Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Archivos de cardiología de México
versión On-line ISSN 1665-1731versión impresa ISSN 1405-9940
Arch. Cardiol. Méx. vol.82 no.3 Ciudad de México jul./sep. 2012
Investigaciones clínicas
Autoimmunity as a possible predisposing factor for Stenotrophomonas maltophilia endocarditis
Autoinmunidad como posible factor predisponente para endocarditis por Stenotrophomonas maltophilia
Jorge R. Carrillo-Córdova, Luis M. Amezcua-Guerra*
Department of Immunology, Instituto Nacional de Cardiología Ignacio Chávez, Mexico City, Mexico.
* Corresponding author at:
Department of Immunology,
Instituto Nacional de Cardiología Ignacio Chávez. Juan Badiano 1,
Sección XVI, Tlalpan. Z.P. 14080. México City, México.
Tel.: +52 (55) 5573 2911; fax: +52 (55) 5573 0994.
E-mail address: lmamezcuag@gmail.com (L.M. Amezcua-Guerra).
Received 31 May 2011.
Accepted 13 March 2012.
Abstract
Only 40 cases of Stenotrophomonas maltophilia (S. maltophilia) endocarditis have been reported to date, and there is no description in patients with underlying autoimmunity.
A 23-year-old woman with systemic lupus erythematosus (SLE) overlapping rheumatoid arthritis (RA) and no risk factors for endocarditis was admitted in our hospital because of community-acquired tricuspid valve endocarditis. During hospitalization, she was complicated with pulmonary thromboembolism and pneumonia. Laboratory showed autoimmune diathesis featured by anti-cyclic citrullinated peptide (anti-CCP), anti-Sm, anti-Ro/SSA, anti-cardiolipin, anti-(β2 glycoprotein 1, and antinuclear antibodies, rheumatoid factor (RF), low complement, lymphopenia and C-reactive protein (CRP) of 425 mg/L.
S. maltophilia grew in serial blood culture sets. Empirical broad-spectrum antimicrobials were ineffective until trimethoprim/sulfamethoxazole (TMP/SMX) was added to therapy. One month after admission, the patient underwent successful surgical replacement of the tricuspid valve and the subsequent course was satisfactory, allowing her to be discharged 14 days after. Nowadays, she remains free of complaints and her cardiac, renal and pulmonary functioning is stable. Noteworthy is that all auto-antibodies have been persistently raised over time.
Here, we present a compilation of the available information about S. maltophilia endocarditis, and suggest that autoimmunity could be included as a novel predisposing factor for S. maltophilia endocarditis.
Keywords: Endocarditis; Stenotrophomonas maltophilia; Systemic lupus erythematosus; Rheumatoid arthritis; Mexico.
Resumen
Actualmente sólo existen 40 casos descritos de endocarditis por Stenotrophomonas maltophilia (S. maltophilia), ninguno de los cuales tenía una enfermedad autoinmune subyacente.
Una mujer de 23 años con imbricación de lupus eritematoso sistémico (LES) y artritis reuma-Artritis reumatoide; toide (AR), ingresó a nuestro Instituto por endocarditis de la válvula tricúspide adquirida en México comunidad. La paciente presentó como complicaciones tromboembolia pulmonar y neumonía, se encontraron anticuerpos contra péptidos cíclicos citrulinados (anti-CCP), antinucleares, anti-Sm, anti-Ro/SSA, anti-cardiolipina y anti-(β2 glucoproteína 1, factor reumatoide (FR), complementopenia, linfopenia y proteína C reactiva (PCR) de 425 mg/L.
Se observó crecimiento de S. maltophilia en hemocultivos seriados y el tratamiento antimicrobiano empírico no fue efectivo, sino hasta que se incluyó trimetoprim/sulfametoxazol (TMP/SMX). Después de un mes de hospitalización, la paciente fue sometida a remplazo quirúrgico de la válvula tricúspide y la evolución clínica subsecuente fue satisfactoria, permitiendo su egreso 14 días después. Actualmente, la paciente se encuentra asintomática y su función cardiaca, renal y pulmonar es estable. Llama la atención que todos los autoanticuerpos han permanecido elevados a través del tiempo.
Presentamos una recopilación de la información disponible acerca de la endocarditis por S. maltophilia, y proponemos que la autoinmunidad podría ser incluida como un nuevo factor de predisposición para esta entidad.
Palabras Clave: Endocarditis; Stenotrophomonas maltophilia; Lupus eritematoso sistémico; Artritis reumatoide; México.
Introduction
Stenotrophomonas maltophilia (S. maltophilia)isa Gram-negative, aerobic bacillus widely distributed in the environment. Although not highly virulent, S. maltophilia exhibits intrinsic resistance to multiple antibiotics and it is emerging as a relevant nosocomial pathogen isolated with increased frequency from diverse clinical specimens.1
Infective endocarditis by S. maltophilia is poorly characterized because only forty patient cases have been published to date. Prosthetic valves, chronic rheumatic carditis, indwelling vascular catheters, and intravenous drugs are well recognized as predisposing factors.2,3 However, there is no description for this infection in patients with autoimmune diseases. This seems to be the case of a patient with systemic lupus erythematosus (SLE) overlapping rheumatoid arthritis (RA), a rare entity often called ''rhupus syndrome'',4 who developed community-acquired S. maltophilia endocarditis.
Case presentation
In September 2007, a 21 year-old woman with a nine month history of arthritis in hands, wrists, knees and ankles was admitted in our hospital. Anti-cyclic citrullinated peptide antibodies (anti-CCP) and rheumatoid factor (RF) were positive. RA was diagnosed and a regimen including sulfasalazine, 2 g/day; prednisone, 5 mg/day; and hydrox-ychloroquine, 200mg/day was started with satisfactory response in the next six months. After that, she was lost from the follow-up in our outpatient clinic and an external physician diagnosed SLE because of new onset of malar rash and photosensitivity. Previous treatment was stopped and deflazacort, 4mg/day, was started.
In March 2009, the patient was admitted again in our hospital for one week with dyspnea, fever, chills, malaise, and chest pain. Examination revealed 115/bpm, 39 °C temperature, and tricuspid systolic murmurs on heart auscultation. Arthritis in hands, wrists, knees, and ankles, as well as photosensitivity and malar rash were found; whereas conjunctival hemorrhages, Janeway lesions, Osler nodes or Roth spots were absent. Noteworthy is that no invasive procedures were performed before admission and the patient had no risk factors for tricuspid valve endocarditis.
Laboratory screening showed hemoglobin 7.4g/dL, leukocytes 10.4 × 103/µL, platelets 86 × 103/µL, and high-sensitivity C-reactive protein (CRP) of 425 mg/L (normal value<9.2mg/L). Trans-thoracic echocardiography showed one vegetation, 21 x 16 mm, in the septal leaflet of the tricuspid valve, atrial intracavitary thrombi, and elevated pulmonary pressure (64mmHg); findings were confirmed by transesophageal echo. Multifocal pulmonary thromboembolism was confirmed by CT-angiogram and enoxaparin was initiated. Suspicion of infective endocarditis was raised and empiric intravenous antimicrobials, cephalothin and amikacin, were added to therapy.
On the fifth hospital day, S. maltophilia susceptible to ceftazidime, trimethoprim/sulfamethoxazole (TMP/SMX) and carbapenem grew in serial blood culture sets (automated BACTEC system®, Becton Dickinson, FR) obtained at admission and, three days after, cephalothin was changed to ceftazidime. Additionally, laboratory showed proteinuria > 3g/day, low C3 and C4 complement proteins, lymphopenia, positive antinuclear antibodies (speckled pattern, 1:5120 dilution), anti-Sm, anti-Ro/SSA, anti-cardiolipin and anti-β2 glycoprotein 1, as well as persistence of anti-CCP and RF. SLE overlapping RA was confirmed and prednisone, 60mg/day, was added; also, antiphospholipid syndrome (APS) was suggested due to pulmonary thromboembolism and circulating antiphospholipid (aPL) antibodies.
Patient's clinical course was complicated by cough, sputum, and persistent fever. A new CT-scan showed left basal pulmonary consolidation suggestive of pneumonia and antimicrobial therapy was changed to meropenem and TMP/SMX, with gradual resolution of symptoms in the following 10 days. Two subsequent blood culture sets were sterile.
One month after admission, she underwent surgical replacement of the tricuspid valve with a home-made, bovine pericardium prosthetic valve. Surgery was successful and the clinical course satisfactory. Patient was discharged 14 days after valve replacement on treatment with TMP/SMX, Coumadin, prednisone at 20mg/day, hydroxichloroquine at 200mg/day, and azathioprine at 100 mg/day.
Histology of the tricuspid valve showed unspecific changes consisting of chronic neutrophil infiltrates, large areas of neovascularization, and calcified fibrin thrombi. Whereas Libman-Sacks endocarditis and lupus valvulitis were ruled out. Culture of native valve was sterile.
After 18 months of follow-up in the outpatient clinic, the patient remains free of cardiac complaints and her renal and pulmonary functioning is stable, although finger deformities (Z-thumbnail and hallux valgus) and marginal bone erosions (metacarpal-phalangeal joints) have been detected. Interestingly, all autoantibodies have been persistently raised over time, including those APS-related antibodies.
Discussion
To date, there are only 40 cases of S. maltophilia endocarditis described worldwide, almost half of them in the past five years.2,3,5-8 Here, we describe the first case associated with autoimmunity, and take the opportunity to compile available information about S. maltophilia endocarditis.
Endocarditis by S. maltophilia endocarditis develops under specific conditions, with approximately 90% of patients bearing at least one predisposing factor. Established risks include prosthetic valves, intravenous drugs, endovascular devices, and chronic rheumatic carditis (Table 1). While all these factors were absent in our patient, an autoimmune diathesis characterized by SLE, RA and APS was noticeable. Moreover, she was not receiving immuno-suppressants other than low-dose corticosteroid before the onset of endocarditis. This suggests that immune disturbances associated with systemic autoimmunity may constitute a debilitating factor for the development of S. maltophilia endocarditis.
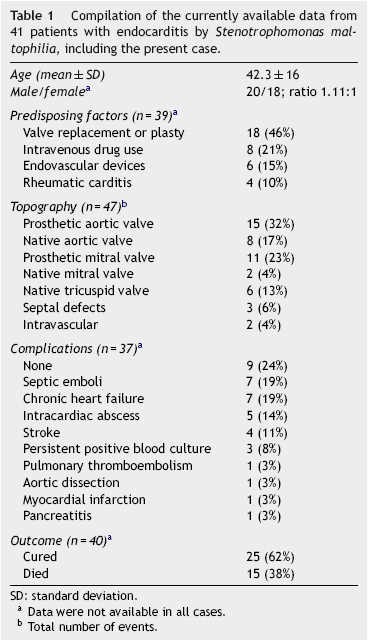
Patients with S. maltophilia endocarditis are often complicated by both infectious and thromboembolic events (Table 1). In this regard, our patient developed pneumonia and pulmonary thromboembolism (perhaps septic emboli) in presence of circulating aPL antibodies. Whether these aPL antibodies were part of the autoimmune diathesis or induced by infection remains unclear, although a role for S. maltophilia infection in the induction of aPL antibodies is provocative. In this line of thought, diverse aPL antibodies induced by infection have been related to endothelial activation, thrombin generation, impairment of fibrinolysis, and major embolic events in patients with infective endocarditis other than S. maltophilia.9 All aPL antibodies have persisted raised over time in our patient, suggesting that there was a pathogenic role in thrombotic events instead of being just an epiphenomenon.
Therapy for S. maltophilia infection is troublesome because the broad resistance to antimicrobials that is characteristic of this pathogen.1 Antibiotic treatments used for bloodstream infections, such as vancomycin and broad-spectrum (β-lactams, have no effect on S. maltophilia. This may partially explain the high mortality (38%) and frequency of complications (~90%) reported in S. maltophilia endocarditis (Table 1). Currently, combined antimicrobial therapy is indicated with TMP/SMX as the agent of choice.3 As could be noticed in our patient, infection control was achieved only when TMP/SMX was included within the antimicrobial scheme and the patient underwent surgery.
To the best of our knowledge, this is the first report on endocarditis due to S. maltophilia in a patient with systemic, overlapping autoimmunity and no other identifiable predisposing factors. We suggest that autoimmunity could be included in the list of predisposing factors for S. maltophilia endocarditis, and believe that physicians need to be aware of this rare but life-threatening condition that seems to be increasing its incidence. Moreover, a high index of suspicion is required for the detection of this organism and the consequent early introduction of TMP/SMX in the antimicrobial therapy.
The present manuscript is derived from protocol number 08-622, which was revised and accepted by the institutional ethics committee.
Funding
This manuscript was supported with personal funding.
Conflict of interest
There are no conflicts of interest to disclose.
References
1. del Toro MD, Rodríguez-Baño J, Herrero M, et al. Clinical epidemiology of Stenotrophomonas maltophilia colonization and infection: a multicenter study. Medicine (Baltimore) 2002;81:228-39. [ Links ]
2. Katayama T, Tsuruya Y, Ishikawa S. Stenotrophomonas maltophilia endocarditis of prosthetic mitral valve. Intern Med 2010;49:1775-7. [ Links ]
3. Bayle S, Rovery C, Sbragia P, et al. Stenotrophomonas maltophilia prosthetic valve endocarditis: a case report. J Med Case Reports 2008;2:174. [ Links ]
4. Amezcua-Guerra LM. Overlap between systemic lupus erythematosus and rheumatoid arthritis: is it real or just an illusion? J Rheumatol 2009;36:4-6. [ Links ]
5. Müller-Premru M, Gabrijelcic T, Gersak B, et al. Cluster of Stenotrophomonas maltophilia endocarditis after prosthetic valve replacement. Wien Klin Wochenschr 2008;120:566-70. [ Links ]
6. Takigawa M, Noda T, Kurita T, et al. Extremely late pacemaker-infective endocarditis due to Stenotrophomonas maltophilia. Cardiology 2008;110:226-9. [ Links ]
7. Rostoff P, Paradowski A, Gackowski A, et al. Stenotrophomonas maltophilia pacemaker endocarditis in a patient with d-transposition of the great arteries after atrial switch procedure. Int J Cardiol 2010;145:e92-5. [ Links ]
8. Carmona JA, Velasco MJ, Zambrana JL, et al. Tricuspid infective endocarditis caused by Stenotrophomonas maltophilia in a patient without risk factors. Med Clin (Barc) 2004;123:357. [ Links ]
9. Kupferwasser LI, Hafner G, Mohr-Kahaly S, et al. The presence of infection-related antiphospholipid antibodies in infective endocarditis determines a major risk factor for embolic events. J Am Coll Cardiol 1999;33:1365-71. [ Links ]














