Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista de la Sociedad Química de México
versión impresa ISSN 0583-7693
Rev. Soc. Quím. Méx vol.46 no.1 Ciudad de México ene./mar. 2002
Investigación
Phytotoxicity of crude alkaloid fractions from Erythrina americana
Rosario García-Mateos,1 Cecilia Beatriz Peña-Valdivia,2* and Marcos Soto-Hernández2
1 Área de Química. Preparatoria Agrícola. Universidad Autónoma Chapingo, México 56230, Chapingo.
2 Botánica Colegio de Postgraduados, Carretera México-Texcoco km 35.5, Montecillo México 56230, México. Phone/Fax: + (595) 2 02-47. E-mail: cecilia@colpos.colpos.mx
Recibido el 10 de enero del 2001.
Aceptado el 26 de septiembre del 2001.
Resumen
Se evaluaron los efectos de extractos alcaloideos obtenidos de las semillas de Erythrina americana en la germinación de frijol (Phaseolus vulgaris L.) y maíz (Zea mays L.), así como en las actividades fotoquímicas de cloroplastos aislados de chícharo (Pisum sativum L.). Los extractos no afectaron la germinación, pero concentraciones bajas de alcaloides libres (< 2.5 mg / mL) inhibieron la síntesis de ATP y el consumo de protones inducido por luz en los cloroplastos aislados. Los alcaloides liberados (< 5 mg / mL) estimularon la síntesis de ATP, pero no afectaron el consumo de protones inducido por luz en los cloroplastos aislados. Aunque podría ser necesaria mayor evidencia experimental, los resultados mostraron la baja fitotoxicidad de las fracciones crudas de alcaloides de E. americana.
Palabras clave: Metabolitos secundarios, germinación, fotosíntesis.
Abstract
Effects of alkaloid extracts from Erythrina americana seeds were evaluated on common bean (Phaseolus vulgaris L.) and maize (Zea mays L.) germination and also on the photochemical activities of isolated chloroplasts of pea (Pisum sativum L.). The alkaloid mixtures did not affect the germination, but low concentrations of free alkaloids (< 2.5 mg / mL) inhibited the ATP synthesis and the light induced proton uptake in isolated chloroplasts. The liberated alkaloid fraction (< 5 mg / mL) stimulated the ATP synthesis, but did not affect the proton uptake. Although more experimental evidence could be necessary, these results demonstrated a low phytotoxicity of the crude alkaloid fractions of E. americana.
Keywords: Secondary metabolites, germination, photosynthesis.
Introduction
There is an increasing body of evidence to suggest that alkaloids affect negatively the growth of microorganisms, plants and both vertebrate or invertebrate animals [1]. The multiplicity and variety of susceptible target suggests mechanisms of action for the alkaloids that are either common to all types of organism [2], or perhaps depend on paralleled modes of action. Wink and Latz-Brunning [3], referred to many basic plant processes that might be targeted by allelochemicals, e.g. membrane stability, protein synthesis, photosynthetic electron transport and light induced proton uptake. These authors developed bioassays to elucidate the mode of action of some alkaloids that inhibited radicle growth in cress. Many of these alkaloids where found to be inhibitors in more than one assay, but some were active in none. It has been shown that the Fabaceae family has allelophatic activity against plants, and this plant family is recognized by its alkaloid production. Some of this alkaloids are weak inhibitors of germination e.g. quinolizidine alkaloids, found in lupinus (Lupinus spp.) [4]. Others alkaloids inhibit the germination of lettuce (Lattuca sativa L.) and lawngrass mixture [1] and affect the germination of various other species [5].
The Erythrina genus contains some toxic alkaloids which are found mainly in the seeds [6]. Hastings [7] observed low and moderate inhibition of corn and bean germination, respectively, in a bioassay of E. americana bark; beside, some details about the LD50 Erythrina genus alkaloids on experimental animals are known [8, 9]. However, it can be say that information about allelophatic effect of Erythrina genus in plants is scarce. The knowledge of its eventual phytotoxicity could be used in natural herbicide studies. The focus of the present work was to explore the effect of the crude alkaloid fraction on germination of common bean (Phaseolus vulgaris L.) and maize (Zea mays L.), and also on ATP synthesis and light induced proton uptake in isolated chloroplasts from pea (Pisum sativum L.) leaves.
Results and discussion
Identification of the alkaloids. α-Erythoidine and β-erythoidine were the most abundant alkaloids in the "free" fraction (0.55 mg per 100 g of dry weight), whereas in the "liberated" alkaloids fraction (0.47 mg per 100 g of dry weight) were identified erysodine, erysovine and erysopine (Fig. 1). The relative alkaloid proportions were calculated from the GC peak areas and the time retention of the alkaloids: 82.6 mm2 and 30.25 min to α-erythoidine and 17.4 mm2 and 23.70 min to β-erythoidine in the free alkaloid fraction; 39.6 mm2 and 17.85 min to erysodine, 56.5 mm2 and 18.50 min to erysovine and 3.6 mm2 and 17.75 min to erysopine in the liberated alkaloids fraction, respectively. This results confirm the composition of the fractions of E. americana seed previously reported [8, 10].
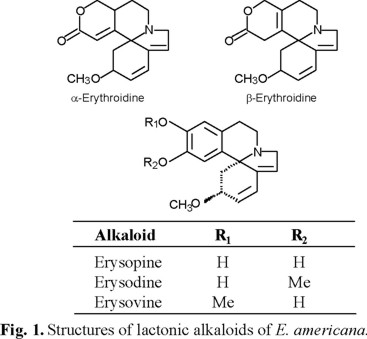
Effect of the alkaloids on bean and maize seeds germination. Neither fraction, "free" or "liberated" alkaloids, affected the percentage of germination of bean and maize (Fig. 2 and 3). The percentage of germination ranged between 90 and 100 % and there were not any significant difference between the controls and the alkaloid treatments. These results differ from those reported by Hastings [7], who stressed that in a bioassay E. americana bark induced low and moderate inhibition on corn and bean germination, respectively. This author did not provide information about the type of extract used; however, the small differences between the effects on seed germination of seeds and bark extracts of E. americana could result of the different composition, since bark contain furanoid derivative, flavonoids, one polyphenol, tannins, and terpenoids [17].
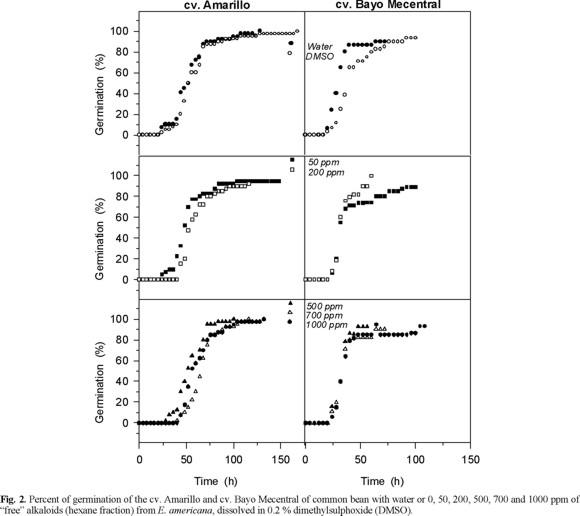
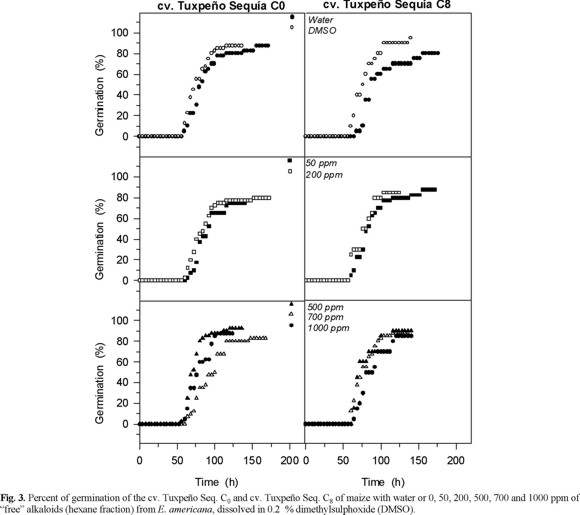
Effects of the "free" alkaloids on ATP synthesis and on light induced proton uptake in isolated pea chlorolplasts. Low concentration of free alkaloids (< 2 mg / mL) inhibited the ATP synthesis (photophosphorylation) and the light induced proton uptake, but higher concentration (3.5-7 mg / L) had the opposite effect, e.g. they stimulated the ATP synthesis and proton uptake (Fig. 4). The ATP synthesis could be inhibited indirectly by a proton gradient depression or directly by inhibition of the ATP synthase (CF0-CF1 complex) [13, 18]. The inhibition of the photophosphorylation by low concentrations (≤2.5 mg mL-1) of the free alkaloids fraction could be a result of the depression of the proton electrochemical gradient, because also inhibited the proton uptake (Fig. 4). These results showed that low concentrations (2.5 mg mL-1) of free alkaloids fraction of E. americana affect negatively the photochemical activities of the chloroplasts. However, the stimulation of photophosphorylation and proton pump with concentrations higher than 3 mg mL-1 suggests that only one of the lactonic alkaloids could be inhibitor of the photochemical activities in isolated chloroplast and the other could alleviated this effect. Another possibility could be the synergetic effect of the mixture dimethylsulfoxide (solvent) and alkaloids, which is hardly detected with the control treatments. To this respect, it has been observed that solvents used for dissolving chemical effectors inhibit energy transducting reactions [13, 19].
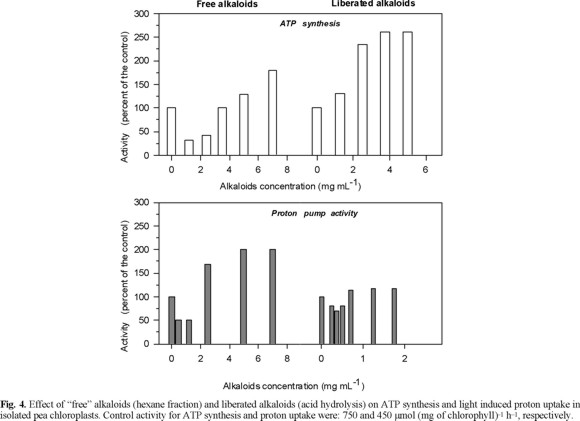
Effect of the "liberated" alkaloids fraction on the ATP synthesis and light induced proton uptake in isolate pea chloroplasts. The "liberated" alkaloid fraction (1-5 mg mL-1) activated ATP synthesis (150 %), but none of the tested concentrations affected the proton uptake (Fig. 4). The increase of photophosphorylation induced for the "liberated" alkaloid fraction could be a result of the direct interaction of the alkaloids with the CF0-CF1, which result in an ATP synthase activation without effect on the proton uptake [13]. Although with the available information it is not possible to explain how this activation could occur. These alkaloids could induce some structural conformation of the CF0-CF1 complex, by modifying the polarity of the complex microenvironment, which produce an activation.
All these results showed a low phytotoxicity of the crude alkaloids of E. americana in the physiological processes evaluated: germination, photophosphorylation and light induced proton uptake on the tested plant material, and they contrast with those obtained in other studies with experimental animals in which a high toxicity of the α and β-erythroidines is described [16, 20, 21, 22]. However there are some similarities between the results of the present study and those obtained by Garcia-Mateos et al. [8], who described the LC50 of the free and liberated alkaloids on Daphnia magna (393 and 79 ppm, respectively) and also the toxicity of the liberated alkaloids to Panagrellus redivivus.
Conclusions
This work showed that "free" (methanol soluble) and "liberated" (acid hydrolysis liberated) alkaloid fractions extract from E. americana seeds do not affect significantly the photochemical activities in isolated chloroplasts. Beside, these fractions do not affect the germination of common bean and maize seeds. Although more experimental evidence could be necessary, including the effects of this extracts on other plant species, like weeds, and in other biochemical processes, like lipids, carbohydrates and nitrogen metabolism, it was demostrated the low phytotoxicity of the crude alkaloids of E. americana.
Experimental
Plant materials. The seeds of E. americana were collected in Oaxtepec, Morelos (40 km southwest Mexico City). Voucher specimen of the plant were deposited at the CHAPA Herbarium (Especialidad de Botánica, Colegio de Postgraduados) under the number 112. The authenticity of the sample was certified by the curator of the Herbarium of the Especialidad de Botanica, Colegio de Postgraduados. The dried seeds were milled manually and the alkaloids were extracted exhaustively. The Bayo Mecentral and Amarillo cultivars of common bean were developed in the National Institute for Research in Agriculture, Forestry and Animal Husbandry, Mexico, and reproduce in an experimental plot at Colegio de Postgraduados, Montecillo, Mexico, located at 19° 29' North latitude and 98° 53' West longitude, and 2250 m above sea level, with an average annual temperature of 15.9 °C and a mean rainfall of 691 mm [9]. The Tuxpeño Drought C0 and Tuxpeño Drought C8 cultivars of maize were obtained from the International Maize and Wheat Improvement Center (CIMMYT), Mexico.
Preparation of the alkaloid fractions. Alkaloids were extracted from the seed flour by the same method previously used [10]. Intact seeds were airdried at 70 ºC and milled, the flour was extracted for 48 hours with hexane by Soxhlet extraction. Alkaloids in the hexane fraction were washed with 1M sulphuric acid (3 × 50 mL), and the aqueous acidic phase was adjusted to pH 8 using solid NaHCO3. Finally, an extraction with CH2Cl2 (3 × 100 mL) gave the hexane "free" alkaloid fraction (0.22 %). The defatted flour of each fraction was next extracted in a Soxhlet for 48 hours with MeOH, the extract was evaporated under vacuum, and the residue taken up in 0.2 % sulphuric acid. The acidic solution was extracted with CH2Cl2 to remove traces of fat. The aqueous phase pH was raised to 8 with NaHCO3 and was extracted with CH2Cl2 (3 × 100 mL) to give a methanolic fraction containing the "free" alkaloids; the acidic hydrolysis of the methanolic residue gave the "liberated" alkaloids (obtained by hydrolysis of alkaloids occurring as glycosides). The solvent of each sample was evaporated and the residue was dried in a desiccator and weighed.
Identification of the alkaloids. The alkaloids were identified previously on base of their Rf values, UV fluorescence, color reaction after treatment with cromogenic reagents (e.g. Dragendorff reagent), co-chromotography with authentic samples of Erythrina alkaloids and subsequently with combined gas chromatography-mass spectrometry (GC/MS). The alkaloid-trimethylsilylderivatives (free alkaloids and liberated alkaloid fractions, separately) were obtained and identify by GC/MS as was previously described [20]. One or two mg of crude alkaloid mixture were treated with N,O-bis (trimethylsilyl)acetamide (25 µL) in acetonitrile (25 µL), using a Teflon lined screw-cap vials to prevent evaporation, during 30 min [20]. Mass spectra were determined on a JMS-AX 505 HA (JEOL) mass spectrometer coupled with a GC Hewlett Packard 5890, Series II, equipped with a flame ionization detector and a PAS 1701 silicone capillary column, (25 m × 0.32 mm × 0.25 um), Hewlett Packard (Palo Alto, California), via a two-stage Watson-Biemann separator. The temperature of the ion source was 220 °C and the acceleration and ionization potentials were 3 kV and 70 eV, respectively. Alkaloids were identified by comparison with α- and β-erythroidines, erysovine, erysodine and erysopine trimethylsilylderivatives, which were obtained using the same method previously described [20].
Evaluation of maize and bean seeds germination. The effect of the "free" alkaloid fraction was evaluated on common bean (cv. Bayo Mecentral and cv. Amarillo) and maize (cv. Tuxpeño Drought C0 and cv. Tuxpeño Drought C8) seeds germination as was previously described [21]. The germination was evaluated using 25 seeds on a Petri dish, at 25 ± 1 °C in the dark, and four replications were included. The alkaloids were dissolved on 0.2 % DMSO. A stock solutions of alkaloids (5 g L-1) was used to obtain 30, 200, 500, 700 and 1000 mg L-1 solutions and 25 mL of each solution was added to each Petri dish. Also, the treatments with water or DMSO (without alkaloids) were included. Seed germination was quantified each 12 h during ten days.
Evaluation of photophosforilation (ATP synthesis) and light-dependent proton uptake (proton pump activity) in isolated pea chloroplasts. Chloroplasts were isolated from fresh leaves of pea growing in a greenhouse, as was previously described [12]. Light-dependent ATP formation and light-dependent proton uptake were quantified in a suspension of freshly lysed chloroplasts as the pH of the medium rose 8.0 and 6.0, respectively, as was previously described [12, 22]. These photochemical activities were evaluated in 3 mL of an assay medium containing chloroplasts (10 mg of chlorophyll mL-1), 0.1 mM sorbitol, 0.01 M KCl, 0.05 mM methylviologen and 1 mM tricine, plus 1 mM and 3 mM KH2PO4 in the case of ATP synthesis. The suspension was stirred and maintained at 22 °C, and after one minute of illumination (400 µmol photons m-2 s-1) the light source was switched off, and the total pH change of the suspension was quantified by titration with a calibrate solution of 0.1 N HCl. The pH was measured with a microelectrode connected to an expanded scale potentiometer [12]; and light-dependent pH changes were recorded by a 2210 LKB recorder.
Statistical analysis. The statistical procedures included analysis of variance (ANOVA) and multiple comparison of means by Tukey test. Data analysis were performed with the Statistical Analysis System (SAS) software [23].
Acknowledgement
The authors gratefully acknowledge the provision of the maize seeds (cv. Tuxpeño Drought C0 and Tuxpeño Drought C8) by the International Maize and Wheat Improvement Center (CIMMYT), Mexico.
References
1. Stermitz, F.R.; Bebfsky, G.N.; Ng, D.; Singer, M.C. J. Chem. Ecol. 1989, 15, 521-530. [ Links ]
2. Wink, M., in: The Alkaloids, Vol. 43, Cordell, G., Ed., Academic Press, San Diego, 1993, 1-118. [ Links ]
3. Robinson, T. Science 1974, 184, 430-435. [ Links ]
4. Wink, M.; Latz, B., in: Insights into Allelopathy. Dakshini, M., K. M.; Einhelling, F. A., Eds., American Chemical Society, Symposium Series No. 582, American Chemical Society, Washington, D.C., 1995, 117-126. [ Links ]
5. Muzquiz de la Cuadra, M. 1998. Personal communication.
6. Hargreaves, R. T.; Johnson, R. D.; Millington, D.S.; Mondal, M. H.; Breavers, W.; Becker, L.; Young, C.; Rinehart, K. L. Lloydia 1974, 37, 569-580. [ Links ]
7. Hastings, R. B. Econ. Bot. 1990, 44, 336-348. [ Links ]
8. García-Mateos, R. Estudio químico-biológico de los alcaloides de Erythrina. Tesis Doctoral, Programa de Fisiología Vegetal, Colegio de Postgraduados. Montecillo, Méx. México, 1996. [ Links ]
9. García-Mateos, R. M.; Soto-Hernández, M.; Martínez-Vázquez, M. Ciencia Ergo Sum 2000, 7, 166-170. [ Links ]
10. Abdullah, M. I.; Barakat, I. E.; Games, D. E.; Ludgate, P.; Mavragaiis, V. G.; Ratnayake, V. U.; Jackson, A. H. Ann. Missouri Bot. Gard. 1979, 66, 533-540. [ Links ]
11. Domínguez, X. A.; Alarcón, J. B. J. Ethnopharmacol. 1985, 13, 139-156. [ Links ]
12. Peña-Valdivia, C. B.; Rodríguez-Flores, L.; Tuena de Gómez-Puyou, M.; Lotina-Hennsen, B. Biophys. Chemist. 1991, 41, 169-174. [ Links ]
13. Peña-Valdivia, C. B.; Lotina H., B., Boletín de Educación Bioquímica 1991, 8, 52-61. [ Links ]
14. Tuena de Gómez-Poyou, M.; Ayala, G.; Darzon, A.; Gómez-Poyou, A. J. Biol. Chem. 1984, 259, 9472-9478. [ Links ]
15. Craig, L. E., in: The Alkaloids, Vol. V, Manske, R. H. F.; Holmes H. L., Eds., Academic Press, London, 1955, 265-293. [ Links ]
16. Lozoya, X.; Lozoya, M. Flora medicinal de México. 1. Plantas indígenas. Instituto Mexicano del Seguro Social. México, D. F. 1982, 174-192. [ Links ]
17. Payne, L. G.; Foley, J. P., in: Chromatography and Pharmaceutical Analysis, Ahuja, S., Ed., American Chemical Society Symposium Series No. 512, American Chemical Society, Washington, D.C., 1992, 85-99. [ Links ]
18. García, E. Modificaciones al Sistema de Clasificación de Köppen. Instituto de Geografía, UNAM, México, D.F., 1988. [ Links ]
19. Games, D. E.; Jackson, A. H.; Khan, N. A.; Millington, D. S. Lloydia 1974, 37, 581-588. [ Links ]
20. Soto H. M.; Jackson, A. H. Planta Medica, 1994, 60, 175-177. [ Links ]
21. Ellis, R. H. Handbook of Seed Technology. Principles and Methodology. Vol. I, International Board for Plant Genetics Resources, Rome, 1985. [ Links ]
22. Dilley, R. A., in: Methods in Enzimology, Vol. 24, San Piero A., Ed., Academic Press, New York, 1972, 68-74. [ Links ]
23. SAS Institute. SAS/STAT User´s Guide. SAS Institute, North Carolina, 1986. [ Links ]














