Services on Demand
Journal
Article
Indicators
Related links
Share
Revista de investigación clínica
On-line version ISSN 2564-8896Print version ISSN 0034-8376
Rev. invest. clín. vol.59 n.1 Ciudad de México Jan./Feb. 2007
Artículo original
Association between polymorphisms of the androgen and vitamin D receptor genes with prostate cancer risk in a Mexican population
Asociación entre polimorfismo de andrógenos y receptores de vitamina D con riesgo de cáncer prostático en una población mexicana
Brenda Patino–García,*, ,
, Carlos Arroyo,*,
Carlos Arroyo,*, ,
, Hector Rangel–Villalobos,
Hector Rangel–Villalobos, Elena Soto–Vega,
Elena Soto–Vega, Jose Salvador Velarde–Félix,*,
Jose Salvador Velarde–Félix,*, Fernando Gabilondo,** Lucila Sandoval–Ramírez,* Luis Eduardo Figuera*,
Fernando Gabilondo,** Lucila Sandoval–Ramírez,* Luis Eduardo Figuera*,![]()
* División de Genética, Centro de Investigación Biomedica de Occidente–Instituto Mexicano del Seguro Social, Guadalajara.
 Programa de Doctorado en Genética Humana, Centro Universitario de Ciencias de la Salud–Universidad de Guadalajara.
Programa de Doctorado en Genética Humana, Centro Universitario de Ciencias de la Salud–Universidad de Guadalajara.
* Departamento de Cirugía, Hospital Universitario de Puebla, Benemérita Universidad Autónoma de Puebla.
 Laboratorio de Genética Molecular, Centro Universitario de la Ciénega (UdeG).
Laboratorio de Genética Molecular, Centro Universitario de la Ciénega (UdeG).
 Unidad de Enfermedades Autoinmunes y Sistémicas, Hospital Regional No. 36, Instituto Mexicano del Seguro Social de Puebla.
Unidad de Enfermedades Autoinmunes y Sistémicas, Hospital Regional No. 36, Instituto Mexicano del Seguro Social de Puebla.
 Centro de Medicina Genómica, Servicios de Salud de Sinaloa.
Centro de Medicina Genómica, Servicios de Salud de Sinaloa.
** Departamento de Urología, Instituto Nacional de Ciencias Médicas y Nutrición Salvador Zubirán.
 Both authors participated equally in the work and writing of this paper.
Both authors participated equally in the work and writing of this paper.
Correspondence and reprint request:
Carlos Arroyo, PhD
Department of Surgery Hospital Universitario de Puebla,
Benemérita Universidad Autónoma de Puebla
25 Poniente y 13 Sur, Col. Volcanes, Puebla, Pue.
Phone and fax: 52 222 243–1542
E–mail: jcakl@yahoo.com
Recibido el 6 de enero de 2006.
Aceptado el 15 de noviembre de 2006.
ABSTRACT
Introduction. Prostate cancer (PCa) is a worldwide health issue, because of its high incidence and mortality. Its etiology is complex and includes certain risk factors such as age, hormonal status, ethnic origin and family history of PCa. Genetic predisposition is proposed as a major risk factor and there are several controversial reports on the association of PCa and gene polymorphism such as the receptors of the androgen receptor (AR) and the vitamin D (VDR).
Objective. To evaluate the CAG triplets repetitions in the first exon of the AR and polymorphisms in the restriction site Taql in the VDR in Mexicans with PCa.
Material and methods. A total of 68 Mexicans with histopathological diagnosis of PCa and 48 healthy Mexican with normal prostate specific antigen and rectal exam where included. 10ml of peripheral blood were extracted to isolate DNA and the polymorphisms were evaluated with specific primers for the AR and VDR.
Results. The allelic and genetic distributions of the AR and VDR polymorphisms were consistent with the Hardy–Weinberg equilibrium, and there were no statistical differences between the PCa patients and controls (p > 0.05). However, there was a statistical difference between the number of CAG repeats in younger patients with PCa compared to controls (p = 0.045) but when the young patient group was compared versus the elder group there was not stadistically difference (p = 0.085), but the results showed a tendency towards less repetitions of CAG in elder patients. Concerning the VDR, when we analyzed the patients with PCa and a bad pathological prognosis they had a less frequent genotype of TT (p = 0.03).
Conclusions. Our results suggest an association between the VDR and AR gene polymorphisms, and the hystopathological score and age at diagnosis in Mexican patients with PCa, respectively. However, it is important to confirm these results in a larger scale study.
Key words. Prostate cancer risk. Androgen receptor. Vitamin D receptor. Prostate cancer genetics.
RESUMEN
Introducción. El cáncer de próstata (PCa) es un problema de salud mundial, tanto por su elevada incidencia como mortalidad. Su etiología es compleja e incluye factores de riesgo reconocidos como la edad, estado hormonal, origen étnico y antecedentes familiares de PCa. El fondo genético es un factor de riesgo y existen reportes controversiales de la asociación de PCa y polimorfismos en los genes como son los receptores de vitamina D (VDR) y el de andrógenos (AR).
Objetivo. Evaluar las repeticiones de tripletes de CAG en el primer exon del AR y polimorfismos en el sitio de restricción Taql en el VDR en mexicanos con PCa.
Material y métodos. Se incluyeron 68 mexicanos con diagnóstico histopatológico de PCa y 48 mexicanos con niveles normales de antígeno prostático y tacto rectal normal. Se les extrajo 10 mL de sangre periférica para aislar DNA y mediante olígos específicos se evaluaron los polimorfismos mencionados.
Resultados. La distribución alélica y genotípica de los polimorfismos en el AR y VDR fueron consistentes con el equilibrio de Hardy–Weinberg, y no mostraron diferencias significativas entre los casos y controles (p > 0.05). Sin embargo, el número de repeticiones de CAG en el AR fueron estadísticamente diferentes en pacientes jóvenes con PCa comparados con los controles (p = 0.045), cuando se comparó el grupo de pacientes de jóvenes contra aquellos mayores de 60 años no se encontró diferencia estadísticamente significativa (p – 0.085); sin embargo, se observó una tendencia de un número menor de repetidos CAG en pacientes mayores con PCa. Por otra parte, al comparar VDR en los pacientes con PCa de mal pronóstico por el patrón histológico tenían menor frecuencia de genotipos TT (p – 0.03).
Conclusiones. Nuestros resultados sugieren una asociación entre los polimorfismos de los genes del VDR y AR, y el patrón histológico y la edad al diagnóstico en pacientes mexicanos con PCa, respectivamente. Sin embargo, es necesario confirmar estos resultados en un estudio con mayor número de pacientes.
Palabras clave. Riesgo de cáncer de próstata. Receptores andrógenos. Receptor de vitamina D. Genética de cáncer de próstata.
INTRODUCTION
Prostate cancer (PCa) is the most common cancer diagnosed in older men, and it is the first cause of cancer death in men older than 50, with a rate of 25.5 per 100,000 Mexicans in 2001. In Mexico, PCa represents 16.6% of all cancers in men, being the third cause of cancer in men older than 40 and the first tumor in men over 50. In 2001 the PCa incidence was of 5,911 and caused 4,030 deaths.1 Despite its high morbidity, the etiology of PCa remains largely unknown. The only established risk factors are age, race and a family history of PCa. Some studies have implicated other risk factors like androgens, diet, physical activity, sexual activity, inflammation and obesity, but it is not clear what are their roles in PCa etiology. Some researchers have proposed that about 42 % of the PCa risk is due to genetic causes.2 Taken alone, the different susceptibility genes present low absolute risk; however, there are some genes that have been suggested to be involved in the incidence and progression of sporadic PCa. Among them are the androgen receptor (AR) and vitamin D receptor (VDR) genes3,4 but the exact mechanisms by which these hormones affect this process are unknown.
The AR gene is located on the chromosome X, its N–terminal domain has a transactivation function codified in the exon 1, which contains two polymorphic repeats: CAG and GGC at the 5' and 3' portion, which codify for polyglutamine and polyglycine, respectively.5 The length of the CAG repeats varies among individuals, and it is believed that these polymorphisms are related to the transcriptional activity of the AR gene. The CAG repeat has a normal length range between 8 and 31 units, with an average of 20.6 There are several studies that have revealed an association between the shorter alíeles with a more active AR and this alíele is also predominant in the population at higher risk for PCa, as well as a more aggressive pattern of Pea.7 However there are other reports that have not found this association.8,9
The VDR is a member of the steroid/thyroid hormone nuclear receptor family, and plays an important role in the proliferation and differentiation of normal and malignant cells.10 In several studies polymorphism in the VDR gene has been reported to be associated with PCa. The hormone activity requires binding to the VDR, which contains several genetic polymorphisms that have been associated with the risk of PCa. The VDR gene consists of nine exons and has several polymorphisms in intron 8 and exon 9. There are several restriction fragment length polymorphisms that have been studied in the VDR gene which include: TaqI, BsmI, Apal, poly(A), and Fokl.11 Recent studies have shown that homozygous patients for a less active VDR form have 4 to 5 times increased risk of localized Pea.12 Among the identified markers, the TaqI polymorphism in exon 913 is one of the most promising prospects, however, in pooled data on current reports of the VDR gene polymorphisms and risk of PCa the results are still inconclusive.14 In this paper we studied the influence of the AR and VDR polymorphisms in sporadic PCa Mexican patients and healthy Mexican controls.
MATERIALS AND METHODS
Subjects
We included a total of 68 Mexican patients (born in Mexico from Mexican parents and grand parents) with hystophatological diagnosis of PCa by transrectal biopsy, with no family history of PCa. The control group consisted of 48 healthy Mexican men older than 65, with no previous history of prostate pathology at screening, with normal serum prostate–specific antigen (less than 4 ng/mL) and normal digital rectal examination. In case of doubt an ultrasound–guided biopsy was performed with no evidence of PCa in any of the twelve cores. All the patients and controls gave their informed consent to participate in this study, according to the protocol previously approved by the Ethics Committee of the IMSS Biomedical Research Center.
Genetic analysis
DNA isolation. Genomic DNA was extracted from 10 mL of peripheral blood by the methods of phenol/ chloroform.
The study of CAG repeat polymorphism in exon 1 of the AR gene was genotyped as described by Correa, et al.9 Reaction mixtures containing 50 ng of genomic DNA, 10 pmol of each oligonucleotide primer (5'–TCC AGA ATC TGT TCC AGA GCG TGC–3' and 5'–CTC TAC GAT GGG CTT GGG GAG AAC–3'), and 0.5 unit of Taq polymerase. PCR conditions consisted of 27 cycles of 95 °C for 45 seconds, 70 °C for 30 seconds, and 72 °C for 30 seconds. The initial denaturation and final elongation steps were lengthened to 3 min. The PCR product obtained were analyzed on a 6% polyacrylamide gel. The size of the amplified AR was 234 bp.
For the VDR gene Taql polimorphism, a 745 bp DNA fragment was isolated using the forward primer in the intron 8, 5'–CAG AGC ATG GAC AGG GAG CAA –3' and the reverse primer, 5'–CAC TTC GAG CAC AAG GGG CGT TAG –3' in exon 9. Thirty–two cycles of PCR were performed; each cycle consisted of 94°C for 30 seconds, 67 °C for 30 seconds, and 72 °C for 30 seconds. The initial denaturation and final extension were carried out for 7 min. The amplified product was digested with 0.2 /xL of the enzyme Taql for 90 min at 65°C and electrophoresed on a 2 % agarose gel stained with ethidium bromide, and visualized under UV light. The VDR alíeles were named as TT, Tt and tt, where uppercase and lowercase letters represent the absence and presence of restriction sites. The size of the amplified VDR was 740 bp, when the T alíele was present there was restriction and the products sizes were of 495 bp and 245 bp; compared to the t alíele with the restriction site present with products sizes of 290 bp and 205 bp.
Statistical methods
The agreement of genotype distribution to the Hardy–Weinberg expectations was checked by exact tests. Alíele frequencies were estimated by gene counting method. Genetic differentiation between groups and controls was established by pairwise comparisons by exact tests. Odds ratios (ORs) were computed for specific alíeles. T student test was also applied to compare the AR CAG repeats. Patients were subdivided into three prognosis groups according to their histopathological Gleason score, and also according to their age at diagnosis, into less than 60 and older than 60 year old.
RESULTS
Cases were slightly younger (62.93 ± 17.8 years old) compared with control (69.92 ± 10.17 years old) but these were not statistically different.The observed and expected genotype frequencies for the AR and VDR polymorphisms were consistent with those predicted by the Hardy–Weinberg equilibrium.
The average AR CAG repeats was 8.79 ± 2.65 for the PCa group and 8.28 ± 2.34 for the control group, when the t Student was applied, it did not reveal any statistical difference (p > 0.05). Neither was there any statistical difference when PCa patients were compared according to their histopatological Gleason score, with similar mean values between groups (7.12 ± 2.57 for the good, 8.46 ± 2 for the moderate, and 8.43 ± 3.11 for the worse pathology pattern).
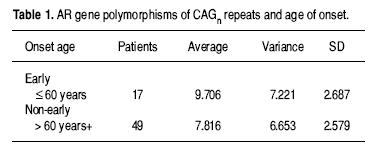
The PCa patients were also grouped depending on their age at diagnosis (less than 60 and older) making them comparable to the control group that is older than 60 years old. When we compared the number of AR CAG repeats between the younger age at diagnosis of PCa and controls, there was a statistical difference (p = 0.045 IC 95% –3.46 to –0.0.4). There was not statistical difference between PCa groups, younger than 60 compared to the older than 60 (p = 0.085 IC 95% –3.53 to –0.54), however this results only showed a tendency to a lower average of CAG repeats in the elder PCa patients at diagnosis (U–Mann–Whitney test, p = 0.060) (Table 1). Finally, we carried out a Spearman correlation test for the number of CAG repeats with the age at diagnosis and the pathology prognosis in PCa patients, with no statistical difference. However, these results must be considered cautiously because of the small sample size studied (Table 2).
The VDR alíeles showed no statistical differences among the PCa group and controls. When PCa patients were grouped according to their histopathology Gleason score, controls and PCa patients with good and moderate pathological pattern had a similar distribution for the VDR gene polymorphism (p > 0.05). However, PCa patients with poor prognosis presented a lower frequency of TT genotypes, compared to controls, in both allelic frequencies (p = 0.0301; OR 2.25, IC 95% 1.0719–4.7294) and genotype distribution (p = 0.0377; OR 0.364 IC 95% 0.1027–1.048). These results indicated that it was 1.75 times more probable to find genotypes Tt and tt in PCa patients with poor prognosis than in controls. On the other hand, PCa patients were compared according to their pathological prognosis: the patients with poor prognosis showed a diminished frequency of the TT genotype (p = 0.03, OR 3.37, IC 95% 1.06–10.71) associated with an increase on the Tt genotype (p = 0.05, OR 0.32, IC 95% 0.10–1.04) compared to the moderated prognosis group (Table 2). The small sample of PCa patients with good histopathological prognosis limited the statistical power of the tests (n = 8). A greater sample size will be needed in order to confirm these associations.
DISCUSSION
PCa is a worldwide health issue, not only because its high incidence and mortality, but also because up to date there is no complete knowledge of its cause and risk factors. As it is well known, PCa is multifactorial, in which there is an interaction of genetic susceptibility and potentially co–dependent variables such as diet, environmental exposure, age and DNA sequence polymorphisms that are responsible for the risk of Pea.15 In this paper we evaluated two polimorphisms in two receptor genes that might be associated with PCa in a Mexican population, which to our knowledge is the first study in Mexico with the benefit of a population of a specific race.
The studies of genetic–association are of great value in understanding the pathway of disease; this is why the AR gene has been studied extensively due to the hormonal dependency of PCa and its involvement in androgen regulation, metabolism and their related pathways. It has been shown that there is an inverse relationship between CAG length and AR transactivation function, which translates into the shorter the CAG alíeles are better in activating gene transcription.16 Coetzee and Ross17 hypothesized that the racial differences seen in PCa susceptibility may be due in part to variability in the AR CAG repeat length.6 However, because the association between AR repeat length polymorphisms and PCa is small, other factors also must be involved.
Our results were only marginally suggestive for an association of the CAG repeat (treated as a continuous variable) and PCa risk, with a maximal effect when the size is smaller than previously suggested. Irvine et al.18 reported that increased CAG length inhibited both basal and coactivator–mediated AR transactivation activity in cultured prostate epithelial cells that were cotransfected with AR constructs of up to 42 repeats and pl60 coactivator expression vectors. This could explain how a decreased repeat length may make the prostate more vulnerable to chronic androgen stimulation, leading to increased proliferative activity, which may enhance the rate of somatic mutations.19 On one hand, it has also been shown that the AR repeat polymorphisms is associated with the incidence of other androgen–related clinical conditions; specifically, a high number of repeats seem to adversely influence fertility and spermatogenesis20 and bone density.21 On the other hand, fewer CAG repeats are associated with increased risk of baldness22 and benign prostatic hyperplasia,23–26 although this has not always been confirmed.27,28 Concerning PCa, there are contradictions between the different papers in the literature, some authors report an inversely proportional relation between the number of CAG repeats (alíele length) and risk of pca29,30 aggregivity,31 and risk variations according to the smoking status.32 Other researchers have not found this association between CAG length and an increase in PCa risk. This is the case of our results, in which we found a high frequency in the distribution alíeles 7, 9, and 10, which did not represent any significant differences between the two groups (p = 0.913), in brief, we did not find an influence of the AR alíeles and PCa risk in Mexicans, which complies with previous reports of different populations.
Concerning the race differences, Balic et al, found that the prevalence of shorter CAG repeat was lower in Hispanic men compared to other white men, and that these short CAG repeats are associated with an increased PCa risk in Hispanic men.33 However, their results do not correlate with ours; this could be partly explained because the Hispanic population, however does share a common a Spanish background, that has a great variability accounted by different native Amerindian populations, which result in a group relatively heterogeneous. Because of the characteristics and number of our patients, these differences could have not been detected.
Finally, although the number of CAG repeats has been associated to an earlier age (< 60 years old) at diagnosis,8 we identified a relationship between a greater number of CAG repeats and an earlier age at diagnosis (p = 0.0123), which does not agree with previous reports. This variation could be explained by a high ethnic variability that was explained earlier.
Regarding Dihidroxyvitamin D or calcitrol, it is well known its primary effect on bone and calcium homeostasis. However, it has also been shown that calcitrol acts via the VDR, a nuclear transcription factor that belongs to the steroid hormone receptor family, which is involved in the regulation of in vitro cell proliferation and differentiation.34 The VDR, which dimerizes with the retinoid X receptor, binds to la,25 (OH)D3 promoter sites and regulates the transcription of many genes in more than 30 different tissues, including the prostate, thereby emphasizing its pleiotropic actions. The above with the epidemiological association of reduced ultraviolet radiation exposure and vitamin D insufficiency that are associated with the PCa risk.35
There are other biochemical data supporting a link between 1,25(OH)2D3 and PCa by direct inhibitory effects on PCa cells, by an influence on apoptosis, cell differentiation, cell cycle arrest, growth factors, inhibition of metastases and angiogenesis in a cell specific manner.36 So, it has been proposed that VDR gene could influence the behavior of PCa and other cancers, including ovarian, breast, melanoma, sarcoma, and colon.
There are several studies in the literature that have shown that inherited polymorphisms in the 3'UTR of the VDR gene correlate with its transcriptional activity and mRNA stability in mini–gene receptor constructs. There are other studies that have determined the VDR genotypes in different populations. Most of these studies have not detected TaqI polymorphism associated to PCa or benign prostatic hyperplasia.37 We did not find a statistical difference among PCa and healthy individuals in distribution of the VDR alíeles. However, there are other papers that have only shown a weak but statistically significant association between TaqI polymorphism and PCa risk, however there is no strong association between the VDR genotype alone and lethal metastatic PCa in the literature. We however did find that when the PCa patients were grouped according to their histopathology Gleason score, the patients with poor prognosis presented a lower frequency of TT genotypes, which indicates that it is 1.75 times more probable to find genotypes Tt and tt in PCa patients with poor prognosis than in controls, and a poor prognosis pattern with a diminished frequency of the TT genotype. These are interesting findings; however caution must be taken because a larger cohort of Mexicans should be studied in order to confirm this association.
It is important to take into account that among the limitations of this study is the limited number of patients, the follow–up of the PCa patients in order to better understand the prognosis, as well as the limitation on the different polymorphisms, this is why we consider that there is a need for a more thorough study of Vitamin D and androgen receptor polymorphisms in the Mexican population.
CONCLUSIONS
The present study on Mexican men did not reveal any significant difference in AR polymorphism in PCa patients compared to controls, neither an association between the t alíele in the VDR an the risk of PCa, which agrees with other studies in the literature. However, when the genotypes and alíeles were correlated with PCa pathology prognosis, there was a significant difference between patients with a poor prognosis and the rest of our population. This has not been previously reported, therefore it is important to confirm our results in other populations as well as study the steps involved in the levels of vitamin D and the Gleason score.
Our results show that there is a role of the VDR and the AR that could influence the development and progression of PCa, however, this relationship is very complex because of the ethnic and environmental influence. This is the first study in a Mexican population with PCa and genetic polymorphisms; further studies with larger sample sizes should be done to confirm these observations.
ACKNOWLEDGEMENTS
We are very grateful with Lidia Cerda–Aguilar, Dr. Luis Mora, and Luis Antonio Paez–Riveros for their important assistance and collaboration in this work.
REFERENCES
1. Dirección General de Epidemiología. Registro Histopatológico de Neoplasias Malignas, CD ©2004. www.dgepi.salud.gob.mx [ Links ]
2. Hsing AW, Chokkalingam AP. Prostate cancer epidemiology. Front Biosci 2006: 1(11): 1388–413. [ Links ]
3. Ko YJ, Devi GR, London CA, Kayas A, Reddy MT, et al. Androgen receptor down–regulation in prostate cancer with phosphorodiamidate morpholino antisense. J Urol 2004; 172(3): 1140–4. [ Links ]
4. Suzuki H, Shimbo M, Ichikawa T. New biomarkers for prostate cancer. Nippon Rinsho 2005; 63(2): 351–9. [ Links ]
5. Hakimi JM, Schoenberg MP, Rondinelli RH, Piantadosi S, Barrack ER. Androgen receptor variants with short glutamine and glycine repeats may identify unique subpopulations of men with prostate cancer. Clin Cancer Res 1997; 3: 1599–608. [ Links ]
6. Hsing AW, Gao YT, Wu G, Wang X, Deng J, et al. Polymorphic CAG and GGN repeat lengths in the androgen receptor gene and prostate cancer risk: a population–based case–control study in China. Cancer Res 2000; 60: 5111–116. [ Links ]
7. Mowszowicz I, Mestaver C. Mutations of the androgen receptor gene a possible role in prostate cancer. Bull Cancer 1997; 84(2): 183–8. [ Links ]
8. Lange EM, Chen H, Brierley K, Livermore H, Wojno KJ, et al. The polymorphic exon 1 androgen receptor CAG repeat in men with a potential inherited prediposition to prostate cancer. Cancer Epidemiol Biomarkers Prev 2000; 9: 439–42. [ Links ]
9. Correa–Cerro L, Wohr G, Haussler J, Berthon P, Drelon E, et al. (CAG)nCAA and GGN repeats in the human androgen receptor gene are not associated with prostate cancer in French–German population. Eur J Hum Genet 1999; 7: 357–62. [ Links ]
10. Tayeb MT, Clark C, Haites NE, Sharp L, Murray GI, et al. Vitamin D receptor, HER–2 polymorphisms and risk of prostate cancer in men with benign prostate hyperplasia. Saudi Med J 2004; 25(4): 447–51. [ Links ]
11. Bid HK, Mishra DK, Mittal RD. Vitamind–D receptor (VDR) gene (Fok–1, Taq–1 and Apa–1) polymorphisms in healthy individuals from north indian population. Asian Pac J Cancer Prev 2005; 6(2): 147–52. [ Links ]
12. Cheteri MB, Stanford JL, Friederichsen DM, Peters MA, Iwasaki L, et al. Vitamin D receptor gene polymorphisms and prostate cancer risk. Prostate 2004; 59(4): 409–18. [ Links ]
13. Correa–Cerro L, Berthon P, Haussler J, Bochum S, Drelon E, et al. Vitamin D receptor polymorphisms as markers in prostate cancer. Hum Genet 1999; 105: 281–7. [ Links ]
14. Ntais C, Polycarpou A, Ioannidis JP. Vitamin D receptor gene polymorphisms and risk of prostate cancer: a meta–analysis. Cancer Epidemiol Biomarkers Prev 2003; 12(12): 1395–402, . [ Links ]
15. Macoska J. Ancestry, genetic susceptibility, E/Caqdherin160A and prostate cancer risk– is there an association? J Urol 2006; 176(2): 435–6. [ Links ]
16. Chamberlain NL, Driver ED, Miesfeld RL. The length and location of CAG trinucleotide repeats in the androgen receptor N–terminal domain affect transactivation function. Nucleic Acids Res 1994; 2: 3181–6. [ Links ]
17. Coetzee GA, Ross RK. Re: Prostate cancer and the androgen receptor. J Nati Cancer Inst 1994; 86: 872–3. [ Links ]
18. Irvine RA, Ma H, Yu MC, Ross RK, Stallcup MR, et al. Inhibition of p160–mediated coactivation with increasing androgen receptor polyglutamine length. Hum Mol Genet 2000; 9: 267–74. [ Links ]
19. Nelson KA, Witte JS. Androgen receptor CAG repeats and prostate cancer. Am J Epidemiol 2002; 155: 883–90. [ Links ]
20. Yoshida KI, Yano M, Chiba K, Honda M, Kitahara S. CAG repeat length in the androgen receptor gene is enhanced in patients with idiopathic azoospermia. Urology 1999; 54: 1078–81. [ Links ]
21. Zitzmann M, Brune M, Kornmann B, Gromoll J, Junker R, et al. The CAG repeat polymorphism in the androgen receptor gene affects bone density and bone metabolism in healthy males. Clin Endocrinol (Oxf) 2001; 55: 649–57. [ Links ]
22. Sawaya ME, Shalita AR. Androgen receptor polymorphisms (CAG repeat lengths) in androgenetic alopecia, hirsutism, and acne. J Cutan Med Surg 1998; 3: 9–15. [ Links ]
23. Giovannucci E, Stampfer MJ, Chan A, Krithivas K, Gann PH, et al. CAG repeat within the androgen receptor gene and incidence of surgery for benign prostatic hyperplasia in U.S. physicians. Prostate 1999; 39: 130–4. [ Links ]
24. Giovannucci E, Platz EA, Stampfer MJ, Chan A, Krithivas K, et al. The CAG repeat within the androgen receptor gene and benign prostatic hyperplasia. Urology 1999; 53: 121–5. [ Links ]
25. Mitsumori K, Terai A, Oka H, Segawa T, Ogura K, et al. Androgen receptor CAG repeat length polymorphism in benign prostatic hyperplasia (BPH): correlation with adenoma growth. Prostate 1994; 41: 253–57. [ Links ]
26. Shibata A, Stamey TA, McNeal JE, Cheng I, Peehl DM. Genetic polymorphisms in the androgen receptor and type II 5a–reductase genes in prostate enlargement. J Urol 2001; 166: 1560–4. [ Links ]
27. Bousema JT, Bussemakers MJ, van Houwelingen KP, Debruyne FM, Verbeek AL, et al. Polymorphisms in the vitamin D receptor gene and the androgen receptor gene and the risk of benign prostatic hyperplasia. Eur Urol 2000; 37: 234–8. [ Links ]
28. Roberts RO, Bergstralh EJ, Cunningham JM, Hebbring SJ, Thibodeau SN, et al. Androgen receptor gene polymorphisms and increased risk of urologic measures of benign prostatic hyperplasia. Am J Epidemiol 2004; 159(3): 269–76. [ Links ]
29. Xue W, Irvine RA, Yu MC, Ross RK, Coetze GA, et al. Susceptibility to prostate cancer: interaction beetwen genotypes at the androgen receptor and prostate–specific antigen loci. Cancer Res 2000; 60: 839–41. [ Links ]
30. Gastón KE, Kim D, Singh S, Ford H, Mohler JL. Racial differences in androgen receptor protein expression in men with clinically localized prostate cancer. J Urol 2003; 170; 990–3. [ Links ]
31. Yu H, Bharaj B, Vassilikos EJ, Giai M, Diamandis EP. Shorter CAG repeat length in the androgen receptor gene is associated with more agressive forms of breast cancer. Breast Cancer Res Treat 2000; 59: 153–61. [ Links ]
32. Visvanathan K, Helzlsouer KJ, Boorman DW, Strickland PT, Hoffman SC, et al. Association among an ornithine decar–boxylase polymorphism, androgen receptor gene (CAG) repeat length and prostate cancer risk. J Urol 2004; 171(2): 652–5. [ Links ]
33. Balic I, Graham ST, Troyer DA, Higgins BA, Pollock BH, et al. Androgen receptor length polymorphism associated with prostate cancer risk in hispanic men. J Urol 2002; 168(5): 2245–8. [ Links ]
34. Zhao XY, Peehl DM, Navone NM, Feldman D. 1 alpha, 25–dihydroxyvitamin D3 inhibits prostate cancer cell growth by androgen–dependent and androgen–independent mechanisms. Endocrinology 2000; 141: 2548–56. [ Links ]
35. Price DK, Franks ME, Figg WD. Genetic variations in the vitamin D receptor, androgen receptor and enzymes that regulate androgen metabolism. J Urol 2004; 171(2 Suppl.): 45–9. [ Links ]
36. Gsur A, Madersbacher S, Haidinger G, Schatzl G, Marberger M, et al. Vitamin D receptor gene polymorphism and prostate cancer risk. Prostate 2002: 51(1): 30–4. [ Links ]
37. Paiss T, Gschwend J, Autenrieth M, Correa–Cerro L, Haussler J, et al. Vitamin D receptor polymorphisms and prostate cancer risk in an European population. J Urol 1999; 161 (4S): 56. [ Links ]














