Introduction
Many industries, such as metallurgy, foundry, mining, paper, canning, energy and fuel production, electroplating, machinery, plastic, agriculture, photography, aviation, textiles, ceramics, pesticides and fertilizers discharge water containing heavy metals (Han et al., 2006; Farooq et al., 2010). These metals accumulate in the environment over time, with consequent bioaccumulation in the food chain, affecting the environment and human health (Navarro-Aviñó et al., 2007; Singh et al., 2009). In particular, the textile, leather tanning, aviation and steel industries discharge large amounts of metals, mainly chromium (Nikazar et al., 2008).
The U. S. Environmental Protection Agency published a list of PBT (persistent, bioaccumulative and toxic) compounds that must be dealt with in order to reduce pollution, noting that the most toxic heavy metals are cadmium, nickel, copper and zinc. Likewise, the World Health Organization states that chromium, cobalt, lead and mercury are also of concern because of their effects on health (Filipovic-Kovacevic et al., 2000; Correia de Soto and Martín de Armando, 2004; Ziagova et al., 2007). The United Nations Organization for Food and Agriculture in the Codex Alimentarius Commision mentions that the most toxic pollutants that food may contain are heavy metals (FAO, 2011).
Heavy metal elements are defined as having an atomic weight above 44.956 g mol-1 and density greater than 5 g cm-3 in their elemental form and are toxic (Morton, 2006; INE, 2011). Heavy metals exhibit a toxicity range at low concentrations from 0.001 to 10 mg L-1 and they cannot be biodegraded by any method; they can only be bio-transformed to less toxic forms (Cañizares-Villanueva, 2000; Wang and Chen, 2006; Cuizano and Navarro, 2009). Chromium, for example, is considered an environmental pollutant due to its widespread use in various industrial activities (Asamudo et al., 2005). In its Cr3+ form it is an indispensable micronutrient for sugar metabolism, it prevents blood clots, carries out important enzymatic functions in the human body and reduces cholesterol and triglyceride levels; the Cr6+ form, on the other hand, is both highly carcinogenic and mutagenic.
There are several conventional methods for the removal of heavy metals in aqueous media such as chemical precipitation, ion exchange, electrochemical treatment, membrane technology, activated carbon adsorption, liquid-liquid extraction, reverse osmosis, ultrafiltration, and others. These methods are effective at certain concentrations but not when the heavy metal concentration is below 10 mg L-1; some of the treatments also produce a large amount of sludge, which necessitates expensive post treatment (Garbisu et al., 1999; Wang and Chen, 2006; Jonglertjunya, 2008; Cuizano and Navarro, 2009; Cárdenas-González et al., 2010).
The fungus Phanerochaete chrysosporium belongs to the Basidiomycetes division and is a saprophyte white-rot fungus that has been studied for the past 20 years because it is able to remove a wide range of toxic pollutants from the environment: polycyclic aromatic hydrocarbons (PAHs), polychlorinated biphenyls, organochlorine pesticides, heavy metals, textile dyes and nitro-explosives, among others (Kanaly and Hur, 2006). It has been reported as capable of heavy metal biosorption due to the production of significant amounts of biomass able to remove cadmium, lead, nickel and copper, and it is tolerant to certain concentrations of these ions (Say et al., 2001; Kanaly and Hur, 2006; Gómez-Bertel, 2008).
Metal ion uptake by living and dead cells can consist of two modes. The first uptake mode involves surface binding of metal ions to cell wall and extra cellular material. The second mode of metal uptake into the cell through the cell membrane is dependent on cell metabolism and is referred to as intracellular uptake, active uptake or bioaccumulation. For living cells, metal uptake is also facilitated by the production of metal-binding proteins. In the case of living cells, metal uptake is also facilitated by the production of metal-binding proteins (Bishnoi and Garima, 2005). Ezzouhri et al. (2010) studied the presence of lead deposits in the cytoplasm of Penicillium sp. by transmission electron microscopy (TEM) and energy dispersive X-ray spectroscopy (EDX). Their results indicate that at least two mechanisms are involved in lead sorption by fungus biomass. First, lead was bound to the cell surface, and then intracellular accumulation took place.
Trametes versicolor is a fungus that grows on dead wood (Rebelo et al., 2007). When grown in the laboratory, it produces white cottony mycelia attached to the Petri dish. It has thick spores and hyaline hyphae, which are difficult to observe under the microscope (Rebelo et al., 2007). The fungus has the ability to produce different enzymes that can degrade recalcitrant compounds and, for this reason, it is used in biotechnology for bioremediation studies (Congeevaram et al., 2007). Trametes versicolor was used for Cu2+, Pb2+, Cd2+, Ni2+ and Zn2+ biosorption in aqueous solution batch systems, as well as biosorption of organic textile dyes (Fu and Viraraghavan, 2001; Baldrian, 2003; Bayramoglu et al., 2003).
Due to the need for environmental bioremediation and its importance in improving health, the present study aimed to determine growth parameters of Phanerochaete chrysosporium ATCC 32629 and Trametes versicolor ATCC 12679 in liquid YNB and YPD media with different metal ion concentrations in order to compare the ability and selectivity of these fungi to produce biomass and carry out Pb2+, Zn2+ and Cr3+ biosorption.
Materials and methods
Microorganisms
Phanerochaete chrysosporium ATCC 32629 and Trametes versicolor ATCC 12679 fungi were propagated in Petri dishes with potato dextrose agar (PDA) at 26 °C for 42 h and 96 h, respectively. The dishes were then refrigerated at 4 °C.
Preparation of Inoculum
One cm2 of PDA solid medium with fungus culture was taken and then inoculated into 100 mL previously sterilized YPD liquid medium (5 g L-1 yeast extract, 5 g L-1 peptone and 20 g L-1 glucose). The flasks were incubated at 25 °C, 150 rpm. Phanerochaete chrysosporium ATCC 32629 and Trametes versicolor ATCC 12679 were incubated for 42 and 96 hours, respectively.
Growth Kinetics for Metal Biosorption
Growth kinetics were performed in triplicate in Erlenmeyer flasks (batch system) in 500 mL of Difco Yeast Nitrogen Base media (YNB), supplemented with Pb(NO3)2, Zn(NO3)2 6H2O and Cr(NO3)3 9H2O (Sigma Aldrich, purity 98 to 99.0%), at concentrations of 0.25, 0.5, 1, 2, 5, 10, and 20 mg L-1 (ppm) and pH adjusted to 6 ± 0.002 with NaOH (0.1 M). These concentrations were chosen to cover high, medium and low concentrations as stipulated by NOM-001-ECOL-1996, which establishes the maximum permissible limits of pollutants in the wastewater discharged into national waters and properties to protect their quality and possible uses. The cultures were inoculated with 5 g L-1 biomass in exponential phase. Subsequently, the Erlenmeyer flasks were incubated at 25 °C, 150 rpm for 150 hours. Growth kinetics was measured in 5 mL samples taken every 4 hours to be used for subsequent analysis.
Analysis of Metal Ion Biosorption
The concentrations of metal ions were as follows: Pb2+ (0.25, 1 and 2 mg L-1), Zn2+ (5, 10 and 20 mg L-1) and Cr3+ (0.5, 1 and 2 mg L-1). Pb2+, Zn2+ and Cr3+ analyses were carried out according to Method 3005A, described by the Environmental Protection Agency of the United States of America (US EPA, 1987). Ten mL were taken from the supernatant of each of the culture media and diluted 1:10 with distilled water. Then, 5 mL hydrochloric acid and 2 mL concentrated nitric acid were added and the sample heated at 95 °C until the volume was reduced to approximately 20 mL. Subsequently, the digested samples were analyzed by Atomic Emission Spectrophotometer in an Inductively Coupled Plasma ICP machine with an Elemental Thermo Iris Intrepid dual lens (ICP-AES), resolution 0.0009 nm, using argon as the main source. Analysis was controlled with TEVA 1.1\1.01.0 TM software. A calibration curve was prepared with standard Pb2+, Zn2+ and Cr3+ at different concentrations.
Biomass Analyses
Fungus growth was measured in triplicate batch cultures for all strains and monitored by measuring the biomass produced every 4 hours. Biomass was determined gravimetrically by filtering 10 mL culture samples on pre-weighed nitrocellulose filters (pore size 0.65 /j.m; Sartorius AG, Gottingen, Germany). The filters were washed with demineralized water and dried in an oven set at 80 °C. Dry mass was established by repeatedly weighing the filters until a constant value was reached (after 24 hours). Finally, the biomass was determined by dry cell weight.
Calculation of Kinetic Parameters
In order to evaluate the effect of metal ions on the growth of microorganisms, the following kinetic parameters were calculated:
Biomass yield (YX/S, g/g) = (X-X0/S0-S)
Substrate consumption (S, g L-1) = S0 - Sf
Where X0, and X are initial and final biomass concentrations (g L-1) and S0 and S are initial and final substrate concentrations (g L-1).
Specific growth rate (μ, h-1) = 1/X (dX/dt)
Sugar consumption rate (r s, g. g L-1. h-1): = dS/dt
Duplication time (td=h) = μmax/ Ln 2
Substrate Consumption
Substrate consumption (glucose concentration) analysis was carried out with 2 mL medium samples. The samples were centrifuged at 4500xg for 10 min; the supernatant was recovered and determined by the DNS (3, 5-Dinitro salicylic acid) method (after Bernfield, 1955) measured at 540 nm. A calibration curve was drawn up using standard solutions of 0 to 2 mg mL-1 glucose equivalent.
Statistical Analysis
All experimentation was performed in triplicate and the results expressed as mean ± standard deviation.
The analysis of variance in the experimental designs and comparison of means by Fisher test (P < 0.05) were carried out using the Minitab v.16 statistical software (Minitab Inc. State College, PA). The resulting data for Biosorption percentage were subjected to comparison of means by the Tukey test (P < 0.05) using the Minitab v.16 statistical software (Minitab Inc. State College, PA). Graphical means were carried out using statistical analysis software Stat graphics Centurion XVI (c).
Results
Growth Kinetics of Phanerochaete chrysosporium ATCC 32629
The growth kinetics of Phanerochaete chrysosporium ATCC 32629 in YNB medium and YNB medium with different Pb+2 ion concentrations are shown in Figure 1a. The culture with Pb+2 ions reached exponential phase at approximately 36 hours, with significant growth decrease over the 150 hours of culture, contrasting with YNB without metal ions where an exponential growth phase was observed up to 140 hours of culture.

Figure 1 Growth kinetics of Phanerochaete chrysosporium ATCC 32629 in (a) YNB (♦), with different concentrations of Pb+2 ions: 0.25 mg L-1 ( □ ),0.5 mg L-1 (O) and 1 mg L-1 (X); (b) of Zn+2 ions: YNB (♦), 5 mg L-1 (□), 10 mg L-1 (O) and 20 mg L-1 (X); (c) and of Cr+2 ions: 0.5 mg L-1 (□), 1 mg L-1 (O) and 2 mg L-1 (X). Bars means standard deviation for triplicates.
The highest biomass production was achieved in the YNB medium without metal ions (0.90 g L-1), followed by YNB with 0.25 mg Pb+2 L-1 (0.87 g L-1); biomass yields in all other cultures were low (Table 1). Cultures with Pb+2 at concentrations of 0.5 and 1 mg L-1 were found to affect biomass production, an even lower biomass being obtained than with the other two medium concentrations after 24 hours of growth (Table 1). This is likely due to an increase in the Pb2+ concentrations in the culture medium. At 56 hours, decrease in biomass and the start of a consequent death phase were observed (Figure 1a).
Table 1 Kinetic parameters of Phanerochaete chrysosporium ATCC 32629 in YNB medium and YNB medium plus Pb+3, Zn+2 and Cr+2.

In the cultures in YNB medium with Zn+2 ions (Figure 1b), behavior was very similar to that observed with Pb+2 ions. Exponential growth phase ended at approximately 50 hours of culture, directly entering a phase of progressive decline and death, which remained constant until 150 hours of culture. The highest biomass production was obtained in YPN medium with 5 mg Zn+2 L-1, a 27% decrease in biomass occurring in media containing 10 and 20 mg Zn2+ L-1 (Table 1).
In the YNB medium with Cr+3 ions, Phanerochaete chrysosporium ATCC 32629 growth kinetics showed it was better suited to this metal as growth was promoted (Table 1). Cr+3 ions favor biomass production, peaking at 1.35 g L-1, compared to YNB medium without this metal, where less biomass was produced. It is believed that Cr+3 ions present in the culture medium act as a growth cofactor for Phanerochaete chrysosporium ATCC 32629 (Figure 1c), which somehow favors growth and biomass production.
We also note that at low concentrations of Pb+2 and Zn+2 ions (Table 1), the fungus can produce more biomass than with higher concentrations of these metal ions. Normally, Phanerochaete chrysosporium ATCC 32629 is cited as a fungus with low growth rate and biomass production, a fact we can note with the growth rates presented in Table 1.
Substrate (glucose) consumption measurement showed that Phanerochaete chrysosporium ATCC 32629 consumed virtually all the glucose (20 g L-1) in all culture media with and without metal (data not shown). However, substrate consumption rate was different when metal ions are added to the medium, probably because the fungus modifies its metabolism and glucose is consumed according to cell necessity (Table 1).
Biosorption Kinetics of Pb+2, Zn+2 and Cr+3 Ions by Phanerochaete chrysosporium ATCC 32629
P. chrysosporium ATCC 32629 metal ion biosorption results are shown in Table 2. We observed variable metal biosorption ability by this fungus, dependent on the concentration and type of metal used. Cultures carried out with an initial concentration of 0.25 ppm Pb2+ showed that at 24 h growth kinetics the fungus achieved a maximum biosorption of 1.1% of Pb2+, with a specific uptake (qe) of 0.0027 mg metal per gram of biosorbent.
Table 2 Biosorption percentage and specific metal uptake by Phanerochaete chrysosporium ATCC 32629 in YNB medium.

Different letters in the same line indicate statistically significant differences (P < 0.05).
Furthermore, the kinetics of P. chrysosporium ATCC 32629 biosorption at different concentrations of Zn2+ showed that 20 g L-1 of the fungus increased uptake of this metal (Table 2); however, no significant variation was observed in the percentage of biosorption compared with the other concentrations tested.
Table 2 showsthat atthe 0.25mg Pb+3 L-1 concentration the fungus achieved a maximum biosorption of 1.1% and a specific uptake (qe) of 0.0027 mg metal g-1 biosorbent, although it was not statistically different from the other studied concentrations. In Figure 2, we show that P. chrysosporium ATCC 32629 is capable of Cr3+ biosorption; however, at concentrations of 2 g L-1 the fungus had a significantly lower percentage of biosorption (Table 2) relative to the other concentrations tested.
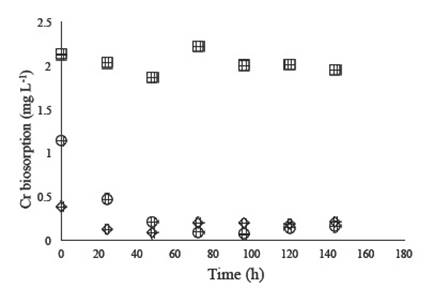
Figure 2 Cr+3 ion biosorption by Phanerochaete chrysosporium ATCC 32629 in YNB at 0.5 mg L-1 (◊%), 1.0 mg L-1 (O) and 2.0 mg L-1 (□).
In spite of the low biosorption of Pb2+ and Zn2+ ions by Phanerochaete chrysosporium ATCC 32629 in the YNB medium, the fungus was capable of Cr+3 ion biosorption after 144 h growth kinetics, exhibiting maximum biosorption of 94.2% at a concentration of 1 ppm Cr+3. The same good biosorption of the metal was observed at 0.5 ppm Cr+2 (e.g. Table 2), a satisfactory result compared with the experiments conducted with Pb3+ and Zn2+ ions.
Growth Kinetics of Trametes versicolor ATCC 12679
Trametes versicolor ATCC 13488 growth was highly heterogeneous compared with that observed in P. chrysosporium ATCC 32629 (data not shown). Because of this, it was not possible to quantify the biomass during kinetics, as the fungus formed filamentous cell clumps approximately 2 cm thick and it was virtually impossible to separate the biomass into representative portions of the sample.
With time, the fungus was seen to grow considerably as an aggregate, to the extent that it occupied the entire flask volume. The difficulty of quantifying the biomass generated from Trametes versicolor ATCC 12679 was due to heterogeneous growth, which did not produce a typical cell growth curve like that of P. chrysosporium ATCC 32629. However, despite the form of fungus growth, the substrate (glucose), completely consumed after 48 hours of cultivation, could be measured.
Total biomass from Trametes versicolor ATCC 13488 after 72 hours cultivation in YNB medium without metal ions was approximately 1.28 g L-1, with a biomass yield of 0.064 g g-1. The yields obtained in cultures with metal ions were lower than in YNB medium without, ranging from 0.98 to 1.08 g L-1.
Biosorption Kinetics of Pb+2, Zn+2 and Cr+3 Ions by Trametes versicolor ATCC 13488
Table 3 shows the results of metal biosorption by T. versicolor ATCC 13488, similar to those observed for P. chrysosporium ATCC 32629. The ability of T. versicolor ATCC 13488 for metal biosorption in the culture medium was variable and dependent on the type and concentration of the metal used.
Table 3 Biosorption percentage and specific metal uptake by Trametes versicolor ATCC 13488 in YNB medium.

Different letters in the same line indicate statistically significant differences (P < 0.05).
The cultures carried out with an initial concentration of 0.50 ppm Pb3+ showed that at 24 h growth kinetics (Figure 3), the fungus achieved maximum Pb2+ biosorption of 43.3%, with specific uptake (qe) of 0.1187 mg metal g biosorbent-1 (Table 3). However, at a concentration of 0.25 ppm Pb2+, the fungus exhibited a significantly much higher percentage of biosorption (86.3%) compared to other concentrations. Comparing this result with those of P. chrysosporium ATCC 32629, T. versicolor ATCC 13488 presented greater Pb2+ biosorption and uptake. Most importantly, the higher the concentration of Pb2+, the lower the fungal biosorption capacity for this metal (Table 3). However, at the onset of the death phase, desorption of the metal ion was observed, probably due to fungal cell wall lysis at the time of death. It is important to note that after 24 hours, the kinetics must be stopped to prevent desorption of the previously trapped ion.
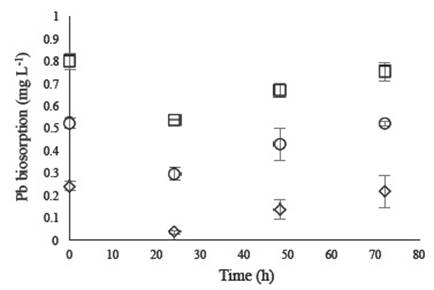
Figure 3 Pb+2 ion biosorption by Trametes versicolor ATCC 13488 in YNB at 0.25 mg L-1 (◊), 0.5 mg L-1 (O) and 1 mg L-1 (□).
Biosorption kinetics of T. versicolor ATCC 13488 with Zn2+ and Cr3+ show decreased uptake of these metals, a considerable reduction in the case of Cr3+ (Figure 3) compared with values obtained with Pb2+. Consequently, the fungus is more susceptible to Zn2+ and Cr3+ ions, although the presence of these metals did not affect its growth.
Discussion
Metal ion biosorption by fungal biomass is a species-specific process. The mechanism of biosorption is selective and, depending on the nature of the fungus used and various external factors, its efficiency differs among species of different fungi used and the metal tested (Baldrian, 2003; Zhang et al., 2007). Fungi normally used for the removal of metal ions produce significant amounts of biomass with large surface area, which facilitates mycelium immobilization in agroindustrial substrates, such as wheat bran (Say et al., 2001; Gómez-Bertel, 2008).
In our study, Phanerochaete chrysosporium ATCC 32629 and Trametes versicolor ATCC 12679 produced significant amounts of biomass in the form of pellets, whose diameters averaged ±2 mm after 8 hours cultivation and ± 5 mm after 48 hours in the presence of Pb2+, Cr3+ and Zn2+ ions. The bigger the pellet, the greater the Cr3+ biosorption by Phanerochaete chrysosporium ATCC 32629 in the culture medium, and the same relationship exists for Pb2+ biosorption by Trametes versicolor ATCC 12679.
It has been reported that Phanerochaete chrysosporium is capable ofbiosorption ofheavy metals such as cadmium, lead, nickel and copper; moreover, this strain is tolerant to certain concentrations of these metal ions (Say et al., 2001; Kacar et al., 2002; Nikazar et al., 2008).
Several kinetic parameters (agitation, temperature, pH, biomass) have been studied to determine the biosorption rate of some metals by Phanerochaete chrysosporium grown in different culture media. Structural biosorbent properties have even been studied to analyze the protein and carbohydrate composition of the cell wall and to measure the charge density on the cell surface and surface area, among others (Falih, 1997; Yalcincaya et al., 2002; Bayramoglu, 2003; Jonglertjunya, 2008).
Maximum biosorption of heavy metals by fungal biomass was observed when pH was around 6 (Say et al., 2001). Stirring is another important factor as high levels of oxygen increase biodegradation rate (Ulmer et al., 1983; Michel et al., 1992; Hossain et al., 2005); the best agitation condition for higher biomass production is 200 rpm (Singh and Chen, 2008). Another factor that may affect biosorption ability is temperature; several reports indicate that fungus incubation between 15 and 45 °C does not affect biosorption capacity (Kacar et al., 2002; Yalcincaya et al., 2002; Arica et al., 2003; Bayramoglu et al., 2003; Iqbal and Edyvean, 2004).
In our study, we observed that Pb2+, Cr3+ and Zn2+ biosorption by Phanerochaete chrysosporium ATCC 32629 and Trametes versicolor ATCC 12679 was variable and we believe that this can be attributed to the culture conditions established in this study (YNB, 150 rpm agitation, 25 °C temperature and 6 initial pH) which influenced biomass production and metal biosorption. These growth parameters, which encourage the uptake of metal ions by these two strains of fungi, were established according to those reported by other researchers. For example, Baldrian (2003) states that the biosorption mechanism is selective and that its effectiveness differs among fungal species used and the metal to be tested. He showed that Phanerochaete chrysosporium biosorption capacity is in function of the type of metal, in the following order: Cu>Pb> Cd.
We therefore think that the low biosorption of Pb2+ and Zn2+ ions by Phanerochaete chrysosporium ATCC 32629 and Cr3+ and Zn2+ ions by Trametes versicolor ATCC 12679 is mainly species-specific. On the other hand, we find that, P. chrysosporium ATCC 32629 and T. versicolor ATCC 12679 are capable of Cr3+ and Pb2+ ion biosorption, respectively, in the YNB medium under the culture conditions established in this study. Another example of this is the Trichoderma atroviride strain, isolated from sewage sludge, which is capable of surviving high concentrations of copper, zinc and cadmium (Lin et al., 2003).
Trametes versicolor is a fungus employed in various studies of bioremediation for Cd2+ biosorption and has been studied in a range from 30 to 700 mg L-1 of this metal. The results showed that this fungus achieved a maximum biosorption rate of 124 mg Cd+2 g biomass-1 at pH 6 (Yalcincaya et al., 2002). Likewise, Trametes versicolor ATCC 13488 has been used for biosorption of Cu2+, Pb2+ and Zn2+ at concentrations of 30 to 400 mg L-1 in a batch system with immobilized strains in carboxymethyl cellulose, with shaking (400 rpm) and incubated at 25 °C. Under these culture conditions, the maximum biosorption capacity observed was 1.51 mmol Cu2+, 0.85 mmol Pb2+ and 1.33 mmol Zn2+ g-1 biosorbent with pH between 4 and 6 (Bayramoglu et al., 2003).
Therefore, comparing the work of Bayramoglu et al. (2003) with our results, we can confirm that Trametes versicolor ATCC 12679 is capable of Pb2+ ion biosorption at concentrations between 400-100 ppm. Furthermore, Trametes versicolor was resistant to, and viable in the presence of, several metals such as cadmium, zinc, nickel, copper, chromium, molybdenum, lead, mercury and tin (Baldrian, 2003). Similarly, strains of Phanerochaete chrysosporium have been studied at different concentrations of metal ions.
We have shown in our work that the capacity for biosorption of metal ions by Phanerochaete chrysosporium ATCC 32629 and Trametes versicolor ATCC 12679 varies; we cannot generalize that these strains are similar. However, because they produce different amounts of biomass, their capacity for metal ion biosorption is not the same. Therefore, it is important to conduct further studies on culture conditions that favor biosorption and ascertain the metabolism of the fungus to achieve greater uptake of metals in the culture medium.
Conclusions
The fungus Phanerochaete chrysosporium ATCC 32629 is tolerant and capable of significant biosorption at exponential and stationary phases at the Cr+3 ion concentration of 1 mg L-1 in YNB medium, although Pb+2 and Zn+2 ion biosorption was not found under the same culture conditions. Trametes versicolor ATCC 12679 was tolerant and capable of Pb+2 ion biosorption at concentrations of 0.25, 0.50 and 1 mg L-1 in YNB medium; however, at onset of the death phase, desorption of the metal ion was observed and the fungus was not capable of Zn+2 and Cr+3 ion biosorption. The two fungi showed promising results and highlighted the importance of environmental biotechnology. These results may be the basis for the development of clean economically viable methods of treating natural water and industrial effluent. It should be noted, however, that the mechanism of the biosorption process has not been fully understood and there are certain gaps in this technology. These mechanisms are now the main focus in the development of future applications in situ.











 nueva página del texto (beta)
nueva página del texto (beta)


