR, EC 1.1.1.202) to PDO (Talarico and Dobrogosz, 1989; Schutz and Radler, 1984). In the same second stage, 3–HPA is used as an alternative hydrogen acceptor for recycling the NAD+, which is generated during hexose catabolism via the 6–phosphogluconate pathway (Talarico and Dobrogosz, 1989; Schutz and Radler, 1984).
Therefore, the aim of the present work was to study the effect of glucose on PDO production using different initial glucose concentrations (20–200 mM), in glycerol co–fermentations in an effort to develop an alternative for the production of this compound.
2 Materials and methods
2.1 Strain
Lactobacillus reuteri ATCC 23272, isolated from rat intestine, was provided in lyophilized form by the Agricultural Research Service (USDA Peoria, IL). The strain was activated in MRS (Man Rogosa and Sharpe; Difco, Detroit, Mich.) medium and kept at 4°C.
2.2 Culture conditions
Maintenance cultures were grown at 37°C and 200 rpm in 250 mL flasks containing MRS medium at pH 5.5 under anaerobic conditions. Lactobacillus samples were cultured in duplicated for determination of growth curve by inoculating 10 mL of the 24–h maintenance culture into 100 mL of newly prepared medium under the same conditions. Other samples were also taken for determination of viable cell counts under different initial glycerol concentrations (60, 100, 150, 200, 250 and 300 mM) according to Doleyres et al., (2005).
2.3 Biomass determination
Bacterial growth was monitored by measurement of the optical density at 600 nm (OD600) and cell dry weight at several stages of fermentations and co–fermentations. Optical density was measured using an Agilent diode–array spectrophotometer (Model G1103 A, China). Cell dry weight was calculated on the basis of OD600 using a previously prepared standard curve (El–Ziney et al., 1998). Specific growth rate was determined using the semi logarithmic plot of OD600 vs time.
2.4 Measurement of substrates and products
PDO and substrates (glucose and glycerol) and the other products (lactate, ethanol and acetate) were determined using a Waters Breeze HPLC system (Milford, MA) fitted with a HPX–87H column (300 mm × 7.8 mm, Bio–Rad, Richmond, CA). Separation was carried out at 50°C with 5 mM H2SO4 as mobile phase at a flow–rate of 0.6 mL/min. Quantification and identification of components were made using a refractive index detector (Waters 410) based on comparison of retention times with those of true standards (purchased from Sigma Chemical Company; St. Louis, MO).
2.5 Fermentation conditions
The effect of initial glycerol concentration on L. reuteri viability was evaluated at 60, 100, 150, 200, 250 and 300 mM in MRS medium without glucose. The effect of glucose on PDO production was tested at 36, 56, 83, 100 and 200 mM, once initial glycerol concentration was defined. All of the fermentations and co–fermentations were carried out in flasks containing 100 mL MRS medium (in glycerol fermentation MRS medium had no glucose and in co–fermentations MRS medium had different glucose concentrations) at 37°C, pH 5.5 (maintained by the addition of 3M H3PO4 or 3M NaOH) and 200 rpm under anaerobic conditions. When co–fermentations were carried out at fermenter level, a 1.5L Biostat V bioreactor (B. Braun Biotech International, Melsungen, Germany) was used under the same conditions, but containing 1L MRS medium. After runs concluded, L. reuteri cells were removed by centrifugation (Eppendorf Centrifuge 5415 C) at 14,000 rpm for 10 min at room temperature, and the supernatants were filtered through 0.45 μm filters to be analyzed by HPLC. Stoichiometric and kinetic parameters were calculated to determine the final biomass concentration and product formation according to Lee (1992).
3 Results and discussion
3.1 Bacterial growth
L. reuteri is a heterofermentative bacterial strain which metabolizes glucose through the Embden–Meyerhof–Parnas (EMP), and the phosphoketolase pathways (Morita et al., 2008). The latter allows L. reuteri to transform xilulose–5P into glyceraldehyde–3P and acetyl–P. Glyceraldehyde–3P is then converted to acetate or lactate through the EMP with a yield of 2 ATP, whereas acetyl–P is reduced to ethanol without ATP formation.
Fig. 1 shows the fermentation kinetics carried out with 100 mM glucose as sole carbon source. As it was mentioned above, L. reuteri through those particular pathways was able to transform glucose into lactate (80.44 mM), ethanol (74.13 mM) and acetate (91.5 mM). It is important to note that acetate was the main product. This high production of acetate is convenient for cells because of the ATP synthesized, which means more energy for growth. According to the calculated stoichiometric and kinetic parameters, the maximum specific growth rate (μmax) reached was 0.3 h–1 and the yield of ATP (YATP/X) was 96.67 mM ATP/gX, when 1.66 × 108 cfu/mL (ln OD600 = 0.25) were inoculated reaching 2.26 × 109 cfu/mL ln OD600 = 2.58). Fermentation conditions were 200 rpm, 37°C, 16 h and pH 5.5 in flasks containing 100 mL MRS medium.
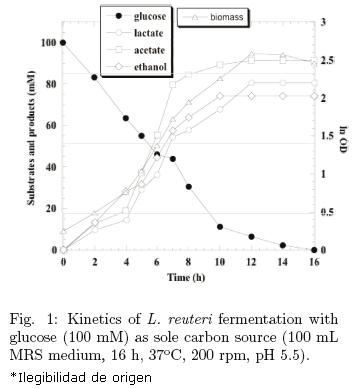
3.2 Effect of initial glycerol concentration
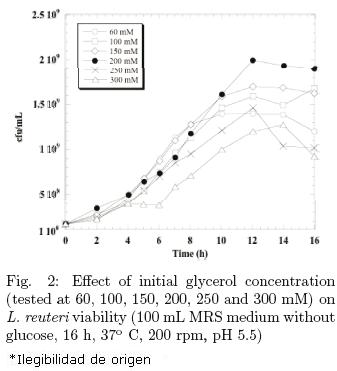
PDO is a product from glycerol fermentation; hence, the effect of initial glycerol concentration is very important. On the other hand, the first step in glycerol fermentation is the production of 3–HPA which may be reduced by the cells of L. reuteri. The concentrations tested were 60, 100, 150, 200, 250 and 300 mM in MRS medium without glucose. As it can be seen in Fig. 2, 200 mM glycerol was the highest concentration at which L. reuteri had the highest viability and thus this concentration was selected for the co–fermentations with glucose. Fermentation conditions were 200 rpm, 37°C, 16 h and pH 5.5 in flasks containing 100 mL MRS medium without glucose. These results are comparable to those reported by Zheng et al. (2008), who found this also to be the best concentration for K. pneumoniae.
3.3 Effect of initial glucose concentration in co–fermentations
Glycerol is metabolized in a dismutation process via two routes, one reductive and one oxidative. The reductive route leads to PDO formation. This route is initiated by glycerol dehydration by the coenzyme B12–dependent GDHt (C. butyricum GDHt is B12–independent), which produces 3–HPA. After this step, 3–HPA is reduced to PDO in a NADH+H+–dependent reaction catalyzed by 1,3–PDOR. The second, oxidative route begins with glycerol oxidation by NAD+–dependent glycerol dehydrogenase (GDH), which produces dihydroxyacetone. Then, dihydroxyacetone is phosphorylated by dihydroxyacetone kinase and further metabolized to CO2, H2, acetate, butyrate, lactate, ethanol, butanol, or 2,3–butanediol (Morita et al., 2008).
For glycerol metabolism, L. reuteri has only the reductive pathway and needs an additional fermentation substrate for growth and generation of the reduction equivalents (Veiga da Cunha and Foster, 1992). The regulation of this pathway is dependent on the availability of fermentable carbohydrates, in particular glucose. When L. reuteri ferments glucose plus glycerol, 1,3–PDOR is synthesized, providing an additional pathway to NADH+H+ disposal and a modified products profile.
Therefore, different initial glucose concentrations were tested at 36, 56, 83, 100 and 200 mM when glycerol was set at 200 mM in all the co–fermentations. Table 1 shows the effect of glucose on PDO production. Clearly, as the initial glucose concentration was increased in MRS medium, greater PDO production was reached. This suggests another pathway for NAD+ regeneration, which is different from the reduction of acetyl–P to ethanol. The latter is observed in Table 1 when ethanol formation decreased from 103.26 mM (56 mM glucose/200 mM glycerol co–fermentation) to 74.13 mM (100 mM glucose/200 mM glycerol co–fermentation) and acetate formation increased from 86.83 mM (36 mM glucose/200 mM glycerol co–fermentation) to 118.17 mM (100 mM glucose/200 mM glycerol co–fermentation). Greater acetate production is convenient for cells because of the ATP formation. The yield of ATP (YATP/X) also increased with the initial glucose concentration (see Table 1).
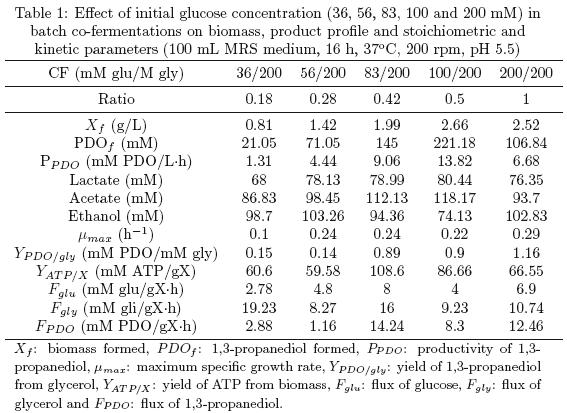
The highest PDO concentration reached was 221.18 mM in 100 mM glucose/200 mM glycerol co–fermentation, in flasks containing 100 mL MRS medium at 37°C, pH 5.5 during 16 h. This result is greater than that reported by Tobajas et al. (2009), who reached 170.7 mM PDO in 111 mM/200 mM glycerol co–fermentation.
Biomass formation was also improved by the initial glucose concentration as well as growth rate (see Table 1). This is explained by the combination of different metabolic pathways and it has already been reported elsewhere. Both glucose (Fglu) and glycerol (Fgly) fluxes have an interesting behavior because they do not seem to fit the glucose/glycerol ratios even when they are seldom exactly the same in 56 mM glucose/200 mM glycerol, 83 mM glucose/200 mM glycerol and 100 mM glucose/200 mM glycerol co–fermentations (see Table 1). This can be explained by insufficient NADH+H+ disposal and 3–HPA being accumulated in the medium with a toxic effect on growing cells. The supporting idea is a low PDO flux (F1,3–PDO) in 36 mM glucose/200 mM glycerol and 56 mM glucose/200 mM glycerol co–fermentations. However, in 83 mM glucose/200 mM glycerol and 100 mM glucose/200 mM glycerol co–fermentations Fglu, Fgly and FPDO were almost half. We assume this idea by the stoichiometric and kinetic parameters calculated for each co–fermentation, which are a powerful tool in metabolic flux analysis (Soto and Páez, 2005).
The effect of the addition of glucose has also been observed in continuous co–fermentations by C. butyricum E5 where a 0.4 glucose/glycerol ratio yielded 0.89 mol PDO/mol glycerol (Malaoui and Marczak, 2001). Lüthi–Peng et al. (2002), also reached the highest PDO production with 100 mM glucose/200 mM glycerol co–fermentation.
In recombinant strains, the effect of glucose in glycerol metabolism has been demonstrated to be positive. Native E. coli does not ferment glycerol to PDO, but it has been reported that E. coli cells transformed with K. pneumoniae dha regulon genes were able to co–ferment glycerol with glucose. PDO yield may be enhanced by co–fermentation of glycerol with sugars, providing that kinetic constraints are overcome. The yield of PDO from glycerol was improved from 0.46 mol/mol with glycerol alone to 0.63 mol/mol with glucose co–fermentation and 0.55 mol/mol with xylose co–fermentation (Saxena et al., 2009).
3.4 Co–fermentations in bioreactor
In order to drive L. reuteri to synthesize larger amounts of PDO, a 1.5 L bioreactor was employed with pH, temperature and stirring rate control. The effective volume was one liter, the stirring rate was 200 rpm, temperature was 37°C, and pH was maintained at 5.5 by the automatic addition of 3M H3PO4 or 3M NaOH. The first co–fermentation was carried out in 100 mM glucose/200 mM glycerol and 19.89 g/L was reached; but in order to investigate further changes if both substrates concentrations were increased, 200 mM glucose/400 mM glycerol co–fermentation was tested and reached a considerably higher PDO concentration: 377.55 mM (see Fig. 3).
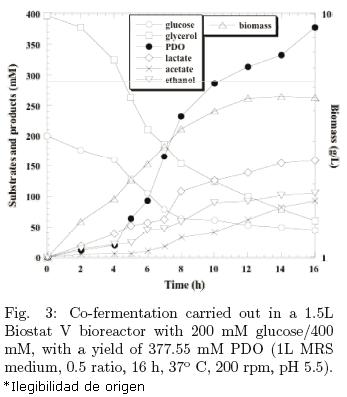
In this co–fermentation, not only a higher concentration of PDO was reached, but also higher biomass and lactate concentrations were attained. The ratios of initial glucose/glycerol concentrations are almost exactly the same to those of flux even when μmax was the same and YATP/X was slightly lower (see Table 1). The non significant change in ethanol and acetate formation might be due to residual glucose and glycerol in the medium. GDHt is inhibited by glycerol and as it is shown in Fig. 2; 200 mM glycerol was the highest concentration at which L. reuteri was able to grow. Therefore, an inhibition may have taken place and the balance in both glucose and glycerol fermentations were affected by environmental and nutritional conditions. On this particular issue, Zhang et al. (2008), showed that partitioning of the carbon flux between the reductive and the oxidative route is robust against environmental conditions. Such tight control at the glycerol content is provided by kinetic parameters of GDH, GDHt and 1,3–PDOR.
In this work, 377.55 mM (28.69 g/L) of PDO were produced in a bioreactor but higher concentrations of PDO have been reached and reported (Table 2). Several K. pneumoniae, C. acetobutylicum and C. butyricum strains have been used for the production of PDO, along with a modified E. coli strain which yielded 135 g/L of PDO (Sanford et al., 2004). Even though L. reuteri production is lower than those listed in Table 2, the advantages offered by this strain are that this is not a pathogenic or engineered strain, with easier and safer co–fermentation conditions than those required in chemical processes, the possibility of using raw glycerol from the biodiesel industry and the resulting probiotic biomass may be considered a value–added to the process.
Conclusions
We found that 37°C, 200 rpm, 16 h and pH 5.5 under anaerobic conditions, are the best conditions for the production of PDO in flasks containing 100 mM glucose and 200 mM glycerol. The highest production of PDO achieved was 377.55 mM (28.69 g/L) in a 1.5 L bioreactor under the same conditions as in flasks but with 200 mM glucose and 400 mM glycerol. Even if this is a relatively smaller production than those reported for other bacterial species, L. reuteri offers the advantages of safe handling and novel applications for glycerol and production of a probiotic biomass; this production rate could also be further optimized.
The stoichiometric and kinetic parameters tested showed that the 0.5 ratio favored the highest PDO production. The flux for substrates and products showed how glucose impacts in PDO formation. The flux calculated in the co–fermentations with highest PDO productions, has the same 0.5 ratio even when high glycerol concentration may have inhibited GDHt.
Acknowledgment
The authors wish to acknowledge the financial support of DGEST (SEP) for this project through the grant 911.08–P
References
Biebl, H., Menzel, K., Zeng, A.P. and Deckwer, W.D. (1999). Microbial production of 1,3–propanediol. Applied Microbiology and Biotechnology 52, 289–297. [ Links ]
Cheng, K.K., Zhang, J.A., Liu, D.H., Sun, Y., Liu, H.J. and Yang, M.D. (2007). Pilot–scale production of 1,3 propanediol using Klebsiella pneumoniae. Process Biochemistry 42(4), 740–744. [ Links ]
Colin, T., Bories, A., and Moulin, G. (2000). Inhibition of Clostridium butyricum by 1,3–propanediol and diols during glycerol fermentation. Applied Microbiology and Biotechnology 54, 201–205. [ Links ]
Doleyres, Y., Beck, P., Vollenweider, S. and Lacroix, C. (2005). Production of 3–hydroxypropionaldehyde using a two step process with Lactobacillus reuteri. Applied Microbiology and Biotechnology 68, 467–474. [ Links ]
El–Ziney, M.G., Arneborg, N., Uyttendaele, M., Debevere, J. and Jakobsen, M. (1998). Characterization of growth and metabolite production of Lactobacillus reuteri during glucose/glicerol cofermentation in batch and continuous cultures. Biotechnology Letters 20(10), 913–916. [ Links ]
Gonzales–Pajuelo, M., Meynial–Salles, I., Mendes, F., Andrade J.C. Vasconcelos, A and Sucaille, P. (2005). Metabolic engineering of Clostridium acetobutylicum for the industrial production of 1,3–propanediol from glycerol. Metabolic Engineering 7, 329–336. [ Links ]
Haas, T., Jaegaer, B., Weber, R., Mitchell, S.F. and King, C.F. (2005). New diol processes: 1,3–propanediol and 1,4–butanediol. Applied Catalysis A: General 280, 83–88. [ Links ]
Lee, J. M. (1992). Biochemical Engineering. Washington State University. Prentice Hall, New Yersey. [ Links ]
Lüthi–Peng, Q., Dileme, F.B. and Puhan, Z. (2002). Effect of glucose on glycerol bioconversion by Lactobacillus reuteri. Applied Microbiology and Biotechnology 59, 289–296. [ Links ]
Malaoui, H. and Marczak, R. (2001). Influence of glucose metabolism by wild–type and mutant strains of Clostridium butyricum E5 grown in chemostatic culture. Applied Microbiology and Biotechnology 55, 226–233. [ Links ]
Morita, H., Toh, H., Fukuda, S., Horikawa, H., Oshima, K., Suzuki, T., Murakami, M., Hisamatsu, S., Kato, Y., Takizawa, T., Fukuoka, H., Yoshimura, T., Itoh, K., O'Sullivan, D.J., McKay, L.L., Ohno, H., Kikuchi, J., Masaoka, T. and Hattori, M. (2008). Comparative genome analysis of Lactobacillus reuteri and Lactobacillus fermentum reveal a genomic island for reuterin and cobalamin production. DNA Research 15, 151–161. [ Links ]
Sanford, K., Valle, F. and Ghirnikar R. (2004). Pathway engineering through rational design. Genetic Engineering News 24(2), 44–45. [ Links ]
Saxena, R.K., Anand, P., Saran, S. and Isar, J. (2009). Microbial production of 1,3–propanediol: Recent developments and emerging opportunities. Biotechnology Advances 27, 895–913. [ Links ]
Schutz, H. and Radler, F. (1984). Anaerobic reduction of glycerol to propanediol–1,3 by Lactobacillus brevis and Lactobacillus buchneri. Systematic and Applied Microbiology 5, 169–178. [ Links ]
Soto, O. and Páez, J. (2005). Balances en procesos de fermentación: análisis de consistencia y de flujos metabólicos. Revista Mexicana de Ingeniería Química 4, 59–74. [ Links ]
Talarico, T.L. and Dobrogosz, W.J. (1989). Chemical characterization of an antimicrobial substance produced by Lactobacillus reuteri. Antimicrobial Agents and Chemotherapy 33, 674–679. [ Links ]
Tobajas M., Mohedano, A.F., Casas, J.A. and Rodríguez, J.J. (2009). Unstructured kinetic model for reuterin and 1,3–propanediol production by Lactobacillus reuteri from glycerol/glucose cofermentation. Journal of Chemical Technology and Biotechnology 84, 675–680. [ Links ]
Veiga da Cunha, M. and Foster, M.A. (1992). Sugar–Glycerol Cofermentations in Lactobacilli: the Fate of Lactate. Journal of Bacteriology 174 (3), 1013–1019. [ Links ]
Willke, T. and Vorlop, K. (2008). Biotransformation of glycerol into 1,3–propanediol. European Journal of Lipid Science and Technology 110, 831–840. [ Links ]
Zhang, Q., Teng, H., Sun, Y., Xiu, Z. and Zeng, A.P. (2008). Metabolic flux and robustness analysis of glicerol metabolism in Klebsiella pneumoniae. Bioprocess and Biosystems Engineering 31, 127–35. [ Links ]
Zheng, Z.M., Chen, K,K., Hu, Q.L., Liu, H.J., Guo, N.N. and Liu, D.H. (2008). Effect of culture conditions on 3–hydroxypropionaldehyde detoxification in 1,3–propanediol fermentation by Klebsiella pneumoniae. Biochemical Engineering Journal 39, 305–310. [ Links ]
Zhu, M.M., Lawman, P.D. and Cameron D.C. (2002). Improving 1,3–propanediol production from glycerol in a metabolically engineered Escherichia coli by reducing accumulation of sn–glycerol–3–phosphate. Biotechnology Progress 18, 694–699. [ Links ]














