Introduction
Breast cancer (BC) is the most frequently diagnosed malignancy and the leading cause of cancer-related deaths in females, with a worldwide estimate of 2.3 million new diagnoses and 685 000 registered deaths in 2020.1 Notably, incidence rates are rapidly increasing in Latin America, where the transitioning of economies have brought on substantial socio-cultural and lifestyle changes that have favored a rise in BC risk factors (e.g. delayed childbearing, smaller family size, high prevalence of physical inactivity, and increased frequency of overweight/obesity).1 In Mexico, the incidence of BC has steadily risen from 23 new cases per 100 000 females in 2010 to 35 per 100 000 females in 2019, and this disease poses a substantial public health issue as the 5-year prevalence nears 100 000 patients.2 Furthermore, BC diagnoses in the country tend to present at a young age, be detected at an advanced stage, and have a high incidence of aggressive clinicopathological features such as triple-negative subtype (TNBC), all of which increase the national burden of this disease.3 Thus, the development of interventions aimed at improving early BC detection and optimizing treatment strategies is urgently needed.
The etiology of BC is complex, but it is estimated that 5-10% of cases are attributable to germline pathogenic variants (PV) in BC susceptibility genes.4 PVs of multiple genes have been associated with an increased risk of BC, which can be broadly classified into three categories: high-penetrance variants (e.g. PVs in BRCA1, BRCA2, PTEN, TP53, CDH1, and STK11) that confer a >5 relative risk for BC, moderate-penetrance variants (e.g. PVs in CHEK2, BRIP1, ATM, and PALB2) that convey a relative risk of 1.5-5 for BC, and low-penetrance variants that contribute to BC risk in a polygenic fashion.5,6 In the last decades, genetic testing for PVs in BC-susceptibility genes has become widely available, though there is limited knowledge about PVs associated with TNBC in Mexico.
The identification of germline PVs in moderate- to high-penetrance genes has substantial clinical implications. In patients with BC, the presence of germline PVs has been associated with certain clinicopathological features and has considerable implications for treatment selection. For example, BRCA1-associated BC often demonstrates early-onset, high histological grade, and estrogen-receptor negative phenotype, as well as enhanced sensitivity to DNA-damaging therapies such as platinum-containing chemotherapy regimens and poly (ADP-ribose) polymerase inhibitors.7,8 Furthermore, the identification of PVs in a BC patient can lead to cascade testing in relatives, enabling the identification of individuals at an increased risk for this disease.9 Patients diagnosed with a genetic predisposition to cancer are candidates for specialized screening strategies and risk-reducing procedures.10 Therefore, genetic testing has the potential to improve the outcomes of BC patients by favoring the early detection of hereditary BC and allowing a case-by-case optimization of treatment strategies.
Currently, genetic testing in Mexico is not routinely provided to BC patients that meet genetic testing criteria. The barriers to implementing genetic cancer risk assessment (GCRA) services in the country include excessive costs, lack of coverage of genetic testing within the public health system, limited awareness of the benefits of identifying PVs, and limited number of physicians with GCRA expertise.11,12 Previous studies in Mexican BC patients unselected for family history have demonstrated that 15% of cases overall are associated with BRCA PVs.13 The frequency of BRCA PVs was higher (>20%) among young TNBC patients in two small studies,14,15 and this molecular subtype accounts for 16-23% of BC cases in Mexico.16,17 The aim of this study is to analyze the spectrum of PVs identified among Mexican patients with TNBC who were enrolled in a prospective registry and underwent genetic testing.
Materials and methods
Mexican patients diagnosed with invasive TNBC between December 2011 and December 2020 in Clinical Cancer Genomics Community Research Network (CCGCRN) collaborating centers were included in this study. The CCGCRN is a large research group with over 40 collaborating sites in the United States and Latin America that employ a common hereditary cancer registry protocol.18 Approximately 20% of patients included in the registry were from Latin American sites; half of these (10% overall) from Mexico. Patients who consented to participate were recruited into a prospective database coordinated by City of Hope Comprehensive Cancer Center (Institutional Review Board # 96144). The protocol was reviewed and approved by the research and ethics committees at each participating institution. After provision of informed consent, a blood sample was obtained and prospective data collection was undertaken, including information regarding personal risk factors, multigenerational family medical history, and clinical characteristics of cancer diagnosis. In Mexico, the sites of patient enrollment included Instituto Nacional de Cancerología, Instituto Nacional de Ciencias Médicas y Nutrición Salvador Zubirán and Hospital General de México in Mexico City, Hospital San José TecSalud and Hospital Zambrano Hellion TecSalud in Monterrey, Nuevo León, Hospital Universitario de Guadalajara in Guadalajara, and Centro de Estudios y Prevención del Cáncer in Juchitán de Zaragoza, Oaxaca.
Most patients enrolled in the CCGCRN registry meet NCCN criteria for genetic testing. All patients included in this study received GCRA and had genetic testing results available at the time of this analysis. Various methods were used for PV detection,12 including a multigene panel test with a custom QIAseq (QIAGEN, Inc., Germantown, MD, USA) amplicon-based panel for detecting breast cancer predisposition variants (ATM, BRCA1, BRCA2, CDH1, CHEK2, NF1, PALB2, PTEN, RAD51C, RAD51D, TP53). The design includes the 5’ and 3’ untranslated regions, full exonal gene coverage, and extends 10 base-pairs into introns, and results in a consistent coverage of over 300-500x. Sequencing was done on the HiSEQ 2500 Genetic Analyzer (Illumina Inc., San Diego, CA), or full BRCA1 and BRCA2 sequencing on the ion torrent platform (Thermo-Fisher). BRCA1 was also analyzed for number variants by multiplex ligation-dependent probe amplification (MLPA; Holland). Sanger re-sequencing was used to confirm PVs.
The statistical analysis was carried out using the STATA version 13.0* software, with patients grouped according to mutational status. Descriptive statistics were performed using frequency and proportions for categorical variables and median and range for quantitative variables. Mann-Whitney U and Fisher exact tests were used for exploring differences according to group category, as appropriate. Statistical significance was defined as a two-sided p-value of < 0.05.
Results
A total of 1 506 Mexican patients with primary BC underwent genetic testing, of which 387 (25.7%) had invasive TNBC and were included in this study. Most patients met NCCN criteria with TNBC diagnosis at ≤ 60 years of age (381/387; 98.5%), or >60 years old and ≥1 close blood relative diagnosed with BC at ≤50 years of age (2/387; 0.5%), or ≥3 diagnoses of BC in patient and/or close blood relatives (1/387; 0.3%). Just three cases (0.8%) did not meet NCCN criteria, all of whom were >60 years old at BC diagnosis. Overall, 245 (63.3%) patients had a multi-gene panel assay performed, while 142 (36.7%) underwent screening for BRCA PVs only.
A total of 113 (29.2%) patients were found to be carriers of PVs in BC-susceptibility genes (table I). As shown in table II, PVs were identified in BRCA1 (n=89, 78.8%), BRCA2(n=17, 15.0%), PALB2 (n=2, 1.8%), ATM (n=1, 0.9%), BRIP1 (n=1, 0.9%), PTEN (n=1, 0.9%), RAD51C (n=1, 0.9%), andTP53 (n=1, 0.9%). In the subset of patients that had a multi-gene panel assay performed, 73 (29.8%) had a PV identified, of which 66 (90.4%) were in BRCA and 7 (9.6%) in other BC-susceptibility genes. Recurrent PVs (identified in ≥ 3 patients) included BRCA1 ex9-12del (n=26, 23.0%), BRCA1 c.2433delC (n=9, 8.0%), BRCA1 ex16-17del (n=6, 5.3%), BRCA1 c.211A>G (n=5, 4.4%), BRCA1c.5030_5033delCTAA (n=3, 2.7%), and BRCA2 c.274C>T (n=3, 2.7%).
Table I Characteristics of included TNBC patients and proportion of patients diagnosed with a germline PV. Mexico, December 2011-December 2020
|
PV carriers (%) n=113 |
Non-PV carriers (%) n=274 |
p-value |
Overall (%) n=387 |
|
|
Site of enrolment | ||||
|
INCan |
35 (31.0) |
112 (40.9) |
0.048 |
147 (38.0) |
|
TecSalud Hospitals |
44 (38.9) |
87 (31.8) |
131 (33.9) |
|
|
Hospital General de México |
20 (17.7) |
26 (9.5) |
46 (11.9) |
|
|
INCMNSZ |
7 (6.2) |
31 (11.3) |
38 (9.8) |
|
|
Hospital Universitario de Guadalajara |
6 (5.3) |
17 (6.2) |
23 (5.9) |
|
|
CEPREC |
1 (0.9) |
1 (0.4) |
2 (0.5) |
|
|
Sex | ||||
|
Female |
112 (99.0) |
274 (100) |
0.292 |
386 (99.7) |
|
Male |
1 (0.9) |
0 |
1 (0.3) |
|
|
Age at BC diagnosis | ||||
|
Median (years) |
37 |
40 |
0.004* |
39 |
|
≤ 30 |
19 (16.8) |
33 (12.0) |
0.142 |
52 (13.4) |
|
31-40 |
57 (50.4) |
112 (41.2) |
170 (43.9) |
|
|
41-50 |
28 (24.8) |
90 (32.9) |
118 (30.5) |
|
|
51-60 |
8 (7.1) |
33 (12.0) |
41 (10.6) |
|
|
> 60 |
1 (0.9) |
5 (1.8) |
6 (1.6) |
|
|
Stage at diagnosis | ||||
|
I |
11 (9.7) |
22 (8.0) |
0.585 |
33 (8.5) |
|
II |
49 (43.4) |
121 (44.2) |
170 (43.9) |
|
|
III |
46 (40.7) |
99 (36.1) |
145 (37.5) |
|
|
IV |
5 (4.4) |
22 (8.0) |
27 (7.0) |
|
|
Missing |
2 (1.8) |
10 (3.7) |
12 (3.1) |
|
Analyses were performed using the STATA version 13.0 software. All p-values were calculated using Fisher exact tests unless otherwise specified.
*Calculated with Mann-Whitney U
TNBC: triple negative breast cancer; PV: pathogenic variant; CEPREC: Centro de Estudios y Prevención del Cancer; INCan: Instituto Nacional de Cancerología; INCMNZ: Instituto Nacional de Ciencias Médicas y Nutrición Salvador Zubirán: BC: breast cancer
Table II PVs identified in Mexican patients with TNBC. Mexico, December 2011-December 2020
|
Gene |
Identified PVs |
|
ATM |
c.3802delG |
|
BRCA1 |
c.66_67delAG c.80+1G>A c.81-1G>A c.211A>G c.220C>T c.241C>T c.815_824dupAGCCATGTGG c.1407_1408delAA c.1674delA c.1860delT c.1961delA c.2433delC c.2806_2809delGATA c.3029_3030delCT c.3037_3038delGA c.3648dupA c.3759_3760delTA c.3858_3861delTGAG c.4065_4068del c.4327C>T c.4862_4871delATACTGCTGG c.4921_4924delGAAA c.5030_5033delCTAA c.5062_5064delGTT c.5074+1G>A c.5123C>A c.5263dupC c.5266dupC c.5346G>A c.5353C>T c.5359T>A ex1-2del ex1-24del ex5-15del ex14-17dup ex14del ex16-17del ex18-19del ex21-22dup ex9-12del |
|
BRCA2 |
c.2653_2656delGACA c.274C>T c.2808_2811delACAA c.3264dupT c.4111C>T c.4889C>G c.5631delC c.6024dupG c.658_659delGT c.6591_6592delTG c.8168A>G c.9699_9702delTATG |
|
BRIP |
c.241delG |
|
PALB2 |
c.108+1G>A c.2167_2168delAT |
|
PTEN |
c.421delC |
|
RAD50 |
c.2980A>T & TP53 c.916C>T |
|
RAD51 |
c.577C>T |
Bold values represent recurrent variants (identified in ≥ 3 patients)
TNBC: triple negative breast cancer; PV: pathogenic variant
The median age at diagnosis for the entire cohort was 39 years (range 21-72). Overall, carriers of PVs were diagnosed with BC at a younger age than non-carriers (37 vs. 40 years, p=0.004). However, when stratifying by mutational status, carriers of BRCA1 PVs were found to be younger than non-carriers (36 vs. 40 years, p<0.001), but no statistical difference was observed between carriers of BRCA2 PVs and non-carriers (41 vs. 40 years, p=0.791). As shown in table I, a tendency towards decreased detection of PVs was observed with increasing age. Notably, BRCA2 and other BC-susceptibility genes represented a greater proportion of PVs among patients aged >40 years at diagnosis compared to younger cases (27 vs. 9.2% and 13.5 vs. 2.6%, respectively).
Regarding disease stage, 33 (8.5%) were stage I, 170 (43.9%) were stage II, 145 (37.5%) were stage III, 27 (7.0%) were stage IV, and for 12 (3.1%) the stage at diagnosis was unknown. The distribution of stage according to mutational status is shown in table III. No statistical differences were found for stage distribution between carriers of PVs and non-carriers (p=0.545).
Table III Disease stage according to mutational status. Mexico, December 2011-December 2020
|
Stage |
Non-carriers (n=274)(%) |
BRCA1 PV carriers (n=89)(%) |
BRCA2 PV carriers (n=17)(%) |
Other PV carriers (n=7)(%) |
|
I |
22 (8.0) |
7 (7.9) |
3 (17.7) |
1 (14.3) |
|
II |
121 (44.2) |
41 (46.1) |
5 (29.4) |
3 (42.9) |
|
III |
99 (36.1) |
34 (38.2) |
9 (52.9) |
3 (42.9) |
|
IV |
22 (8.0) |
5 (5.6) |
0 |
0 |
|
Missing |
10 (3.7) |
2 (2.3) |
0 |
0 |
PV: pathogenic variant
A total of 50 (12.9%) of patients were diagnosed with multiple primary malignant tumors (table IV). The organ of origin of other primary malignancies were breast (n=40, 80.0%), ovary (n=4, 8.0%), and others (n=6, 12.0%). Carriers of PVs had a higher rate of multiple primary malignancies than non-carriers (25/113 [22.1%] vs 25/274 [9.1%], p=0.001)
Table IV Incidence of second primary malignancies in TNBC patients according to mutational status. Mexico, December 2011-December 2020
|
Non-carriers (%) |
BRCA1 PV carriers (%) |
BRCA2 PV carriers (%) |
Non-BRCA PV carriers (%) |
p-value |
|
|
>1 primary cancer diagnoses |
25/274 (9.1) |
16/89 (18.0) |
4/17 (23.5) |
5/7 (71.4) |
<0.001 |
|
Type of second primary diagnosis | |||||
|
Second primary breast cancer |
20 (80.0) |
14 (87.5) |
2 (50.0) |
4 (80.0) |
0.327 |
|
Primary ovarian cancer |
3 (12.0) |
1 (6.3) |
0 |
0 |
|
|
Other primary malignancies* |
2 (8.0) |
1 (6.3) |
2 (50) |
1 (20.0) |
|
Analyses were performed using the STATA version 13.0 software. p-values were calculated using Fisher exact tests.
* The site of origin of other primary malignancies were thyroid, kidney, and gynecologic otherwise unspecified.
TNBC: triple negative breast cancer; PV: pathogenic variant
Discussion
To our knowledge this is the largest study of Mexican patients with TNBC, wherein we documented BC susceptibility gene PV in a large proportion (29.2%) of cases. Consequently, GCRA services should be offered to Mexican TNBC patients as a valuable tool to tailor cancer risk management strategies and direct treatment decisions.
Overall, 27.4% of the entire cohort was found to carry a BRCA PV while only 2.9% of the subset of patients that underwent testing with multi-gene panel assays were identified with PVs in non-BRCA genes. Previous studies have postulated that the TNBC phenotype has a tendency to be associated with hereditary BC.16 It is estimated that 9-21% of unselected cases and up to 40% of early-onset and familial cases of TNBC are associated with PVs in BC predisposing genes.19,20,21,22 The rate of patients with PVs identified in this cohort was high compared to what has been reported in the literature. Couch and colleagues evaluated the prevalence of PVs in a cohort of unselected TNBC patients from Germany, Greece, Finland, United States, and United Kingdom, and identified deleterious mutations in 14.6% (11.2% of patients had a BRCA PV and 3.7% in non-BRCA genes).21 Similarly, Shimelis and colleagues studied a cohort of TNBC patients that were subjected to clinical genetic testing in the United States and found that 12.0% carried a PV in moderate- to high-risk BC susceptibility genes (8.3% in BRCA and 3.7% in non-BRCA genes).23 In Chinese TNBC patients, Sun and colleagues reported that 11.2% of unselected TNBC patients carried a PV in BRCA and 3.8% had a germline PV in other BC-associated genes.22 Likewise, Ma and colleagues reported that 16.0% of Chinese TNBC patients harbored a PV (9.6% in BRCA and 7.6% in non-BRCA genes).24 There is a possibility that our results are due in part to selection bias, as similar rates of PVs in BRCA genes (31%-35%) have been reported in TNBC patients referred for genetic counseling in the United States.25,26 However, our group previously observed that 27% of Mexican TNBC patients aged ≤50 years at diagnosis had a BRCA PV,14 and the results of this study confirm a high rate of BRCA-associated cases in Mexican patients with this molecular subtype.
BRCA1 and BRCA2 represented the majority of PVs identified in this cohort. These high-penetrance genes code for proteins involved in maintaining genomic stability by participating in homologous recombination repair of double-strand DNA breaks.24 Both of these genes have been associated with an increased risk of TNBC (odds ratio [OR] 16.3- 42.9 for PVs in BRCA1 and 5.4-9.7 in BRCA2).23,27 BRCA1 ex9-12del was the most common PV identified in this study, accounting for 25% of BRCA PVs. This PV was first reported in a cohort of Hispanic Americans with a personal history of BC or ovarian cancer in 2007 and is largely considered to have a founder effect in Mexico, representing a regional public health problem.13,28 Other frequent PVs found in this cohort included BRCA1 c.2433delC (p.Lys812Argfs) that has been reported to account for 2.1-7.1% of BRCA1/2 mutations in Hispanics,29 BRCA1 c.211A>G (p.Arg71Gly) that is considered a founder mutation of Spanish origin,30 and BRCA1 ex16-17del that has been described to be common among Latin American BC patients.31
PVs in ATM, PALB2, RAD51C, and BRIP1 accounted for 67% of the PVs identified in non-BRCA1/2 genes. Thus, most of the carriers of PVs in this cohort had defective homologous recombination repair, indicating that alterations in this pathway might be a substantial contributor to the pathogenesis of TNBC. The aforementioned genes have been extensively reported to increase the risk for BC, and three of them (PALB2, BRIP1, and RAD51C) have been associated with high-to moderate TNBC risk (>2 OR). However, ATM PVs have not been demonstrated to convey a greater risk for TNBC,23,27,32 and could be an incidental finding in this cohort. Other PVs identified in this cohort included PTEN which is primarily involved in antagonizing cell proliferation and carries an up to 85% lifetime risk of BC,5 and TP53 that codes for a key controller of DNA damage-induced apoptosis and has been proposed as a TNBC predisposition gene,33 though it is more commonly associated with triple-positive disease in young women.34
Similar to previous studies,21 the median age at diagnosis in this cohort of TNBC patients was relatively young (39 years), with carriers of PVs being diagnosed at an earlier age than non-carriers. The highest prevalence of germline PVs (37%) was found in patients aged ≤ 30 years at BC diagnosis. However, a considerable proportion of patients aged > 60 years at diagnosis (17%) were carriers of PVs in BC-susceptibility genes. Comparable rates of PVs in patients aged 60 years or older with TNBC have been reported in other populations (13-18%), indicating that age alone should not be used as an exclusion criterion for GCRA in TNBC patients.25,35 On the other hand, mutational status was not found to be statistically associated with the distribution of disease stage, which is consistent with previous literature.36 As expected, carriers of PVs experienced a two-fold rate of second primary malignancies than non-carriers (22 vs. 9%). This underscores the clinical utility of detecting PVs in order to provide specialized surveillance strategies to patients with deleterious mutations and offer risk-reducing surgeries that can improve clinical outcomes in this population.
The multi-gene assay only demonstrated a marginally higher PVs detection rate (2.8% non-BRCA PVs) than screening for BRCA1/2 PVs alone. Given that elevated costs and lack of access to genetic tests can act as considerable barriers for the uptake of GCRA in Mexico, incremental genetic screening assays, as employed in this study, may be appropriate for genetic testing in Mexican TNBC patients who meet screening criteria (figure 1) if resources are constrained. First, as nearly 1 in 4 identified PVs corresponded to the BRCA1 Mexican founder mutation (ex9-12del), testing for this variant is an appropriate first step towards genetic screening. In case the patient is not found to be a carrier of BRCA1 ex9-12del, testing limited to BRCA1 and BRCA2 sequencing is an appropriate next step. If no PVs are identified in the first two steps, BRCA1 MLPA should be offered as up to 35% of identified PVs in this cohort corresponded to large genomic rearrangements (i.e., whole exon deletions or duplications) in this gene. A similar approach of initiating genetic screening in Mexican patients with BRCA1 ex9-12del testing was recently proposed by Fragoso-Ontiveros and colleagues because of the low cost (≈10 USD), relatively fast result (≈1 week), and high yield (5% of patients with suspected hereditary breast and ovarian cancer) of this test in Mexico.37 Alternatively, a multi-gene panel assay can be considered as an initial test, particularly in cases with personal or family history suspicious for hereditary cancer syndrome associated with non-BRCA BC-susceptibility genes (e.g., Cowden and Li Fraumeni syndromes). However, commercial multi-gene panel tests are costly at present, did not significantly increase the detection rate of PVs in this cohort, and can include variants that might not be clinically actionable.10 Thus, the performance of these tests should be considered on a case-by-case basis, according to available resources and potential clinical impact. Ultimately, we expect that advances in sequencing technology will continue to lower the cost of multi-gene panels, sparing the need for a stepwise approach.
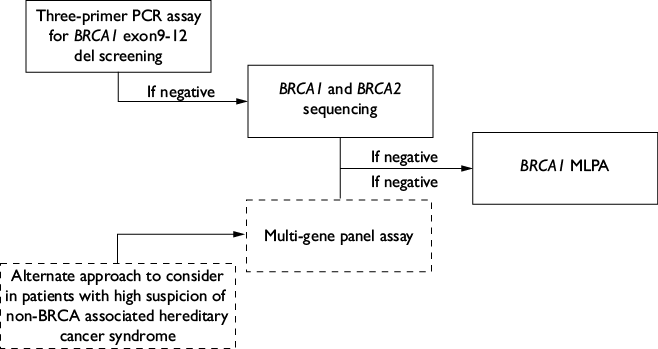
TNBC: triple negative breast cancer; MLPA: multiplex ligation-dependent probe amplification; PCR: polymerase chain reaction
PV: pathogenic variant; NCCN: National Comprehensive Cancer Network; GCRA: genetic cancer risk assessment
Figure 1 Proposed algorithm for the performance of genetic tests in Mexican patients with TNBC that meet NCCN’s criteria for GCRA. Mexico, December 2011 - December 2020
This study has several limitations. Some patients in this cohort underwent genetic testing for BRCA1 and BRCA2 only, representing temporal availability and standard of practice. Therefore, some carriers of PVs in other BC susceptibility genes could have been erroneously classified as non-carriers and we likely underestimated the true prevalence of PVs in this population. Nonetheless, the frequency of PVs in this cohort was higher than what has been reported in the literature. Similarly, the frequency of second primary malignancies might be underestimated given the limited follow-up available for some patients. Despite these limitations, this study provides a valuable estimate of the prevalence of PVs of BC predisposing genes in Mexican women with TNBC who are referred to genetic testing.
Conclusions
A high prevalence of germline PVs in moderate- to high-penetrance BC susceptibility genes is present in Mexican patients with TNBC, with most located in BRCA1 and BRCA2. GCRA is an effective strategy to identify candidates for specialized screening strategies and risk-reducing procedures. Furthermore, the identification of PVs in certain genes can help guide treatment selection and appropriate surveillance strategies in high-risk BC patients. The cost of genetic testing and the uptake of GCRA services can be optimized in Mexican centers by adopting a stepwise approach to genetic testing, starting with low-cost, high-yield assays (e.g., PCR assay for BRCA1 ex9-12del detection) and culminating in multi-gene panel assays in case of high genetic predisposition suspicion but negative results by single/limited gene assays.











 nueva página del texto (beta)
nueva página del texto (beta)


