Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista latinoamericana de química
versión impresa ISSN 0370-5943
Rev. latinoam. quím vol.38 no.3 Naucalpan de Juárez dic. 2010
Effect of photo–oxygenation on biological activities of some commercial egyptian essential oils
A.M.Dawidar*ª, M.Abdel–Mogibª, M.M. Abou–Elzahab, M.A. Berghot, M.Mahfouzª, A.H.El–Ghorabb, Kh. Hussienª
ª Chemistry Dept., Fac, of Sci., Mansoura Univ., Mansoura, Egypt. *Corresponding author E–mail: adawidar@yahoo.com
b Flavour and Aromatic Chem. Dept., National research Center, Dokki, Cairo, Egypt.
Received May 2010.
Accepted December 2010.
ABSTRACT
The components of five common commercial Egyptian essential oils; caraway, cumin, basil, thyme, and rose oils, were identified using gas chromatography/ mass spectrometry (GC/MS) and Kovats retention index. For each oil the main components were reported. Their biological activities, including deoxyribonucleic acid (DNA) damage, protein cleavage, antioxidant, antimicrobial and anti–diabetic effect were tested. The effect of photo–oxygenation reaction on their biological activities, using UV–irradiation in the presence of singlet oxygen sensitizer was investigated.
Keywords: Essential oils; Caraway; Carum carvi; Cumin; Cuminum cyminum; Basil; Ocimum basilicum; Thyme; Thymus vulgaris; Rosa damascema; Free radical scavenging; DNA and protein cleavage; Antioxidants; Antimicrobial; Hypoglycaemia; Photo–oxygenation.
RESUMEN
Se analizaron cinco aceites esenciales comerciales de Egipto: aceite de alcaravea, comino, albahaca, tomillo y rosas, por cromatografía de gases acoplado a espectrometría de masas y utilizando el índice de retención de Kovats para identificar a sus componentes. Se reportan los componentes principales de cada aceite. También se probo la actividad biológica de cada acite sobre el daño al ADN, la ruptura de proteínas, la actividad antioxidante, antimicrobiana y antidiabética. También se investigó el efecto de la fotooxigenación, por irradiación con luz UV. En general la fotooxigenación aumentó el daño sobre el AND y las proteínas, lo que pone de manifiesto la importancia de proteger los aceites esenciales de la luz y la oxigenación.
Palabras clave: Aceites esenciales; Alcaravea; Carum carvi; Comino; Cuminum cyminum; Albahaca; Ocimum basilicum; Tomillo; Thymus vulgaris; Rosa damascema; atrapamiento de radicales libres; rompimiento de ADN y proteínas; Antioxidante; Antimicrobiano; Hipoglucemia; Foto–oxigenación.
INTRODUCTION
Essential oils are of great importance and are used extensively in our daily–life in various domains of human activities such as aromatherapy, food flavoring fragrances, cosmetics and pharmacy. The investigated essential oils are obtained from local market for Caraway (Carum carvi L.); Cumin (Cuminum cyminum L.); Basil (Ocimum basilicum); Thyme (Thymus vulgaris L.) and Rose (Rosa damascema L.). The biological properties of these essential oils as well as their applications have been studied extensively by several workers, (Nicola et al., 2005; Peter, 2001; Micklefield, 2000; Opalchenova, 2003).
The unsaturated compounds of essential oils are easily oxidized to products as alcohols, ketons, acids or aldehydes, depending on the oxidation conditions and consequently there will be a change in their biological properties.
This article aims to study the effect of photo–oxygenation on biological activities of the mentioned commercial essential oils. This was performed by using photolytic singlet oxygen sensitizer. The biological properties that have been investigated are DNA and protein oxidative damage, antioxidant, antimicrobial and antidiabetic activities (Dawidar et al., 2010; Hussien, 2006).
MATERIALS AND METHODS
Chemicals
Caraway, cumin, basil, thyme and rose essential oils were purchased from Harraz Drug stores, Bab El–Khalk, Cairo Egypt. The chemicals used in this study are of analytical reagents or molecular biology grade and were obtained from Sigma, BDH chemicals LTD. Albino rats, with an average body weight of 100 to 120 g, were purchased from National Research Center, Giza, Egypt. The chromosomal DNA was supplied by one of the authors (M. Mahfouz).
Instrumentation
Infrared spectra were measured on thin films cast from chloroform, and performed on Mattson 5000 FT–IR spectrometer by sample oil films. Ultra violet spectra were determined using Unicom UV/Vis, UV spectrophotometer. The photo–oxygenation reaction was carried out by a photo–oxygenation apparatus which consists of a sodium lamp (Phillips G/5812 SON) for photolysis, cylindrical jar (15 w x 20 l x 30 h cm) filled with ethanol, sample tube inserted in the jar and cooling unit with alcoholic thermometer. Dry oxygen was supplied from external cylinder. The distance between sodium lamp and sample tube was 10 cm. Optical rotation of the investigated oils were determined using Euromax microscopes b.v polarimeter (nr. Po 450 model) with tube length 10 cm and sodium lamp. Refractive index were performed by ABBE–Refractometer model G VEB Carl Zeiss JENA.
Isolation of essential oils by hydro–distillation
Each oil (200 g) was placed in a 2 L round–bottom flask with 1 L of deionized water. The solution was steam–distilled for 4 h. The distillate was extracted with 100 mL of dichloromethane. The extract was dried over anhydrous sodium sulfate and the solvent was removed by using a rotary evaporator.
Infrared absorption bands of caraway oil
IR, [v,(oil film),cm–1]: 3350, br. band (OH), 3019 (CH, aromatic, str.), 2964, 2933, 2875 (CH, aliphatic, str.), 1702 (non conjugated CO), 1673 (conjugated CO), 1606, 1575, 1459 (C=C, aromatic and aliphatic), 1382 (isopropyl moiety), 1305, 1211, 1172 (C=C–O–C–aliphatic),1054, 833 (terminal methylene group), 727.
Ultra Violet absorption of caraway oil
UV, λmax. (ethanolic solution), nm: 317 and 259.
Infrared absorption bands of Cumin oil
IR, [v, (oil film), cm–1]: 3380 – 3494 br. band (OH, str,), 2964, 2933, 2875, 2831 (CH, aromatic and CH aliphatic), 2724 (CH, aldehydic proton, str.), 1671, 1698 (non conjugated and conjugated CO), 1605, 1573 (C=C aromatic and aliphatic), 1459, 1430, 1388 (isopropyl moiety), 1305, 1284, 1211, 1172 (C=C–O–C–aliphatic ), 1054, 835 (terminal methylene group), 757, 727.
Ultra Violet absorption of Cumin oil
UV, λmax. (ethanolic solution), nm: 318 and 260.
Infrared absorption bands of Basil oil
IR, [v, (oil film), cm–1]: 3455 br. band (OH), 2954, 2927, 2867 (CH, aromatic and aliphatic, str), 1722 (non conjugated CO) 1639, 1612, 1511 (C=C aromatic and aliphatic), 1450, 1373 (isopropyl moiety), 1299, 1245, 1035, 995 (propenyl group), 914, 815.
Ultra Violet absorption of Basil oil
UV, λmax. (ethanolic solution), nm: 285, 278 and 251.
Infrared absorption bands of Thyme oil
IR, [v, (oil film),cm–1]: 3388–3229 br. band (OH), 2962 (CH, aromatic, str.), 2927, 2877 (CH, aliphatic, str.), 1444, 1429, 1376 (isopropyl moiety), 1288, 1228, 1157, 808, 759.
Ultra Violet absorption of Thyme oil
UV, λmax. (ethanolic solution), nm: 281, 275 and 242.
Infrared absorption bands of Rose oil
IR, [v (oil film), cm–1]: 3434–3377 br. band (OH, str.), 3011 (CH, aromatic, str.), 2965, 2927, 2879 (CH, aliphatic, str.), 1663 (conjugated CO), 1451, 1379 (isopropyl group), 1217, 1047, 762, 700 and 668.
Ultra Violet absorption of Rose oil
UV, λmax. (ethanolic solution), nm: 247, 285 and 304.
GC/MS identification of essential oils components
The components of the essential oils were identified by GC/MS instrument (Varian GC interfaced to Finnigan SSQ 7000 Mass Selective Detector, MSD) with ICIS V2.0 data system for MS identification of the GC components. The column used was DB–5 (J & W Scientific, Folosm, CA) cross–linked fused silica capillary column (30 m long, 0.25 mm internal diameter) coated with polydimethylsiloxane (0.5 pm film thickness). The oven temperature was programmed from 50ºC for 3 min., at isothermal, then heating by 7 ºC / min. to 250 ºC and isothermally for 10 min., at 250 ºC. Injector temperature was 200 ºC and the volume injected was 0.5 pl. Transition–line and ion source temperature were 250 ºC and 150 ºC respectively. The mass spectrometer had a delay of 3 min. to avoid the solvent peak and then scanned from m/z 50 to m/z 300. Ionization energy was set at 70 eV. Kovats retention index were determined by co–injection of the sample with a solution containing homologous series of n–hydrocarbons (C9–C22) in a temperature programmed run identical to that described for the samples under investigation.
The separated components were identified by matching with the NIST mass–spectral library data, comparison of the Kovats retention index with those of authentic components and with authentic spectra (Adams, 1995). The percentage composition of the essential oil was computed in each case from GC peak areas without correction.
Photo–oxygenation
A solution of each of caraway, cumin, basil, thyme, rose essential oils, d–carvone and geraniol (1 ml) in chloroform (50 ml) and a few milligrams of hematoporphyrine (HP) was irradiated externally by mean of a sodium lamp (Philips G/5812 SON) at –20 °C for 24 hrs. During the irradiation a continuous stream of dry oxygen was allowed to pass through the reaction mixture at a slow rate to avoid the solvent evaporation. The hydroperoxide formation was followed up by TLC (silica gel 60 GF 254, 10% Potassium iodide as a spray reagent). The solvent was removed at 20 °C/0.1 torr to give oily material. The crude photoproduct was purified from hematoporphyrine (HP) by silica gel CC using petroleum ether, ethyl acetate (3:2) as an eluent.
DNA Assay
A solution of essential oil (10μl) in 10 μl 1% dimethyl sulfoxide (DMSO) was mixed with 10 μl of chromosomal DNA solution. The mixture was incubated for 3 hours at 37°C. DNA was analyzed by using horizontal agarose gel electrophoresis. The gels were photographed using a digital camera. The same procedure was repeated with photo–oxygenated essential oils and authentic sample before and after photo–oxygenation (Dawidar et al., 2010; Hussien, 2006).
Protein Assay
A solution of essential oil (10μl) in 10 μl 1% DMSO was mixed with 10 μl of bovine serum albumin solution. The mixture was incubated for 3 hours at 37°C. The electro–phoresis apparatus was connected to the power supply with 80 V for 2 hours. The gel was photographed after staining. The same procedure was repeated with photo–oxygenated essential oils and authentic sample before and after photo–oxygenation (Dawidar et al., 2009).
Antioxidant Activity
Assay of superoxide dismutase activity: Superoxide dismutase (SOD) activity was assayed by the procedure of Niskikimi et al., (1972).
1,1'–Diphenylpicrylhydrazyl (DPPH) assay: In this spectrophotometric assay, a stable DPPH radical is used as a reagent (Miliauskas et al., 2004). Inhibition of DPPH in percent (I %) was calculated. Both methods were used in vitro and tests were carried out in triplicate.
Antimicrobial Activity
The investigated samples were individually tested against four bacterial strains including Bacillus thuringenesis, Bacillus subtillus as Gram positive and Escherichia coli, Klebsiella pneumonia as Gram negative, as well as two fungal strains; Apsergillus flavus and Trichoderma harzianum. The discagar diffusion method was employed for the determination of antimicrobial activities of the investigated samples. Filter paper discs (6 mm in diameter) were impregnated with 50 pl of the oil and placed on the inoculated plates. The procedure used by Lacobellis et al., (2005) was applied. All tests were performed in duplicate.
Glycaemic activity
Chemicals and experimental animals Streptozotocin (STZ) was obtained from sigma. Other chemicals used were purchased from EDWIC. All experiments were performed using adult male albino rats, with an average body weight of 100 to 120 g, purchased from National Research Center, Giza, Egypt. The rats were housed in steel mesh cage and provided with commercial diet and tap water, and allowed to adapt to the environment for one week after their arrival before the experiment started.
Experimental procedure
Diabetes was induced by intravenous injection of streptozotocin (65 mg/Kg body weight; STZ was dissolved in 0.05M sodium citrate buffer, PH 4.5) into a lateral lail vein. The control rats were injected with the same volume of vehicle. Three days after the injection, the diabetic animals were randomly assigned to two groups: STZ–diabetic untreated rats (group 1) and STZ–diabetic treated rats (group 2). The group 2 individually received investigated oils by gavages (2 ml/ kg body weight). During two hours of treatment, the animals were housed in steel mesh cage and were given free access to water and a powdered diet. After 2 hours blood samples were collected from the medial canthus of the eye for measuring blood glucose levels by One Touch blood glucose meter from life scan (Eddouks et al., 2004) (Johnson & Johnson Company USA). The procedure used by (Cheng–Tzu Liu et al., 2006) was applied.
RESULTS AND DISCUSSION
GC/MS and Kovats retention index analyses were performed for the five investigated essential oils (Tables 1). The identification of d–carvone was supported by IR (1673 cm–1An absorption band appeared at 1720 cm–1, characteristic to the carbonyl group of both perilladehyde and isomenthone. d–Carvone in caraway essential oil, was detected by UV absorption (λmax 317 nm).
In agreement with the cumin oil, it was reported by Georgiev et al. (1988) that Bulgarian, Egyptian and Turkish cumin essential oil components were cuminalde–hyde (22–46%), p–cymene (9–23%), β–pinene (15–18%) and γ–terpinene (4–23%).
It is important to report that the sesqui–terpenes, caryophyllene, khusimone, carotol, guaiol, cedranone, cadinol, which were identified from the volatile fraction of commercial basil oil were not detected by Karaway et al., (1974), Hoertrammer et al., (1964) in a laboratory prepared Egyptian basil oil. Egyptian basil oil, basil volatile fraction and laboratory prepared sample, resembles type 2 of Benin basil, type 2 of Italian basil, type 3 of Turkish basil, type 1 of Iranian basil and European type basil oil, all of them contain linalool and methyl chavicol as main constituents (Panuwat et al., 2003). The oils differed especially with respect to the relative amounts of linalool, methyl chavicol and the sesquiterpenes.
The aromatic odour of the thyme oil is mainly related to thymol and carvacrol. The thyme commercial oil investigated here belongs to the thymol chemotype; which may be adulterated by origenum oil (carvacrol type oil) (Schulz et al., 2003). Literature survey revealed no information about the effect of photo–oxygenation on the biological properties of the essential oils. The photo–oxygenation was evidenced from the change of densities (Table 2). No significant changes in the refractive indices were observed, before and after photo–oxygenation. The hydro–peroxide formation was detected by TLC and potassium iodide, as well as by the IR spectrum.
Reactive oxygen species such as free radicals like hydroxyl radical ( OH) or non free radical such as singlet oxygen (1O2) have a damage effect on deoxy ribonucleic acid (DNA) and proteins. This damage may cause aging, cancer and many other di–seases (Gülçin et al., 2003). Table 3 reports the results for the five investigated essential oils, in addition to d–carvone and geraniol as the major constituents of caraway and rose oils. The table revealed that the effect of the oils before photo–oxygenation was in the following descending order: rose> basil> thyme=cumin> caraway, while the protein oxidative damage order was rose = thyme > basil = cumin. Caraway and d–carvone showed no effect on both DNA and protein. After photo–oxygenation, caraway, cumin, basil, thyme and rose essential oils all showed strong DNA and protein cleavage effect and also d–carvone, while geraniol showed a moderate effect. In the protein oxidative damage, all showed strong effect except caraway and rose which showed no effect. Generally, photo–oxygenation enhanced the damage effect of both DNA and protein.
OH) or non free radical such as singlet oxygen (1O2) have a damage effect on deoxy ribonucleic acid (DNA) and proteins. This damage may cause aging, cancer and many other di–seases (Gülçin et al., 2003). Table 3 reports the results for the five investigated essential oils, in addition to d–carvone and geraniol as the major constituents of caraway and rose oils. The table revealed that the effect of the oils before photo–oxygenation was in the following descending order: rose> basil> thyme=cumin> caraway, while the protein oxidative damage order was rose = thyme > basil = cumin. Caraway and d–carvone showed no effect on both DNA and protein. After photo–oxygenation, caraway, cumin, basil, thyme and rose essential oils all showed strong DNA and protein cleavage effect and also d–carvone, while geraniol showed a moderate effect. In the protein oxidative damage, all showed strong effect except caraway and rose which showed no effect. Generally, photo–oxygenation enhanced the damage effect of both DNA and protein.
Antioxidants act as radical–scavengers, and inhibit lipid peroxidation and other free–radical mediated process. Therefore, they are able to protect the human body from several diseases (Epe et al., 1993). In our study two complementary test systems were used which are 1,1–diphenyl–2–picrylhydrazyl (DPPH) (Miliauskas et al., 2004) and superoxide dismutase (SOD) (Niskikimi et al., 1972) like activity assays to evaluate the investigated essential oils, before and after photo–oxidation.
The antioxidant bioassay results for the investigated essential oils are reported in table 4.
Before photo–oxygenation, the DPPH assay indicated that the investigated regular essential oils have free radical scavenging activity (as antioxidant effect) in the following descending order; rose > thyme > basil > cumin > d–carvone > caraway > geraniol. The SOD assay showed that caraway and d–carvone do not have effect, but cumin, thyme and geraniol on the other hand exhibited large free radical scavenging activity. After photo–oxygenation, DPPH assay indicated that the investigated photolytic essential oils have free radical scavenging activity in the descending order; basil > d–carvone > thyme > cumin>caraway = rose > geraniol. The SOD assay showed also that generally the antioxidant property is enhanced by photo–oxygenation in the descending order; cumin > d–carvone > basil > thyme > caraway > geraniol = rose. The increasing of free radical scavenging activity by photo–oxygenation may be explained by the probable increasing of phenolic content (Al–Ismail and Aburjai, 2004).
It was reported by Tomaino et al., (2005) that basil and thyme essential oils exhibited good antioxidant properties which is in agreement with our results (Table 4). Al–Ismail and Aburjai (2004) reported that the higher content of phenolic compounds means the higher antioxidant activity except in the case of the water extract of dill seeds, which had the lowest phenolic content but showed higher antioxidant activity than anise seed extract. It is worthwhile mentioning that cumin and basil essential oils were reported by Aruna and Sivara–makrishnan (1996) as anticarcinogenic.
Essential oils as antimicrobial agents received comprehensive studies by several authors (Nicola et al., 2005; Hudaib, 2002). The investigation of the five commercial essential oils before and after photo–oxygenation as antimicrobial agents were performed against the fungal (Aspergillus flavus and Triechoderma harzianum) and bacterial (Gram positive: Bacillus thuringenesis, Bacillus subitilus and Gram negative: Escherichia coli, Klebsiella pneumonia) strains. The obtained results are reported in table 5.
After photo–oxygenation, caraway and basil oils acrtivity was not changed, d–carvone, and geraniol showed a complete inhibition of fungal grow; while for cumin, rose and thyme oils weak increase of inhibition effect was observed.
The antimicrobial activity of cumin oil could be due to the high level of cuminalde–hyde (23.37%), whereas the antimicrobial activity of caraway oil is apparently due to d–carvone (42.61%). This is in agreement with Lacobellis et al., (2005). It is most likely that the decreasing effect of the photo–oxygenation reaction on the antimicrobial activity of some investigated samples probably due to the conversion of the alcoholic derivatives to the corresponding aldehydes or ketons.
The search for hypoglycaemic agents from plants used in traditional medicine was reported (Fararh et. al, 2004). In this article, we tried to test caraway, cumin, basil , thyme and rose essential oils as hypoglycaemic agents, along with d–carvone and geraniol as major individual constituents on streptozotocin (STZ)– diabetic rats, via induction by intravenous injection (65 mg/kg body weight in citrate buffer, pH 4.5) in a lateral tail vein. For measuring blood glucose levels, a blood droplet was collected from the eye medial canthus after anesthetization using diethyl ether, and measured by "One–Touch" blood glucose meter. The results (Table 6 and Fig. 1) indicated that before photo–oxygenation, the tested samples are arranged in the hypo–glycemic descending order; cumin > thyme > basil > rose=d–carvone > caraway>geraniol. After photo–oxygenation, caraway oil efficacy was enhanced as hypoglycaemic agent. On the other hand for cumin and d–carvone hypoglycaemic effect was inverted by photo–oxygenation.
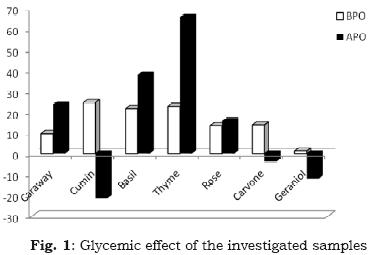
Several workers have studied various essential oils from plant origin as glycaemic agents. For example, it was documented by Fararh et al. (2002), that essential oil of Nigella sativa has hypoglycaemic effect in STZ–induced diabetic rats and rabbits. Others also reported a hypoglycaemic effect of garlic oil in diabetic animals (Cheng–Tzu Liu et al., 2006), and in diabetic patient it was shown that garlic oil can correct hypo–glycaemia (Duncan, 1999), Significant and potent hypoglycaemic activity was observed by myrtle oil in diabetic rats (Sepici et. al., 2004). The aqueous extract of caraway (Carium carvi) produced a significant decrease on blood glucose levels in STZ–diabetic rats (Eddouks et al., 2004).
CONCLUSION
In addition to identification of the volatile components of oils of caraway, cumin, basil, thyme, and rose using GC/MS and Kovats retention index, biological activities, including DNA damage, protein cleavage, antioxi–dant, antimicrobial and anti–diabetic effect were studied for d–carvone, geraniol and the 5 oils before and after photo– oxygenation of the oils. The chemical composition could be of value in identifying adulteration and different chemo–types of these oils.
Generally, photo–oxygenation enhanced the damage effect of both DNA and protein, which revealed the importance of essential oils to protect from light and oxygen. The tested oils act as free radical scavengers, both before and after photo–oxygenation. The increasing of free radical scavenging activity by photo–oxygenation may be explained by the probable increasing of phenolic content.
The antimicrobial studies indicated that caraway and basil are good antifungal agents, while d–carvone and geraniol are antibacterial against Gram + bacteria. After photo–oxygenation, caraway and basil oils acrtivity was not changed, d–carvone, and geraniol showed a complete inhibition of fungal grow; while for cumin, rose and thyme oils weak increase of inhibition effect was observed
As hypoglycemic agents, the tested samples are arranged in the descending order, before photo–oxygenation: cumin > thyme > basil > rose= d–carvone > caraway>geraniol; after photo–oxygenation, caraway oil efficacy was enhanced as hypoglycaemic agent. On the other hand for cumin and d–carvone hypoglycaemic effect was inverted by photo–oxygenation.
REFERENCES
Adams, R. P. (1995) Identification of volatile oil components by gas chromatography/mass spectroscopy. USA: Allured Publishing Corporation. [ Links ]
Al–Ismail, Kh. M.; Aburjai, T. (2004) Antioxidant activity of water and alcohol extracts of chamomile flowers, anise seeds and dill seeds. J. Sci. Food Agric., 84: 173–178. [ Links ]
Aruna, K.; Sivaramakrishnan, V. M. (1996) Anticarcinogenic effects of the essential oils from cumin, poppy and basil. Phytother. Res. 10: 577–580. [ Links ]
Burt, S. (2004). Essential oils: their antibacterial properties and potential applications in foods–a review. International Journal of Food Microbiology. 94: 223–253. [ Links ]
Cheng–Tzu, L.; Pei–Linn, W.; Chong–Kuei, L.; Hunry, H.; Lee–Yan, S. (2006) Antidiabetic effect of garlic oil but not diallyl disulfide in rats with streptozotocin–induced diabetes. Food and Chemical Toxicology 44: 1377–1384. [ Links ]
Dawidar, A. M.; Abdel–Mogib, M; El–Ghorab, A. H.; Hussien, Kh. (2009). Chemical composition and effect of photooxygenation on biological activities of Egyptian commercial spearmint and dill essential oils. Mansoura J. Chem. 36: 49–60. [ Links ]
Dawidar, A.; Hussien, Kh. (2010) Essential oils Composition, photo–oxidation, Bioactivity. Cairo, Egypt: Osiris Bookstore. [ Links ]
Duncan, M. G. (1999) The effects of nutritional supplements on the treatment of depression, diabetes, and hypercholesterolemia in the renal patient, J. Renal Nutr. 9: 58–62. [ Links ]
Eddouks, M.; Lemhadri, A.; Michel, J. (2004) Caraway and caper: potential anti–hyperglycemic plants in diabetic rats. J. Enthopharma. 94:143–148. [ Links ]
Epe, B.; Haring, M.; Ramaiah, D.; Stopper, H.; Abou–Elzahab, M. M.; Adam, W.; Saha–Moller, C. R. (1993) Carcinog. 14: 2271–2276. [ Links ]
Fararh, K. M.; Atoji, Y.; Shimizu, Y.; Shiina, T.; Nikami, H.; Takewaki, T. (2004) Mechanisms of the hypoglycaemic and immunopotentiating effects of Nigella sativa L. oil in streptozotocin–induced diabetic hamsters, Research in Veterinary Science 77: 123–129. [ Links ]
Fararh, K. M.; Atoji, Y.; Shimizu, Y.; Takewaki, T. (2002) Isulinotropic properties of Nigella sativa oil in Streptozotocin plus Nicotinamide diabetic hamster, Research in Veterinary Science 73: 279–282. [ Links ]
Georgiev, E.; Badr, F. H. M.; Kostadinova, S. I. (1988) Comparative studies on Bulgarian, Egyptian and Turkish cumin (Cuminum cyminum) Nauchni Tr. –Visch, Inst. Khranit. Vkusova Prom. St., Ploudiv 35: 239–248. [ Links ]
Gülçin, I.; Oktay, M.; Kireçci, E.; Küfrevioglu. O.I. (2003) Food Chemistry 83: 371–382. [ Links ]
Hoertrammer, L.; El Hamidi, A. E.; Richter, G. (1964) J. Pharm. Sci. 53: 1033–1036. [ Links ]
Hudaib, M.; Speroni, E.; Di Pietra, A. M.; Cavrini, V. (2002). GC/MS evaluation of thyme (Thymus vulgaris L.) oil composition and variations during the vegetative cycle J. Pharm. Biomed. Anal. 29: 691–700. [ Links ]
Hussien, Kh. (2006) Chemical and biological studies on terpenoids of some commercially available essential oils, Ph.D.Thesis; Mansoura university, Egypt. [ Links ]
Iacobellis, N. S.; Lo Cantore, P.; Capasso, F.; Senatore, F. (2005). Antibacterial activity of Cumium cyminum L. and Carum carvi L. essential oils. J. Agric. Food Chem. 53: 57–61. [ Links ]
Karawya, M. S.; Hashim, F. M.; Hifnawy M. S. (1974). Oils of jasmin, rose and cassie of Egyptian origin. Bull. Fac. Pharm., Cairo Univ. 13: 183–192. [ Links ]
Lacobellis, S.; Pietro, L.; Francesco, C.; Felice. S. (2005) J. Agric. Food. Chem. 53: 57–61. [ Links ]
Micklefield, G. H.; Greving, I.; May, B. (2000). Effects of peppermint oil and caraway oil on gastroduodenal motility. Phytotherapy Research 14: 20–23. [ Links ]
Miliauskas, G.; Venskutonis, P.R.; Beek. T.A. (2004). Screening of radical scavenging activity of some medicinal and aromatic plant extracts. Food Chemistry 85: 231–237. [ Links ]
Niskikimi, M.; Roa, N.A.; Yagi. K. (1972). The occurrence of superoxide anion in the reaction of reduced phenazine methosulfate and molecular oxygen. Biochem. Biophy. Res. Commun. 46: 849–854. [ Links ]
Opalchenova, G.; Obreshkova, D. (2003). Comparative studies on the activity of basil–an essential oil from Ocimum basilicum L.–against multidrug resistant clinical isolates of the genera Staphylococcus, Enterococcus and Pseudomonas by using different test methods. Journal of Microbiological methods, 54: 105–110. [ Links ]
Panuwat, S.; Joseph, M.; Kees, S.; Stephen, B.W. (2003) J. Agric. Food Chem., 51: 3197–3207. [ Links ]
Peter, K. V. (2001). Handbook of Herbs and Spices, first published, Woodhead Publishing Limited and CRC Press LTC. Cambridge England. [ Links ]
Schulz, H.; Quilitzsch, R.; Kruger, H. (2003). Classification of Essential Oils with FT–IR Spectroscopy in Routine Quality and Process Control. Journal of Molecular Structure, 661–662: 299–306. [ Links ]
Sepici, A.; Gürbüz, I.; Çevik, C.; Yesilada, E. (2004). Hypoglycaemic effects of myrtle oil in normal and alloxan–diabetic rabbits. Journal Ethnopharmacology. 93: 311–318. [ Links ]
Tomaino, A.; Cimino, F.; Zimbalatti, V.; Venuti, V.; Sulfaro, V.; De Pasquale, A.; Saija, A. (2005). Influence of heating on antioxidant activity and the chemical composition of some spice essential oils. Food Chemistry. 89: 549–554. [ Links ]














