Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Hidrobiológica
versão impressa ISSN 0188-8897
Hidrobiológica vol.17 no.1 Ciudad de México Abr. 2007
Artículos
Littoral free living nematode fauna of Socorro Island, Colima, Mexico
Nematofauna de vida libre en el litoral de Isla Socorro, Colima, México
Alberto de Jesús-Navarrete
Departamento de Aprovechamiento y Manejo de Recursos Acuáticos. El Colegio de la Frontera Sur (ECOSUR), A.P. 424. Chetumal Q. Roo, México C. P.77900. E-mail: alberto@ecosur-qroo.mx
Recibido: 15 de noviembre de 2005
Aceptado: 21 de septiembre de 2006
Abstract
In order to study the littoral nematode fauna in the Mexican Pacific, sediment samples were collected in three sites at Socorro Island, during November 1997. A total of 30 species of nematodes, from three orders and 15 families were determined. Order Chromadorida had 20 species, with Metachromadora (Chromadoropsis) sp. and Rhips sp. as the most abundant genus (19 and 17 ind./0.008 m2, respectively). Monhysterida had six species and Enoplida four species, being Enoplolaimus sp. the most abundant genera with 14 ind./0.008 m2. Higher number of species was found at Bahia Blanca (24). Six species are considered cosmopolitan; two tropical and 21 are undescribed. All species are first records for the Mexican Pacific Ocean.
Key words: Colima, Mexico, Nematodes, Pacific Ocean.
Resumen
Con el fin de estudiar la nematofauna litoral en el Pacífico mexicano, se colectaron muestras de sedimento en tres sitios de Isla Socorro durante noviembre de 1997. Un total de 30 especies de nemátodos, pertenecientes a tres órdenes y 15 familias, fueron identificadas. El orden Chromadorida tuvo 20 especies, siendo Metachromadora (Chromadoropsis) sp. y Rhips sp. los géneros más abundantes (19 y 17 ind./0.008 m2, respectivamente). El orden Monhysterida tuvo seis especies y el Enoplida cuatro especies, siendo Enoplolaimus sp. el género más abundante con 14 ind./0.008 m2. El mayor número de especies se encontró en Bahía Blanca (24). Seis especies se consideran cosmopolitas, dos tropicales y 21 especies no están descritas. Todas las especies representan los primeros registros para el Pacífico mexicano.
Palabras clave: Colima, México, Nemátodos, Océano Pacífico.
Introduction
The free-living marine nematode fauna has been studied in many regions of the world; however, the research effort in some geographical regions has not been extensive, particularly in the tropics (Coull, 1988; Boucher & Lambshead, 1995). In the Northeast Pacific, Allgen (1947) studied the nematode fauna of La Jolla, California; Chitwood (1960) studied nematodes from Northern California; and Murphy (1962) described three new species from the coast of Oregon. In Southern California, Jones (1964) re-described Bolbella californica and Hope (1967a, b) described three new species of nematodes from the West coast of North America. However, there are very few reports dealing with free living marine nematodes in subtropical latitudes. Allgen (1947) studied the nematodes off the Panama coast; and Gerlach (1955) described new nematode species from San Salvador. In the Clarion-Clipperton fracture zone of the Eastern Pacific, Renaund-Mornant & Gourbault (1990) described the abyssal meiobenthos, including some nematodes; and more recently Lambshead et al. (2003) addressed the use of nematodes as biomonitoring organisms, including information about their biodiversity.
Nematodes are considered between the most abundant and diverse group on Earth (Gaston, 1991; Lambshead & Boucher, 2003) and the study of their distribution and abundance are important in order to understand the biogeographic aspects of the group (Heip et al., 1982; Boucher & Lambshead, 1995). Additionally, nematodes have been proposed as bioindicators under environmental stress (Platt, 1984; Lambshead, 1986; Warwick & Clarke, 1998). In spite of this importance, the study of marine nematode fauna in the Mexican Pacific Ocean is absent.
The earliest scientific studies carried out at Socorro Island were related with algae distribution (Albert & Lyon, 1930), hydrology (Villalobos, 1960), mollusk distribution, (Bratcher & Burch, 1971) and fishes (Hernández & Pagés, 1976). Recently, physical and faunistic characteristics were reviewed by Llinas et al. (1993); whereas Mille-Pagaza et al. (2003) studied the decapod community in the littoral zone, but research on the coastal meiofauna and particularly nematodes has been missed. This is the first report dealing with marine nematodes from the Mexican Pacific, and the main goal is to describe the littoral free-living nematode fauna of Socorro Island, in order to increase the knowledge of regional biodiversity in this poorly studied area.
Materials and methods
Study Area
The Revillagigedo archipelago is located at 18° 20' and 18° 48' N, 110° 45'and 114° 50' W. Socorro, Clarion, San Benedicto and Roca Partida are the main islands, of which the largest (210 km2) is Socorro. It is a marine volcano (1040 m altitude), with plain, ravines and lava cords, descending to the sea level (Fig. 1).
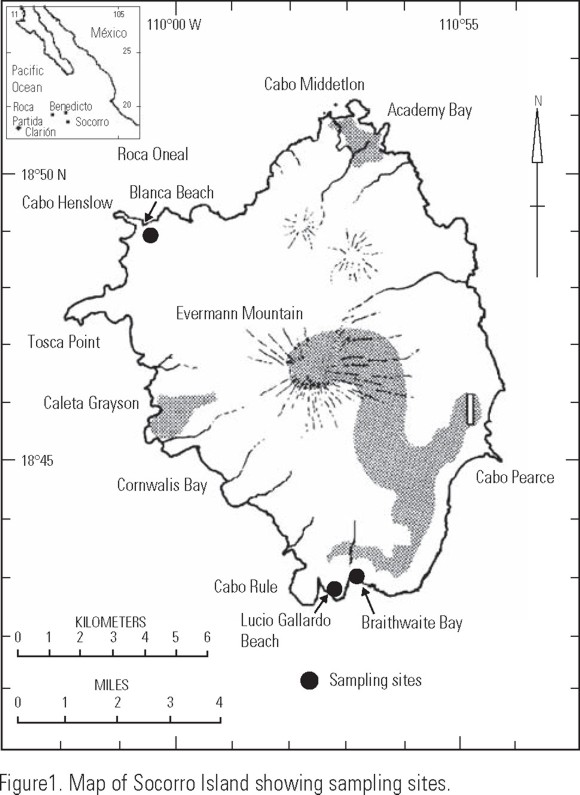
Samples were collected during the oceanographic cruise SURPACLIPP-1, on board R/V "El Puma" during November 1997, in which a littoral survey to Socorro Island was carried out. Five sediment samples were collected manually in three sites using a 5 cm diameter PVC corer (area 0.008 m2) that was introduced 10 cm in the sediment. Two corers were collected at Braithwaite Bay (BB), two at Blanca Beach (Bb), and one at Lucio Gallardo Beach (LGb), in the supra-littoral, intertidal and infra-littoral zones (0-1.5 m depth) (Fig. 1). Nematode density was expressed as ind./0.008 m2. Samples were fixed immediately with a 10% buffered formalin solution. In the laboratory, meiofauna was extracted by successive decantation, collecting organisms in a 35 µm mesh (Hulings & Gray, 1971). Nematodes were manually separated from the meiofauna and mounted on glass slides with anhydrous glycerine. Genera determination was done using pictorial keys (Platt & Warwick, 1983), whereas species were determined with the original descriptions. Systematic hierarchy was that proposed by Lorenzen (1981). Feeding type (FT) of each species was determined following Wieser's classification: selective (1A), non-selective (1B), deposit feeders, epistrate feeders (2A) and predators/omnivores (2B) (Wieser, 1953).
Sediments were analysed to medium grain size (Mgs) expressed as (φ) scale, standard deviation (STD) and classification (CLAS), according to Buchanan (1984). Organic matter percentage (% OM) content was determined by mass loss after combustion; sediments were placed in a furnace and combusted to 550° C for one hour (Dean, 1974).
Results
A total of 156 nematodes, belonging to 30 species from three orders and 15 families were collected from Socorro Island. Chromadorida had eight families and 20 species, Monhysterida three families and six species and Enoplida three families and four species. Six species are considered cosmopolitan; two tropical and 21 are in a description process, and they will be published posteriorly.
Among the most important Chromadorida families were: Desmoscolecidae and Chromadoridae (5 spp.), followed by Desmodoridae (3 spp.) and Comesomatidae (2 spp.). Metachromadora (Chromadoropsis) sp. and Rhips sp. were the species with higher abundance (19 and 17 ind./0.008 m2 representing a density of 2375 and 1751 ind./10 cm2 , respectively).
In the Monhysterida, Xyalidae was the richest family (4 spp.), followed by Sphaerolaimidae and Linhomoeidae with one species each. Three families of the Enoplida were present: Ironidae (2 spp.), Oncholaimidae and Thorascostomopside with 1 species each. Enoplolaimus sp. was the most abundant species with 14 ind./0.008 m2, other genera were poorly represented.
Enoplolaimus sp., Paracyatholaimus paramucronata, and Theristus sp.1 had a wide distribution, since they were collected in three of the five sites at Socorro Island (Table 1). Blanca Beach (BbI) had the higher number of species (24). Braithwaite Bay (BBI) had four species and Lucio Gallardo Beach (LGb) only two species (Table 1).
Sediment at Lucio Gallardo Beach was very coarse volcanic sand (VCS) (1.23 mm medium size); whereas at two sites at Braithwaite Bay, sediment was coarse sand (CS) (1.18 mm medium size). Both sites possessed well sorted sediments composed by spherical sand particles. At Blanca Beach, sediment was medium sand (MS) (0.45 mm) and it was poorly sorted. Organic matter was less than 2% in all stations (Table 2).
More than one third (36.7%) of the species was epistrate feeders, 26.7% were selective deposit feeders, 23.3% were selective filters, whereas 13.3% were predators.
Discussion
Some of the species found in Socorro Island have been reported in other marine sites around the world, and they can be considered cosmopolitan (Boucher & Lambshead, 1995). In the Pacific Ocean, Spilophorella paradoxa De Man, 1888, Syringolaimus striatocaudatus De Man, 1888, Viscosia glabra Bastian, 1865 and Euchromadora vulgaris Bastian, 1865, have been reported at Contadora and Perla Islands in Panama, San Diego Bay and La Jolla, in California (Allgen, 1947). Cheironchus vorax Cobb, 1917, has been reported in Australia but not in America's Pacific Ocean areas (Fisher, 2003). Cheironchus vorax specimens recollected during this survey were more related to the redescription of the species collected in the Gulf of Mexico by Castillo-Fernandez & Decramer (1993); however, they differ in the number of precloacal supplements, but this could be product of the plasticity in this nematode's structure (Tchesunov & Okhlopkov, 2006).
Thalassironus britannicus De Man, 1889, recorded in Socorro, is more similar to the specimens from Japan in terms of their De Man ratios (Yoshimura, 1980), than to T. britannicus reported by Chitwood (1960) in California.
Differences in the species number among the three sites of sampling might be related with the very coarse sediments present in Braithwaite Bay and Lucio Gallardo Beach, where a low diversity was observed. Well sorted sediments have less interstitial spaces than sediments poorly sorted ones, and it can support high species diversity (Gray, 2000). It is possible that desiccation conditions between the sub-littoral and littoral area by effect of tides, influence nematode distribution.
Nematode densities at Socorro Island ranged between 125 to 2375 ind./10 cm2 , which are similar to densities reported in other coastal areas. In Australia, Fisher (2003) reported a density of 609-2744 ind./10 cm2 for a nematode assemblage associated with marine seagrasses. In the West Indian Ocean, Muthumbi et al. (2004) recorded nematode densities between 660 to 1928 ind./10 cm2 at 50 m depth. Our values were superior to Norwegian Sea, where a nematode density of 107-204 ind./10 cm2 was reported (Jensen, 1988).
A similarity of species in shallow waters has been noted as a characteristic of marine nematodes (Castillo-Fernández & Lambshead, 1990). In coastal environments, a dominance of Chromadorida and Monhysterida is frequent and this fact has been related with the ecological conditions, especially food availability (Jensen, 1984; 1985; Fisher, 2003). At Socorro Island, the same pattern was observed, where Chromadoridae was dominant. A high percentage of epistrate feeders (36.67%) were evident in the sampling sites, and it could indicate high diatom abundance. This fact has been observed in other temperate and subtropical latitudes (Tietjen, 1969; Ndaro & Olafsson, 1999). In shallow waters, Dichromadora and Neochromadora have been associated with diatom feeding, using the teeth to pierce or break the frustule (Jensen, 1984; Romeyn & Bowman, 1983). However, nematodes have a feeding diversity that produce a partition of food resources in the ecosystem, and this generate a higher diversity (Platt & Warwick, 1983). It is evident by the presence of filter feeding and predators in our samples. Species with wide distribution could be related with food availability; organic mater, in the case of deposit feeders, and benthic diatoms in the case of epistrate feeders (Moens & Vincx, 2000).
All species reported in this survey represent new records for Mexico and for the Eastern Tropical Pacific. Of the total species registered, 21 probably are new species to science (68%), but in some cases, it will be necessary to have more specimens to elucidate their descriptions. However, it is clear that the collections of more samples at Socorro Island will contribute to a better understanding of marine nematode fauna distribution.
Acknowledgments
I wish to express my gratefulness to the collector, Ana Noemí Canales Cáceres† and the SURPACLIP-1 Chief Scientist Vivianne Solís, for allowing me to study this interesting material. Comments of Jacobo Schmitter-Soto, David González and two anonymous reviewers improved the manuscript substantially.
References
ALBERT, S.W. & G.M. LYON. 1930. Marine algae of the Revillagigedo Islands Expedition in 1925. Proceedings of California Academic Science 19(11):109-215. [ Links ]
ALLGEN, C. 1947. West American Marine Nematodes. Vidensk Medd fra Dansk Nathurh Foren 110:65-219. [ Links ]
BOUCHER, G. & P. J. D. LAMBSHEAD.1995. Ecological Biodiversity of Marine Nematodes in samples from Temperate, Tropical and Deep-Sea Regions. Conservation Biology 9:1594-1604. [ Links ]
BRATCHER, T. & R. R. BURCH.1971. The Terebridae (Gastropoda) of Clarion, Socorro, Cocos and Galapagos Islands. Proceedings of California Academic Science 37(21):537-566. [ Links ]
BUCHANAN, J.B. 1984. Sediment analysis In: A. Holme & R. McIntyre (Eds). Methods for the study of Marine Benthos. Blackwell London, pp. 41-65. [ Links ]
CASTILLO-FERNÁNDEZ, D. & W. DECRAMER. 1993. Cheironchus paravorax n.sp. and Cheironchus vorax Cobb, 1917 from the Campeche Sound, an oil producing zone in the Gulf of Mexico (Nemata: Selachinematidae). Bulletin Institute Royal Science Natural Belgique, Biologie 63:55-64. [ Links ]
CASTILLO-FERNÁNDEZ, D. & P. J. D. LAMBSHEAD.1990. Revision of the genus Elzalia Gerlach, 1957. (Nematoda: Xyalidae) including three new species from an oil producing zone in the Gulf of Mexico, with a Discussion of the sibling species problem. Bulletin British Museum Natural History 56(1):63-71. [ Links ]
CHITWOOD, B.C. 1960. A preliminary contribution on the marine nemas (Adenophorea) of Northern California. Transactions of the American Microscopy Society 79(4)347-383. [ Links ]
COULL, B.C. 1988. Ecology of the marine meiofauna. In: R. Higgings & H. Thiel. (Eds). Introduction to the study of Meiofauna. Smithsonian Institution Press, Washington, pp. 18-38. [ Links ]
DEAN, W. E. 1974. Determination of carbonate and organic matter in calcareous sediments and sedimentary rocks by loss on ignition: comparison with other methods. Journal of the Sedimentary Petrology 448(1):242-248. [ Links ]
FISHER, R. 2003. Spatial and temporal variations in nematode assemblages in tropical seagrass sediments. Hydrobiologia 493:43-63. [ Links ]
GASTON, K. J. 1991.The Magnitude of global insect species. Conservation Biology 5:283-296. [ Links ]
GERLACH, S. A. 1955. Zur Kenntnis der freilebenden marinen Nematoden von San Salvador. Aus dem Zoologischen Institut der Universität Kiel 158:249-303. [ Links ]
GRAY, J. S. 2000. The measurement of marine diversity, with application to the benthic fauna of the Norwegian continental shelf. Journal Experimental Biology and Ecology 250:23-49. [ Links ]
HEIP, C., M. VINCX, N. SMOL & G. VRANKEN, 1982. The Systematics and Ecology of Free-living Marine Nematodes. Helminthological Abstracts 51(1):1-31. [ Links ]
HERNÁNDEZ, E. R. & A. G. PAGÉS.1976. Catálogo de los peces marinos mexicanos. Secretaria de Industria y Comercio, Subsecretaría de Pesca. Instituto Nacional de la Pesca 520 p. [ Links ]
HOPE, W. D.1967A. Free-living marine nematodes of the genera Pseudocella Filipjev, 1927, Thoracostoma marion,1870, and Deontostoma Filipjev,1916 (Nematoda: Leptosomatidae) from the west coast of North America. Transactions of the American Microscopy Society 86 (3):307-334. [ Links ]
HOPE, W. D. 1967B. A review of the genus Pseudocella Filipjev, 1927 (Nematoda: Leptosomatidae) with a description of Pseudocella triaulolaimus n.sp. Proceedings of the Helminthological Society of Washington 34(1):6-12. [ Links ]
HULINGS, N. C. & J. S. GRAY. 1971. A manual for the study of Meiofauna. Smithsonian Institution Press 83 p. [ Links ]
JENSEN, P. 1984. Ecology of benthic and epiphytic nematodes in brackish waters. Hydrobiologia 108:201-217. [ Links ]
JENSEN, P. 1985. The Nematode fauna in the Sulphide-Rich Brine seep and Adjacent Bottoms of the East Flower Garden, Nw Gulf of México. I. Chromadoria. Zoologica Scripta 14(4):247-263. [ Links ]
JENSEN, P. 1988. Nematode assemblages in the deep-sea benthos of the Norwegian Sea. Deep Sea Research 35:1173-1184. [ Links ]
JONES, G. 1964. Redescription of Bolbella californica Allgen, 1951 (Encheliididae: Nematoda) with notes on its ecology off Southern California. Pacific Science 18(2): 160-165. [ Links ]
LAMBSHEAD, P. J. D. 1986. Sub-catastrophic sewage and industrial waste contamination as revealed by marine nematode faunal analysis. Marine Ecology Progress Series 29:247-260. [ Links ]
LAMBSHEAD, P. J. D. & G. BOUCHER. 2003. Marine nematodes deep-sea biodiversity - hyper-diverse or hype? Journal of Biogeography 30(4): 475-485. [ Links ]
LAMBSHEAD, P. J. D., C. J. BROWN, T. J. FERRERO, L. E. HAWKINS, C. R. SMITH & N. J. MITCHELL. 2003. Biodiversity of nematode assemblages from the region of the Clarion-Clipperton Fracture Zone, an area of commercial mining interest. BMC Ecology 2003; 3: 1. Published online 2003 January 9 doi: 10.1186/1472-6785-3-1. [ Links ]
LLINAS, J., D. LLUCH-COTA, D. CASTELLANOS & A. ORTEGA-RUBIO. 1993. La Isla Socorro, Revillagigedo, México. In: Salazar-Vallejo, S.I. y N.E. González. (Eds) Biodiversidad Marina y Costera de México CONABIO-CIQRO, pp 520-534. [ Links ]
LORENZEN, S. 1981. Entwurf eines phylogenetischen systems der freilebenden Nematoden. Veröff Instut Meeresforch Bremen Supplement 7:1-472. [ Links ]
MILLE-PAGAZA, S., A. PEREZ-CHI & M. E. SANCHEZ SALAZAR. 2003. Littoral decapods of Socorro Island, Revillagigedo archipelago, Mexico. Revista de Biologia Tropical 51 (1):175-182. [ Links ]
MOENS, T. & M. VINCX. 2000. Temperature and salinity and food thresholds in two brackish water bacteriovorus nematode species: assessing niches from food absorption and respiration experiments. Journal of Experimental Marine Biology and Ecology 243:137-154. [ Links ]
MURPHY, D.G. 1962. Three undescribed nematodes from the coast of Oregon. Limnology and Oceanography 7(3):365-382. [ Links ]
MUTHUMBI, A., A. VANREUSEL, G. DUINEUELD, K. SOETAERT & M. VINCX. 2004. Nematode community structure along continental slope off the Kenyan coast, Western Indian Ocean. International Review Hydrobiologia 89(2):188-205. [ Links ]
NDARO, J. G. M. & E. OLAFSSON. 1999. Soft bottom fauna with emphasis on nematode assemblages structure in a tropical intertidal lagoon in Zanzibar eastern Africa. I. Spatial variability. Hydrobiologia 405:133-148. [ Links ]
PLATT, H. M. 1984. Classify Worms and Spot Pollution: Marine nematodes may reveal the 'health' of the oceans-but only if we are able to distinguish one species from another. New Scientist 28-29. [ Links ]
PLATT, H. M. & R. WARWICK. 1983. A Synopsis of the Freeliving Marine Nematodes Part I. British Enoplids. Cambridge University Press, London 307 p. [ Links ]
RENAUD-MORNANT, J. & N. GOURBAULT. 1990. Evaluation of abyssal meiobenthos in the eastern Central Pacific (Clarion-Clipperton fracture zone) Progress. Oceanography 24: 317-329. [ Links ]
ROMEYN K. & L. A. BOWMAN. 1983. Food selection and consumption by estuarine nematodes. Hydrobiological Bulletin 17(2):103-109. [ Links ]
TCHESUNOV, A. V. & J. R. OKHLOPKOV. 2006. On some Selachinematidad nematodes (Chromadorida: Selachinematidae) deposited in the collection of the Smithsonian National Museum of Natural History. Nematology 8(1):21-44. [ Links ]
TIETJEN, J. H. 1969. The ecology of shallow water meiofauna in two New England estuarios. Oecologia 2:251-291. [ Links ]
VILLALOBOS, F. A. 1960. Notas acerca del aspecto hidrobiológico de la parte sur de la Isla. In: Adem, J. E., L.Cobo, F. Blásquez, A. Miranda, T Villalobos Herrera y L. Vásquez. Isla Socorro, Archipiélago de las Revillagigedo. Monografias del Instituto de Geofísica. UNAM, pp. 153-180. [ Links ]
YOSHIMURA, K. 1980. Free-living marine nematodes from Kii Peninsula. Publications of Seto Marine Biology Laboratory 25:39-49. [ Links ]
WARWICK, R. M. & K. R. CLARKE. 1998. Taxonomic distinctness and environmental assessment. Journal of Applied Ecology 35:532-543. [ Links ]
WIESER, W. 1953. Die beziehung zwischen mundnhlengstalt emahrungweise und workommen bei freilebenden marinen nematoden. Ark Zoologie 4:439-484. [ Links ]














