INTRODUCTION
Electronic cigarettes, also known as “e-cigarettes,” “e-cigs,” “cigalikes,” “e-hookahs,” “mods,” “vape pens,” “vapes” or “tank systems,” refer to a device that delivers nicotine through the respiratory system, thus their proposed technical name of electronic nicotine delivery system (ENDS)1,2. A recently released ENDS, launched by Phillip Morris as IQOS, was designed to produce an aerosol by heating tobacco without burning it. This “heat-not-burn” device is a novel ENDS different from e-cigs. ENDS not only disembarked widely in the tobacco consumer’s market but also has introduced a wedge in the scientific smoking-cessation and tobacco-control advocacy communities.
The first commercially available e-cigs device was developed in China, in 2003, as an alternative to smoking in places where smoking regular cigarettes were prohibited3,4. It was named Ruyan, a Chinese word for “resembling smoking,” and unlike nicotine patches, gum or lozenges it was not designed as a pharmacologic tool for smoking cessation but to deliver “enjoyable” nicotine and overcome regulations5.
E-cigs are by definition handheld devices that generate an aerosol, improperly named as “vapor,” by heating a solution or e-liquid. The generic e-cig is a battery-powered device that heats a metal coil to atomize the e-liquid drawn by wicks, typically made of cotton or silica, from a cartridge or a refilling reservoir1,2,4. Although e-cigs are diverse in terms of design, technology, and liquid filling system, they all operate similarly. Activation is triggered by pressing a button or by airflow sensor in the mouthpiece so that the generated vapor is disposed by inhalation into the respiratory system, resulting in variable exposures, deposition, and absorption depending on the composition, device and inhalation patterns1,2 (Fig. 1).

Figure 1 Structure of an e-cigarette. E-cigarettes have some basic components as shown, although new generations have changed considerably the shape. Most current models differ considerably from a cigarette. 1: Battery: usually rechargeable, and as other batteries with the possibility of leaks and explosions. 2: Heating coil: amount of vapor depends on temperature, and new devices can modify it. 3: Vaporizing chamber: includes a wick in touch with e-liquid, with different flavors and nicotine content. 4: Mouthpiece.
Composition of the aerosol, particles suspended in gas, is determined by temperature and the substances contained in the heated liquid. E-liquids are solutions that contain vegetable glycerin (VG), propylene glycol (PG), nicotine in variable concentrations, flavoring agents, and other non-nicotine compounds2,4. Up to 7700 different flavoring components in e-cigarettes have been described in the market1.
Although manufacturers have proposed standardization for e-liquid, they offer scarce accurate information about the chemical components in the e-liquid and the e-cigarette aerosol1,6. >80 compounds have been found in e-liquid and aerosols, while PG and VG are the liquid vehicle through which the heating process generates formaldehyde, acetaldehyde, and acrolein by pyrolysis. Other identified substances are nicotine, acetone, benzaldehyde, siloxanes, reactive oxygen species, volatile organic compounds, polycyclic aromatic hydrocarbons, and tobacco-specific nitrosamines (TSNAs), including N-nitrosonornicotine (NNN)2,6,7. Even when the nicotine concentration, as well as that of other components, is listed on the labels of e-cigarette cartridges and refill solutions, large differences have been described between brands, and in some e-liquids labeled as “no nicotine,” a high nicotine content was found. Total TSNAs, NNN, nicotine-derived nitrosamine ketone (NNK), and N'-nitrosoanatabine have also been found with 3 order of magnitude variation between different brands. Nickel-chromium, chromium-aluminum-iron, copper, silver, zinc, tin, or manganese have been described as metal nanoparticles in the e-liquid and vapor as toxins resulting from the device’s components and the heating process. Nickel contained in the e-cig aerosol was 2-100 times higher than in tobacco smoke8.
E-cigarette aerosol is not produced by combustion. Therefore, neither smoke nor carbon monoxide is emitted. Although there is no side steam vapor, in chamber studies and model café environments, a low level of most of the vapor components can be found in the air9. Furthermore, serum cotinine was similar in non-smoking bystanders’ exposure to aerosol and smokes using machine-smoked e-cigarettes and cigarettes10.
E-cig’s aerosol is a source of high exposure of the human respiratory system to fine particles (100-160 nm); their size and concentration are similar to those of tobacco smoke11. Since the composition, inhalation and exposure pattern to e-cig aerosol and tobacco smoke are very different, the health consequences and risks should not be evaluated with the same model.
E-cigs and tobacco cigarettes have similar patterns of particle deposition in the lungs, and nicotine is rapidly absorbed and delivered to the brain. Early pharmacologic reports found that nicotine delivery was significantly slower with e-cigs compared to regular cigarettes, but more recent evidence shows that nicotine levels are influenced by e-cig generation, inhalation patterns, and users’ experience1,12. In some late-generation devices, users can increase the heating temperature and therefore, modify the aerosol composition by increasing up to 2.5-fold the nicotine, formaldehyde, acetaldehyde, and acetone content as well as carbonyl compounds2,7,12,13.
Although the toxins’ concentration of e-cig’s aerosol is significantly lower than that in tobacco cigarette smoke, there is no clinical evidence to support the long-term use so far14-16.
EPIDEMIOLOGY
Epidemiological definition of e-cigarette vapor exposure is challenging since the term user may include from those who use it daily to those who have only used it once or twice4,17-19. Some studies report e-cigarette users as ever, regularly or daily users but in any case, the device, filling system, and/or e-liquid used are considered. The heterogeneity and imprecision of this exposure must be taken into consideration as a source of inaccuracy while appraising vaping as a health risk factor.
E-cigarettes were launched in China, in 2003, but they were commercially available in the marketplace in the United States and Europe since 200720. Based on a 2014 market report on brands (466), sales (3000 M US$) and forecast of sales for 2030 to increase by a factor of 17, the use of e-cigarettes are apparently escalating21.
Estimations of e-cigarette use around the world are questionable due to the definition of prevalence. The most reliable data are from International Tobacco Control (ITC)20. In a survey to current and former smokers older than 18 years from 10 different countries between 2009 and 2013, 34% were aware of the e-cigarette, and 4% had tried it20. The authors observed considerable cross-country variation by year of data collection, for awareness (33-88%), and present use of e-cigarettes (<1-14%).
E-cigarette use among young people is a major public health concern globally since it may lead or progress to conventional smoking of cigarettes22,23. In Europe, e-cigarette use was associated with younger age and heavier tobacco use. Regular e-cigarette prevalence was also associated with the current (20.3%), former (4.7%), and never (1.2%) tobacco smoker condition24. The prevalence of ever, current (at least 1 in the past 30 days), and regular (at least 20 in the past 30 days) e-cigarette use reported in the US, in 2017, was 7.7%, 2.1%, and 0.9%, respectively25. Since 2014, self-report of e-cigarette use among youth exceeded conventional cigarette’s consumption1.
In Mexico, ENCODAT (National Drugs, Alcohol and Tobacco Use Survey, Encuesta Nacional de Consumo de Drogas, Alcohol y Tabaco) revealed that the prevalence of use of e-cigarettes for 2016-2017 was 0.6%, lower in women (0.4%) than in men (0.9%). These figures estimate that 5 million persons ever experimented with e-cigarettes, and almost 1 million are regular users26. In 2015, 10% of secondary school students tried e-cigarettes22. A 2016 survey among teenagers 11 and 16 years old in Mexico City, Monterrey and Guadalajara revealed a prevalence of having ever experienced e-cig of 35% and 31%, and regular use, 14% and 13%, respectively; of them, 5-7% declared having initiated tobacco smoking after experiencing with e-cigarettes27,28.
The characteristics of ever users of e-cigarettes are similar to those reported for conventional cigarettes: male gender, heavier tobacco consumption, family and friends smoking tobacco, and tendency for drug-seeking22,29.
Two waves of the National Adult Tobacco Survey (2012-2013 and 2013-2014) explored characteristics of tobacco quitters and of those who switched to e-cigarettes. Male gender (odds ratio [OR] 1.2; 95% confidence interval [CI], 1.0-1.5) and younger age (18-24 years old; OR 3.6, 95% CI 2.4-5.5) were associated with e-cigarette use before quitting30.
Tobacco and e-cigarette consumers declared to use e-cigarettes as an aid to quit smoking (52.6%), to avoid second-hand tobacco smoke (16.1%), and also as a harm-reduction tool (8%)19,22.
Regulation of ENDS varies significantly between countries, and to date, there is no consensus among the ITC community20. While in the UK there are few restrictions and ENDS is widely available, the US Food and Drug Administration (FDA) proposed regulations similar to those for traditional tobacco products, and FCTC’s COP recommended objective-based regulations (prevent initiation in non-smokers and youth, reduce ENDS’ health risks and second-hand emissions, prevent unproven health claims, protect tobacco control activities, avoid commercial interests, in particular, and of tobacco industry)31.
THE HEALTH RISK DILEMMA OF E-CIGARETTES
Since Doll’s report, in 1954, uncontroversial evidence has been growing regarding the burden of health consequences of smoking tobacco. However, a global health response consistent with the magnitude of the challenge was launched only half a century later32. Along the XXI century, the world will still be facing hundreds of millions of deaths as a toll of nicotine addiction and tobacco industry revenues. The tobacco documents released in 1998 as a response to a legal action revealed the ignominious and systematic efforts of these cartels to prevent the decline of their trade33. In fact, one of the strategies of tobacco manufacturers in the 60s was to launch light and low-tar cigarettes as less harmful smoking. Subsequent studies not only failed to demonstrate the health benefits of smoking light and low-tar cigarettes versus standard cigarettes but also they observed an increase in the risk as well1,34.
Faced with the categorical evidence of the burden of tobacco deaths, the tobacco industry developed a broad and comprehensive strategy to preserve and expand their market. Creating doubt and controversy around the health risks of tobacco, and promoting falsely safer smoking products were cornerstones of this strategy. During the 70 s and 80 s, filtered or “light” and “low” tar or “low” nicotine cigarettes created among smokers a wrong impression of being less harmful than others, which may have curbed their motivation to quit smoking. The WHO Framework Convention on Tobacco Control Art 11 addressed and promoted parties to regulate tobacco product packaging and labeling to avoid by any means false, misleading, and deceptive messages32.
E-cigarette is now reloading the dilemma of low-risk nicotine consumption through the respiratory system. The cornerstone argument of e-cigarette supporters is that the vapor’s concentration of toxic substances is significantly lower than that of tobacco smoke35-37.
By modeling analysis, a selected number of drug experts estimated the harm of nicotine products, concluding that e-cigarette’s relative harm is 5% compared to cigarettes. Some limitations to this argument should also be addressed apart from the conflict of interest warned by journals’ editors. First, there is a clear inductive fallacy in the paper by Nutt et al., since harm reduction was estimated based on the authors’ opinion while they recognized the lack of hard evidence for the harm of most of the nicotine products35. Second, the process for recruiting experts was not specified, resulting in potentially serious selection bias. Finally, asking the experts to assign a scale value based on their subjective perception of harm for each product, and the difference in damage between the most and least harmful products in 16 dimensions, leads to several criticisms, as has been observed38.
This paper was the pillar for the Public Health England e-cigarette report, translated into two questionable statements: “e-cigs are 95% safer than smoking” and “tobacco health burden can be reduced by 95% if all smokers move to e-cigarettes”39.
MECHANISMS OF DAMAGE OF E-CIGARETTES
There is no doubt that the concentration of ingredients in e-cigarettes’ aerosol is significantly lower than that of combustible cigarettes, but this does not mean that it is “harmless vapor.” As described, e-cigarettes are a novel source of high concentrations of submicron-sized particles, and users are subjected to an unusual and original model of inhalation exposure since the inhalation pattern of e-cigarettes in terms of length, volume, and puffing frequency is different from that of conventional cigarettes. The aerosol generated “in vitro” has particle sizes in the range of 100-600 nm, similar to the conventional cigarettes. Other studies described a bimodal particle size distribution: 11-25 nm and p 96-175 nm11. One report estimated that 6.25 × 1010 particles generated would be deposited in the respiratory system while most particle depositions would occur in the alveolar region2. Ultrafine particles, <100 nm, were described to cause DNA damage, pro-inflammatory cytokine expression and production of free oxygen radicals11.
Among the aerosol’s contents described above, potent carcinogens such as NNN and NNK were identified in the vapors generated from a different brand of e-cigarettes40. TSNAs are related to curing and processing tobacco, or possibly with the addition of tobacco flavorings1,41. Formaldehyde, acetaldehyde, and acrolein are well-known potent irritants and toxics and have been found exceeding the National Institute of Occupational Safety and Health recommended level for short-term exposure6. There is also growing concern for the presence of heavy metals in e-liquids as potential carcinogens42.
Flavoring components of e-cigarettes are major contributors of carbonyl species production2. In one report, diacetyl (DA, the most well-known artificial flavoring) and acetyl propionyl were found in 28.3% of a sample of e-liquids. These substances were associated with bronchiolitis obliterans that cause fixed obstructive lung disease1. Nicotine concentrations in the e-cigarette vapor vary more widely than in conventional cigarette’s smoke1. Most e-liquids, even those labeled as “nicotine free,” contain nicotine and 60-70% of the nicotine is the one released by the aerosol9,13. E-liquid labels report the nicotine concentration, but this is inaccurate. Goniewicz et al. estimated an average of 82.8 mg of nicotine per 100 mL of aerosol in an 18-mg nicotine cartridge, and in those e-cigarettes in which 15 puffs are equivalent to smoking one cigarette41. A typical cigarette delivers approximately 2 mg of nicotine to its smoker, and the lethal dose LD50 is 60 mg2. The evidence supports that nicotine availability in e-cigarettes is effective to saturate brain receptors and to avoid abstinence symptoms43,44.
The systemic and airways’ changes observed after exposure to e-cigarette aerosol may be explained by different mechanisms, some of which are still under debate and research:
Aldehydes (formaldehyde and acrolein) exposure has been associated with altered epithelial response, mucus hypersecretion, activation, and degranulation of neutrophils and induction of neutrophil apoptosis12.
Exposures to e-cig aerosol induce measurable oxidative and inflammatory responses in lung cells and tissues, and in bronchial epithelial cells cause acute toxicity and reduce the antiviral response45,46. E-cigarette users show increased proteins secretion in sputum related to the innate defense functions of leukocytes, bronchial inflammation, and structural damage. These include neutrophil elastase, proteinase 3, azurocidin 1, and myeoloperoxidase as well as other secondary neutrophil granule proteins12.
Exposure to e-cigarette induces platelet aggregation and upregulates expression of CD41, CD42b, and CD62p, independently of nicotine content and exposure time possibly due to fine particulate matter. These facts may be the hallmarks of cardiovascular and other systemic diseases47.
E-cigarettes have been linked to lung and systemic damage (Table 1), with consistent evidence and biologic plausibility that the constituents of e-cigarettes’ aerosol cause airway irritation, bronchitis, cough, phlegm, bronchoconstriction, platelet dysfunction, and carcinogenic changes among others (Fig. 2).
Table 1 Components of e-cigarettes and potential damage.
| Components | Potential damage | |
|---|---|---|
| Metal material | Batteries and heating element: nichrome wire (80% nickel, 20% chrome), kanthal, iron, chromium, aluminum, ceramic, silica. | Carcinogen, respiratory, and reproductive toxicant; respiratory disease and autoimmune dysfunction. |
| E-liquids | Nicotine | Concentrations range from 0 to 50 mg/mL. |
| Impair antibacterial defense, alter macrophage activation. | ||
| Pyrolysis of blanks: one or mixture solvent (PG or VG) | Mixture PG/VG produced more ROS than either alone resulting in inflammation, cytotoxicity, and increased endothelial cell permeability. | |
| Flavorings (tobacco, menthol, candy, beverage themed) | “Primary irritants” | |
| Cinnamon increases cytokine IL-8 | ||
| Diacetyl: bronchiolitis obliterans | ||
| Not all have been tested for safety when inhaled. | ||
| Aerosol | TSNAs, NNN | Potent carcinogens |
| Acrolein | Increase risk of lung cancer, asthma, COPD. | |
| Glycidol | Probable carcinogen | |
| Formaldehyde | Epithelial response and increasing mucin secretion | |
| VOCs | Irritation, headaches, organ damage | |
| PAHs | Carcinogens |
PG: propylene glycol, VG: vegetable glycerin, ROS: reactive oxygen species, TSNAs: tobacco-specific nitrosamines, NNN: N-nitrosonornicotine, COPD: chronic obstructive pulmonary disease, VOCs: volatile organic compounds, PAHs: polycyclic aromatic hydrocarbons.
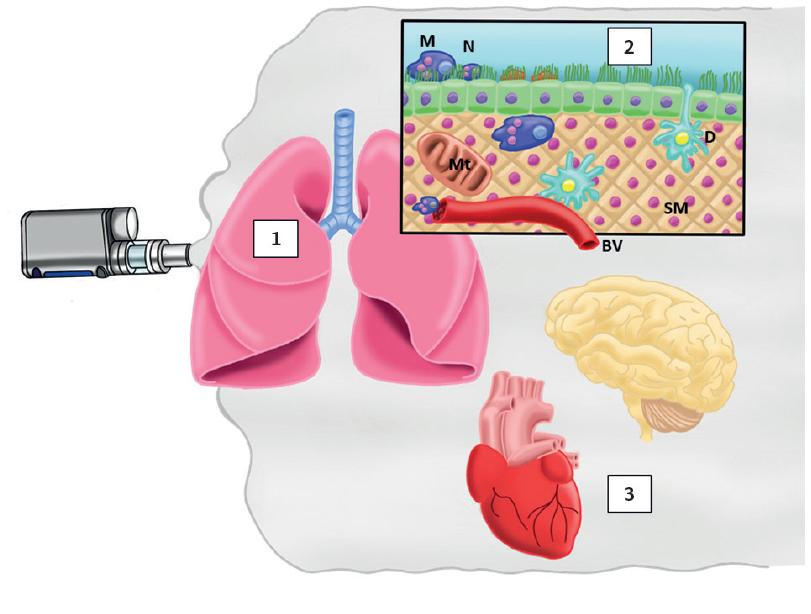
Figure 2 Probable mechanisms of lung abnormalities with e-cigarette. The combination between high temperature and several components of e-aerosol including particulate matter (1) has the potential of generating pro-inflammatory effects, production of reactive oxygen species in lung with varied consequences such as airway hyperreactivity, increased airway resistance, decreased antimicrobial activity, decreased alveolar development, endothelial disruption, and fragmentation of fibroblasts (2). Systemic changes are produced by nicotine with increases in heart rate and blood pressure, as well as an addiction with all consequences including tolerance, craving, abstinence, and change to normal cigarettes or inhalation of other liquids containing other drugs (3).
IN VITRO DAMAGE BY E-CIGARETTES AND EVIDENCE IN ANIMAL MODELS
There is emerging evidence of possible pathways and toxicities of e-cig aerosols, but there are still many important gaps to understand fully the burden of harms from this distinctive and varied exposure2. In vitro exposure of human bronchial cells to e-cigarettes’ carrier solution containing PG and/or VG decreased cell metabolic activity, bronchial cell viability and cilia activity, and increased mitochondrial oxidative stress (consistent with reduced glutathione levels)2,13. Some reports suggest that artificial flavoring and nicotine concentrations may influence these dysfunctions45,48-50. In vivo, short-term exposure to e-cigarette aerosols in mice models reproduced similar changes resulting in oxidative stress, macrophage-mediated inflammation, and lung injury with increased capillary permeability45,51. Furthermore, exposure to e-cigarette aerosol in mice showed impaired bacterial and influenza A clearance, increase in lung viral titers, and deleterious effects in morbidity and mortality51. Other report highlighted the increase of bacterial load, especially of methicillin-resistant Staphylococcus aureus, pneumococcal adhesion to upper and lower airway epithelial cells, and increased macrophages in bronchoalveolar lavage samples, but with impaired bacterial phagocytosis and increased alveolar-capillary permeability2,52.
One report explored long-term exposure to e-cigarette aerosol containing nicotine in a mouse model and human bronchial epithelial cells and revealed changes as a causative factor of progression of chronic obstructive pulmonary disease (COPD), including cytokine expression, airway hyperreactivity, and lung tissue destruction53.
SYSTEMIC IMPACT OF E-CIGARETTES IN HUMANS
Nicotine induces stimulation of the sympathetic nervous system, with tachycardia, increase in blood pressure and cardiac output, leading to an increase in myocardial oxygen consumption as well as vasoconstriction of cutaneous and coronary blood vessels54. Increased cardiac sympathetic activity associated to regular smoking was described 20 years ago and, lately, similar effects were observed for e-cigarette as well as oxidative stress and inflammation signaling55-57. Recently, evidence of population-based e-cigarette impact on cardiovascular health has been reported56,57.
There is considerable evidence that carcinogenic substances and some compounds causing DNA damage and mutagenesis have been detected in e-cigarette aerosols, but there is no available evidence so far that e-cigarette use is associated with cancer, abnormal fetal development, or immune defects leading to increased risk of respiratory infections18. However, the period of observation since the massive launch of e-cigarettes is too short to analyze the occurrence of cancer. On the other hand, there is usually a time gap between the description of toxicologic impacts on in vitro or in vivo models and the description of clinical or epidemiologic outcomes. The diversity of the devices and exposures is also barriers to categorize the exposure18.
Other reported risks associated with e-cigarette use include device explosions, accidental, and intentional poisoning with PG and nicotine overdose in children, increased circulating endothelial progenitor cells, and potential acute endothelial injury, and nickel contact dermatitis2,18.
EVIDENCE OF PULMONARY DAMAGE ASSOCIATED WITH E-CIGARETTE EXPOSURE IN HUMANS
While evidence of median and long-term adverse effects of e-cigarette exposure on the respiratory system emerges, it is desirable to review and monitor intermediate outcomes, such as alterations in lung structure and function and respiratory symptoms18. Most of the information comes from dual users (smoking tobacco and e-cigs) or individuals switching completely from conventional tobacco smoking to e-cigarettes, thus avoiding a valid comparison of users of e-cigs versus unexposed individuals2,58,59. Studies in healthy e-cigarette users revealed an increase in oxidative stress, nitric oxide deficiency, and endothelial/vascular dysfunction; acute and short-time exposure to PG in aerosol from artificial smoke generators resulted in ocular and respiratory symptoms and deleterious lung function in healthy non-asthmatic patients, but this evidence, in contrast with the effect of tobacco smoke, is considered marginal13,60,61.
Exposure to e-cigarette aerosol has been associated with respiratory symptoms in healthy individuals, changes in respiratory physiology and host defense, and with increased symptoms in asthma, cystic fibrosis (CF), and COPD2.
There is growing evidence that adolescents who were exposed to e-cigarettes more often have cough and phlegm (OR 2.1, 95% CI 1.8-2.5)62,63. Adolescents using e-cigarettes more frequently report not only respiratory symptoms but also school absenteeism18,64. Airway exposure to nicotine-containing e-cigarette vapor inhibits bronchial and nasal mucociliary clearance, with the production of a cough and rhino-nasal symptoms, compared with individuals unexposed to e-cigarette aerosol18,65.
Short-term exposure to e-cigarette with and without nicotine in healthy adults increases airway resistance and reduces nitric oxide in exhaled air (fractional exhaled nitric oxide, FeNO)1,18,66. This evidence is consistent with a report that revealed decreased pulmonary function (decrease in forced expiratory volume in one second [FEV1], FEV1/forced vital capacity; and increase in airflow resistance) after exposure to aerosolized PG in healthy humans2,10,66. So far, there is neither evidence supporting long-term safety nor improvements in lung function in smokers who switch to e-cigarettes, as is observed in quitters67.
Recent evidence reports parenchymal and bronchial inflammation, lung damage and toxicity (e.g., lipoid pneumonia), as well as impaired systemic inflammation signaling, and defense mechanisms associated with e-cigarette exposure12,58.
E-CIGARETTE AND MORE COMMON LUNG DISEASES
A recent report revealed that the prevalence of ever use of e-cigarettes was higher among adults with one or more comorbidities, compared with those without comorbidities, and in smokers with COPD, asthma, and cardiovascular disease compared to “healthy” smokers68. Use of e-cigarette was associated with higher odds of asthma symptoms, considering cigarette smoking and marijuana use69. Adolescents with asthma or CF who use e-cigarettes were more likely to have respiratory symptoms and exacerbations18,64. Common flavoring agents in e-cigarettes are recognized as “primary irritants” of mucosal tissue of the respiratory tract; airway irritants and sensitizers have been reported to cause occupational asthma64.
In patients with COPD, the use of e-cigarettes has been associated with more cough and phlegm, more exacerbations and possibly a more rapid decline in lung function, even after adjusting for tobacco smoking and age70.
E-cigarettes have been shown to lower indoor air quality, and non-smokers have been found to absorb nicotine from second-hand vaping comparable to passive smoking2,9. Although passive exposure to e-cigarette vaping does not include combustion of toxic agents, there is significant secondhand exposure to nicotine in the exposure chamber, leading to serum cotinine levels among those exposed that can be higher than levels obtained from conventional cigarettes9.
SMOKING CESSATION WITH E-CIGARETTES
Two Cochrane reviews had explored in 2014 and 2016 the effectiveness of e-cigarettes for smoking cessation and safety, and both agreed on the low quality of the evidence based only on a small number of studies; therefore, further research was suggested71-75. The latter meta-analysis revealed that abstinence was higher among e-cigarette users compared with placebo, but no effect was observed comparing e-cigarettes with nicotine patches. Despite the scarce information, supporters of e-cigarettes promote them as a better option to smoking or to reduce cigarette consumption in a harm-reduction strategy, while skeptic tobacco-control actors alert of a boost in the nicotine market by attracting youths that may later start smoking regular cigarettes73,76,77.
In fact, a major public health concern is e-cigarettes arising as a novel, high-tech pathway to nicotine addiction through early experimentation by children and adolescents, since e-cigarettes saturate the nicotinic receptors in the brain as much as conventional cigarettes do28,69,78.
Recent “aggressive” and restriction-free e-cigarette advertisements in internet sites, social media and movies resulted in a growing awareness, perception of safer behavior than smoking, and increased appeal and interest to try e-cigarettes79,80. Data from the US, Canada, United Kingdom, and Australia obtained in the ITC project revealed a higher prevalence of e-cigarette use among young and non-daily smokers, as a consequence of feeling that vaping was safe. However, teens and young adults from the US who have used e-cigarettes were in a significantly higher risk of smoking regular cigarettes in the following years (OR 3.87; 95% CI, 1.86-8.06), a risk also found in a new longitudinal study in Great Britain (OR 1.34; 95% CI, 1.05-1.72; p=0.018)29,78,81. A recent meta-analysis confirmed that e-cigarette experimentation among never smokers almost quadrupled the chances of becoming a smoker at follow-up82.
Flavoring or taste in e-cigarette not only stimulated experimentation and initiation behaviors but also increased the risk of smoking combustible cigarettes83,84. These facts may partially explain the dramatic increase in e-cigarette use among youths observed in the US, UK, Italy, Poland, South Korea and Finland, and among other countries.
A key public health challenge is e-cigarette initiation in a never-smoker youth, as it may not only cause harm to the respiratory system but also lead to smoke regular cigarettes and vape other substances, like cannabis derivatives.
The awareness of e-cigarettes’ potential health risk, their regulation as tobacco products, and the promotion of banning must be considered among other actions to prevent the evolving scenario3,18. In the US, FDA e-cigarette policies resulted in a significant decline of e-cigarette use among young persons, in 201685.
In established smokers, an e-cigarette may be less deleterious to the respiratory system, but so far there is insufficient evidence of whether e-cigarettes increase the likelihood of smoking cessation19. Furthermore, existing reports fail to compare e-cigarettes with state-of-the-art proven therapies.
CONCLUSION
For >50 years, a solid body of incontrovertible evidence revealed that smoking cigarettes are a cause of disease in almost every organ of the body1. In the early 60s, it was a challenge to prove the real burden of tobacco on global health, as the evidence required decades to emerge entirely; in the meantime, millions of deaths occurred. Now, e-cigarettes emerged, spread promptly, and challenge the scientific, and public health community, since they have to act to control and regulate the market based on public health, even with incomplete proofs and evidence, which are needed without delay7,86.
ENDS, and particularly e-cigarettes, are devices that effectively deposit nicotine in the brain and generate nicotine addiction. These e-cigarette aerosols contain fewer toxins than tobacco smoke, but a safety comparator must be clean air breathing. There is growing evidence of the presence of a variety of toxic products in vaporizing liquids in e-cigarettes that result in chemical, morphologic, and functional deleterious effects in in vitro and in vivo models12,47. Evidence for acute respiratory damage and toxicity is evolving, but data on mid- and long-term effects are still lacking as well as standardization models to compare different devices.
It is urgent to regulate e-cigarette design, e-liquid and aerosol composition, health warnings, marketing, promotion, sales, taxation, and secondhand vapor exposure at least at a level equivalent to that of regular tobacco products22,78.











 nueva página del texto (beta)
nueva página del texto (beta)


