Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Hidrobiológica
versión impresa ISSN 0188-8897
Hidrobiológica vol.19 no.2 Ciudad de México may./ago. 2009
Artículos
The effect of protein and energy levels in diet on the antioxidant activity of juvenile redclaw Cherax quadricarinatus (von Martens, 1868)
Efecto del nivel de proteína y energía en la actividad antioxidante de juveniles de langosta de agua dulce Cherax quadricarinatus (von Martens, 1868)
Edilmar Cortés–Jacinto1*, Ángel I. Campa–Córdova2, Felipe Ascencio2, Humberto Villarreal–Colmenares1 and Ramón J. Holguín–Peña2
1 Laboratorio de Nutrición Experimental, Centro de Investigaciones Biológicas del Noroeste (CIBNOR), Mar Bermejo 195, Playa Palo Santa Rita, La Paz, B. C. S. 23096, México. *E–mail: ecortes04@cibnor.mx
2 Laboratorio de Biotecnología de Organismos Marinos, CIBNOR.
Recibido: 8 de marzo de 2007.
Aceptado: 20 de junio de 2009.
ABSTRACT
This study determined the influence of dietary crude protein and energy levels on growth, survival, and antioxidant response of superoxide dismutase (SOD) in juvenile redclaw Cherax quadricarinatus. Six practical diets containing three crude protein (CP) levels (28, 35, and 40%) and two energy levels (~18 and 20 kJ · g–1) were tested. The control group was fed a commercial shrimp diet. The highest SOD activity (34.8 U·mg–1 protein) occurred in the diet with 35% CP and 18 kJ g–1. Soluble protein in tissue of redclaw fed 28 and 35% CP increased, compared to the control. The best mean final weight (9.23 g), weight gain (788%), and the lowest feed conversion ratio (1.3) occurred with the 35% CP diet. We conclude that a dietary CP content of 35% and 18 kJ · g–1 is recommended for production and best immune responses in redclaw.
Key words: Cherax quadricarinatus; redclaw, SOD; protein, grow–out.
RESUMEN
Se realizó un estudio para evaluar el efecto de los niveles de proteína y energía sobre el crecimiento, supervivencia y la respuesta antioxidante de la enzima superóxido dismutasa (SOD) en juveniles de Cherax quadricarinatus. El experimento incluyó seis dietas prácticas con tres niveles de proteína cruda (28, 35 y 40%) y dos niveles de energía (~18 y 20 kJ·g–1). El grupo control fue alimentado con alimento comercial para camarón. La actividad SOD más alta (34.8 U·mg–1 prot.) se presentó en juveniles alimentados con 35% de proteína cruda (PC) y 18 kJ·g–1. La proteína soluble en tejido de langosta de agua dulce alimentada con 28 y 35% de PC fue superior con respecto a los acociles del grupo control. El mejor peso final promedio (9.23 g), la mejor ganancia en peso (788%) y el factor de conversión alimenticia mas bajo (1.3) se presentó en acociles alimentados con 35% de PC. Se concluye que una dieta conteniendo 35% de PC y 18 kJg–1 de energía es recomendada para la producción y además incrementa la respuesta inmune de los acociles en su fase de juvenil.
Palabras clave: Cherax quadricarinatus, langosta de agua dulce, SOD, proteína, crecimiento.
INTRODUCTION
A proper balance of protein and energy input is required to supply calories and amino acids for rapid growth and to produce redclaw meat that is high in protein and low in fat (Cortés–Jacinto et al., 2004). The nutritive value of formulated feeds depends on the digestibility of the components and the resulting diets (Campaña–Torres et al., 2005, 2006).
The nutrient requirements, feeding frequency, and physiological energetic of redclaw Cherax quadricarinatus (von Martens, 1898) are becoming clearer with the advance of investigations of this species (Cortés–Jacinto et al., 2003a, 2003b; Thompson et al., 2004; 2005; Díaz et al., 2006). It is now known that juveniles require feed with protein content between 31–34% (Webster et al. 1994; Ponce–Palafox et al., 1998; Cortés–Jacinto et al., 2003a), while pre–adults require about 25.6% (Cortés–Jacinto et al., 2004).
Recently, the interaction of diet and health was investigated (Vega–Villasante et al., 2006). Evidence suggests that a nutritional diet with pyridoxine and the immune status are tightly linked in abalone (Chen et al., 2005). However, a diet supplemented with astaxanthin was effective in rapidly eliminating reactive oxygen species (ROS), which is essential to the health and survival of all higher animals. Animals have evolved antioxidant defenses, including non–enzymatic and enzymatic mechanisms (Pan et al., 2003). Enzymatic antioxi–dant defenses include ascorbate peroxidase, glutathione reductase, catalase, and peroxidases. These enzymes efficiently remove hydrogen peroxide from cells, as does superoxide dismutase (SOD), which scavenges the superoxide anion (Muñoz et al., 2000). SOD, a cytosolic enzyme specific for scavenging superoxide radicals, is involved in protective mechanisms within injured tissue following oxidative processes and phagocytosis (Pan et al., 2003). The complex antioxidant system of aerobic organisms not only prevents the effect of ROS, but also plays a vital role in protecting cells from oxidative stress. It has been used as a potential indicator of oxidative stress in marine organisms (Chien et al., 2003; Zenteno–Savin et al., 2006). Zenteno–Savin et al. (2008) indicated that dietary protein and lipid levels affect the oxidative stress response in juvenile redclaw.
SOD activity has been widely studied in fishes (Li et al., 2003). In crustaceans, recent discoveries concerning cellular SOD, which plays an important role in immunity (Arun & Subramanian, 1998; Pan et al., 2003; Chen et al., 2005) and modulating oxidative responses leads to increased or decreased SOD activity (Campa–Córdova et al., 2005).
This study evaluated the effects of three protein levels at two energy levels to increase or decrease SOD activity and growth of redclaw.
MATERIALS AND METHODS
Experimental diets. Six experimental diets were tested for 65 days, combining three levels of crude protein (28, 35, and 40%) with either a low or a high level of energy (~18 and 20 kJ g–1.). The ratio of protein–to–energy (P:E) ranged from 14.7 to 21.3 mg protein kJ g–1. The diets were formulated with highly digestible ingredients, similar to those that were prepared and used in experimental diets designed for juvenile redclaw (Cortés–Jacinto et al., 2003a). The proximate analyses of the diets (AOAC, 1995) are given in Table 1. Gross energy of the diets was measured in an adiabatic bomb calorimeter (Model 1261, Parr, Moline, IL, USA). Juvenile redclaw were initially fed a ration of 5% of their biomass each day. Two feedings, each consisting of 50% of the diet, were fed at 08:00–08:30 h and 16:00–16:30 h, as described by Cortés–Jacinto et al. (2003b).
Experimental procedure. Juvenile redclaw Cherax quadricarinatus were obtained from a pond at the CIBNOR research facility. Initially, stocking was 20 redclaw m–2 (each weighing 1.04 ± 0.30 g) in 1,500–L fibreglass tanks. Each experimental diet was randomly assigned to three tanks. Juveniles that died during the first three days were replaced with reserve specimens held under identical conditions. Water quality was monitored and maintained within recommended limits for redclaw (Villarreal, 2000). Temperature was set at 27.5 ± 0.9 °C with two 300–W heaters (Aquarium Pharmaceuticals, Chalfont, PA, USA) and the light–dark photoperiod was 14:10. A 5–hp blower (Sweetwater, Apopka, FL) and an air stone in each tank provided aeration to maintain dissolved oxygen levels above 6 mg·l–1, pH was measured once a day with a pH meter (American Marine®, Ridgefield, C T, USA); N–nitrate, N–nitrite, and total ammonium nitrogen were measured weekly with a spectrophotometer (model DREL–2010; The Hach Company, Loveland, CO, USA). Uneaten feed and feces were siphoned from each tank daily. After siphoning, 30% of the tank water was replaced with fresh tap water.
At the end of the feeding trial, the redclaw in each tank were weighed in bulk. Weight gain as a percent in each tank was calculated as: WG = 100 × (final weight – initial weight)/initial weight; Survival Rate was calculated as: %SR = (final number of specimens in each tank/initial number of specimens in each tank) × 100; protein efficiency ratio was calculated as: PER = (weight gain/protein intake); feed efficiency was calculated as: FE = (weight gain × 100)/dry weight of feed offered); and feed conversion ratio was calculated as: FCR = (feed intake/weight gain).
After final weighing, three specimens were randomly sampled from each tank. After dissection over ice, the muscle was rapidly excised from each specimen, placed in Eppendorf tubes on ice, and muscle samples were measured for SOD activity (Campa–Cordova et al., 2005).
SOD extraction and activity assay. Redclaw from the same batch were collected from a production pond at CIBNOR as the control to measure SOD. These specimens were fed a commercial shrimp diet of pellets with 36.7% protein and 12.6% lipids (PIASA®, La Paz, B.C.S., Mexico), similar to the diet used by Díaz et al. (2006).
For SOD extraction cells were disrupted by adding 100 mg frozen shrimp muscle in a mechanical homogenizer containing 0.5 mL phosphate buffer (50 mM, pH 7.8). The homogenate was centrifuged (5,724 g for 5 min at 4 °C); the supernatant was recovered and heated for 5 min at 65 °C. Another supernatant sample was obtained from a second centrifugation of crude extract and stored at –20 °C. Samples were maintained on ice at all times.
SOD activity was determined according to Beauchamp and Fridovich (1971), using nitro blue tetrazolium (NBT) in the presence of riboflavin. For this, a 2–ml reaction mixture (0.1 mM EDTA, 13 µM methionine, 0.75 mM NBT, and 20 µM riboflavin in 50 mM phosphate buffer at pH 7.8) and 0 to 100 µL crude extract were placed under fluorescent light for 2 min or until A560 in control tubes reached 0.2 to 0.25 OD. The specific activity (units per mg protein) was calculated using a computer program described by Vazquez–Juárez et al. (1993).
Protein. Total soluble protein content was determined according to Bradford (1976), using a protein dye reagent (Bio–Rad, #500–0006) and bovine serum albumin (Sigma, #A–9430) as a standard. Optical density was determined at 595 nm with a microplate reader (Bio–Rad, Model 550).
Statistical analysis. Each experimental diet was fed to three groups of redclaw, using a completely randomized design. Results were analyzed by one–way ANOVA, using the Tukey test to measure the differences. Survival percentages were transformed by the square root of the arcsine before statistical analyses (Statistica, StatSoft, Tulsa, OK). Values of p < 0.05 were considered significantly different (Zar, 1999).
RESULTS
Over the duration of the study, water quality parameters averaged (±SD) were: temperature, 27.5 ± 0.9 °C; dissolved oxygen, 6.1 ± 0.09 mg l–1; total ammonia nitrogen, 0.11 ± 0.06 mg l–1; nitrite, 0.08 ± 0.04 mg l–1; nitrate, 5.6 ± 0.06 mg l–1; pH, 7.90 ± 0.13. These values were within acceptable limits for indoor production of redclaw (Villarreal 2000, Cortés–Jacinto et al., 2003b).
Weight gain, survival, protein efficiency ratio, feed efficiency, and feed conversion ratio for each diet group are presented in Table 2. Mean final weights, weight gain, and feed efficiency of juveniles in treatment 35LE were significantly higher (p < 0.05). Survival was affected by the experimental treatments, but diets with 31LE, and 28HE did not show significant differences. Feed conversion ratio (FCR) was significantly higher in the treatment with the lowest (28LE) and highest gross energy (35HE and 40HE) for diets containing 20.2, 20.2, and 16.9 kJ g–1. Diets with 28 and 35% CP (28HE and 35LE) showed the highest protein efficiency ratios (PER). Redclaw fed with treatments 35HE, 40LE, and 40HE had the lowest survival rates.
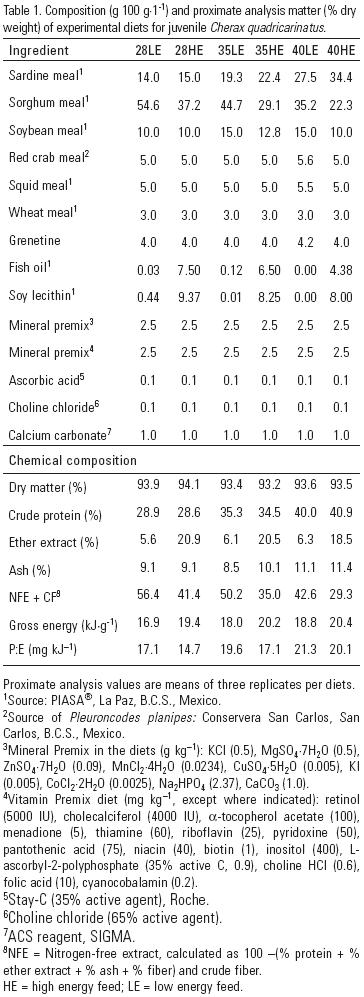
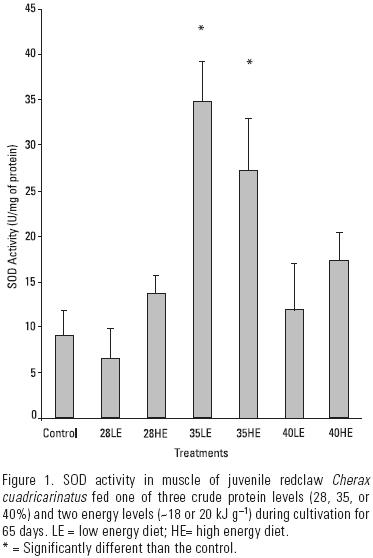
Exposure to three different levels of protein and two levels of energy induced a significant increase in SOD activity in muscle at the end of the 65–day experiment. The highest SOD activity reached 34.8 U mg–1 protein using treatment 35LE (Fig. 1), followed by 27.2 U mg–1 protein using treatment 35HE. Treatments 28LE and the control group showed the lowest SOD activity, reached 6.6 and 9.0 U mg–1 protein, respectively. Fig. 2 shows the soluble protein content in muscle of juvenile redclaw at 65 days. Significant increase of protein content in muscle occurred when juveniles were exposed to treatments 28LE and 35HE (1.36 and 1.4 mg ml–1, respectively).
DISCUSSION
Our understanding of crustacean nutritional needs has greatly expanded over the last few decades (Shiau, 1998). However, research is still necessary, with many gaps remaining. Optimal dietary protein level for redclaw is influenced by the balance of dietary protein to energy (Cortés–Jacinto et al., 2004) and digestibility of the dietary protein (Campaña–Torres et al., 2005, Campaña–Torres et al., 2006). Dietary energy content (18.9 kJ g–1) for better growth of juvenile redclaw was reported by Cortés–Jacinto et al. (2003a). In this study, the best productivity in terms of weight gain, feed efficiency, and feed conversion ratio occurred with treatment 35LE (18.0 kJ g–1). Treatment 28LE had significantly lower weight gain, probably because protein and energy supply was low and used for protein synthesis and somatic growth. This implies that diets in this low lipid series were limiting, with protein oxidized to meet the redclaw's energy demand at the expense of somatic growth. Pilot studies with the giant perch Lates calcarifer (Bloch, 1790) fed low–protein, high–energy diets gave disappointing results and may have made the fish more susceptible to disease (Williams et al., 2003). The protein efficiency ration tended to decrease with an increase of dietary protein. The results of treatment 35LE (2.2 kJ g–1) was similar to what was reported by Cortés–Jacinto et al. (2003a) for juvenile redclaw (2.8 kJ g–1).
An interesting finding was low survival in redclaw fed with treatments 40LE and 40HE. These diets had the lowest SOD activity (Table 2; Fig. 1). Since nutritional status is the most important factor influencing immune defense mechanisms of animals (Chen et al., 2005), deficient or excessive levels of nutrients can alter the immune system, and cause cell stress (Folkers et al., 1993; Matozzo et al., 2005; Villa–Cruz et al., 2009). Hence, nutritional status is a good health indicator of crustaceans; inadequate levels of nutrients may result in reduced growth rates, poor feed conversion ratios, decreased resistance to stress, and reduced capability to heal wounds (López et al., 2003; Sánchez et al., 2005). Increased SOD activity in juvenile redclaw is a response to changes in lipid composition of hemocyte cell membranes, which enhance production of cell–activating factors (cytokines or chaperonins) that improve phagocytic capability (Itami et al., 1998). Fridovich (1995) linked increased SOD activity to increased longevity and increased tolerance to ischemic or reperfusion events or to factors that induce oxidative stress. Zenteno–Savin et al. (2008) found that levels of dietary protein and lipid have a differential effect on specific tissue membrane composition, affecting lipid peroxidation levels in different ways in gills, muscle, and the hepatopancreas in redclaw. Downs et al. (2001) reported increased levels of Mn–SOD, glutathione, heat shock proteins, and ubiquitin in the grass shrimp Palaemonetes pugio (Holthuis, 1949) after heat stress. Campa–Córdova et al. (2005), reported increased SOD activity in the juvenile whiteleg shrimp Litopenaeus vannamei (Boone, 1931) when exposed to immuno–stimulants. Thus, increases in antioxidant SOD activity reflect its important role in clearing excessive ROS (Li et al., 2003) and in the immune responses in invertebrates (Chen et al., 2005).
The increase in soluble protein in juvenile redclaw after exposure to treatments 28HE and 35LE may promote generation of other immuno–proteins, offering more tolerance to stress and less susceptibility to diseases (Downs et al., 2001). Additionally, optimal requirements in diet could enhance the immune and detoxifying systems to more efficiently mitigate stress conditions in cultivation ponds, where diseases are frequent (López et al., 2003).
Some authors report on the importance of shrimp tissues as sites of recognition and removal of bacteria and viruses (Martin et al., 1993; Jiravanichpaisal & Miyazaki, 1994; De la Peña et al., 1995). In our study, the 35LE treatment induced the highest SOD activity in juvenile redclaw. Arun and Subramanian (1998) studied SOD activity in different tissues of the freshwater prawn Macrobrachium malcolmsonii (H. Milne–Edwards, 1844), reporting 16.02 U·mg–1 protein in muscle of subadult prawns and 32.16 U·mg–1 protein in the hepatopancreas. They concluded that higher antioxidant values in the hepatopancreas are a consequence of multiple oxidative reactions. Hence, this may be the site of greatest free–radical generation. Values of SOD activity found by Arun and Subramanian (1998) were similar to those observed in juvenile redclaw. The protein–to–energy ratio was the only difference in the diets. Digestibility of animal–derived and plant–derived ingredients vary, as reported by Campaña–Torres et al. (2005). Digestibility differences can lead to variations in the response of redclaw. Chien et al. (2003) enhanced antioxidant capacity of the giant tiger prawn Penaeus monodon (Fabricius, 1798) by including astaxanthin in the diet. Mercier et al. (2009) reported tolerance to handling stress in the whiteleg shrimp Litopenaeus vannamei when fed highly unsaturated fatty acids and this supplement increased immune response capacity. Thus, we suggest that enhancing the diet in juvenile redclaw increased SOD activity.
The use of biomarkers to evaluate nutritional requirements in aquaculture species could be an important approach to manage diets more efficiently and evaluate whether diets are assimilated at adequate rates. This will contribute to our understanding of metabolic routes of nutrients and monitoring immune responses of invertebrates with enhanced diets.
ACKNOWLEDGMENTS
We thank Sonia Rocha for chemical analyses, Sandra de La Paz and Mildred Cortés for maintaining the experimental system. Malta Texo de México, Sinaloa, provided the squid meal. This research was funded by CONACYT grant 2888–1, and CIBNOR grant AC2.5. E. Cortés–Jacinto received a postdoctoral fellowship from CONACYT at the Universidad de Sonora (DICTUS), Sonora, Mexico. The authors thank L. R. Martínez–Córdova for constructive comments and the anonymous reviewers for some very insightful comments.
REFERENCES
AOAC, 1995. Official Methods of Analysis, 16th Ed. Association of Official Analytical Chemist, Gathersburg, MD. 1234 p. [ Links ]
Arun, S. & P. Subramanian. 1998. Antioxidant enzymes in freshwater prawn Macrobrachium malcolmsonii during embryonic and larval development. Comparative Biochemistry Physiology B 121 (3): 273–277. [ Links ]
Beauchamp, Ch. & I. Fridovich. 1971. Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Analytical Biochemistry 44 (1): 276–287. [ Links ]
Bradford, M. M. 1976. A rapid and sensitive method for quantification of microgram quantities of protein using the principle of protein dye binding. Analytical Biochemistry 72 (1–2): 248–254. [ Links ]
Campa–Córdova, A .I., N. Y. Hernández–Saavedra, G. Aguirre–Guzmán & F. Ascencio. 2005. Immunomodulatory response of superoxide dismutase in juvenile American white shrimp (Litopenaeus vannamei) exposed to immunostimulants. Ciencias Marinas 31 (4): 661–669. [ Links ]
Campaña–Torres, A., L. R. Martínez–Córdova, H. Villarreal–Colmenares & R. Civera–Cerecedo. 2006. Carbohydrate and lipid digestibility of animal and vegetal ingredients and diets for juvenile Australian redclaw crayfish, Cherax quadricarinatus. Aquaculture Nutrition 12 (2): 103–109. [ Links ]
Campaña–Torres, A., L. R. Martínez–Córdova, H. Villarreal–Colmenares & R. Civera–Cerecedo. 2005. In vivo dry matter and protein digestibility of three plant–derived and four animal–derived feedstuffs and diets for juvenile Australian redclaw, Cherax quadricarinatus. Aquaculture 250 (3–4): 748–754. [ Links ]
Chen, H., K. Mai, W. Zhang, Z. Liufu, W. Xu & B. Tan. 2005. Effects of dietary pyridoxine on immune responses in abalone, Haliotis discus hannai Ino. Fish Shellfish Immunology 19 (3): 241–252. [ Links ]
Chien, Y. H., C. H. Pan & B. Hunter. 2003. The resistance to physical stresses by Penaeus monodon juveniles fed diets supplemented with astaxanthin. Aquaculture 216 (1–4): 177–191. [ Links ]
Cortés–Jacinto, E., H. Villarreal–Colmenares, R. Civera–Cerecedo & L. R. Martínez–Córdova. 2003a. Effect of dietary protein level on growth and survival of juvenile freshwater crayfish Cherax quadricarinatus (Decapoda: Parastacidae). Aquaculture Nutrition 9 (4): 207–213. [ Links ]
Cortés–Jacinto, E., H. Villarreal–Colmenares & M. Rendon–Rumualdo. 2003b. Efecto de la frecuencia alimenticia en el crecimiento y sobrevivencia de juveniles de langosta de agua dulce Cherax quadricarinatus (von Martens, 1868) (Decapada: Parastacidae). Hidrobiológica 13 (2): 151–158. [ Links ]
Cortés–Jacinto, E., H. Villarreal–Colmenares, R. Civera–Cerecedo & J. Naranjo–Páramo. 2004. Effect of dietary protein level on monosex growth and survival of pre–adult of freshwater crayfish (Cherax quadricarinatus). Aquaculture Research 35 (1–3): 71–79. [ Links ]
De La Peña, L. D., T. Nakai & K. Muroga. 1995. Dynamics of Vibrio sp. PJ in organs of orally infected kuruma prawn, Penaeus japonicus. Fish Pathology 30 (1): 39–45. [ Links ]
Díaz, F., G. Escalante, A. D. RE & E. Sierra. 2006. Fisiología energética de Cherax quadricarinatus alimentado con dos dietas, expuesto a un régimen constante y fluctuante de temperatura. Hidrobiológica 16 (1): 35–44. [ Links ]
Downs, C. A., J. E. Fauth & C. M. Woodley. 2001. Assessing the health of grass shrimp (Palaemonetes pugio) exposed to natural and anthropogenic stressors: A molecular biomarker system. Marine Biotechnology 3 (4): 380–397. [ Links ]
Folkers, K., M. Morita & J. Mcree. 1993. The activities of coenzyme Q10 and vitamin B6 for immune responses. Biochemical and Biophysical Research Communications 193 (1): 88–92. [ Links ]
Fridovich, I. 1995. Superoxide radical and superoxide dismutases. Annual Review of Biochemistry 64: 97–112. [ Links ]
Itami, T., M. Asano, K. Tokushige, K. Kubono, A. Nakagawa, N. Takeno, H. Nishimura, M. Maeda, M. Kondo & Y. Takahashi. 1998. Enhancement of disease resistance of kuruma shrimp, Penaeus japonicus after oral administration of peptidoglycan derived from Bifidobacterium thermophilum. Aquaculture 164 (1–4): 277–288. [ Links ]
Jiravanichpaisal, P., T. Miyazaki & C. Limsuwan.1994. Histopathology, biochemistry, and pathogenicity of Vibrio harveyi infecting black tiger prawn Penaeus monodon. Journal of Aquatic Animal Health 6 (1): 27–35. [ Links ]
Li, X., Y. Liu, L. Song & J. Liu. 2003. Responses of antioxidant systems in the hepatocytes of common carp (Cyprinus carpio L.) to the toxicity of microcystin–LR. Toxicon 42 (1): 85–89. [ Links ]
López, N., G. Cuzon, G. Gaxiola, G. Taboada, M. Valenzuela, C. Pascual, A. Sánchez & C. Rosas. 2003. Physiological, nutritional, and immunological role of dietary b 1,3 glucan and ascorbic acid 2–monophosphate in Litopenaeus vannamei juveniles. Aquaculture 224 (1–4): 223–243. [ Links ]
Martin, G.G., D. Poole, C. Poole, J.E. Hose, M. Arias, L. Reynolds, N. Mckrell& A. Whang. 1993. Clearance of bacteria injected into the haemolymph of the panaeid shrimp, Sicyonia ingentis. Journal of Invertebrate Pathology 62 (3): 308–315. [ Links ]
Matozzo, V., M. Monari, J. Foschi, T. Papi, O. Cattani & M. G. Marin. 2005. Exposure to anoxia of the clam Chamelea gallina I. Effects on immune responses. Journal of Experimental Marine Biology and Ecology 325: 163–174. [ Links ]
Mercier, L., I. S. Racotta, G. Yepiz–Plascencia, A. Muhlia–almazán, R. Civera, M. F. Quiñones–ArreoLa, M. Wille, P. Sorgeloos& E. Palacios. (In Press). Effect of diets containing different levels of highly unsaturated fatty acids on physiological and immune responses in Pacific white leg shrimp Litopenaeus vannamei (Boone) exposed to handling stress. Aquaculture Research. [ Links ]
Muñoz, M., R. Cedeño, J. Rodríguez, W. P. Van Der Knaap, E. Mialhe & E. Bachère. 2000. Measurement of reactive oxygen intermediate production in haemocytes of the penaeid shrimp, Penaeus vannamei. Aquaculture 191 (1–3): 89–107. [ Links ]
Pan, C. H., Y. H. Chien & B. Hunter. 2003. Alterations of antioxidant capacity and hepatopancratic enzymes in Penaeus monodon (Fabricus) juveniles fed diets supplemented with astaxanthin and exposed to Vibrio damsela challenge. Journal Fish Society Taiwan 30 (4): 279–290. [ Links ]
Ponce–Palafox, J. T., J. L. Arredondo & M. A. Moreno. 1998. The effects of varying dietary protein levels on growth and survival of juvenile and pre–adult redclaw (Cherax quadicarinatus). The biodiversity crisis and crustacea. In: Von Vaupel, K.J.C. and Schram, F. (Eds.) Proceedings of the 4th International Crustacean Congress, A.A. Balkeman, Amsterdam, Netherlands, Vol. 2, pp. 715–719. [ Links ]
Sánchez, D. R., J. M. Fox, A. L. Lawrence, F. L. Castille & B. Dunsford. 2005. A methodology for evaluation of dietary feeding stimulants for the Pacific white shrimp, Litopenaeus vannamei. Journal of the World Aquaculture Society 36 (1): 14–23. [ Links ]
Shiau S. Y. 1998. Nutrient requirements of penaeid shrimps. Aquaculture 164 (1–4): 77–93 [ Links ]
Thompson, K. R., L. A. Muzinic, L. S. Engler & C. D. Webster. 2005 Evaluation of practical diets containing different protein levels, with or without fish meal, for juvenile Australian red claw crayfish (Cherax quadricarinatus ). Aquaculture 244 (1–4): 241–249. [ Links ]
Thompson, K. R., L. A., Muzinic, L. S. Engler, S. R. Morton & C. D. Webster. 2004. Effects of feeding practical diets containing various protein levels on growth, survival, body composition, and processing traits of Australian red claw crayfish (Cherax quadricarinatus) and on pond water quality. Aquaculture Research 35 (7): 659–668. [ Links ]
Vázquez–Juárez, R., F. Vargas–Aalbores & J. L. Ohoa. 1993. A computer program to calculate superoxide dismutase activity in crude extracts. Journal of Microbiological Methods 17 (3): 239–244. [ Links ]
Vega–Villasante, F., U. Bécquer–Zúñiga, N. Hernández, H. Nolasco–Soria & O. Carrillo–Farnés. 2006. La langostilla roja (Pleuroncodes planipes Stimpson, 1860) (Crustacea: Galatheidae), como alimento funcional en el crecimiento, supervivencia y composición corporal de larvas de (Litopenaeus schmitti Burkenroad, 1936) (Crustacea: Penaeidae). Hidrobiológica 16 (3): 241–249. [ Links ]
Villa–Cruz V., J. Davila, M. T. Viana & R. Vazquez–Duhalt. 2009. Effect of broccoli (Brassica oleracea) and its phytochemical sulforaphane in balanced diets on the detoxification enzymes levels of tilapia (Oreochromis niloticus) exposed to a carcinogenic and mutagenic pollutant. Chemosphere 74: 1141–1155. [ Links ]
Villarreal, H. 2000. El cultivo de la langosta de agua dulce. Una oportunidad para la diversificación acuícola. In: Bonilla, Z. & I. Burciaga (Eds.). Memorias III Simposium Internacional de Acuacultura. 5–7 de Octubre, 2000. Culiacán, Sinaloa, pp. 110–135. [ Links ]
Webster, C. D., L. S. Goodgame–Tiu, J. H. Tidwell & D. B. Rouse. 1994. Evaluation of practical feed formulations with different protein levels for juvenile red claw crayfish (Cherax quadricarinatus). Transactions of the Kentucky Academy of Science 55: 108–112. [ Links ]
Williams, K. C., C. G. Barlow, L. Rodgers, I. Hockings, C. Agcopra & I. Ruscoe. 2003. Asian seabass Lates calcarifer perform well when fed pelleted diets high in protein and lipid. Aquaculture 225 (1–4): 191–206. [ Links ]
Zar, J. H. 1999. Biostatistical Analysis. Prentice Hall, Upper Saddle River, NJ. 663 p. [ Links ]
Zenteno–Savín, T., R. Saldierna & M. Ahuejote–Sandoval. 2006. Superoxide radical production in response to environmental hypoxia in cultured shrimp. Comparative Biochemistry Physiology C 142 (3–4): 301–308. [ Links ]
Zenteno–Savín T., E. Cortés–Jacinto, J. P. Vázquez–Medina & H. Villarreal–Colmenares. 2008. Oxidative damage in tissues of juvenile crayfish (Cherax quadricarinatus von Martens, 1868) fed different levels of proteins and lipid. Hidrobiológica 18 (2): 147–154. [ Links ]














