Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista latinoamericana de química
versión impresa ISSN 0370-5943
Rev. latinoam. quím vol.38 no.3 Naucalpan de Juárez dic. 2010
Antifungal activity of extracts and terpene constituents of aerial parts of Juniperus Lucayana
Yarelis Ortiz Núñezª*, Iraida Spengler Salabarriab, Isidro G. Colladoc, Rosario Hernández–Galánc
ª Instituto de Investigaciones Fundamentales en Agricultura Tropical Alejandro de Humboldt (INIFAT), Calle: 2, Esq.1, Santiago de las Vegas, CP 17200, Cuba. *To whom correspondence should be addressed: Tel.: +53 7 57 90–10; Fax: + (53–7) 57 90–14; e–mail: yareliso@inifat.co.cu
b Centro de Estudios de Productos Naturales, Universidad de La Habana, Zapata y G. Vedado, Cuba.
c Departamento de Química Orgánica, Facultad de Ciencias, Universidad de Cádiz, 11510 Puerto Real, Cádiz, Spain.
Received July 2010.
Accepted December 2010.
ABSTRACT
Ethanolic and hexane extracts from the aerial parts of Juniperus lucayana were assayed against the phytopathogenic fungus Botrytis cinérea by the poisoned food technique. The hexane extract showed to have a higher antifungal activity than ethanolic extract. Fractionation of hexane extract by silica gel open column chromatography and HPLC afforded seven known compounds sandaracopimaric acid (1), 4–epi–dehydroabietic acid (2), oplopanone (3), oplodiol (4), nephtediol (5), 7α–hydroxycallitrisic acid (6), and 7–oxocallitrisic acid (7). Compound 1 showed a significative fungal growth inhibition during the assay. The effect of compounds 2–7 on spore germination fungus was also studied.
Keywords: Juniperus lucayana, terpenoids, antifungal activity, Botrytis cinerea, growth inhibition, spore germination.
RESUMEN
Los extractos etanólicos y n–hexánico obtenidos a partir de las partes aéreas de Juniperus lucayana fueron evaluados sobre el hongo fitopatógeno Botrytis cinerea por el método de envenenamiento del medio. El extracto n–hexánico mostró una mayor actividad antifúngica que el etanólico. El fraccionamiento del extracto n–hexánico mediante cromatografía de columna abierta sobre gel de silice y CLAE permitió el aislamiento de siete compuestos conocidos, el ácido sandaracopimárico (1 ), el ácido 4–epi–dehidroabiético(2), la oplopanona (3), el oplodiol (4), el neftediol (5), el ácido 7α–hidroxicallitrisico (6), y el ácido 7–oxocallitrisico (7). El compuesto 1 presentó una inhibición significativa del crecimiento del hongo durante el experimento. Adicionalmente se estudió el efecto de los compuestos 2–7 sobre la germinación de esporas del hongo.
Palabras clave: Juniperus lucayana, terpenoides, actividad antifúngica, Botrytis cinerea, inhibición del crecimiento, germinación de esporas.
INTRODUCTION
In recent years, public pressure to reduce the use of synthetic fungicides in agriculture has increased (Shafique et al., 2007). Concerns have been raised about both the environmental impact and the potential health risk related to the use of these compounds. In contrast, natural product–based fungicides have the ability to decompose rapidly, reducing risk to the environment with the added advantage in that they have both unique modes of action and low mammalian toxicity (Duke et al, 2003; Ortiz et al., 2008). During our search for antifungal substances from Cuban plants, we found that the ethanolic extracts from the wood and aerial parts of Juniperus lucayana Britton (Cupressaceae) exhibited antifungal activity against Botrytis cinerea, with IC50 values ranging from 125 to 250 μg/mL (Ortiz et al., 2004). This fungus is a serious pathogen that attacks economically important crops such as lettuce, carrots, tobacco, strawberry and grapes (Aleu et al., 2001; Daoubi et al., 2005). Juniperus species have been extensively investigated as a source of natural products with potential antimicrobial, acaricidal and insecticidal activities (Karaman et al., 2003; Schmidt, 2004; Barrero et al., 2005). To our knowledge, no fungicidal studies had been carried out on this species. In previous works we reported the bioassay–guided fractionation of etha–nolic extract from the wood of J. lucayana which afforded three new sesquiterpenes along with six known sesquiterpenes and two flavonoids (Ortiz et al., 2007). In this work, we reported the antifungal activity of the n–hexane and ethanolic extracts from the aerial parts of J. lucayana, as well as fractionation of the n–hexane extract by open column chromatography and HPLC to afford the isolation of seven compounds, first time reports for this species.
MATERIAL AND METHODS
General experimental procedures
The melting points (m.p.) were determined on a Reichert–Thermovar apparatus. The optical rotations were measured in CHCl3 solution on a Perkin–Elmer 341 polari–meter. IR spectra were recorded on a Mattson Genesis spectrophotometer, series FT–IR. 1H and 13C NMR measurements were obtained on Varian Inova 400 and 600 MHz NMR spectrometers, using SiMe4 as the internal reference. HPLC was performed with a Hitachi/Merck L–6270 apparatus equipped with an UV/Vis detector (L 4250) and a differential refractometer detector (RI–71). TLC was performed on Merck Kie–segel 60 F254 layers, 0.2 mm thick. Sepha–dex LH–20 and Si–gel (Merck) were used for column chromatography. Semi–preparative HPLC purification was conducted using a Si–gel column Lichrospher Si–60 column (10 μm, 1 cm wide, 25 cm long).
Plant material
J. lucayana was collected at the Institute of Fundamental Research on Tropical Agriculture ''Alexander von Humboldt'' in Santiago de las Vegas, Cuba, in March, 2005 and identified by Dr. Pedro Sánchez. A specimen of this plant is deposited in the herbarium of the ''Instituto de Ecología y Sistemática de las plantas en Cuba'' with the number HAC–42498.
Extraction and isolation
The air–dried powdered aerial parts (460 g) of J. lucayana were extracted using successively n–hexane and ethanol in a Soxhlet apparatus, for 4 h. The solvent was evaporated under reduced pressure to yield 31.7 g of an ethanol crude extract ( JFetd). The n–hexane extract was cooled at –10 0C for 24 h, yielding an insoluble fraction. The defatted hexane extract (JFhex, 15.0 g) was fractionated by Sephadex LH–20 column chromatography using a mixture of n–hexane, CHCl3, and MeOH (2:1:1) yielding six fractions (F1–6). TLC revealed that fractions F–3 and F–4 contained the same products. These fractions were purificated by means of column chromatography on Si–gel with CHCl3, and MeOH and CHCl3 mixtures containing increasing percentages of MeOH to give the compounds 1 (91.2 mg) and 2 (19.0 mg). Fraction F–1 was subjected to Sephadex LH–20 column yielding three fractions (F7–9), Fraction F–8 was purificated by column chromatography on Si–gel elu–ting with MeOH and CHCl3 mixtures containing increasing percentages of MeOH to give 15 fractions (F10–24). Finally, fractions F–11, F–19, and F–20 were thus purificated by means of semi–preparative HPLC column [mixture of acetone and n–hexane (20:80), 2.8 mL. min–1] to afford compounds 3 (8.4 mg), 4 (4.3 mg), 5 (11.2 mg), 6 (1.9 mg), and 7 (4.4 mg).
Microorganism
B. cinerea culture used in this work, Bc 2100, was obtained from the ''Colección Española de Cultivos Tipos (CECT)'', Universidad de Valencia, Facultad de Biología, Spain, where a culture of this strain is on deposit.
Antifungal assays
Poison food technique
Extracts and fractions (F1–6) were dissolved in ethanol to give final of 500 mg/ L for fractions and concentrations ranging from 125 to 500 mg/L for extracts. Compound 1 was evaluated at 150 mg/L. Antifungal assays were then carried out in accordance with the poison food technique (Soundharrajan et al, 2003). The solutions were added to a glucose–malt–peptone–agar [61 g/L of glucose (20 g)–malt (20 g)–peptone (1 g)–agar (20 g), pH 6.5–7.0]. The final ethanol concentration was identical in both control and treated cultures. The medium was poured into sterile plastic Petri dishes measuring 9 cm in diameter and 1.0–cm diameter my–celial discs of fungus cut from an actively growing culture were placed at the center of the agar plates. Inhibition of radial growth was measured during 6 days. Growth inhibition was calculated as the percentage of inhibition of radial growth relative to the negative control. Two independent assays were conducted, each in triplicate. The results are shown as mean values of colony diameters; (±SD).
Spore germination
Compounds 2–7 were dissolved in ethanol–water (1:1) at 100 and 200 mg/L. B. cinerea conidia used for this experiment were collected from cultures of fungi growing in the PDA media. The conidia were collected from the plates with 1 mL sterile distilled water, passed through a glass wool filter to remove hyphae, diluted, counted, and immediately used in the bioassay. Spore germination assays were carried out on multi–well microscope slides containing 10 wells. Each well was filled with 3 μL of the compound solutions at 100 and 200 mg/L, sterilized by filtration, together with conidia (2 μL, to a final concentration of 1 x 105 conidias/mL) of the fungus to be tested. Assays with sterile distilled water and water–ethanol, were used as controls. The plates were incubated at 25 °C and after 8 h, numbers of germinated and non–germinated conidias were counted in a light microscope and the spore germination percentage was calculated. Three wells per compounds concentration were prepared, and three areas per well were recorded (each containing 50 conidia). The experiment was performed by duplicate.
RESULTS AND DISCUSSION
Ethanolic and hexane extracts from the aerial parts of Juniperus lucayana were assayed against the phytopathogenic fungus B. cinerea, using the poisoned food technique (Soundharrajan et al., 2003). As observed in Figure 1 , the hexane extract (JFhex) proved to be more active than ethanolic extract (JFetd), at different concentrations evaluated. The hexane extract exhibited a significant inhibition in fungal growth at concentrations of 250 and 500 mg/L (50.1 and 61.5%, respectively), while the ethanolic extract was less effective. In both extracts, in concentrations lower than 250 mg/L, a delay in fungal growth was observed.
Although the doses recorded in our experiments are high (125–500 mg/L) these results are highly relevant, since a fungicidal effect is not previous observed in products derived from aerial parts of J. lucayana.
As mentioned before, the hexane extract was more effective on B. cinérea than ethanolic extract. Therefore, we decided as a first step to isolate the active principles from hexane extract. The hexane extract was chromatographed over a Sephadex LH–20 column (n–hexane/CHCl3/MeOH, 2:1:1). All fractions obtained, F1–6, were also tested against B. cinérea using the same assay. Fractions F–1, F–3, and F–4 exhibited a 59.2, 71.5 and 70% inhibition in the growth of this fungus, respectively, at concentrations
of 100 mg/L (see Figure 2). On the basis of their antifungal activity and their thin–layer chromatographic profiles, fractions F–1, F–3 and F–4 were purified by a combination of Si–gel column and semi–preparative HPLC analysis affording seven known compounds. The structures of the compounds were identified by physical and spectroscopic data measurement (mp,  , IR, 1H NMR, 13C NMR and 2D NMR) and by comparing the data obtained with published values, such as sandaracopimaric acid ((–)–pimara–8(14), 15–dien–19–oic acid) (1) (Edwards et al., 1960; Dang et al., 2005), 4–epi–dehydroabietic acid (2) (San Feliciano et al., 1992), oplopanone (2a–methyl–5a–isopropil–7α–oxomethylbiciclo[4.3.0]nonan–2p–ol) (3) (Su et al., 1995; Kuo et al., 2002), oplodiol (7–eudesmene–1P,4P–diol) (4) (Minato and Ishikawa, 1967; Tchuendem et al., 1999), nephtediol ((1S, 4R, 7S)–germacra–5E, 10(15)–diene–1,4–diol) (5) (Kitagawa et al., 1987), 7a–hydroxycallitrisic acid (7a–hy–droxyabieta–8,11,13–trien–19–oic acid) (6) (De Pascual et al., 1983) and 7–oxocallitrisic acid (7–oxoabieta–8,11,13–trien–19–oic acid) (7) (Prinz et al., 2002) (see Figure 3).
, IR, 1H NMR, 13C NMR and 2D NMR) and by comparing the data obtained with published values, such as sandaracopimaric acid ((–)–pimara–8(14), 15–dien–19–oic acid) (1) (Edwards et al., 1960; Dang et al., 2005), 4–epi–dehydroabietic acid (2) (San Feliciano et al., 1992), oplopanone (2a–methyl–5a–isopropil–7α–oxomethylbiciclo[4.3.0]nonan–2p–ol) (3) (Su et al., 1995; Kuo et al., 2002), oplodiol (7–eudesmene–1P,4P–diol) (4) (Minato and Ishikawa, 1967; Tchuendem et al., 1999), nephtediol ((1S, 4R, 7S)–germacra–5E, 10(15)–diene–1,4–diol) (5) (Kitagawa et al., 1987), 7a–hydroxycallitrisic acid (7a–hy–droxyabieta–8,11,13–trien–19–oic acid) (6) (De Pascual et al., 1983) and 7–oxocallitrisic acid (7–oxoabieta–8,11,13–trien–19–oic acid) (7) (Prinz et al., 2002) (see Figure 3).
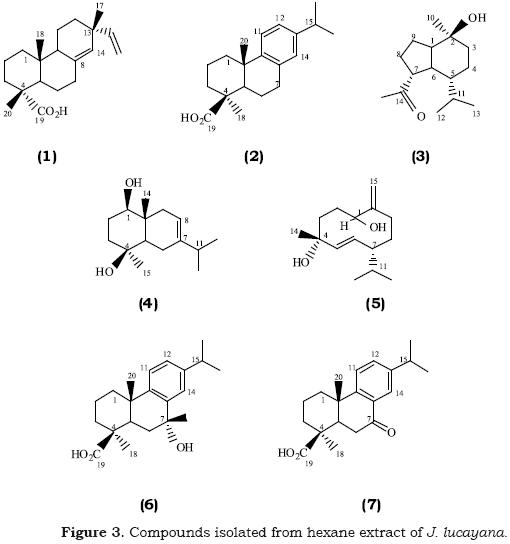
Compound 5 is isolated for the first time from a Juniperus species, meanwhile compounds 1, 2, 3, 4, 6, and 7 have been previously obtained from the genus Juniperus (Kuo et al., 1994; San Feliciano et al., 1995; Fang et al., 1996). The isolation of all this compounds from J. lucayana has not before been reported to the best of our knowledge.
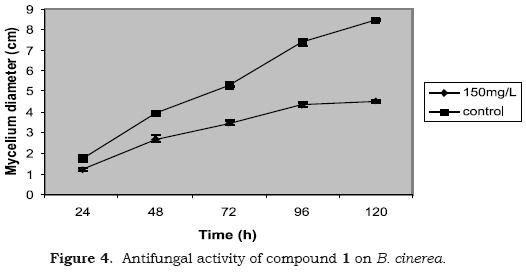
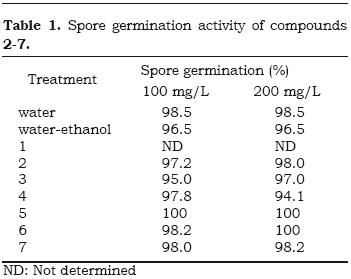
The antifungal properties of the compounds 1–7 was evaluated for first time against B. cinerea, as described in the part experimental. The sandaracopimaric acid (1), evaluated by poison food technique, reduced the radial fungus growth (70.8% growth inhibition) in the first day of assay. However, it showed a weak activity after five days of experiment (52.7% growth inhibition) (see Figure 4). Compounds 2–7 did not inhibited the spore germination of B. cinerea at concentrations tested (Table 1).
CONCLUSIONS
The ethanolic and hexane extracts from J. lucayana showed an important antifungal effect against B. cinerea. The presence of sandaracopimaric acid (1) contributes to this effect. Further studies on the isolated of additional compounds responsible of the activity of the extract are required.
REFERENCES
Aleu, J., Hanson, J.R., Hernández–Galán, R., Collado, I.G. (2001) Biotransformation of the fungistatic sesquiterpenoids patchoulol, ginsenol, cedrol and globulol by Botrytis cinerea. Journal of Molecular Catalysis B: Enzymatic 11: 329–334 [ Links ]
Barrero, A.F., Quílez del Moral, J., Lara, A., Herrador, M. (2005) Antimicrobial activity of sesquiterpenes from the essential oil of Juniperus thurifera Word. Planta Medica 71: 67–71. [ Links ]
Dang, N.H., Zhang, X.F., Zheng, M.S., Son, K.H., Chang, H.W., Kim, H.P., Bae, K., Kang, S.S. (2005) Inhibitory constituents against cyclooxygenases from Aralia cordata Thunb. Arch Pharm Res. 28: 28–33. [ Links ]
Daoubi, M., Deligeorgopoulou, A., Macias, A.J., Galán, R., Hitchcock, P., Hanson, J., Collado, I.G. (2005) Antifungal Activity and Biotransformation of Diisophorone by Botrytis cinerea. J. Agric. Food Chem. 53: 6035–6039. [ Links ]
De Pascual, J., San Feliciano, A., Miguel del Corral, J.M., Barrero, F.A. (1983) Terpenoids from Juniperus sabina. Phytochemistry 22: 300–301. [ Links ]
Duke, S.O., Baerson, S.R., Dayan, F.E., Rimando, A.M., Schffler, B.E., Tellez, M.R., Wedge, D.E., Schrader, K.K., Akey, D.H., Arthur, F.H., De Lucca, A.J., Gibson, D.M., Harrison, H.F.Jr., Peterson, J.K., Gealy, D.R., Tworkoski, T., Wilson, C.K., Moris, J.B. (2003) United States Department of Agriculture–Agricultural Research Service research on natural products for pest management. Pest. Manage. Sci. 59: 708–717. [ Links ]
Edwards, O.E., Nicolson, A., Rodger, M.N. (1960) Structure of sandaracopimaric acid. Canadian Journal of Chemistry 38: 663–7. [ Links ]
Fang, J.M., Chen, Y.C, Wern, B., Cheng, Y.S. (1996) Terpenes from heartwood of Juniperus chinensis. Phytochemistry 41 : 1361–1365. [ Links ]
Karaman, I.,  ahin, F., Güllüce, M., Ö
ahin, F., Güllüce, M., Ö ütçü, H.,
ütçü, H.,  engül, M., Adιgüzel, A. (2003) Antimicrobial activity of aqueous and methanol extracts of Juniperus oxycedrus L. Journal of Ethnopharmacology 85: 231–235. [ Links ]
engül, M., Adιgüzel, A. (2003) Antimicrobial activity of aqueous and methanol extracts of Juniperus oxycedrus L. Journal of Ethnopharmacology 85: 231–235. [ Links ]
Kuo, C.L., Fang, J.M., Cheng, Y.S. (1994) Abietanes from leaves of Juniperus chinensis. Phytochemistry 35: 983–986. [ Links ]
Kuo, Y.H., Lo, J.M., Chan, Y.F. (2002) Cytotoxic components from the leaves of Schefflera taiwaniana. Journal of the Chínense Chemical Society 49: 427–431. [ Links ]
Kitagawa, I., Cui, Z., Son, B.W., Kobayashi, M., Kyogoku, Y. (1987) Marine natural products XVII. Nephtheoxydiol, a new cytotoxic hydroperoxy–germacrane sesquiterpene, and related sesquiterpenoids from an Okinawan soft coral of Nephthea sp. (Nephtheidae). Chem. Pharm. Bull. 35: 124–135. [ Links ]
Minato, H. and Ishikawa, M. (1967) Studies on sesquiterpenoids. Part XV. Structure and Absolute Configuration of Oplodiol, a new Sesquiterpene Alcohol from Oplopanax japonicus (Nakai). Journal of the Chemical Society (C): 423–427. [ Links ]
Ortiz, Y., Hernández–Galán, R., Spengler, I., Rodríguez, Y., Collado, I.G. (2004) Actividad fungicida de cuatro especies cubanas de plantas sobre Botrytis cinerea Pers. Estudio fitoquímico. Revista Cubana de Química 16: 204–206. [ Links ]
Ortiz, Y., Hernández–Galán, R., Spengler, I., Rodríguez, Y., Collado, I.G. (2007) Sesquiterpenes from the wood of Juniperus lucayana. Phytochemistry 68: 2409–2414. [ Links ]
Ortiz, Y., Spengler, I., Rodríguez, Y., Collado, I.G., Hernández–Galán, R., (2008) Screening Study of Potential Lead Compounds for Natural Products Based Fungicides from Juniperus lucayana. Natural Product Communications 3: 469–473. [ Links ]
Prinz, S., Mullner, U., Heilmann, J., Winkelmann, K., Sticher,O., Haslinger, E., Hufner, A. (2002) Oxidation Products of Abietic Acid and Its Methyl Ester. J. Nat. Prod. 65: 1530–1534. [ Links ]
San Feliciano, A., Del Corral, J.M.M., Gordaliza, M., Salinero, M.A. (1992) Diterpenoides neutros y compuestos aromáticos de las hojas de Juniperus phoenicea subsp. turbinata. Anales de química 88: 512–516. [ Links ]
San Feliciano, A., Medarde, M., Gordaliza, M., Lucas, M.J. (1995) Structure elucidation of germacrane alcohols from Juniperus communis subsp. hemisphaerica. Journal of Natural Products 58: 1059–1064. [ Links ]
Shafique, S., Javaid, A., Bajwa, R., Shafique, S. (2007) Effect of aqueous leaf extracts of al–lelopathic trees on germination and seed–borne mycoflora of wheat. Pak. J. Bot. 39: 26192624. [ Links ]
Schmidt, R.J. (2004) Botanical Dermatology Database–CUPRESSACEAE: pp.1–18. [ Links ] Soundharrajan, R.S., Velusamy, R.R., Ramasamy, R., Selladurai, M., Srinivasan, N. (2003) Antifungal activity of some essential oils. J. Agric. Food Chem. 51: 7596–7599. [ Links ]
Su, W.C., Fan, J.M., Cheng, Y.S. (1995) Sesquiterpenes from leaves of Cryptomeria japonica. Phytochemistry 39: 603–607. [ Links ]
Tchuendem, M.H.K., Mbah, J.A., Tsopmo, A., Ayafor, J.F., Sterner, O., Okunjic, C.C., Iwu, M.M., Schuster, B.M. (1999) Anti–plasmodial sesquiterpenoids from the African Reneilmia cincinnata. Phytochemistry 52: 1095–1099. [ Links ]














