Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Hidrobiológica
versión impresa ISSN 0188-8897
Hidrobiológica vol.12 no.2 Ciudad de México dic. 2002
Article
Biochemical characterization of crude extract from Pleuroncodes planipes (Crustacea: Galatheidae) as potential feed additive, considerations for a new fishery along the Mexico Pacific coast
Caracterización bioquímica del extracto crudo de Pleuroncodes planipes (Crustacea: Galatheidae), como un aditivo alimenticio potencial, consideraciones para una nueva pesquería en la Costa del Pacífico de México
Fernando Vega-Villasante1, Héctor Nolasco1, Adyary Fallarero2 and Olimpia Carrillo-Farnes2,3
1 Centro de Investigaciones Biológicas del Noroeste, S.C. (CIBNOR). La Paz, B.C.S. México.
2 Facultad de Biología, Universidad de La Habana. La Habana, Cuba.
3 Centro de Investigaciones Marinas. Universidad de La Habana, Cuba.
Recibido: 8 de abril de 2002
Aceptado: 31 de julio de 2002
Abstract
The red crab Pleuroncodes planipes, commonly called "langostilla," is arguably the most abundant benthonic decapod of west coast Mexico, with natural productivity estimated at 735,000 metric tons/year. At the present time, it has not been exploited commercially. The implementation of a new fishery in Mexico, based on the capture of red crab, offers diversification of the fishing industry. In this work a raw extract of langostilla, obtained by mechanical pressing of the whole organism, was analyzed as a potential feed ingredient or additive for cultured marine species. The lyophilized red crab extract possessed: 1) enzymatic activity of proteasa, trypsin, amylase, and lipase; 2) no inhibition of serine-proteases; 3) antioxidant capacity against lipoperoxidation and superoxide ions produced by xanthine oxidase reaction; and 4) activity of insulin-like peptides. The amino acid profile shows high levels of arginine, lysine, and methionine. The chemical score of extract protein was compared with M. japonicus larvae, whole body fish tissue, and short-necked clam tissue. The digestibility of the product was 95% taking purified casein as reference. These biochemical and nutritional characteristics are discussed. These results offer a new use of red crab and the potential of a new fishery in Mexico.
Key words: Pleuroncodes planipes, red crab, nutrition, crustacean, Mexican fishery.
Resumen
La langostilla roja Pleuroncodes planipes es posiblemente el decápodo bentónico más abundante de la costa oeste de México, con una productividad natural estimada en 735,000 toneladas métricas al año, sin embargo este recurso aún no ha sido explotado comercialmente. En este trabajo el extracto crudo de la langostilla, obtenido por prensado mecánico del organismo completo, es analizado como un ingrediente potencial o aditivo alimentario para la nutrición de especies marinas en cultivo. El extracto liofilizado de la langostilla demostró poseer i) actividad enzimática digestiva de proteasas, amilasas y lipasas, ii) ausencia de inhibición de proteasas de tipo serínico, iii) actividad antioxidante hacia la lipoperoxidación de tejido cerebral de rata y de los iones superóxido producidos por la reacción de la xantina-oxidasa, iv) actividad de péptidos tipo insulina. El perfil de aminoácidos presentó niveles altos de arginina, lisina y metionina. El cómputo químico de la proteína del extracto fue comparada con proteína de larva M. japonicus, tejido completo de pescado y de almeja. La digestibilidad del producto fue de 95.7% tomando la caseína purificada como referencia. Estas características bioquímicas y nutricionales y su relevancia se discuten en el trabajo. Estos resultados abren la perspectiva de la captura de langostilla como una nueva actividad pesquera en México.
Palabras clave: Pleuroncodes planipes, langostilla, nutrición, crustáceos, pesquería mexicana.
Introduction
Researchers at the Centro de Investigaciones Biológicas del Noroeste (CIBNOR) in Mexico have been studying the biology, ecology, and use of the langostilla red crab (Pleuroncodes planipes) for 13 years. Red crab may be the most abundant benthonic decapod of Mexico. Ehrarhdt and Ramírez (1982) estimated productivity at 205,000 metric tons, but more recently, Aurioles-Gamboa et al., (1995) reported productivity of 735,000 metric tons/year for the coasts of the Baja California peninsula. Presently, it has not been commercially exploited in Mexico. They proposed that 40,000 tons o bento-pelagic langostilla/year could be captured during the initial phase of the fishery without negatively impacting the resource and still generate a sustainable fishery that provides a sufficient biomass for industrial use.
In fishing terms, the west coast of southern Baja California has been exploited for abalone, lobster, clam, sardine, and tuna. There is considerable interest in implementing a new fishery in Mexico. The capture of langostilla offers diversification of fishing activities, with the possibility of generating additional employment and attracting foreign currency and investment to this region of Mexico (Civera et al. 1998).
The uses of langostilla will depend mainly on their size. Langostilla presents three main phases: pelagic, bentho-pelagic, and benthonic, and all phases can be found throughout the year (Civera et al., 1998). Aurioles-Gamboa et al., (1995) suggested that the fishery should focus on the capture of benthonic populations that are over 32 mm (cephalothoracic length) for the production of fresh-frozen tail for human consumption, as occurs in Chile. The by-products (cephalothorax and exoskeleton) could be used to obtain meals, enzymes, pigments, and chitin that could be used to obtain meals, enzymes, pigments, and chitin that could be used as additives to balance feeds (Civera et al., 1998). Langostilla (22-32 mm cephalothoracic length) that populate the continental platform can be exploited. To avoid depletion of the resource, the recommended period for capture is April-September (Aurioles-Gamboa et al., 1995). The smaller langostilla could provide diverse food products, pharmaceuticals, biotechnological ingredients, and aquaculture feed (Spinelli et al., 1974; Civera et al., 1998).
Major capital investment would be required to prevent rapid decomposition of whole langostilla at ambient temperature. Therefore, we considered as alternative strategy to study the characteristics of the body fluid obtained by mechanic pressing immediately after capture. The single-step process produces two fractions, body juice and pressed mass. The pressed mass can be used to obtain flour, pigments, and chitin (under study) and the body juice as a food additive. The goal of the present study was to characterize the body juice extract of langostilla is an attractive prey for many marine environment, langostilla is an attractive prey for many marine species (Aurioles-Gamboa et al., 1995), since langostilla contains considerable protein content (Civera et al., 1998).It also has a relative heavy exoskeleton, which decreases protein content of whole langostilla. It has a relatively big cephalothorax that includes ocular peduncules (site of hormone synthesis) and large digestive gland (hepatopancreas) that is rich in enzymes, lipids, and other nutrients. These characteristics supported our hypothesis that langostilla extract (exoskeleton free) has high protein content, digestive enzymes, hormones, oils, pigments, and other nutrient component, and has value as a food additive in aquaculture.
The longer-range goal of this study is to develop technologies for the integration and expansion of uses of langostilla, while promoting a sustainable commercial fishery of this abundant marine resource. Additionally, diversifying fisheries along the Pacific coast would reduce pressure on current commercial species.
Material and methods
Langostilla were captured in March 1999 and April 2000 with a shrimp trawler boat (450 hp), BIP II, with a shrimp trawler net at a depth of 50 m in Magdalena Bay, on the Pacific coast of Baja California Sur. Langostilla were maintained alive condition at 8°C until the trawler reached San Carlos harbor.
The langostilla extract was obtained by mechanical pressing (manual screw press) of whole langostilla. Langostilla total extract (LTE), yielding an average of 420 ml of extract per kg of whole langostilla, was frozen and stored at −40°C until further processing. For analysis purposes, LTE was thawed, then centrifuged (centrifuge Eppendorf 5804R, Germany) at 5,000 g for 5 min to separate big tissue particles and other undesirable particles, such as sand (product of the capture). LTE was then lyophilized (Virtis Benchtop, Gardiner N.Y. USA) (conditions: 20 h, −48°C, 46 MT), and the product (LLTE) stored at −10°C until used.
The proximal analysis (dry matter, crude protein [N x 6.25] crude fat, ash, crude fiber, nitrogen free extract) of the LLTE was performed according to AOAC (1990) standards. Gross energy was measured with an automatic calorimeter (Parr Isoperivolt 1261, U.S.A.).
The activity of nonspecific proteases in the LLTE was determined by using 2.5% azocasein as substrate and LLTE (500 mg/ml), as described in Vega-Villasante et al., (1995).
Amylase activity of the LLTE was measured by using soluble starch (1%) as substrate as described by Vega-Villasante et al., (1993). Lipase activity was determined by using b-naphthyl caprylate as substrate, according to the method of Versaw et al., (1989). Determination of serine-type inhibitors in LLTE, which are stable in trichloroacetic acid (TCA), was performed as follows: 1 g LLTE was suspended in 10 ml of 5% TCA, incubated 30 min at 25°C in water bath (Benchtop water bath, VWR 1220, USA) and then centrifuged at 10,000 g for 20 min at 5°C. The pH of the supernatant was adjusted to 8.0 with 10N NaOH and centrifuged at 10,000 g for 10 min at 5°C. The resulting preparation is called TCA extract. One volume of bovine trypsin (1 mg/ml, in Tris-HCl, 50 mM, pH 8.0) was mixed with one volume of TCA extract (1:1, v/v) and incubated for different times (10, 20, 30, and 40 min) at 25°C. The remaining proteinase activity was measured according to the method described by Vega-Villasante et al., (1995).
In vitro digestibility of LLTE was measured according to the method described by Hsu et al., (1977) using an aqueous solution of LLTE with a protein concentration of 20% (p/v). Caseis (SIGMA,C5890) at the same concentration was used as control. The digestibility was determined as the decrease of pH units over 10 minutes. The profile of amino acids of LLTE was determined in an automatic amino acids analyzer (Alpha Plus II, LKB, Sweden). The samples of LLTE were hydrolyzed with HCl 6N and analyzed by following the methodology of LKB Biochemicals and the standardized operation proceedings of PNOAC (01.001.94). The chemical score of the protein was determined by the method described by Gallardo et al., (1989). The antioxidant capacity of LLTE was determined by measuring its inhibitory activity over the spontaneous and induced lipoperoxidation of rat brain homogenates according to the technique of Ohkawa et al. (1979). Also, by measuring the capacity of different concentrations of LLTE (1, 5, 10, and 50 mg of LLTE/ml) to inhibit the reduction of nitro tetrazolium blue (NBT) by superoxide radicals released by the xanthine oxidase reaction according to the method described by Fridovich and Mc Cord (1969). Insulin-like activity present in LLTE was determined by radio immune assay (RIA) according to the method described by Gallardo (1998).
The effect of different concentrations of LLTE (1,10, 50, and 100 µg of LLTE/ml) on growth promotion in cellular culture of mouse (Balb-c) fibroblasts and human carcinoma cells was measured according to the methodology described by Florestin et al. (1983)
Results
Chemical analysis and gross energy of langostilla extract
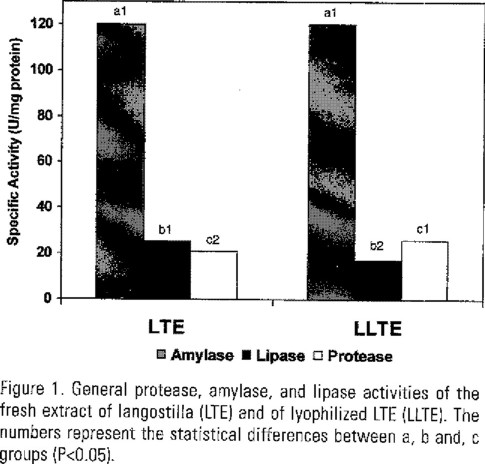
Lyophilized langostilla total extract (LLTE) has a high concentration of protein and low levels of crude fiber. The chemical analysis and gross energy of LLTE is shown in Table 1. In Figure 1, protease, amylase, and lipase activities of LTE and of LLTE are shown. Amylase was similar in both treatments, whereas lipase diminishes in LLTE. Protease activity of LLTE increased.

In Figure 2, activity of bovine trypsin and bovine trypsin incubated with TCA-extract of LLTE are shown. No inhibitory effect of TCA-extract on trypsin activity was found. In vitro di are shown. Concentration of insulin-like activity in LLTE as measured by RIA analysis, was 852 pg/ml.
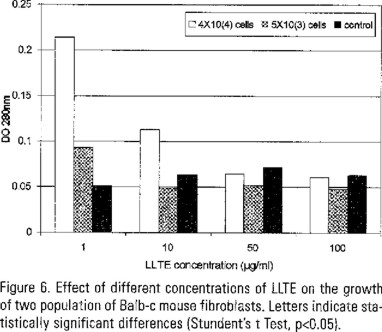
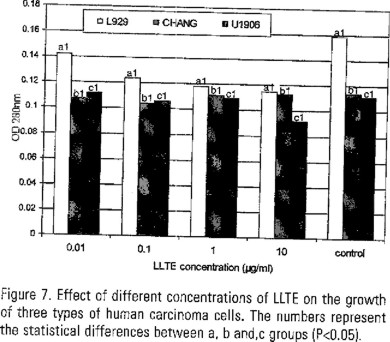
The effect of different concentration of LLTE on the growth of two populations (1x104 and 5x103) of mouse fibroblasts is shown in Figure 6. A concentration of 1 µg LLTE/ml produced the highest cell proliferation. The effect of different concentrations of LLTE on the growth of three types of human carcinoma cells is shown in Figure 7. None concentration produced cel proliferation.
Discussion
Until now studies of the use of langostilla have focused on the use of langostilla flour as a partial or total substitute for fish, shrimp head and soy meal in feeds for shrimp Farfantepenaeus californiensis and Litopenaeus vannamei. The effect on survival and growth and in vivo digestibility of nutrients has been measured (Civera et al. 1998). Langostilla is a viable ingredient for partial substitution of the three meals mentioned above. At certain levels, the replacement may increase growth, increase proteolytic activity of the hepatopancreas and improve the digestibility of feed protein and lipids (Civera et al., 1998).
In some studies, flours, extracts, or factions of marine organisms have been used as ingredients or feed additives. For example, squid flour seems to have a growth-promoting effect on shrimp (Cruz-Suárez and Guillaume 1987); hepatopancreas extracts of shrimp Litopenaeus schmitti (Hepatopancreatine) at low concentrations (1%) in shrimp diets promote better weight gain in shrimp postlarvae (Forrellat-Barrios 1998), and adding peptides with "insulin-like" activity from the hepatopancreas of the lobster Panulirus argus stimulated the growth of L. schmitti larvae when given in microcapsulated form in a balanced conventional diet (Galindo et al., 1995). However, adding natural feed antioxidant compounds in shrimp culture is still uncommon.
The main goal of using feed additives in aquaculture is to increase the growth rate of cultured species. Growth-stimulating substances are incorporated in very low quantities with no special effect on composition of the diet (Forrellat-Barrios 1998).
The broadest meaning of the term feed-additive would include all natural or synthetic, nutritive or non-nutritive, and physiologically active or inert chemicals added directly to feeds during processing to perform a specific function. Examples include antioxidants, vitamins, minerals, flavoring agents, coloring agents, emulsifiers, stabilizers, bleaching agents, coloring agents, acidulants, nutritive and non-nutritive sweeteners, and leavening agents (Carrillo et al., 2000).
In our results, LLTE had several biochemical characteristics that support its potential use a dietary ingredient or as a food additive.
Specifications for marine products used in aquaculture must include proximate analyses, despite its limited value in predicting nutritional value of the products. Proximate chemical composition of LLTE indicate that protein is the major nutrient of LLTE. Almost all growth factors identified in products used in aquaculture are proteins or peptides.
Digestive enzymes have been of great interest in food additive directed to aquaculture. Many studies have been done on purified or unpurified enzymes in the diet of aquatic organisms (Dabrovsky 1977; Maugle et al., 1983; Lauf and Hoffer 1984; Munilla-Moran et al., 1990; Akiyama et al., 1991; Cardenette et al. 1993; Buchanan et al., 1997; Forrellat-Barrios 1998). According to Carrillo-Farnés (1994), digestive enzymes have been used as feed additives under the following hypotheses: 1) they increase the enzymatic activity in the digestive tract, 2) increase the digestibility of food, 3) activate zymogens of endogenous proteasas, 4) help gut epithelium defoliation, and 5) diminish inflammatory processes. LLTE demonstrates interesting concentrations of three types of digestive enzyme proteases, amylases, and lipases, and suggests that LLTE could be a good source of exogenous digestive enzymes for helping digestion processes. In recent studies by Forrellat-Barrios (1998), incorporation of a lyophilized extract of the midgut-gland of Litopenaeus schmitti (hepatopancreatine) in artificial feeds has a beneficial effect on growth rate when added to the diet of L. schmitti larvae.
Delivering of essential amino acids for most cultured marine species is done by adding protein that guarantees the minimum requirements of the animal. However, few of these ingredients alone contribute all the required essential amino acids. Nutrition science considers an ideal protein source to reflect the profile of amino acids of the muscle of the species to be fed (Guillaume 1997). Table 1 compares the amino acid profile of the hole body protein of langostilla (A and B) and the amino acid profile of LLTE (C). The chemical score of LTE protein was developed to establish the limiting amino acids by taking the amino acid profile of M. japonicus larvae, whole-body fish tissue, and short-necked clam tissue as reference. The limiting amino acid in all cases was valine with 25%, 13%, and 15%. LLTE is a good source of arginine (score of 98%), which is the first limiting amino acid of many protein sources used in crustacean nutrition (Guillaume 1997).
Free radicals are defined as chemical species that posess one or more unpaired electrons. The term "Reactive Oxygen Species" (ROS) collectively describes free radicals such as O2°, OH°, and non-radical oxygen compounds such as H2O2 and (HOCl). These reactive oxygen intermediates may participate in reactions that give rise to free radicals species. Unstable free radical species attack cellular components, causing damage to lipids, proteins, and DNA, which can initiate a chain of events causing disease. However, organisms possess a number of mechanisms both to control the production of ROS and to limit or repair the tissue damage (Burr 1994). Di Mascio et al., (1989, 1992) have examined the quenching effects of carotenoids against O2°, and reported the effectiveness of astaxanthin. Miki (1991) and Miki et al., (1994) revealed that astaxanthin, a common carotenoid found in marine animals, shows strong quenching activity against O2°. This quenching activity is approximately 100 times stronger than a-tocopherol, a common antioxidant in plants and animals. Astaxanthin is commonly in crustaceans in free and esterified form or as carotene-proteins.
Cremades et al., (2001) studying the process for obtaining carotenoproteins from crawfish (Procambarus clarkii) discussed the high content of carotenoproteins (mainly astaxanthin) in this species (162.27 µg/g) (together with its content in essential amino acids and in ?-fatty acids) make it an important protein source for the nutrition of patients needing protein of high-quality rather than large amounts and/or an antioxidant source, as in the case of cancer, AIDS, hepatic, and renal patients and the elderly.
The langostilla is a resource rich in carotenoids (10.0-16.0 mg/100 g LLTE), mainly in astaxanthin (about 90% of the carotenoid content) (Castro-González et al.,1995). LLTE is suggested as a potential source of antioxidants.
LLTE showed high antioxidant activity by inhibiting lipid peroxidation of rat brain tissue and also by inhibiting the superoxide radicals produced by the xanthine-oxidase reaction. These results suggest that LLTE, when used as an ingredient or additive in artificial feeds, could prevent diseases of aquatic organisms, which may be exposed to stressful environmental conditions such as crowding, nutritional deficits, toxic products, and heavy metal exposure.
A decade ago, researchers thought that only vertebrates had insulin and regulator peptides as a responses to specific metabolic and physiological requirements. Identification of hormones and bioregulator peptides in invertebrates, homologous to vertebrate insulin, gastrin-cholecystokinin, and vasopresin-oxytocine has increased in recent years (Le Roith et al., 1980; Sanders 1983a; Favrel et al., 1991; Geraerts et al., 1991; Sevala et al., 1993; Chuang and Wang 1994; Cancre et al., 1995; Chen et al., 1996). These studies have increased knowledge of signal transmission mechanisms in the inner cell and the regulation of intermediate metabolism of invertebrate species, particularly those of commercial or scientific interest. This subject is still not completely understood, but could be applied to growth acceleration shortening of maturation periods, and disease resistance (Ratafia 1995).
In crustaceans, Sanders (1983a, b, c) has done the most representative studies identifying peptides that are immunologically similar to human insulin in the midgut gland, hemolymph, and proventriculus of the lobster Homarus americanus. These peptides have similar functions to that of mammals to stimulate glucogenesis in muscle, regulate intermediate metabolism of proteins and lipids, maintain osmoregulation, promote growth, and initiate cell differentiation, but having no participation on maintenance of glucose levels in hemolymph (King and Kahn 1981; Sanders 1983c).
In the search for growth-stimulating substances, Gallardo (1998) found a peptide similar to vertebrate insulin in the Cuban lobster Panulirus argus. This peptide has been tested as a growth stimulator when included in microcapsulated diets for juvenile white shrimp Litopenaeus schmitti. A significant growth-promoting effect was found. This peptide has the capacity to stimulate DNA synthesis in the midgut gland. LLTE showed, by RIA, a high insulin-like activity. This suggests the existence of peptides similar to insulin, with possible potential as a growth promoter. Related to this, the results from cell culture demonstrated that LLTE at very low concentrations could activate the growth of mouse fibroblasts. However, this promotion on cell proliferation could not be found in carcinoma cell cultures. This effect and the participation of insulin-like peptides is now under study.
Our results as well as those of other researchers mentioned above suggest viable and sustainable uses of the langostilla resource, which will promote the development of a broader-based and sustainable Mexican fishery and enhance other economic sectors, including transformation of marine-source products, improvement of feed products, and advancement of aquaculture industries.
Our research group is interested in langostilla as a future source of nutrients for the shrimp culture industry. This industry is of great importance in Mexico because the rapid growth of this activity places a severe demand on the conventional sources of high quality feed ingredients. Additionally, shrimp cultivation is now in competition with domestic animal production. The search for alternative ingredients that can be used solely for aquaculture purposes is one of the research goals of several groups. Nevertheless, our findings as well as studies by other groups suggest that langostilla could serve, not only as a nutrient source for aquatic species, but for human nutrition as well. Langostilla by-products as a safe source of human nutrients is a goal of our studies in the near future. In the present work, we focused mainly on aspects of raw extract of langostilla as a potential feed ingredient or additive directed to the marine crustacean aquaculture industry. There is substantial reason for considering these same results as a preliminary study for its use in human nutrition.
Acknowledgements
This study was supported by Cuba-Mexico collaboration projects funded by CONACyT and CIBNOR of Mexico. The authors acknowledge the important help of Martha Deas and Celeste Arranz of the National Endocrinology and Metabolic Diseases Institute, Gabriel Coto, Head of the Dept. of Research and Development at the Placental Histotherapy Center, Miriam Ríos of the Oncology and Radiobiology Institute (all of them located in Havana, Cuba) and Claudina Zaldivar, Georgina Orue, and Osmara Díaz for technical advice. Specially thanks to Oli Chong for moral support. Ira Fogel at CIBNOR edited the English language text.
References
Akiyama, D., W. Dominy y L. Lawrence, 1991. Penaeid shrimp nutrition for the commercial feed industry. In: D. Akiyama and R. Tan (Eds.), Proceedings of the Aquaculture Feed Processing and Nutrition Workshop. American Soy Bean Association. pp. 80-98. [ Links ]
AOAC, 1990. Official methods of analysis. 15th edition. Association of Official Analytical Chemistry. Washington D.C. 1094 p. [ Links ]
Aurioles-Gamboa, D., 1995. Distribución y abundancia de la langostilla bentónica (Pleuroncodes planipes) en la plataforma continental de la costa occidental de Baja California. In: D. Aurioles-Gamboa and E. Balart (Eds.), La Langostilla: Biología, Ecología y Aprovechamiento. Centro de Investigaciones Biológicas del Noroeste. México. pp. 59-92. [ Links ]
Buchanan, J., H. Z. Sarac, D. Poppi y R. T. Conway, 1997. Effect of enzyme addition to canola meal in prawn diets. Aquaculture 151: 29-35. [ Links ]
Burr, M. L., 1994. Antioxidants and cancer. Journal of Human Nutrition and Dietetics 7: 409-416. [ Links ]
Cancre, I., A. Van Wormhoudt e Y. Le Gal, 1995. Effects of cellular growth factors on crustacean hepatopancreas cell suspensions. Journal of Marine Biotechnology 2: 83-87. [ Links ]
Cardenette, G., A. Morales, F. Moyanu, A. Sanz y M. De la Higuera, 1993. Adición de enzimas exógenas como medio de mejora de la utilización digestiva de las materias primas en dietas para truchas arcoiris (Onchorhyncus mykiss) WAS 93, European Aquaculture Society Special Publication No.19: 211 p. [ Links ]
Carrillo-Farnés, O., 1994. Producto multienzimático del hepatopáncreas de camarón: reactivo y suplemento dietético. Proceedings of the Second International Symposium of Aquatic Nutrition. Monterrey, N. L. México. pp. 21-26. [ Links ]
Carrillo, O., Vega-Villasante, F., Nolasco y H. N. Gallardo 2000. Aditivos alimentarios como estimuladores del crecimiento de camarón. In: L. E. Cruz-Suárez, D. Ricque-Marie, M. Tapia-Salazar, M. A. Olvera-Novoa y R. Civera-Cerecedo (Eds.). Avances en Nutrición Acuícola V. Memorias del V Simposium Internacional de Nutrición Acuícola. 19-22 Noviembre, 2000. Mérida, Yucatán., México. [ Links ]
Castro-González, M. I., S. Carrillo-Domínguez, F. Pérez-Gil Romo y C. Calvo-Carrillo, 1995. Composición Química de la langostilla y procesos tecnológicos. In: D. Aurioles-Gamboa and E. Balart (Eds.): La Langostilla: Biología, Ecología y Aprovechamiento. Centro de Investigaciones Biológicas del Noroeste, México. pp. 163:177. [ Links ]
Chen, Ch., J. Jack y R. S. Garofalo, 1996. The Drosophila insulin receptor is required for normal growth. Endocrinology 137: 846-856. [ Links ]
Chuang, N. N. y P. C.Wang, 1994. Characterization of insulin receptor from the muscle of the shrimp Penaeus japonicus (Crustacea: Decapoda). Comparative Biochemistry and Physiology 108(B): 289-297. [ Links ]
Civera, R., E. Goytortúa, H. Nolasco, F. Vega-Villasante, E. Balart, E. Amador, G. Ponce, G. Colado, J. Lucero, C. Rodríguez, J. Solano, A. Flores-Tom, J. G. Monroy y Coral, 1998. Uso de la Langostilla roja Pleuroncodes planipes en la nutrición de organismos acuáticos. IV International Symposium of Aquatic Nutrition. La Paz, B.C.S. México. Vol. 2: 1-16. [ Links ]
Cremades, O., E. Ponce, R. Corpas, J. F. Gutiérrez, M. Jover, M. C. Alvarez-Ossorio, J. Parrado y J. Bautista, 2001. Processing of Crawfish (Procambarus clarkii) for the Preparation of Carotenoproteins and Chitin. Journal of Agricultural and Food Chemistry 49: 5468-5472. [ Links ]
Cruz-Suárez, L. E. y J. Guillaume, 1987. Squid protein effect on growth of four penaeid shrimp. Journal of the World Aquaculture Society 18:209. [ Links ]
Dabrovsky, K., 1977. A study of the application of proteolytic enzymes in fish food. Aquaculture 12: 349-360. [ Links ]
Deshimaru, O., 1985. Nutritional quality of compounded diets for prawn Penaeus monodon. Bulletin of the Japanese Society of Science and Fisheries 51: 1037-1044. [ Links ]
Di Mascio, P., S. Kaiser y H. Sies, 1989. Lycopene as the most efficient biological carotenoid singlet oxygen quencher. Archives of Biochemistry and Biophysics 274: 532-538. [ Links ]
Di Mascio, P., A. R. Sundquist, T. P. A. Devasagayam y H. Sies, 1992. Assay of lycopene and other carotenoids as singlet oxygen quenchers. Methods in Enzymology 213: 429-438. [ Links ]
Ehrarhdt, N. E. y P. Ramírez, 1982. Evaluación de los recursos demersales accesibles y redes de arrastre de fondo en la Península de Baja California, México durante 1979 y 1980. INP. Serie Científica 23: 10-46. [ Links ]
Forrellat-Barrios, A., 1998. El hepatopáncreas del camarón: fuente de enzimas digestivas para el cultivo del camarón. Tesis Doctoral. Facultad de Biología. Universidad de La Habana, Cuba. 111 p. [ Links ]
Favrel, P., G. Kegel, D. Sedlmeier, R. Keller y A. Van Wormhoudt, 1991. Structure and biological activity of crustacean gastro intestinal peptides identified with antibodies to gastrin/ cholecystokinin. Biochemie 73: 1233-1239. [ Links ]
Florestin, I., V. Chung y M. Danigny, 1983. Immunomodulatory effects of bestatin in immunocompetent mice. Progress in Cancer Chemoimmunotherapy pp. 212-225. [ Links ]
Fridovich I. y J. M. Mc Cord, 1969. Superoxide dismutase. An enzymic function for erythrocuprein (hemocuprein). Journal of Biological Chemistry 244: 6049-6055. [ Links ]
Galindo, J. G., N. Gallardo, A. Medina y C. Villagrama, 1995. An immunoractive insulin from lobsters a growth factor for the shrimp Penaeus vannamei. Rivista Italiana Acquacoltura 30: 159-162. [ Links ]
Gallardo, N. Y., 1975. Aprovechamiento integral de la langostilla "Pleuroncodes planipes". Tesis de Maestría, Escuela Nacional de Ciencias Biológicas, I. P. N., México. [ Links ]
Gallardo, N., R. González, O. Carrillo, O. Valdés y A. Forrellat, 1989. Una aproximación a los requerimientos de aminoácidos esenciales de Penaeus schmitti. Revista de Investigaciones Marinas 10: 259-267. [ Links ]
Gallardo, N., 1998. Peptides similar to mammal insulin in the lobster Panulirus argus Latreille (Crustacea: Decapoda): Analysis of the biological activity and characterization of possible receptors. Tesis de Doctorado. Facultad de Biología. Universidad de La Habana, Cuba. 142 p. [ Links ]
Geraerts, J., A. B. Smit, K. W. Li, R. H. Vreugdengill y H. Van Heerik-Luizen 1991. Neuropeptide gene families that control reproductive behaviour and growth in molluscs. In: N. N. Osborne (Ed). Current Aspects of the Neurosciences. pp. 255-304. [ Links ]
Guillaume, J., 1997. Protein and Amino acids. In: L. R. D'Abramo (Ed). Crustacean Nutrition. World Aquaculture Society. Baton Rouge, Louisiana. pp. 26-49. [ Links ]
Hsu, H. W., D. L. Vavak, I. D. Satterlee y G. A. Miller, 1977. A multienzyme technique for estimating protein digestibility. Journal of Food Science 42: 1269-1273. [ Links ]
King, G. L. y R. C. Kahn, 1981. Non-parallel evolution of metabolic and growth promoting functions of insulin. Nature 292: 644-646. [ Links ]
Lauf, M. y R. Hoffer, 1984. Proteolytic enzymes in fish development and the importance of dietary enzymes. Aquaculture 37: 335-345. [ Links ]
Le Roith, D., J. Shiloach, J. Roth y M. A. Lesnik, 1980. Evolutionary origin of vertebrate hormones: substances very similar to mammalian insulins are native to unicellular eucaryotics. Proceedings of the Natural Academy of Science USA 77: 6184-6188. [ Links ]
Maugle, P. D., D. Deshimaru, T. Katayama y K. L. Simpson, 1983. Effect of microencapsulated amylase and bovine trypsin as dietary supplement on groth and metabolism of shrimp. Bulletin of the Japanese Society of Science and Fisheries 49: 1421-1427. [ Links ]
Miki, W., 1991. Biological functions and activities of animal carotenoids. Pure Applied Chemistry 63: 141-146. [ Links ]
Miki, W., N. Otaki, N. Shimidzu y A. Yokoyama, 1994. Carotenoids as free radical scavengers in marine animals. Journal of Marine Biotechnology 2: 35-27. [ Links ]
Munilla-Moran, R., J. R. Satrk y A. Barbour, 1990. The role of exogenous enzyme in digestion in culture turbot larvae (Scophtalmus maximus L.). Aquaculture 88: 337-350. [ Links ]
Ohkawa, H., N. Oishi y K. Yagi, 1979. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Analytical Biochemistry 95: 351-358. [ Links ]
Ratafia, M., 1995. Aquaculture today, a world wide status report. World Aquaculture 26: 18-24. [ Links ]
Sanders, B., 1983a. Insulin-like peptide in the lobster Homarus americanus. I. Insulin immunoreactivity. General and Comparative Endocrinology 50: 366-373. [ Links ]
Sanders, B., 1983b. Insulin-like peptide in the lobster Homarus americanus. II. Insulin-like biological activity. General and Comparative Endocrinology 50: 374-377. [ Links ]
Sanders, B., 1983c. Insulin-like peptide in the lobster Homarus americanus. III. No glucostatic role. General and Comparative Endocrinology 50: 378-382. [ Links ]
Sevala, V. M., V. L. Sevala y A. S. M. Saleuddin, 1993. Hemolymph insulin-like peptides (ILP) titers and the influence of ILP and mammalian insulin in the amino acid incorporation in the mantle collar in vitro in Helisoma (Mollusca). Biological Bulletin 185: 140-148. [ Links ]
Spinelli, J., L. Lehman y D. Wieg, 1974. Composition, processing and utilization of Red Crab Pleuroncodes planipes as an agricultural feed ingredient. Journal of the Fisheries Research Board of Canada 31: 1025-1029. [ Links ]
Teshima, S., A. Kanazawa y A. Yamashita, 1986. Dietary value of several proteins and supplemental amino acids for larvae of the prawn Penaeus japonicus. Aquaculture 51: 225-235. [ Links ]
Vega-Villasante, F., H. Nolasco y R. Civera, 1993. The digestive enzymes of the Pacific Brown Shrimp Penaeus californiensis. I. Properties of amylase activity in the digestive tract. Comparative Biochemistry and Physiology 106(B): 547-550. [ Links ]
Vega-Villasante, F., H. Nolasco y R. Civera, 1995. The digestive enzymes of the Pacific Brown Shrimp Penaeus californiensis. I. Properties of protease activity in the whole digestive tract. Comparative Biochemistry and Physiology 112(B): 123-129. [ Links ]
Versaw, W. K., S. L. Cuppett, D. D. Winters y L. E. Williams, 1989. An improved colorimetric assay for bacterial lipases in nonfat dry milk. Journal of Food Science 54: 1557-1558. [ Links ]
Wilson, R. P. y C. B. Cowey, 1985. Amino acid composition of hole body tissue of rainbow trout and Atlantic salmon. Aquaculture 48: 373-376. [ Links ]














