Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Hidrobiológica
versión impresa ISSN 0188-8897
Hidrobiológica vol.17 no.1 Ciudad de México abr. 2007
Notas científicas
Antibacterial activity in the hemolymph of the catarina scallop Argopecten ventricosus
Actividad antibacteriana en la hemolinfa de la almeja catarina Argopecten ventricosus
Antonio Luna-González1, Alfonso Maeda-Martínez2, Ángel Campa-Córdova2, and Javier Orduña-Rojas1
1 Centro Interdisciplinario de Investigación para el Desarrollo Integral Regional, Km. 1 Carretera a Las Glorias, Guasave, Sinaloa, México. C. P. 81101. E-mail: aluna@ipn.mx
2 Centro de Investigaciones Biológicas del Noroeste (CIBNOR), Mar Bermejo 195. Col. Playa Palo de Santa Rita. La Paz, B.C.S., 23090 México.
Recibido: 10 de febrero de 2006
Aceptado: 19 de octubre de 2006
Abstract
We conducted a search for antibacterial peptide like activity in hemolymph of Argopecten ventricosus. Pre-purification of peptides was done by reverse phase HPLC. Hemolymph acidic supernatant was loaded into a column packed with C18 matrix. Stepwise elutions were performed with 5, 50, 80, and 100 % acetonitrile (ACN) in 0.05 % trifluoroacetic acid (TFA) over 550 min at a flow rate of 0.2 ml/min. Absorbance was monitored at 280 nm. Eluted fractions were concentrated under vacuum. Vibrio alginolyticus 138-2 was used as a model to test fractions. Growth inhibition zone were observed at 5 and 50 % ACN. This is the first report of antimicrobial peptide-like activity in hemolymph of a pectinid species.
Keywords: Antimicrobial peptide, Argopecten ventricosus, hemolymph, immunity, Vibrio alginolyticus.
Resumen
Se realizó una investigación para demostrar que la hemolinfa de Argopecten ventricosus presenta actividad tipo péptidos antimicrobianos. La prepurificación de los péptidos se realizó por medio de HPLC, con una columna de fase reversa. El sobrenadante ácido de la hemolinfa fue cargado en una columna empacada con matriz C18. Las eluciones escalonadas se realizaron con 5, 50, 80 y 100 % de acetonitrilo (ACN), en 0.05 % de ácido trifluoroacético (ATA), durante 550 min y un flujo de 0.2 ml/min. La absorbancia se monitoreó a 280 nm. Las fracciones eluídas se concentraron al vacío. Vibrio alginolyticus 138-2 se usó como modelo para probar la actividad antimicrobiana de las fracciones. Se observaron zonas de inhibición del crecimiento bacteriano en las fracciones con 5 y 50 % de ACN. Este es el primer reporte de actividad tipo péptido antimicrobiano en la hemolinfa de una especie de pectínido.
Palabras clave: Argopecten ventricosus, hemolinfa, inmunidad, péptido antimicrobiano, Vibrio alginolyticus.
Bacterial diseases, most of which are caused by bacteria belonging to the genus Vibrio, have been reported as a limiting factor in mollusc aquaculture (Sainz et al., 1999). Efforts have been done in the search of strategies to prevent the infestation of bacteria, as well as in the research of the immunological mechanisms of cultured organisms. The study of antimicrobial peptides (APs) is one of the mayor topics in the field of immunology because they represent a key element of the innate immune response of many organisms (Boman, 1995). APs are small molecules (less than 10 kDa) that tend to display a broad-spectrum antimicrobial activity and possess cationic charge at physiological pH (Boman, 1995). Their positive charge presumably facilitates interactions with the negatively charged bacterial membrane and/or acidic bacterial cell walls, whereas their amphibolic character enables membrane permeabilization (Nissen & Nes, 1997). Here we report the presence of antimicrobial peptide-like activity in the hemolymph of the scallop Argopecten ventricosus (Sowerby II, 1842).
Organisms (150, 56.2 ± 2.1 mm) were collected from Bahia Magdalena, B.C.S., Mexico. In the laboratory, specimens were placed in 1,100 litre fibreglass tank containing areated seawater at 24±1 °C, 36 %thou salinity and fed with a microalgae mixture.
Individuals were bled 1 ml from the posterior adductor muscle by inserting a 27-gauge needle attached to a 1 ml sterile plastic syringe. Samples from 150 specimens were pooled in sterile glass tubes and kept on ice. Hemolymph was diluted (v/v) with a solution of 0.1 % trifluoroacetic acid (TFA) in the presence of aprotinin (1.5 µM, final concentration) and phenylthiourea (20 µM, final concentration) (Charlet et al., 1996). Acidic extraction was performed for 30 min in an ice-cold water bath under gentle stirring, and the extract resultant was centrifuged at 10,000 x g for 20 min at 4 °C (Charlet et al., 1996).
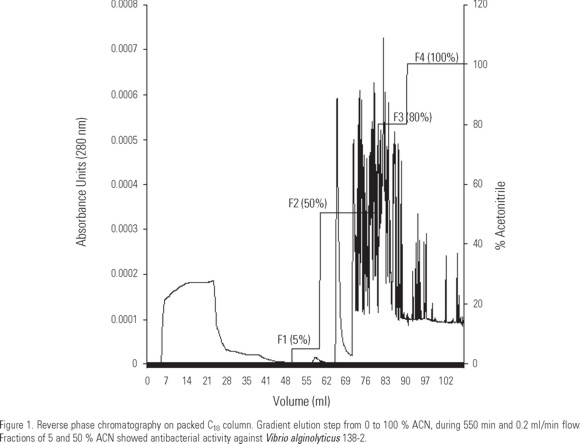
Reverse phase HPLC was performed in an Äkta equipment (Pharmacia). After centrifugation, acidic supernatant was loaded into a 50 cm column (Pharmacia) packed with 5 ml of C18 matrix and equilibrated with acidified water (0.05 % TFA). Stepwise elutions were performed with 5, 50, 80, and 100 % acetonitrile (ACN) in 0.05 % TFA over 550 min at a flow rate of 0.2 ml/min. Absorbance was monitored at 280 nm. The eluted fractions corresponding to each ACN percentage were concentrated under vacuum (SpeedVac System AES 2010, Savant), reconstituted and pooled in 60 µl MilliQ water.
Cultures of Vibrio alginolyticus 138-2 were done in LB broth medium supplemented with 0.5, 1, 3, 5 and 8 % NaCl and grown with constant shaking at 37 °C for 0, 2, 4, 8, 12, 16, and 24 h. Each sample was grown separately in 1 ml volume (990 µl of LB broth plus 10 µl of the stock at -80 °C). Growth was estimated by reading absorbance of the cultures in a Spectronic Genesys 2 spectrophotometer at 580 nm.
Vibrio alginolyticus cells from a 24 h culture in LB broth were harvested by centrifugation (9,000 x g for 20 min, Beckman GS-15 R centrifuge), washed twice in a 0.5 % NaCl sterile saline solution and resuspended in the same solution to achieve an optical density (580 nm, Genesys 2 spectrophotometer) of 1. To assess the number of bacteria per mililiter, a count of viable colony forming units (CFU) was performed by 10-fold serial dilutions.
Antibacterial activity of reverse phase chromatography fractions was monitored by radial diffusion assay (Lehrer et al., 1991). A volume (50 µl) of diluted bacterial stock containing ≈106 CFU was added to 8 ml of melted sterile LB-agarose (LB: 1 % bactotrypton, 0.5 % yeast extract, 0.5 % NaCl, 0.6 % agarose, pH 7.0). After hand shaking, the bacteria with LB-agarose were poured onto a compartment Petri dish. Five microliters of fractions, positive control (penicillin diluted 1:1000 in MilliQ water), and negative control (MilliQ water) were added in small wells (3 mm diameter). The plates were incubated for 24 h at 37 °C and the diameter of the clear zone surrounding the wells was measured with a ruler.
Vibrio alginolyticus can grow over NaCl concentrations from 0.5 to 8 %, showing faster growth response at lower salt content. In view of these Results and due to the possible presence of some salt sensitive peptides in the eluted fractions (Goldman et al., 1997; Lee et al., 1997), LB media supplemented with 0.5 % NaCl was chosen to grow the Vibrio strain in fractions tested. Inhibition of antimicrobial activity of cationic peptides by salt is not well known. It is possible that simple charge competition might inhibit the initial interactions between a cationic peptide and the negatively charged bacterial membrane (Lehrer et al., 1993).
Results (Fig. 1) showed a 12 mm diameter clear zone in fraction F1 (5 % ACN), 21 mm in fraction F2 (50 % ACN) and 30 mm in positive control (penicillin treatment). Negative control (MilliQ water), F3 (80 % ACN), and F4 (100 % ACN) fractions did not showed clearing zones. The antibacterial activity found in the hemolymph of A. ventricosus indicates that this species could have a marked antimicrobial peptide activity as it has been shown by another bivalve molluscs like Mytilus edulis Linnaeus, 1758 (Charlet et al., 1996), Mytilus galloprovincialis Lamark, 1819 (Mitta et al., 1999), Modiolus modiolus Linnaeus, 1758 (Haug et al., 2004), and Mytilus edulis chilensis Hopé, 1854 (Mercado et al., 2005). APs from marine bivalves have been normally isolated from the immune cells, haemocytes (Charlet et al., 1996; Mitta et al., 1999). However, antimicrobial peptide activity has been also detected in gills of M. edulis chilensis (Mercado et al., 2005) and in extracts from several tissues of M. modiolus, including plasma, haemocytes, labial palps, byssus, mantle, and gills (Haug et al., 2004).
In this paper we report for first time the presence of antimicrobial peptide-like activity in A. ventricosus hemolymph.
We are grateful to Dr. H. Tokuda (University of Tokyo. Tokyo, Japan) for generous Vibrio alginolyticus 138-2 strain donation. We thank to Dr. Ira Fogel and Dra. Melina López-Meyer for English improvement of the manuscript.
References
BOMAN, H.G. 1995. Peptide antibiotics and their role in innate immunity. Annual Review of Immunology 13: 61-92. [ Links ]
CHARLET, M., S. CHERNYSH, H. PHILIPPE, C. HÉTRU, J. HOFFMANN & P. BULET. 1996. Innate immunity, isolation of several cysteine-rich antimicrobial peptides from the blood of mollusc, Mytilus edulis. Journal of Biological Chemistry 271: 21808-21813. [ Links ]
GOLDMAN, M. J., G.M. ANDERSON, E.D. STOLZENBERG, U.P. KARI, M. ZASLOFF & J. WILSON. 1997. Human β-defensin-1 is a salt-sensitive antibiotic in lung that is inactivated in cystic fibrosis. Cell 88: 553-560. [ Links ]
HAUG, T., K. STENSVAG, O.M. OLSEN, E. SANSDDALEN & O.B. STYRVOLD. 2004. Antibacterial activities in various tissues of the horse mussel, Modiolus modiolus. Journal of Invertebrate Pathology 85(2): 112-119. [ Links ]
LEHRER, R.I., M. ROSENMANN, S.S.L. HARWIG, R. JACKSON & S. EISENHAUER. 1991. Ultrasensitive assays for endogenous antimicrobial poly-peptides. Journal of Immunological Methods 137: 167-173. [ Links ]
LEHRER, R.I., A.K. LICHTENSTEIN & T. GANZ. 1993. Defensins: antimicrobial and cytotoxic peptides of mammalian cells. Annual Review of Immunology 11: 105-128. [ Links ]
LEE, I.H., Y. CHO & R.I. LEHRER. 1997. Effects of pH and salinity on the antimicrobial properties of clavanins. Infection and Immunity 65: 2898-2903. [ Links ]
MERCADO, L., P. SCHMITT, S.H. MARSHALL & G. ARENAS. 2005. Gill tissues of the mussel Mytilus edulis chilensis: A new source for antimicrobial peptides. Electronic Journal of Biotechnology 8(3): 284-290. [ Links ]
MITTA, G., F. HUBERT, T. NOEL & P. ROCH. 1999. Myticin, a novel cysteinerich antimicrobial peptide isolated from haemocytes and plasma of the mussel Mytilus galloprovincialis. European Journal of Biochemistry 265: 71-78. [ Links ]
NISSEN, J. & I.F. NES. 1997. Ribosomally synthesized antimicrobial peptides: their function, structure, biogenesis, and mechanism of action. Archives of Microbiology 167: 67-77. [ Links ]
SAINZ, J.C., A.N. MAEDA & F. ASCENCIO. 1999. Experimental vibriosis induction with Vibrio alginolyticus of larvae of the cataria scallop (Argopecten ventricosus=circularis) (Sowerby II, 1842). Microbial Ecology 35: 188-192. [ Links ]














