Introduction
Schwannomas are benign nerve sheath neoplasms, originating from schwann cells concentrically wrapped around peripheral and cranial nerves forming their myelin sheath. They are predominantly located in the posterior fossa; arising from the vestibular nerve at the cerebellopontine angle1. Olfactory groove presentation is rare with the exact origin being controversial2. Their neuroimaging characteristics are not specific; thus, the differential diagnosis is wide including other tumors with higher incidence and similar characteristics. Microscopically these neoplasms exhibit distinct ultrastructural features and immunophenotypic profile. CD34 (human hematopoietic progenitor cell antigen) is expressed in neurofibromas and can be used to differentiate them from schwannomas, that are expected to be completely negative or exhibit low expression, exclusively in Antoni B areas2-5.
Herein we present a case of an olfactory groove cellular schwannoma, with an unusual and unexpected diffuse CD34 positivity highlighting potential diagnostic difficulties. The origin and diagnostic challenges are extensively discussed through a literature review.
CLINICAL SUMMARY
Clinical presentation
A 51year-old female presented with a history of drug resistant, frequent and gradually intensifying occipital headache and nausea for a month. The symptoms were referred as intensifying in the upright position. She exhibited a gradual neurological deterioration 48-hours prior to admission in the form of bradypsychia, moderate memory dysfunctions (Mini-MentalTest: 12/30, most affected abilities were orientation in time and place, attention, concentration and short-term memory), and reduced strength bilaterally in upper and lower extremities (4/5). No café au lait spots were registered. Nasopharyngeal endoscopy did not reveal any visible lesion. Past medical history was unremarkable.
Imaging assessment
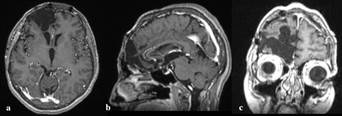
A Computerized-Tomography (CT) scan was performed revealing the presence of an anterior fossa tumefaction, without any accompanying malformations. Magnetic-ResonanceImaging (MRI) confirmed the presence of a heterogeneous contrast-enhancing neoplasm consisted of an admixture of solid and cystic formations (Figure 1).
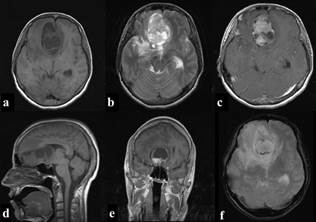
Figure 1 Brain MRI: Axial (a) and sagittal (d) T1 weighted slices presenting a frontal fossa neoplasm of heterogenous intensity, including solid areas and a significant number of cystic degenerative formations. A subfalcine herniation, associated with the volume effect can also be detected. Axial (b) T2 weighted slice confirming the aforementioned and highlighting the presence of significant perilesional edema. Axial (c) and coronal (e) T1 weighted, post-gadolinium images showing heterogeneous enhancement. A slight circumferential blooming is also registered in axial (f) T2 weighted gradient echo.
Treatment
A bifrontal osteoplastic craniotomy was performed, with complete microsurgical excision of a yellow, well-defined, smooth surfaced, and well-vascularized intradural extra-axial mass, infiltrating the dura. There was no immediate correlation between the ipsilateral olfactory nerve and the mass. Surgical time was approx. 4 hours. Multiple tissue samples were acquired from peripheral and central parts of the neoplasm, while the rest was excised employing an Ultra-Sonographic Aspirator. The patient remained in Intensive Care Unit for a 24h gradual reduction of sedation. Post-surgery CT assessment confirmed the total excision. She was discharged two weeks post-surgery with amelioration of symptoms and complete recovery after a month of physical therapy. At that time, she returned to her previous functional and working status with a Modified Rankin Scale of 0 and Karnofsky Perfomance Status of 90 points. Since then she is free of symptoms, with no neurologic or mental function alterations and no recurrence after five years (Figure 2).
Pathological findings
Initial assessment of hematoxylin and eosin (HE) - stained sections showed mild reactive glial proliferation, and the presence of a uniform dense population of spindle to slightly epithelioid cells with ovoid nuclei, small nucleoli and sparse cytoplasm infiltrating the dura (Figure 3). Three mitotic figures in 10 high power fields were identified whereas none was atypical. Architecturally the tumor had a fascicular growth pattern with occasional eosinophilic protein drops. No significant cytologic atypia or infiltration of the brain parenchyma was registered, however small foci of necrosis were noted. Lymphocytic infiltration and foam macrophage cells were evident.
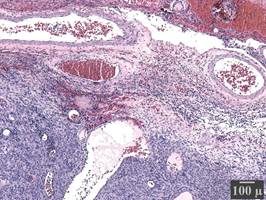
Figure 3 Histopathological investigation: Hematoxylin and eosin stain (x40) of sections acquired from the peripheral part of the neoplasm. Tumor cell infiltration of the dura matter (an endosteal layer of dense fibrous connective tissue, containing a significant number of blood vessels).
Immunohistochemically, the tumor was diffusely positive for vimentin, S-100 protein and CD34, whereas it was negative for calponin, CD99, EMA, desmin, ALK and GFAP. Collagen type IV highlighted the basal lamina, whereas the proliferation index by Ki-67 was low (<5%). The tumor was rich in reticular fibers.
The diffuse positivity for CD34 raised concern for solitary grade II dural hemangiopericytoma (solitary fibrous tumor- SFT) and an expert opinion was requested from a specialized center in the USA. In the reference laboratory the initial immunohistochemical profile was confirmed; in addition, the tumor was diffusely positive for SOX10 and negative for STAT6 and MelanA. These findings essentially confirmed the neural crest origin of the tumor and excluded an SFT. The final diagnosis of cellular schwannoma with unusual CD34 diffuse positivity was rendered (Figure 4).
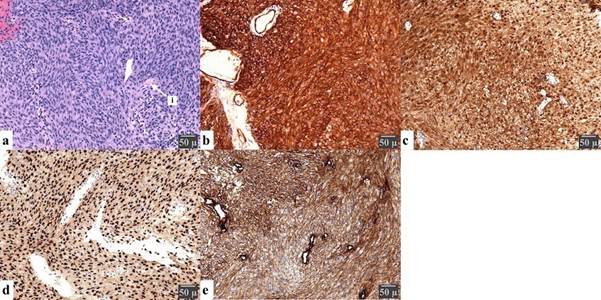
Figure 4 Histopathological investigation: Sections present a cellular spindled to slightly epithelioid cellular proliferation with no significant atypia, low mitotic activity, and foci of necrosis on Hematoxylin and eosin staining (a), arranged in Antoni A (hypercellular) and Antoni B (hypocellular) areas with interspersed Varocay bodies (i). Immunochemical stains show that the neoplasm is diffusely positive for vimentin, S-100(c), SOX10(d), Collagen type IV(e) and CD34(b) while being negative for EMA, desmin, ALK, STAT6, and GFAP with low (<5%) positivity for Ki67 and less than 3/10 HPF nuclear movements. A scale is provided to present the magnification.
Discussion
Intracranial schwannomas are benign, slowly growing nerve sheath tumors, with a wide age range (mean 31) and male predominance2. They are commonly located in the posterior fossa, arising from the vestibulocochlear nerve; accounting for approximately 80% of the cerebellopontine angle tumors. Olfactory groove localization is rare, with a total of 54 cases being reported until 20236-8. Patients present with varying symptomatology including headache, visual deficits, moderate cognitive disturbances, rhinorrhea, and anosmia/hyposmia2. The imaging findings vary, from solid tumors, in the majority of cases, to less frequently tumors with cystic components, with contrast enhancement varying as well2. The aforementioned may result in interpretation difficulties and confusion with other tumors commonly found in the region such as meningiomas9 and esthesioneuroblastomas. In our case based on the neuroradiologic findings, localization, neurologic status and similar cases reported in the literature the diagnosis of esthesioneuroblastoma was favored.
The literature closely associates schwannomas in this region with the olfactory nerve, with more than half of the cases reporting symptoms of the olfactory apparatus, thus the term olfactory schwannoma is commonly employed2. However, similarly to the optic nerve and in contrast to other cranial and peripheral nerves, schwann cells do not reside in the nerve sheaths of the olfactory nerve. Their structural, neurotrophic and phagocytotic functions are substituted by glial cells known as Olfactory Ensheathing Cells (OEC). Tumors arising from the latter are rare, and their characteristics resemble those of schwannomas, with no reliable methods of distinguishing between them10. However, Olfactory Ensheathing Cell tumors are not included in the World Health Organization classification, thus their existence is questionable. Other authors have proposed that mesenchymal pial cells may potentially transform into schwann cells11, explaining the rare intra-axial presentation, but not the extra-axial one, which is lately associated with the trigeminal nerve’s meningeal or anterior ethmoidal branches2,12. The intraoperative findings presented (lack of association with the olfactory nerve; dura infiltration) support the meningeal branches origin theory.
Microscopically the tumor was encapsulated and composed of a well-differentiated and uniform population of Schwann cells. The ordinary schwannomas are commonly biphasic, with compact areas (Antoni A), occasional nuclear palisading (Verocay bodies), and foci of loosely arranged tumor cells (Antoni B). In the Antoni A areas, the cells exhibit a modest eosinophilic cytoplasm, no detectable cell borders, and normo-chromatic elongated tapered nuclei. Antoni B areas contain a cobweb-like network of tumor cells with collections of lipid-laden histiocytes and thick-walled, hyalinized blood vessels. Literature points toward a predomination of Antoni A areas in neoplasms arising from the 8th cranial nerve, while intestinal localization is associated with almost complete absence of Antoni B areas13. In the cellular subtype, as in our case, Antoni A areas are predominate with Antoni B areas constituting <10% of the total tumor14.
Schwannomas are expected to show consistent diffuse positivity for S-100 protein, SOX10 and collagen type IV, CD34 negativity, apart from focal mild to moderate staining in areas of necrosis, degeneration and in Antoni B areas5,15,16. The transmembrane phosphoglycoprotein CD34, is mainly expressed by embryonic hematopoietic cells, including endothelial lymphoid and myelogenous strings, and also by various tumors of vascular, lipomatous, fibroblastic origin and nerve sheath tumors, where it is believed to be expressed by endoneurial and perineurial fibroblasts, whose number significantly increases during nerve damage, thus it was proposed as a potential marker in the evaluation of supportive cell population in pathological nerves15. Others have proposed the presence of a dendritic CD34 reactive, non-schwanian cell population in peripheral nerve sheath tumors serving supporting functions17. Literature implies that these cells are unrelated to schwann cells, however some authors also defy their association with fibroblast populations18. This stain may also be subject to the stage or form of nerve sheath differentiation. An interesting decrease in reactivity has been reported in highly malignant peripheral nerve sheath tumors when compared to low-grade ones19.
CD34 expression is consistent with neurofibromas, presenting a significantly greater population of positive cells at the peripheral part and within the fibrotic and myxoid areas5, 17,18. On the contrary, schwannomas are for the most part completely negative or focally positive at most, presenting a few slender cells in the peripheral zone and to some spindle and stellate cells in the Antoni B areas 3,5,17,18.However, neurofibromas are expected to present characteristic bland spindle cells, smaller than those of a neurilemoma, with thin, wavy nuclei, situated in a loose myxoid collagen stroma13. Moreover, they are composed out of a heterogenous admixture of a variety of components, including perineurial and perineurial-like cells, fibroblasts, mast cells and a significantly smaller overall population of schwann cells when compared to that of a schwannoma. The differentiation between neurofibroma and schwannoma, as in the presented case, may be based upon the morphological characteristics as described. Furthermore, some authors proposed the combinations of additional immunostains to perform an adequate differentiation among histological mimics, such as calretin, CD34 and CD 57 4,6. In summary olfactory groove schwannomas are rare and their origin remains controversial as they are not associated with the olfactory nerve. Our findings support the assumption that they arise from the meningeal trigeminal branches. Their non-specific clinical presentation and imaging characteristics can create potential diagnostic problems. Intriguingly this case expressed diffuse CD34 positivity which is not expected in this tumor raising interesting questions regarding histogenesis. Literature associates this with a dendritic, non-schwanian cell population, assumingly fibroblasts, serving supportive functions. The potential employment in the evaluation of stage or form of nerve sheath differentiation remains to be investigated.
Statement of ethics
Written informed consent regarding the publication of data and images was acquired from the patient herself. The provided neuroradiological depictions were anonymized to secure patient identity with respect to the General Data Protection European Union Regulation 2016/679. The conduction of the study is in accordance with the World Medical Association Declaration of Helsinki. No Ethical Committee approval is required as this is an observational retrospective study.











 nueva página del texto (beta)
nueva página del texto (beta)