Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Hidrobiológica
versión impresa ISSN 0188-8897
Hidrobiológica vol.18 supl.1 Ciudad de México ago. 2008
Dominance shift of zooplankton species composition in the central Strait of Georgia, British Columbia during 1997
Cambio en el zooplancton dominante del estrecho de Georgia, British Columbia durante 1997
Martha J. Haro–Garay1, 2*, Leonardo Huato Soberanis2
1 Department of Earth and Ocean Sciences, University of British Columbia, 6270 University Blvd., Vancouver, B.C., Canada V6T 1Z4. * E–mail: mharo@cibnor.mx.
2 Present address: Centro de Investigaciones Biológicas del Noroeste (CIBNOR), Mar Bermejo 195, Col. Playa Palo de Santa Rita, La Paz, Baja California Sur 23090, México.
Recibido: 8 de enero de 2007
Aceptado: 11 de septiembre 2007
ABSTRACT
A multivariate analysis of the zooplankton was completed during the 1997 annual cycle in the central Strait of Georgia, Canada. Sampling was conducted monthly in a single location (49° 15' 0" N, 123° 44' 9" W). The study used the species as environment descriptors, and examined possible patterns of species associations. Principal components analysis showed two groups of species coinciding with changes in vertical salinity structure arising from two phases of the Fraser River runoff. Group I was dominant during fall–winter to early spring related to low–runoff season. Group II was dominant during late spring to summer, and related to high–runoff season. Notable results were the scarcity of Neocalanus plumchrus and Euphausia pacifica, typically dominant species of zooplankton, coincident with river runoff levels lower than previous years and with an early phytoplankton bloom. We speculate that these results are a consequence of El Niño 1997 event, combined with the climate change expressed as a decreased freshwater runoff. Low abundance of Neocalanus plumchrus and Euphausia pacifica affected the zooplankton biomass. Consequently, substitute dominant P. pacifica and Cyphocaris challengeriprobable play an important role in trophic dynamics while N. plumchrus and E. pacifica are scarce. Both amphipods feed on small zooplankters that feed on nanoplankton, concentrating food energy and biomass from small zooplankters. This suggests that P. pacifica and C. challengeri are an important link in the trophic ecology of the Strait of Georgia. In conclusion, shifts in zooplankton species dominance highly likely have an effect on juvenile zooplanktivorous salmon specie.
Key words: Community structure, Neocalanus plumchrus, Parathemisto pacifica, Cyphocaris challengeri, El Niño 1997.
RESUMEN
Se realizó un análisis multivariado del zooplancton de la parte central del estrecho de Georgia, Canadá durante 1997, con muestreos mensuales en una localidad (49° 15' 0" N, 123° 44' 9" W). Las especies se usaron como descriptores del ambiente para examinar posibles patrones de asociación. Mediante análisis de componentes principales se encontraron dos grupos coincidentes con cambios en la estructura vertical de la salinidad inducidos por las fases de flujo intensa y débil del Río Fraser. El grupo I estuvo presente de septiembre a marzo durante la fase de flujo débil del río. El grupo II durante la fase de flujo intensa de mayo a julio. Fue notoria la escasez de Neocalanus plumchrus y Euphausia pacifica, especies dominantes típicas, coincidente con niveles de flujo del Río Fraser inferiores a años previos, y con un florecimiento de fitoplancton temprano. Se especula que estos resultados son consecuencia de El Niño 1997, combinado con cambio climático expresado en la baja descarga del río. La baja abundancia de Neocalanus plumchrus y Euphausia pacifica afectó la biomasa zooplanctónica, en su lugar dominaron Parathemisto pacifica y Cyphocaris challengerique depredan al zooplancton pequeño que se alimenta de nanoplancton, concentrando la energía y biomasa del zooplancton pequeño. Esto sugiere que Parathemisto pacifica y Cyphocaris challengeri son un elemento de enlace importante en la ecología trófica del Estrecho de Georgia cuando N. plumchrus y E. pacifica son escasas. Se concluye que los cambios en la dominancia de especies muy probablemente tienen efecto sobre el salmón juvenil y los peces zooplanctófagos.
Palabras clave: Estructura de la comunidad, Neocalanus plumchrus, Parathemisto pacifica, Cyphocaris challengeri, El Niño 1997.
INTRODUCTION
Zooplankton organisms are a pivotal component of marine ecosystems, as they link primary production with higher trophic levels due to its role as food supply for planktivorous fish (Brodeur, 1990). The composition and abundance of zooplankton define the type and quantity of preys that may limit survival of young fish. Changes in the species composition of zooplankton may shift trophic pathways of the ecosystem trophic structure, thus affecting fisheries (Cushing, 1978). In the Strait of Georgia, five species of Pacific salmon feed on zooplankton during their juvenile stages: Oncorhynchus gorbuscha Walbaum, 1792, O. keta Walbaum, 1792; O. kisutch Walbaum, 1792, O. nerka Walbaum, 1792 and O. tshawytscha Walbaum & Artedi, 1792, (Lebrasseur et al., 1969; Healey, 1980; Beacham, 1986). In feeding experiments cultured pink salmon O. gorbuscha up to 34 mm fork length preferred the copepod Neocalanus plumchrus Marukawa, 1921 (LeBrasseur et al., 1969), whereas the larger wild pink salmon consumed the pelagic amphipod Parathemisto pacifica Stebbing, 1888 (Beacham, 1986). Sockeye and chum species feed on P. pacifica, both during juvenile and the adult stages (Beacham, 1986). Inspection of zooplankton and stomach contents of fish, revealed a high abundance of amphipods upon which chum salmon were feeding, and of herring that were feeding on copepods (Parker & Kask, 1972a, 1972b). Also, juveniles of O. kisutch, O. tshawytscha and O. mykiss (Walbaum, 1792) at the riverine and estuarine plumes of the Fraser River were feeding on juvenile fish, including clupeoids, whereas juvenile feed on copepods and amphipods (St. John et al., 1992). The type of available zooplankton is an important factor for salmon feeding ecology, and presumably biological production in the Strait of Georgia.
The zooplankton composition was studied during an annual cycle in the Strait of Georgia, a highly productive estuary/fjord located on the Pacific coast of Canada between mainland British Columbia and Vancouver Island (Parsons et al., 1970; Harrison et al., 1983). A seasonal riverine outflow from the Fraser River controls the estuarine circulation, salinity structure and its local plankton dynamics (Parsons & LeBrasseur, 1969; 1970; LeBlond, 1983). Two periods of high runoff occur during the year, the freshet in early summer due to seasonal snow melt, and the winter runoff associated with the peak in coastal precipitation (Thomson, 1981).
Phytoplankton production has two seasonal blooms during spring and summer (Parsons & LeBrasseur, 1969; Stockner et al., 1979). Following these, the zooplankton biomass also peaks twice a year, during late spring and late summer (Harrison et al., 1983). Copepods, in particular Neocalanus plumchrus and the euphausiid Euphausia pacifica Hansen 1911 dominate the zooplankton (Harrison et al., 1983). This work presents changes in structural patterns of the zooplankton community composition during an annual cycle in the central Strait of Georgia, during 1997, a year with the influence of an El niño event.
MATERIALS AND METHODS
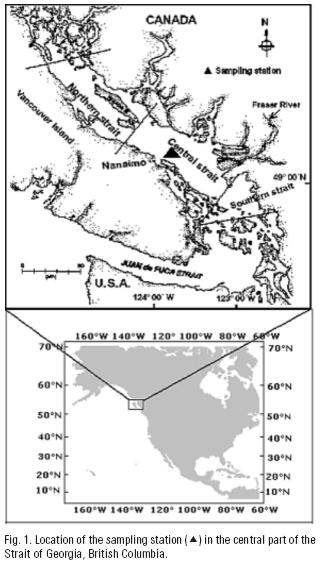
A station located at 49° 15' 0" N, 123° 44' 9" W in the Strait of Georgia, British Columbia, was selected to obtain monthly zooplankton and hydrographic data during 1997 (Fig. 1). The station was located in the area most influenced by the Fraser River, with a depth ca. 400 m; thus both the influence of freshwater in the area and the population of Neocalanus plumchrus would be properly sampled. Samples were obtained during daytime between 10 and 14 hours with oblique plankton hauls using a Bongo net with 202 urn mesh, equipped with a "General Oceanic" flow meter. Mean volume sampled was 73.77 L. Samples were preserved with 5% formalin–seawater buffered with borax. In the laboratory, five replicate 1 ml subsamples were extracted from each sample. Zooplankton contained in sub samples were identified according to specialized literature, and amphipods were identified following Bowman and Gruner (1973) criteria. All organisms were counted under a stereoscopic microscope (Wild M20) and standardized to number of individuals per 1000 m3 seawater (Postel et al., 2000). The zooplankton community composition was analyzed through principal component analysis (PCA), using the software Statistica v5 (StatSoft, Inc.). PCA was performed after abundance values were transformed by applying a double square root, this allowing inclusion of rare species in the analyses (Clarke & Warwick, 1994). The analysis focused on zooplankton species as descriptors of the environment (Ter Braak, 1983). The first three components were selected for analysis considering that the cumulative variance of consecutive components was closest to the total variance (Daultrey, 1976; Dillon & Goldstein, 1984). Results of PCA were plotted and presented with the respective (PSU) salinity data and compared. Salinity was selected over temperature because the Strait of Georgia is a salt–controlled environment (Thomson, 1981). Wet weight zooplankton biomass was obtained by sieving each sample trough 60 urn mesh, allowed to drip 24 hours, and then weighted in an analytical balance. Chlorophyll–a and hydrographic data were provided by Elisabeth Bornhold, and the Department of Fisheries and Oceans, Canada. Chlorophyll–a and zooplankton biomass were used, respectively as indicators of primary and secondary activity.
RESULTS
The monthly pattern of zooplankton Chlorophyll–a variation and during 1997 is shown in Fig. 2. Chlorophyll–a is had the highest value in April, with a small peak in July. Zooplankton peak biomass coincided with April maximum concentration of Chlorophyll–a. It presented a secondary peak in October, apparently not related to chlorophyll a increase. PCA analysis indicated that 83% of the variation in the composition and abundance of zooplankton species was accounted for by the first three components, 67.14% in the first, 8.33% in the second, and 7.43% in the third component. It detected two main zooplankton groups clustered by month during 1997 (Fig. 3). The group I of species dominates during fall–winter to early spring. This group clustered September, October, November, December, January, February and March (Fig. 3). Group II of species dominates from late spring through summer and grouped May to July. The most relevant species contributing to the total variance of each of the three first components are listed in Table 1. Group I was featured by the dominance of the amphipod Cyphocaris challengeri Stebbing, 1888 and the copepod Pseudocalanus minutus Röyer, 1875 and a co–dominance of the amphipod Parathemisto pacifica and the copepods Metridia pacifica Brodsky, 1950, Oithona similis Claus, 1863 and Calanus pacificus Brodsky, 1948 (Fig. 3).
In group II (May–July) the amphipods Cyphocaris challengeri and Parathemisto pacifica, and the cladoceran Podon polyphemoides Leuckart, 1859, the megalopa larvae, the ostracod Conchoecia elegans Sars, 1866, and Polychaetae larvae were codominant (Fig. 3).
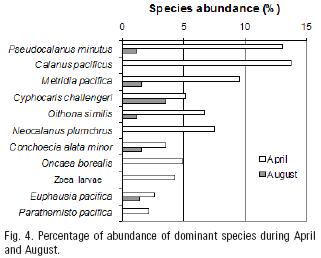
April and August were isolated by the PCA plot from the other groups. Abundant species, common to both months, were Pseudocalanus minutus, Metridia pacifica, Cyphocaris challengeri, Oithona similis and Euphausia pacifica. In this small group Parathemisto pacifica, Calanus pacificus, Neocalanus plumchrus, Oncaea borealis Sars, 1918 and zoea larvae occurred in April only (Fig. 4).
Salinity data are presented in Figure 5. It is represented as isopleths, grouping monthly data as a convenient way to show the vertical salinity structure over the annual cycle. Vertical salinity structure indicates the influence of Fraser River water near the coast of Nanaimo during August 1997, where the sampling station was located. As the spring and summer progressed, increasingly less saline water dominated the upper 10–20 m, reaching its maximum Freshet influence in mid August. This was also the month of maximal stratification, and is bracketed by a progressive deepening of the 28 PSU isoline. It started in April when the 28 isoline was near the surface and reached the maximal depth during late summer in August. From August to December post–Freshet water with 28 PSU tended to surface again (Fig. 5).
DISCUSSION
The PCA of zooplankton species collected during 1997 yielded two main groups that corresponded with a shift of the Fraser River runoff. These species had a high positive contribution to the three first components and represent ecologically dominant species (Jongman et al., 1995) within the zooplankton community. The most important group (Group I) in terms of variance, and therefore with highest diversity (Ter Braak, 1983) was represented by species collected from September to March. This group occurs when conditions of low runoff, limited light, and low plankton production prevail in the Strait of Georgia (Harrison et al., 1983). This fall–winter group is formed by carnivores forms such as Cyphocaris challengeri, Parathemisto pacifica (see Haro, 2004), Oithona similis (see Raymont, 1983), facultative detritivores such as Pseudocalanus minutus (see Poulet, 1976) and Calanus pacificus (see Dilling et al., 1998) and omnivore Metridia pacifica (see Wong, 1988). These species do not feed exclusively on phytoplankton. Therefore, their dominance during fall–winter, a period of low phytoplankton production seems reasonable, and could be explained by their feeding habits. Flagellates and ciliates are abundant during fall and winter (Stockner et al., 1979; Harrison et al., 1983). These forms could provide a food source for these zooplankters. The second group (Group II) included species occurring during late spring to summer (May–July). It coincides with the period when the annual freshet due to the seasonal snow melt occurs and favours intense runoff conditions in the Strait of Georgia (Thomson, 1981). Spring and summer are the most luminous part of the year in temperate areas, and also the most productive season in the Strait of Georgia (Parsons et al., 1970; Harrison et al., 1983). There is usually an early spring phytoplankton bloom in the area (Stockner et al., 1979; Harrison et al., 1983). However in 1997 the spring bloom occurred in the late spring, and the typically dominant copepod Neocalanus plumchrus (Parsons & LeBrasseur, 1969; Fulton, 1973) was not among the most abundant species.
Neocalanus plumchrus was present during April only, but it was about half as abundant as Calanus pacificus, usually a species of moderate abundance in the Strait of Georgia (Harrison et al., 1983). It was during this period, in July, when the expected summer phytoplankton bloom occurred (Parsons et al., 1970); however the zooplankton peak (Harrison et al., 1983) did not follow. April and August outstand separately in the PCA; they are regarded as related to transitional conditions; from spring to summer in April, and from summer to fall in August (Fig. 5). Hydrographically they correspond with a shift in vertical salinity structure from homogeneous to stratified conditions in April and with the change from highly stratified to homogeneous conditions in August (Fig. 4).
Overall vertical salinity changes observed during 1997 coincided with the usual seasonality of Fraser River runoff, as measured at Marguerite, British Columbia (MELP, 1998): Low flow (500–1000 m3 s–1) dominated from November 1996 to March 1997; high flow (3000–3500 m3 s–1) dominated from May to mid July; and moderate–low flow returned from August on. Considering that the reported average of the Fraser River flow is 13000 m3 s–1 (Morrison et al., 2002), the year of 1997 (MELP, 1998) was one of a very low flow. Furthermore, a lower Fraser River total annual flow occurring earlier in the year is among properties of climate that have been observed changed during the last century (http://www.gov.bc.ca/wlap). To this respect, it has been proposed that climate changes will affect river runoff that will affect the estuarine environment by changing estuarine hydrography and plankton dynamics, this in turn will cascade affecting estuarine water residence time, seawater dilution, vertical stratification, nutrient delivery and phytoplankton growth rates, as researches proposed climate changes will affect the estuarine environment (Moore et al., 1997; Howarth et al., 2000). Consequently affecting zooplankton production and reducing survival of subsequent trophic levels due to effects on the food chain.
Lower runoff levels during 1997 were coincidental with scarcity of the characteristic dominant zooplankton species (i. e., Neocalanus plumchrus and Euphausia pacifica), and a change in species composition. Species expected to dominate the zooplankton were the copepods N. plumchrus, Calanus marshallae Frost 1974, C. pacificus, and Pseudocalanus minutus (Parsons & LeBrasseur, 1969) and the euphausiid E. pacifica (Harrison et al., 1983) They are the most abundant zooplankton species, and therefore basically responsible for the transfer of energy from the primary producers to higher consumers in the Strait of Georgia (Parsons & LeBrasseur, 1969; Harrison et al., 1983). Variation in their abundance influences zooplankton biomass, except for P. minutus, which is extremely numerous but very small. N. plumchrus and E. pacifica had very low densities in 1997 as compared to 1996 and 1998 (Bornhold, 1999). For N. plumchrus, this was considered to be a consequence of its presence in the surface layer at the progressive waning of an early–spring phyto–plankton bloom (Bornhold, 1999); it is speculated that this condition prevented copepods from taking full advantage of this food source. The early phytoplankton bloom in 1997 was apparently related to a strong El Niño event (Macdonald, 2000). Its local influence apparently affected primary production through the early onset of the phytoplankton bloom in the Strait of Georgia, as it had happened under previous El Niño conditions in 1992 (Yin et al., 1997).
In conclusion, the timing in the development of a described spring bloom (Yin et al., 1997) together with the zooplankton dominant species must be a significant factor in the trophodynamic phasing in food chains. We speculate that a combined effect of climate changes, reducing the Fraser River flow and an El Niño event most probably caused environmental conditions during 1997 in the Strait of Georgia. Biologically this was observed as a scarcity of characteristic dominant zooplankton species, and a change in species composition. Low abundance of Neocalanus plumchrus and Euphausia pacifica affected the zooplankton biomass and consequently may have influenced the trophic ecology in the Strait. They both are part of the diets of juvenile salmon, and their lower abundance may diminish prey availability for salmon species that feed in the Strait. On the other hand, Parathemisto pacifica, the local most abundant hyperiid amphi–pod in the Strait, may play an important role in trophic dynamics at times when N. plumchrus and E. pacifica are scarce. Both P. pacifica and Cyphocaris challengeri feed on small zooplankters that are mostly omnivorous and/or facultative predators that feed on nanoplankton (Haro, 2004). By concentrating food energy and biomass from small zooplankters, amphipods may represent an alternative source of energy for fish species that consume them. This suggests P. pacifica and C. challengeriare an important link in the trophic ecology of the Strait of Georgia. Shifts in zooplankton species dominance most likely have an effect on Pacific salmon species that feed on zooplankton during their juvenile stages.
ACKNOWLEDGEMENTS
Alan. G. Lewis provided advice and support during this research and Paul .E. Harrison supplied the zooplankton samples for this work. CONACYT and the Instituto Politécnico Nacional, México provided partial funding for this study. Elisabeth A. Bornhold provided Chlorophyll–a data. Robin Williams (Department of Fisheries and Oceans) provided hydrographic data. Melissa Evanson collaborated with field sampling. Robin Williams provided Chlorophyll–a and hydrographic data. The Pacific Biological Station from the Department of Fisheries and Ocean at Nanaimo, British Columbia provided ship and support for collecting zooplankton samples. Mr. I. Fogel provided valuable editing work to a previous draft. Dr. Eduardo Suarez, Dr. Jorge Ciros and an anonym reviewer provided insightful comments to a previous manuscript.
REFERENCES
Beacham, T. D. 1986. Type, quantity, and size of food of pacific salmon (Oncorhynchus) in the Strait of Juan de Fuca, British Columbia. Fisheries Bulletin 84: 77–89. [ Links ]
Bornhold, E. A. 1999. Interannual and interdecadal patterns in timing and abundance of phytoplankton and zooplankton in the Strait of Georgia, B. C. M. Sc. Thesis. Department of Earth and Ocean Sciences. The University of British Columbia. Vancouver, Canada. 122 p. [ Links ]
Bowman, T. E. & H. E. Gruner. 1973. The families and the genera of Hyperiidea (Crustacea, Amphipoda). Smithsonian Contributions to Zoology 146: 1–64. [ Links ]
Brodeur, R. D. 1990. A synthesis of the food habits and feeding ecology of salmonids in marine waters of the North Pacific. (INPFC Doc.). FRI–UW–9016. Fish. Res. Inst., Univ. Washington, Seattle. 38 p. [ Links ]
Clarke, K. R. & R. M. Warwick. 1994. Change in marine communities. An approach to statistical analysis and interpretation. Plymouth Marine Laboratory, UK. Natural Environment research Council, UK. Bourne Press Limited, Bournemouth. 144 p. [ Links ]
Cushing, D. H. 1978. Biological effects of climate change. Rapports et Procés – Verbaux des Réunions du Conseil International pour l'Exploration de la Mer 173: 107–116. [ Links ]
Daultrey, S. 1976. Principal components analysis. Concepts and techniques in modern geography series No. 8. Norwich: Geo Abstracts Ltd, University of East Anglia, Norwich. 50 p. [ Links ]
Dilling, L, J. Wilson, D. Steinberg & A. Alldredge. 1998. Feeding by the euphausiid Euphausia pacifica and the copepod Calanus pacifi–cus on marine snow. Marine Ecology Progress Series 170: 189–201. [ Links ]
Dillon, W. R. & M. Goldstein. 1984. Multivariate analysis. Methods and applications. John Wiley and Sons, New York. 587 p. [ Links ]
Fulton, J. D. 1973. Some aspects of the life history of Neocalanus plumchrus in the Strait of Georgia. Journal of the Fisheries Board of Canada 30: 811–815. [ Links ]
Haro, G. M. J. 2004. Diet and functional morphology of the mandible of two planktonic amphipods from the Strait of Georgia, British Columbia, Parathemisto pacifica (Stebbing, 1888) and Cyphocaris challengeri(Stebbing, 1888). Crustaceana 76: 1291–1312. [ Links ]
Healey, M. C. 1980. The ecology of juvenile salmon in the Strait of Georgia, British Columbia. In: McNeill, W. C. (Ed.), Salmonid Ecosystems of the North Pacific. Oregon State University Sea Grant College Program and Oregon State University Press, pp. 203–230. [ Links ]
Harrison, P. J., J. D. Fulton, F. J. R. Taylor & T. R. Parsons. 1983. Review of the biological oceanography of the Strait of Georgia: Pelagic environment. Canadian Journal of Fisheries and Aquatic Sciences 40: 1064 –1094. [ Links ]
Howarth, R. W. D. P. Swaney, T. J. Butler & R. Marino. 2000. Climatic Control on Eutrophication of the Hudson River Estuary. Ecosystems 3: 210–215. [ Links ]
Jongman, R. H. G., C. J. F. Ter Braak & O. F. R. Van Tongeren. 1995. Data Analysis in Community and Landscape Ecology. Cambridge University Press, Great Britain. 299 p. [ Links ]
LeBlond, P. H. 1983. The Strait of Georgia: functional anatomy of a coastal sea. Canadian Journal of Fisheries and Aquatic Sciences 40: 103–163. [ Links ]
LeBrasseur, R. J., W. E. Barraclough, O. D. Kennedy & T. R. Parsons. 1969. Production studies in the Strait of Georgia. Part III. Observations on the food of larval and juvenile fish in the Fraser River plume, February to May, 1967. Journal of Experimental Marine Biology and Ecology 3: 51–61. [ Links ]
MacDonald, J. S. 2000. Mortality during the migration of Fraser River sockeye salmon (Oncorhynchus nerka): a study of the effect of ocean and river environmental conditions in 1997. Canadian Technical Reports of Fisheries and Aquatic Sciences 2315. Canada. 215 p. [ Links ]
MELP: Ministry of Environment, Lands and Parks, 1998: Government of British Columbia. Canada. River Forecast Centre.Fraser River at Marguerite: http://www.gov.bc.ca/rfc/archive/1998/19980516/dgfrsrun.html. [ Links ]
Moore, M. V., M. L. Pace, J. R. Mather, P. S. Murdoch, R. W. Howarth, C. L. Folt, C. Y. Chen, H. F. Hemond, P. A. Flebbe & C. T. Driscoll. 1997. Potential effects of climate change on freshwater ecosystems of the New England/mid Atlantic region. Hydrological Processes 11: 925–947. [ Links ]
Morrison, J., M. C. Quick & M. G. G. Foreman. 2002. Climate change in the Fraser River watershed: flow and temperature projections. Journal of Hydrology 263:230–244. [ Links ]
Parker, R. R. & B. A. Kask. 1972a. Second Progress Report on studies of the ecology of the outer Squamish estuary May 16–17, 1972. Fisheries Board of Canada Manual Report Series 1193. [ Links ]
Parker, R. R. & B. A. Kask 1972b. Fourth progress report on studies of the ecology of the outer Squamish estuary July 12, 1972. Fisheries Board of Canada Manual Report Series 1195. [ Links ]
Parsons, T. R. & R. J. LeBrasseur. 1969. Production studies in the Strait of Georgia. I. Primary production under the Fraser River plume. February to May, 1967. Journal of Experimental Marine Biology and Ecology 3: 27–38. [ Links ]
Parsons, T. R., R. J. LeBrasseur & W. R. Barraclough. 1970. Levels of production in the Strait of Georgia, British Columbia: A review. Journal of the Fisheries Board of Canada 27: 1251 –1264. [ Links ]
Postel, L., H. Fock & W. Hagen. 2000. Biomass and Abundance. In: Harris, E. P., Wiebe, J., Lenz H. R, Skjoldal R. & Huntley M. (Eds.). ICES Zooplankton Methodology Manual. Academic Press. San Diego, pp. 83–192. [ Links ]
Poulet, S. A. 1976. Feeding of Pseudocalanus minutus on living and non–living particles. Marine Biology 34: 117–125. [ Links ]
Raymont, J. E. G. 1983. Plankton and productivity in the oceans. 2nd Edition. Vol. 2. Pergamon Press, Southampton. 824 p. [ Links ]
St. John, M. A., J. S. Macdonald, P. J. Harrison, R. Beamish & E. Choromanski. 1992. The Fraser River plume: some preliminary observations on the distribution of juvenile salmon, herring and their prey. Fisheries Oceanography 1: 153–162. [ Links ]
Stockner, J. G., D. S. Cliff & K. R. S Shortreed. 1979. Phytoplankton ecology of the Strait of Georgia, British Columbia. Canadian Journal of Fisheries and Aquatic Sciences 36: 657–666. [ Links ]
Ter Braak, C. J. F. 1983. Principal Components biplots and alpha and beta diversity. Ecology 64: 454–462. [ Links ]
Thomson, R. E. 1981. Oceanography of the British Columbia coast. Canadian Special Publications Fisheries and Aquatic Sciences 56: 1–291. [ Links ]
Water, Air and Climate Change Branch, Ministry of Water, Land and air Protection. Government of Canada, http://www.gov.bc.ca/wlap. [ Links ]
Wong, C. K. 1988. Effects of competitors, predators, and prey on the grazing behavior of herbivorous calanoid copepods. Bulletin of Marine Science 43: 573–582. [ Links ]
Yin, K., P.J. Harrison, R.H. Goldblat, M.A. St. John & R. Beamish. 1997. Factors controlling the timing of the spring bloom in the Strait of Georgia estuary, British Columbia, Canada. Canadian Journal of Fisheries and Aquatic Sciences 54: 1985–1995. [ Links ]














