Introduction
The potato (Solanum tuberosum) is one of the most economically and socially important foods, currently as the third most consumed crop worldwide, only surpassed by wheat, and rice (Devaux et al., 2021). In 2019, world potato production was 370 million tons, of which Mexico produced more than 1 million, presenting a per capita consumption of 14.8 kg (FAOSTAT, 2020; Devaux et al., 2021). It is estimated that around 56 % of the national production is used for fresh consumption, processing companies demand 29 %, while 15 % is destined for seed (Mejía and Castellanos, 2018).
In the industrialization of this tuber, approximately 88.9 % of the product is used, the remaining percentage corresponds to the residues generated (Calvache et al., 2022). For the processing of potatoes, both external (size, shape, presence of diseases, defects, deformations, cold and mechanical damages) and internal (total solids, non-reducing and total sugars, flavor, and nutritional value) characteristics are considered to ensure the quality of the final product, those that do not meet the requirements are rejected and considered waste (Ortega and Andrade-Bolaños, 2021).
About 25 % of the waste generated during potato processing is disposed of in sanitary landfills, in unsuitable places or deposited in agricultural plots for degradation, without going through a process that gives them an adds value (Sánchez-Castelblanco and Heredia-Martín, 2021; Calvache et al., 2022). The concern for the use of residues has gained great strength worldwide, in the scientific community and especially at the industrial level, where production processes generate by-products that can be useful in other activities (Quintero et al., 2015), such as use of starch in the case of potato, since, in addition to its importance as a food, it has expansive uses in the industry mainly due to its starch content (Jagadeesan et al., 2020).
Starch is a polysaccharide composed of glucose monomers that form the amylose (20 - 30 %) and amylopectin, the amylose is formed by 1,4-α-D-glucose monomers with a linear structure and small number of long branches, meanwhile, the amylopectin is formed by 1,6-α-D-glucose monomers with a high-density branched chain, these branching occurs every 20 to 23 glucose units (González-Cuello et al., 2016; Kowsik and Mazumder, 2018). The amylose-amylopectin ratio varies significantly between starches depending on their source, such as the type and variety of plant or fruit, as well as growth conditions. Several studies have shown that this variation involves changes in their physicochemical characteristics and interactions with other molecules, resulting in different functional and microscopic properties, as well as different texture and stability of starch products (Lemos et al., 2019). Then, it is possible to recover and make an advanced characterization of the residual potato starch used in the manufacture of potato chips in Mexico.
Potato starch is preferred over others, because its paste has high clarity and neutral flavor (Alvani et al., 2011), attributed to the high content of phosphate esters in the amylopectin chain (Šimková et al., 2013). In 2015, the starches annual world production was approximately 85 million tons (Dupuis and Liu, 2019), with corn, wheat, potato, and rice starch being the main sources, accounting for 84 %, 7 %, 4 % and 1 %, respectively (Basiak et al., 2017). This starch is widely used in industrial sectors, such as food, feed, paper, textiles, laundry finishes, chemical, petrochemical, pharmaceutical industries, bioethanol, construction materials and biodegradable products (Maniglia et al., 2020). In this sense, the objective of this study was to evaluate the physicochemical, morphological, structural, and thermal properties of starch recovered from potato (Solanum tuberosum) residual of chips industry in Mexico, using advanced instrumental techniques, to offer alternatives for the recycling of residual potatoes.
Materials and methods
Starch extraction
The residual potato was collected from a chips production company, located in the industrial zone in the state of Mexico, Mexico. It was then washed, the skin removed, and their size was reduced using a homemade grater. The grated potatoes were soaked for 30 min with constant manual agitation (100 g of potato/200 mL of water), then filtered using a domestic sieve; this process was repeated three times (50 mL of water/100 g of potato). The filtrate was left undisturbed for 1 hour and the starch recovered by decantation and subjected to two washes using distilled water. Finally, the recovered starch was dried at 60 ºC and stored in hermetically sealed plastic bags until use (Vargas et al., 2016).
Proximate composition analysis
Moisture content was determined using a thermobalance (OHAUS™ MB35) according to Pardo et al. (2013), total ash content was determined using the dry ash method (Kirk et al., 1991), and protein content was determined by the Kjeldahl method (factor of 6.25), according to the Official Mexican Standard NOM -F-68-S -1980. Additionally, the Soxhlet method was used to determine the total lipid content (James, 1999) and total carbohydrates were estimated as the percentage remaining to reach 100 % of the proximal composition (Valle et al., 2019).
Amylose content was determined using the methodology of Hoover and Ratnayake (2001). Specifically, defatted starch (25 mg) was dissolved in 10 mL of 90 % dimethylsulfoxide (DMSO), stirred for 20 min, and then maintained for 15 min in a water bath at 85 °C. The mixture was cooled and gauged with 25 mL of distilled water. An aliquot (1 mL) was taken and diluted with 5 mL of iodine solution (0.0025 mol L-1) in potassium iodide (0.0065 mol L-1) (I2/KI) and gauged to 50 mL with distilled water. The absorbance was read at 600 nm using a spectrophotometer (Multiskan™ GO, Thermo Scientific™, USA), mixtures of DMSO and iodine solution in potassium iodide were used as blank. The amylose content of the starch sample was determined using a standard curve prepared from potato amylose and amylopectin mixtures (Sigma Aldrich, Edo. de México), containing 0 - 100, 10 - 90, 20 - 80, 30 - 70, 40 - 60, 50 - 50, 60 - 40, 70 - 30, 80 - 20, 90 - 10 and 100 - 0 % treated in the same way as starch solutions. All determinations were analyzed in triplicates.
Functional properties
Water absorption capacity
To determine the starch water absorption capacity, suspensions of 5 % starch in 40 mL of distilled water were prepared, left to stand for 40 min with stirring every 10 min, and centrifuged (Thermo Scientific™ Sorvall™ ST 8 Centrifuge) at 3250 rpm for 25 min at 25 °C. The supernatant was discarded, and the sediment was weighed to determine the adsorption capacity (eq.1). The test was performed in triplicate (Sindhu et al., 2019).
Solubility and swelling power
The determination of the solubility index (SI) and swelling power (SP) was determined by the method of Meaño et al. (2014) with some modifications. Specifically, 1 % starch suspensions were prepared in 40 mL of distilled water, heated in a water bath (Ecoline Staredit. LAUDA® E100) at 60 ºC, 70 ºC, 80 ºC and 90 ºC for 30 min, then cooled to 25 ºC, and centrifuged (Thermo Scientific™ Sorvall™ ST 8 Centrifuge) at 4000 rpm for 50 min.
The supernatant was discarded, while the gel formed was recovered and dried at 60 °C for 20 hours to determine the SI (eq.2) and the SP (eq.3).
Morphological and optical analysis Scanning electron microscopy (SEM) and elemental analysis (EDS)
Morphological analysis of the starch surface was performed at 100X and 200X using a scanning electron microscope (JEOL, JSM-7600 F, Japan). Starch particles were coated with a thin layer of Au-Pd using a sputter coater (Quorum QI5OR-ES, Sussex, UK) before scanning. The morphological analysis of starch particles was performed to investigate size and shape. In addition, the elemental composition of samples was analyzed using an Oxford X-Max 20 energy dispersive X-ray spectroscopy (EDS) detector and an Oxford wavelength dispersive X-ray spectroscopy (WDS) detector (Acosta-Ferreira et al., 2020).
Dynamic Light Scattering (DLS)
Particle size, polydispersity index (PDI) and zeta potential of starch were determined using a DLS instrument, Zetasizer Nano ZS (Malvern Instruments Ltd., Worcestershire, UK), according to the method reported by Sattari et al. (2017), with some modifications. Specifically, 1.2 mg of the sample was dissolved in 2 mL of 95 % EtOH, subsequently sonicated for 5 min in an ultrasound bath to disintegrate the starch granules, and then placed in a polystyrene cuvette for reading in the equipment. All results were recorded as the average of 5 measurements.
Color
Starch color was determined using a Chroma Meter colorimeter (Minolta CR-400 Japan) calibrated with a white mosaic standard (Y = 94.1; X = 0.3155; Y = 0.3319). Color was read using the CIELab scale and measured (L) luminosity (0 black to 100 white) and chromaticity parameters a* (- 100 green to + 100 red) and b* (- 100 blue to + 100 yellow). Five replicates of the sample were analyzed, placed on the pattern. The color difference (∆E) was calculated using equation 4.
Where ∆L* = L* - Lo, ∆a* = a* - ao and ∆b* = b* - bo. Lo, ao and bo represent the color parameters of the standard, while L*, a* and b* represent the color parameters of the sample (Gonçalves et al., 2020).
Thermal analysis
Differential Scanning Calorimetry (DSC)
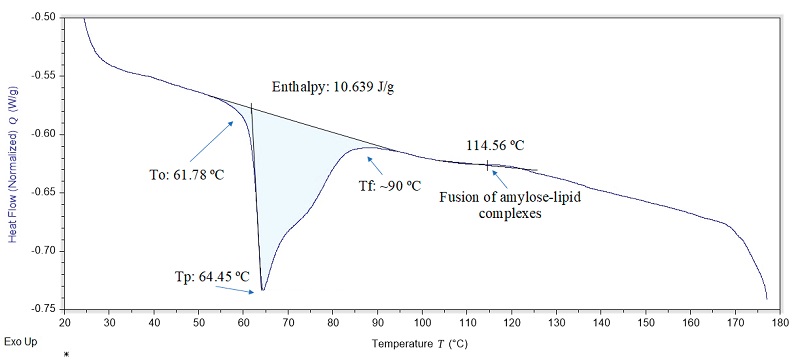
Starch gelatinization properties were analyzed using a Differential Scanning Calorimeter (DSC) on a Discovery model from TA Instruments Inc. (New Castle, DE). Specifically, 10 mg of sample were placed in aluminum cells and sealed under pressure. An empty aluminum cell was used as a reference. Both cells were heated from 20 to 180 °C at a heating rate of 10 °C/min and then rapidly cooled to 20 °C. The thermal parameters determined of the gelatinization peak were, initial temperature (To), maximum temperature (Tp), final temperature (Tf) and gelatinization enthalpy (ΔH), typical of the starch gelatinization process (Rodríguez-Núñez et al., 2014).
Thermogravimetric analysis (TGA/DTGA)
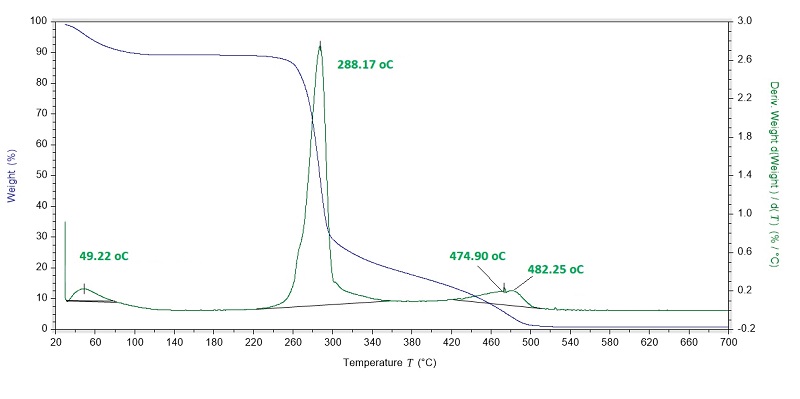
Thermal decomposition of starch was analyzed using a TA Instruments Discovery Model Thermogravimetric Analyzer (TGA) (New Castle, DE). A sample of approximately 20 mg was heated from 50 to 600 °C at a heating rate of 10 °C/min, under an inert N2 atmosphere with a flow rate of 60 mL/min (Rodríguez-Núñez et al., 2014).
NMR spectra
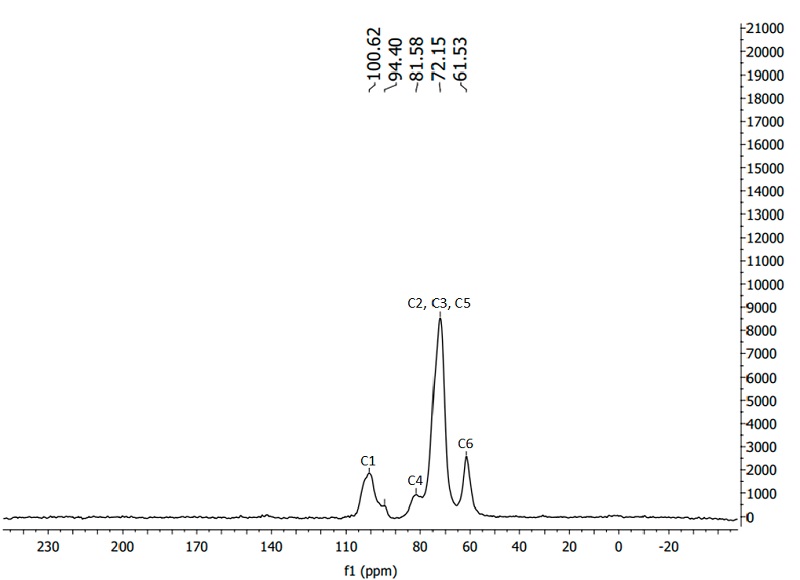
13C nuclear magnetic resonance analyzes were performed on a Bruker Avance III spectrometer (USA, Massachusetts) operating at 500 MHz using CDCl3. NMR spectra were referenced using the residual signal of CHCl3 at 77.26 ppm (chemical shifts) (Gómez and González-García, 2018).
X-ray diffraction (XRD)
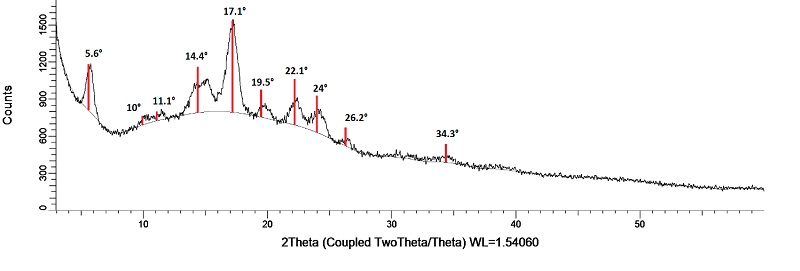
To determine the crystalline structure of starch, a powder sample was placed in a sample holder for X-ray diffractometry. The X-ray diffraction patterns were obtained using a Bruker D8-Advance Bragg diffractometer (Bruker, Billerica, MA, USA) with a Bragg Brentano geometry and monochromatic CuKα radiation (λ = 1.541 A°), operated at 40 kV and 30 mA. The X-Ray diffraction patterns were recorder in reflection mode in an angular range of 5° to 60° (2Ɵ), with a step time of 0.5 s and a step size of 0.02º. The crystallinity was calculated according to equation 5 (Aguilar-Pérez et al., 2020).
FTIR-ATR spectrophotometry
Attenuated total reflectance infrared (FTIR-ATR) spectra of starch were obtained at room temperature using a Thermo Nicolet model Nexus 670-FTIR spectrometer (Madison, WI). The samples were analyzed in the range of 4000 - 600 cm-1, with 4 cm-1 resolution and 100 scans, the OMNIC software was used to obtain the results (Madera-Santana et al., 2016).
Statistical analysis
Data were presented as average of triplicate measurements ± standard deviation. The results obtained in percentage form were transformed to a arcsine to optimize the normality of the data. An analysis of variance (ANOVA) was performed with a completely randomized design (DCA) and a level of significance (p ≤ 0.05), the Tukey test was applied to determine significant differences between means using the SAS statistical analysis software (version 9.3).
Results and discussion
Proximate composition analysis
The proximal composition of the potato starch is shown in Table 1. Similar moisture values were reported by Bravo and Chavarría (2020) with 12.6 % for Leona Blanca variety potato starch, while Acosta et al. (2018) indicated higher values (16.9 - 20.8 %). Dry starches usually have a humidity that ranges between 6 - 16 %; this variation is due to the environment and the drying process employed, however, the humidity should not exceed 13 %, since high levels can cause microbial damages and deteriorate quality (Moorthy, 2002).
The results obtained in this work for ashes, lipids, proteins, and carbohydrates were similar to those reported in previous studies. In this sense, Bravo and Chavarria (2020) reported a proximal composition of potato starch (Leona Blanca variety) with 0.12 % lipids, 0.35 % proteins and 85.87 % carbohydrates, Pardo et al. (2013) mentioned a 0.23 - 0.44 % range for ashes and 0.28 - 0.33 % for protein in potato starch, and Vargas et al. (2016) reported values of 0.43, 0.3 and 98.8 % of ashes, lipids, and carbohydrates, respectively, for potato starch (Única variety).
The variation in the proximal composition of starches has been reported between and within botanical species, since they are influenced by various factors such as growth, development conditions of the plant and the variety used, showing a direct effect on interactions with other molecules, resulting in different physicochemical, functional, and microscopic properties (Martínez et al., 2019a; Lemos et al., 2019).
The amylose content was another important parameter evaluated because it allows to categorized starches as, waxy (< 15 %), normal (20 - 35 %) or high (> 40 %) (Gonçalves et al., 2020). In the present study, starch was identified as normal (24.35 % amylose), similar to those reported by Torres et al. (2020) and Solarte-Montúfar et al. (2019) with ranges of 15.6 - 29.3 % and 24.8 - 26.2 % amylose, respectively, in potato starch of different varieties.
These characterization studies are fundamental since they allow deciding the most promising uses of each starch in the different industrial sectors where it is used, and ruling out those for which its characteristics are undesirable. For example, in the use of starch as a food additive, the amylose-amylopectin ratio influences the clarity or opacity of solutions, amylose also provides greater thermal stability to pastes and also exhibits excellent film-forming properties compared to amylopectin, so in this type of industry a high amylose content becomes a desirable characteristic (Solarte-Montúfar et al. al., 2019).
Functional properties
Water absorption capacity (WAC)
The obtained water absorption capacity (WAC) of potato starch was 1.0163 ± 0.0078 g g-1 (101.63 ± 0.77 %), a value close to that reported by Sarifudin et al. (2020) with 100 %, meanwhile, Cao and Gao (2020) and Chuiza-Rojas et al. (2021), reported a higher water absorption capacity of 1.81 gg-1 and 1.87 g g-1, respectively, for potato starch. Potato starch contains phosphate ester groups that provide a higher WAC and a faster rate of hydration compared to cereals starch, which is reflected in a higher viscosity during cooking. This property indicates the ability of starch to interact with water and to form pastes or gels; in addition, its use as food additive allows determining the texture and quality of food, so that starches that have a high WAC usually intended for this industrial sector, especially as a thickening agent (Solarte-Montúfar et al., 2019; Jiménez-Villalba et al., 2019).
The WAC of starch is influenced by granule size (small granules, characteristic of amylopectin, retain more water than large granules, typical of amylose), and the presence of hydrophilic groups (which have a high-water retention capacity) (Guízar et al., 2008; Babu and Parimalavalli, 2014; Martínez et al., 2015; Sangokunle et al., 2020).
Solubility index and swelling power
The starch, subjected to high temperatures and excess of water, show a gelatinization process due to the breaking of the crystalline structure allowing the union of water molecules with the -OH groups of the amylose and amylopectin through hydrogen bonds, promoting the solubility and swelling processes (Soto and Oliva, 2012). The water solubility index is a measure of the amount of amylose that is released from inside the granule when it begins to lose its structure by the effect of water absorption (Huamani-H et al., 2020); the results obtained in the present work are shown in table 2. In this sense, Sindhu et al. (2019) reported values similar to those obtained in this study, with 31.32 % for buckwheat starch. Meanwhile, Martínez et al. (2015) reported a lower solubility value for potato starch (4.8 - 20.8 %).
An increase in starch solubility index has been reported with increasing temperature (Martínez et al., 2017), however, in the present work the maximum solubility index values were reached between 60 °C and 70 °C, with a decrement at 80 °C and 90 °C. Guizar et al. (2008) reported a similar behavior, with maximum solubility index values between 70 - 80 °C, and a subsequent decrease at 90 °C for sweet potato starch, attributed to the fact that when reaching the maximum solubility, the starch components (amylose-amylopectin) begin to interact with each other forming insoluble complexes, which precipitate decreasing the solubility index.
The swelling power indicates the ability of starch granules to hydrate under cooking conditions (Olu-Owolabi et al., 2011) and can be affected by their minor components (lipids, proteins, etc.) that prevent interaction with water, prior treatment, and processing conditions (Solarte-Montúfar et al., 2019). Martínez et al. (2015) and Babu and Parimalavalli (2014) reported a swelling power of 11 - 26.9 g g-1 for potato starch and 3 - 14 g g-1 for sweet potato starch, respectively, similar to those reported in this work.
As the temperature increases, the intergranular forces of the starch weaken, thus facilitating the penetration of water into the crystalline zone, so that the granules swell and increase in volume. Once the amorphous part is hydrated, the maximum swelling point occurs (Olu-Owolabi et al., 2011; Martínez et al., 2017). The results observed at 80 °C in this work are very close to the final gelatinization temperature (Tf: ~ 90 °C) evaluated by DSC, corroborating thet mentioned by Chacón et al. (2020), who attribute a direct relationship between solubility index and swelling power with starch gelatinization temperature. It is worth mentioning that various studies have attributed that solubility is mainly affected by amylose content, while amylopectin affects swelling power characteristics (Yadav et al., 2019).
Morphological and optical analysis
Scanning electron microscopy (SEM) and elemental analysis (EDS)
The starch sample showed a wide particle size distribution ranging from 14 to 69 µm, as well as an oval shape with a smooth surface (Figure 1). Conglomerates can be observed as a result of electrostatic charges between large and small particles. In this sense, Morales (2012) mentions a size of 5 to 100 µm for potato starch particles, while Ramíreza et al. (2017) reported an average size of 27.1 µm.
Both size and shape turn out to be important in the characterization of starches, since they have a great influence on their properties and applications (Jaiswal and Chibbar, 2017), these characteristics are related to amylose and amylopectin within starch granules, since the amylopectin chains form small oval granules, while amylose tends to form larger granules (Hernández et al., 2017). This is corroborated by Chuiza-Rojas et al. (2021) who reported a size of 6.2 µm and a relatively low amylose content of 19.28 % compared to that obtained in this study (24.35 %), which was reflected in larger particles (14 - 69 µm).
Scanning electron microscopy with energy dispersive detector (EDS) allows knowing part of the simple chemical composition of starch (Table 3). The potato starch sample showed a simple composition of 54.39 % carbon, 45.46 % oxygen, and 0.15 % potassium. Bahrami et al. (2019) reported a similar chemical composition for potato starch, with 59.31 % carbon and 40.69 % oxygen, while Hernández et al. (2017) indicated a composition of 76.43 % carbon and 23.23 % oxygen. The potassium detected in this test is due to the composition of the starch, since the presence of minerals such as calcium, magnesium, sodium, iron, phosphorus, and potassium has been reported (Meaño et al., 2014).
Dynamic Light Scattering (DLS)
DLS allows to determine the nanometric size of particles such as starch, as well as the size distribution (Ji et al., 2016). In this sense, the DLS results showed colloids with a nanometric size of approximately 120 ± 18.87 nm. The DLS procedure has a sonication step where the conglomerate particles are separated, which allowed measuring the size of the colloids present in the sample. Garcia-Gurrola et al. (2019) and Ang et al. (2022) reported a similar procedure used for DLS analysis and demonstrated that the increase in particle size is attributed to associations of amylopectin chains that induce a variation in starch properties, such as rheology.
The determination of the starch particles nanometric size resulted interesting, since these are for example, usually used in drug delivery systems (Zhou et al., 2014), however the characteristics that define the use of nanoparticles also consider the distribution in size and surface charge as they affect physical stability and distribution in vivo (Jimenez, 2022).
The polydispersity index (PDI), which shows the colloidal quality of the particles, was 0.3028 ± 0.12, showing the starch obtained a monodispersed distribution (narrow size distribution) according to previous reports which showed that values less than 0.7 indicate a monodisperse distribution and those in the range of 0.7 to 1 indicate low quality with a large size distribution (Lancheros et al., 2014; García-Gurrola et al., 2019; Dong et al., 2021). Sana et al. (2019) reported a particle size for corn starch of 175 to 248 nm with a PDI of 0.2 to 0.3 and Naderizadeh et al. (2018) indicated a particle size of 230 to 240 nm and PDI of 0.26 for potato starch particles, both studies showed similar PDI values to that obtained in the present study.
The zeta potential is indicative of the magnitude of electrostatic repulsion/attraction between particles that influence the stability of the colloid, the value obtained was 6.14 ± 0.41 mV, indicating an unstable suspension (± 0 - 10 mV); since this superficial charge is close to zero, the particles are considered neutral with a tendency to form aggregations and precipitate. The zeta potential is an important tool when making formulations, since stability tests are shortened by predicting it, reducing time and costs, in addition to improving the useful life and helping to understand the state of the surface of a nanoparticle (Jeong and Shin, 2018; Ahmad et al., 2020). Previous work has reported zeta potential in this range, Chavez da Silva et al. (2017) mentioned a value of - 8.67 mV and Haaj et al. (2014) pointed out values of 3 mV, considering that suspended solids have a neutral charge so close to zero. The variability of the zeta potential of colloids or suspended particles are influenced by the chemical nature of the polymer, pH, solvent, and the stabilizing agent (Piña-Barrera et al., 2021).
Color
Starch color is an important quality parameter, which influences the acceptability of the product by consumers (Too et al., 2022). In this study the colorimetric analysis of starch showed a medium luminosity (40.42 ± 0.44), since sodium metabisulfite or any other antioxidant that favored the whiteness of the starch was not used in the extraction. The values of the parameters a* (0.06 ± 0.02) and b* (2.86 ± 0.09) with an inclination to reddish and yellow color, respectively, indicate a tendency to neutral color (Martínez et al., 2019b). Verma et al. (2018) reported similar values (0.16 for a* and 2.58 for b*), however, they mentioned a higher luminosity (98.81). Likewise, Kumar et al. (2020) reported a luminosity of 96.68 and values of - 0.14 and 1.89 for parameters a* and b*, respectively, in both cases for potato starch.
Thermal analysis
Differential Scanning Calorimetry (DSC)
The DSC thermograms (Figure 2) allowed us to observe the changes that occurred in the potato starch gelatinization process, which presented an initial temperature (To) of said process to 61.78 ºC, while the maximum temperature (Tp) was observed to 64.45 ºC and the final temperature (Tf) was recorded at around 90 ºC, and the enthalpy of gelatinization (ΔH) was 10.96 J/g. These results are similar to those reported by Gonçalves et al. (2020), with values of To (55.1 ºC and 58.2 ºC), Tp (59 ºC and 62 ºC) and Tf (71.4 ºC and 74.4 ºC) and a ΔH of 11.8 J/g and 12.7 J/g, for recovered and commercial potato starch, respectively. On the other hand, Alvani et al. (2011) reported values of To (58.7 ºC to 62.5 ºC), Tp (62.5 ºC to 66.1 ºC) and Tf (68.7 ºC to 72.3 ºC) and ΔH that ranged between 15.1 and 18.4 J/g, for potato starches.
In the present work, an endothermic peak was observed at 114.56 ºC, attributed to the dissociation of the amylose-lipid complex. In this sense, Genkina et al. (2014) reported a peak around 95 ºC for corn starch and mentioned that the fusion of these complexes is a reversible process, contrary to the phase transition of amylopectin. These changes in the transition temperature of starch are attributed to its botanical source, its crystallinity and composition, since the amylose content is negatively correlated (Li et al., 2017; Han et al., 2019). Furthermore, lipids are a determining factor, since their presence results in a gelatinization process at higher temperatures (Alvani et al., 2011).
On the other hand, enthalpy values (ΔH) are positively correlated with crystal concentration, being an indicator of the loss of molecular order. Then, low enthalpy values indicate that less energy is required to start the gelatinization process. Enthalpy is also related to the shape, size, and presence of phosphate esters in starch granules (Alvani et al., 2011; Li et al., 2017; Li et al., 2020; Zhang et al., 2021).
Thermogravimetric analysis (TGA/DTGA)
Thermogravimetric analysis (TGA) and differential thermogravimetric analysis (DTGA) of potato starch describe its thermal stability (Figure 3). Starch shows three zones of weight loss within a temperature range of 20 - 500 °C. The first mass loss of 4.09 % detected at 49.22 °C is attributed to the evaporation of water, the other two regions comprised at 288.17 °C and 474.90 to 482.25 °C, represent the greater mass loss of 53.2 % and 96.17 %, respectively, due to the thermal degradation of starch and proteins (Thakur et al., 2021; Gui et al., 2022), the residual mass (ash) was detected at 600 °C (0.82 %). These results are similar to those reported by Gui et al. (2022), with two mass loss zones between 30 - 150 °C and 150 - 750 °C. Similarly, Li et al. (2022) pointed out two degradation zones in the same temperature ranges, specifically at 70 °C and 300 °C, in both cases for potato starch.
Structural analysis
NMR spectra
Figure 4 shows the 13C NMR spectrum of potato starch in the range of - 20 to 230 ppm. The NMR spectrum detected the presence of five peaks between 60 - 105 ppm. The peak recorded around 60 ppm is due to carbon 6 of the glucose unit, while the wide peak observed between 70 and 80 ppm is related to carbons 2, 3 and 5, C4 was detected at 80 ppm, while carbon 1 presents two peaks around 100 of ppm, thus corroborating the structure of the starch monomer (glucose) (Thérien-Aubin et al., 2007; Šoltýs et al., 2019).
The C1 resonance peak provides information about the conformation of the starch molecule, crystallinity, and double-helix symmetry. For type A crystallinity, the C1 resonance presents three peaks at 102, 101 and 100 ppm, whilst, type B (characteristic of potato starch) presents two peaks at 101 and 100 ppm, as observed in this study. According to Meaño et al. (2016) the amylose content for type B starches (25 - 30 %) is higher compared to type A, as is the size of the granules, which agrees with the results obtained in this investigation. The broad C1 resonance peak around 103 - 104 ppm is typical of single helices, both in amorphous and crystalline V phase, suggesting a higher proportion of short amylopectin chains attributed to degradation (Thérien-Aubin et al., 2007; Pardo et al., 2013; Li et al., 2017).
X-ray diffraction (XRD)
X-ray diffraction of potato starch was performed to examine its amorphous, semi-crystalline or crystalline structure. X-ray patterns identified crystalline structures, characteristic of amylopectin (Pardo et al., 2013), which can classify starch into types A-C. Type A crystallinity starch shows two strong diffraction peaks between 15° and 23° and a doublet at 17° and 18°, type B crystallinity produces a strong diffraction peak at 17° and low intensity peaks between 15 - 24°, while type C is a mixture of A and B (Ma et al., 2022).
As shown in Figure 5, the diffraction showed type B crystallinity patterns typical of potato starch, with a strong diffraction peak at 17.1° and low intensity peaks between a range of 5.6 - 26.2°, corroborating the results obtained by NMR. The degree of starch crystallinity was estimated at 34.73 %, values similar to those reported by Núñez-Santiago et al. (2011) and Gui et al. (2022) who reported 32.3 % and 31.5 % crystallinity, respectively, for commercial potato starch in both cases.
FTIR-ATR spectrophotometry
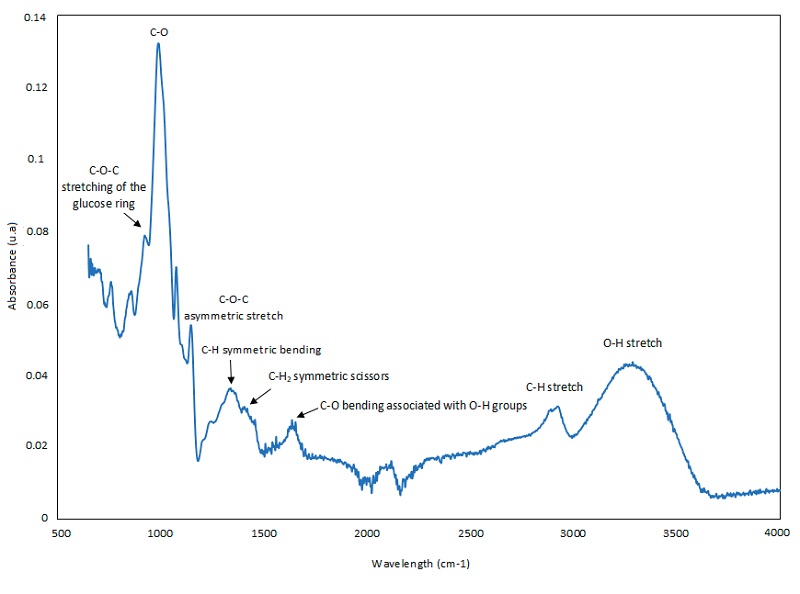
The glucose (starch monomer) structure previously detected with NMR, was corroborated with the detection of their functional groups by FTIR-ATR spectrophotometry (Figure 6). Signals were observed at wave numbers of 763, 858 and 929 cm-1, which are attributed to the stretching of the ether groups (C-O-C) of the glucose ring, in addition, the band observed between 993-1082 cm-1 was due to the C-O bonds stretching. The presence of an absorption band around 1157 cm-1 indicated an asymmetric stretching of the C-O-C groups. Symmetric double vibration of C-H bonds and symmetric deformation of CH2 groups were observed at wavelengths 1381 and 1437 cm-1, respectively. The absorption band around 3300 - 3600 and 2900 cm-1 indicated the presence of hydroxyl functional groups (-OH) and C-H bonds, respectively. The C-O bending associated with the -OH groups showed an absorbance peak around 1648 cm-1. The absorbance peak at 1415 cm-1 implies the presence of a symmetric scissors of the CH2 groups. The unusual CO2 peak (2358 cm-1) observed in the starch IR spectrum could be the result of the measurement conditions (Torres et al., 2015; Abdullah et al., 2018).
Conclusions
The extraction process has influence on the starch color parameters, mainly in the luminosity parameter. The NMR and XRD-ray diffraction allowed the identification of the potato starch crystallinity as type B. The SEM-EDS revealed particles of oval shape and the formation of conglomerates, with an elemental composition of carbon, oxygen, and potassium, and DLS showed a narrow size distribution with a zeta potential close to zero, considering the particles in suspension as neutral. In addition, the FTIR-ATR spectrum corroborated the chemical structure of starch, and the TGA thermogram showed the presence of different stages of weight loss, while the DSC thermogram showed the behavior of the starch gelatinization process, whose temperature showed a direct relationship with solubility and swelling power. The results found in the present work show the relationship between the chemical components of potato starch and its thermal, structural, and morphological behavior, which are important due to the wide range of applications attributed to this product. Residual potato is an excellent source of high-quality starch due to its physicochemical, thermal, and structural characteristics, resulting in a good alternative for the recycling of residual potato from the chips industry in Mexico. In addition, the use of this residual potato presents an excellent alternative to solve the environmental impact of agroindustrial waste.











 nueva página del texto (beta)
nueva página del texto (beta)