Introduction
Guillain-Barré syndrome (GBS) is the most common cause of acute flaccid paralysis worldwide. Twenty percent of patients become seriously disabled and up to 5% die. About 30% of GBS patients become ventilator dependent, with an increased risk of death, associated complications, and worse clinical outcomes.1,2
Scores such as Erasmus GBS Respiratory Insufficiency Score (EGRIS) use clinical data including days from symptom onset and admission, facial or/and bulbar weakness, and Medical Research Council (MRC) sum score to predict respiratory insufficiency within the first week of admission. Additionally, other tools can be used to evaluate pulmonary function (e.g., forced vital capacity and forced expiratory volume in 1s), although special equipment might be required.3,4
Several studies have supported the role of molecular mimicry in disease pathogenesis, by examining a causal relationship between C. jejuni lipo-oligosaccharides and gangliosides. Lymphocytes and neutrophils are associated with systemic inflammation and production of proinflammatory mediators. GBS, as an autoimmune disease, elicits an upregulation in inflammatory and metabolic pathways, with increased production of lymphocytes and neutrophils. Serum markers such as the neutrophil-lymphocyte (NLR) and leuko-glycemic (LGR) ratios have been studied for the severity and prognosis of non-neurological disorders such as myocardial infarction, cancer, and autoimmune diseases.5,6,7 Regarding neurological disorders, little information is available, with some reports in patients with multiple sclerosis and cerebrovascular disease.8,9 Scarce data has been published on the use of these indices and autoimmune peripheral neuropathies. This inflammatory cascade can appear before GBS clinical symptoms.10 In this study, we evaluate NLR and LGR in patients with GBS and their relationship as predictive markers for ventilatory support.
Materials and methods
A cross-sectional study from a prospective cohort of patients with GBS was conducted, from January 2018 to February 2021. GBS diagnosis was made by the National Institute of Neurological Disorders and Stroke (NINDS)11 and all patients had complete blood work on admission to the emergency room (CBC, basic metabolic panel, electrolytes, and liver function tests). Exclusion criteria included patients with diabetes mellitus, previous autoimmune disorders, drug-induced immunosuppression, or HIV infection. Patients with acute bacterial infection (pneumonia, urinary tract infection, etc.) or sepsis on admission, based on the Third International Consensus Definition for Sepsis and Septic Shock, were also excluded.12 General demographic data was obtained, as well as a history of previous infection, symptom-to-admission (GBS-associated symptoms to ER arrival), assessment of muscle strength based on Medical Research Council (MRC) on admission, GBS disability score (GDS) on admission, cranial nerve involvement, autonomic dysfunction, mechanical ventilation (IMV) and length of stay. Nerve conduction studies were performed on admission and the electrophysiological damage mechanism was classified according to Hadden et al. criteria.13 Cerebrospinal fluid (CSF) glucose, protein concentration, and cell count were evaluated. Albuminocytological dissociation was defined as elevated CSF proteins (>45mg/ dL) with low cell count (≤ 50 cells/µL). For each patient, the leuko-glycemic ratio [glycemia (mg/dl) x leukocytes (106/L) /1,000] and the neutrophil-lymphocyte ratio were calculated by neutrophil count divided by lymphocyte count.
The study was approved by the Ethics Committee and the Institutional Review Board. Signed consent was required for study participation.
Statistical analysis
Demographic data were analyzed with descriptive statistics. Kolmogorov-Smirnov test was used for distribution, and medians with standard deviation or medians with interquartile range were obtained, accordingly. A correlation analysis with the Person test was performed. Comparison between patients with and without ventilatory support was performed with student´s t-test or Mann-Whitney U test based on distribution. The chi-square test was used for categorical variables, and Fisher´s exact test was applied when necessary.
A logistic regression analysis was performed for patients with mechanical support, according to TRIPOD consensus.14 Variables included age, dysautonomia, EGRIS score, neutrophil-lymphocyte (NLR), and leuko-glycemic (LGR) ratios. Goodness-of-fit was assessed with Hosmer & Lemeshow test, results were reported in odds ratio with 95% confidence intervals, and the performance of the model was assessed through analysis of area under the curve. Both NLR and LGR performances were obtained with an area under the curve analysis, cut-off values, sensitivity, and specificity were established by the Youden index, and a value of p<0.05 was considered statistically significant. Data were analyzed using SPSS version 22.
Results
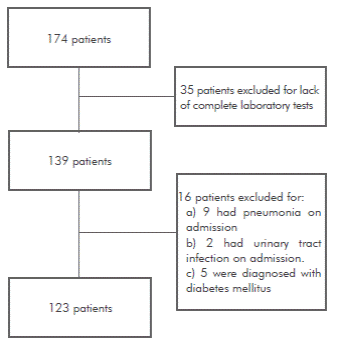
Of 174 patients with GBS, 51 patients were excluded (Figure 1), leaving 123 patients for study analysis. The mean age was 45.5±16.5 years, 77 (62.6%) were men and 37 (30.0%) required ventilatory support. In patients with mechanical ventilation, the mean age was 51.7±18.2 years, and the median time from symptom onset to ventilation was 5 days (IQR 3-5). Moreover, 75.6% had cranial nerve involvement (facial and/or bulbar), a mean MRC sum score of 22.2±16.9, median EGRIS score of 4 (IQR 2-5), median LGR of 1.58 (IQR 0.99-1.99) and median NLR of 6.15 (IQR 4.18-9.23). Baseline demographics patients are shown in Table 1.
Table 1 Comparative analysis between GBS patients with invasive ventilation mechanical (IVM) vs no-IVM
| Patients requiring ventilatory support n=37 | Patients not requiring ventilatory support n=86 | P value | |
|---|---|---|---|
| Age - yr, mean | 51.7±18.2 | 42.9±15.1 | 0.006 |
| Male gender - no. (%) | 20(54) | 57(66.2) | 0.22 |
| Diarrhea, no. (%) | 13(35.1) | 30(34.8) | >0.99 |
| Cranial nerve involvement - no. (%) | 28 (75.6) | 35 (40.6) | <0.001 |
| -Facial nerve- | 24(64.8) | 34(39.5) | 0.011 |
| -Bulbar nerves | 25(67.5) | 21(24.4) | <0.001 |
| Dysautonomia, no (%) | 25(67.5) | 7(8.1) | <0.001 |
| GDS score, median (IQR) | 4(1-4) | 4(2-5) | <0.001 |
| MRC score on admission, mean (SD) | 22.2±16.9 | 35.2 ±16.1 | <0.001 |
| EGRIS score, median (IQR) | 3(2-4) | 5(4-6) | <0.001 |
| Hospital stay (days), median (IQR) | 58(31-83) | 7(5-10.5) | <0.001 |
| Protein count in LCR (mg/dl), median (IQR) | 39(27-100) | 446(32-60) | 0.83 |
| Leucocyte (103/ml), median, (IQR) | 12.51(9.12-16.58) | 9.75(8.20-11.72) | 0.002 |
| Neutrophil (103/ml), median, (IQR) | 8.3 (5.85-11.30) | 6.40(5.45-8.10) | <0.001 |
| Lymphocyte (103/ml), median, (IQR) | 1.60(1.15-1.90) | 2.20(1.60-2.60) | <0.001 |
| Glucose (mg/dl), median (IQR) | 122(110-141) | 106(95.5-118) | <0.001 |
| Sodium (mEq/dl), median (IQR) | 138(135-140) | 138(137-140) | 0.75 |
| Albumin (g/dl), mean SD | 4.28±0.52 | 4.45±0.46 | 0.089 |
| NLR, median, (IQR) | 6.15(4.18-9.23) | 3.1(2.21-4.08) | <0.001 |
| LGR, median, (IQR) | 1.58(0.99-1.99) | 1.02(8.85-1.32) | <0.001 |
The distribution of leukocyte, lymphocyte, neutrophil, and serum glucose counts at admission with respect to the age of the population is shown in Figure 2. In the analysis of correlation between age (years) and the other variables (leukocyte count, lymphocytes, neutrophils, and serum glucose levels) there were no statistically significant results.
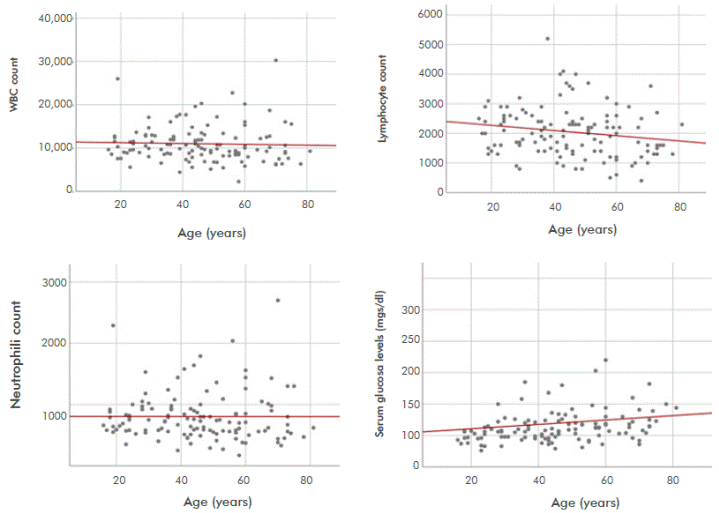
Figure 2 Count of leukocytes, lymphocytes, and serum glucose levels at admission with respect to population age
When comparing both groups, a greater age (51.7±18.2 vs 42.9±15.1, p=0.006), cranial nerve involvement (75.6% vs 40.6%, p<0.001), dysautonomia (67.5% vs 8.1%, p<0.001), MRC score (22.2±16.9 vs 35.2±16.1, p<0.001), and median EGRIS score [3 (IQR 2-4) vs 5 (IQR 4-6), p<0.001] was observed at admission in patients that needed ventilatory support. No statistically significant differences were encountered neither for demyelinating (38.7% vs 42.4%, p=0.82) nor axonal subtype (61.2% vs 50.6%, p=0.39). Both median NLR [6.15 (IQR 4.18-9.23) vs 3.1 (IQR 2.21-4.08), p<0.001] and median LGR [1.58 (IQR 0.99-1.99) vs 1.02 (IQR 8.85-1.32), p<0.001] in patients with ventilatory support were significant.
The multivariable logistic regression analysis demonstrated that the presence of dysautonomia [OR 30.6 (95% CI 6.9-134), p=<0.001], a higher score on the EGRIS scale [OR 2.0 (95% CI 1.3-3.1), p=0.001], and higher NLR [OR 8.6 (95% CI 2.0-36.7), p=0.004] are independent risk factors for invasive mechanical ventilation; the model performance is AUC 0.938, 95% CI (0.89-0.98), p= <0.001 (Table 2).
Table 2 Multivariable analysis for ventilatory support in patients with GBS
| Univariable model | Multivariable model | ||||||
| Patients requiring ventilatory support n=37 | Patients not requiring ventilatory support n=86 | P value | OR (IC 95%) | OR (IC 95%) | P value | ||
| Age - yr | 51.7±18.2 | 42.9± 15.1 | 0.006 | 1.03(1.0-1.06) | 1.06(1.0-1.1) | 0.003 | |
| Symptom-to-admission -median (days) (IQR) | 5(3-5) | 6(4-10) | 0.003 | 0.87(0.78-0.98 | |||
| Cranial nerve involvement - no. (%). | 28(75.6) | 35(40.6) | <.001 | 4.5(1.9-10.7) | |||
| MRC score - mean (SD) | 22.2±16.9 | 35.2 ±16.1 | <0.001 | 0.95(0.92-0.97) | |||
| Dysautonomia, no (%) | 25(67.5) | 7(8.1) | <0.001 | 23(8.3-66) | 30.6(6.9-134) | <0.001 | |
| EGRIS score, median (IQR) | 3(2-4) | 5(4-6) | <0.001 | 1.9(1.4-2.4) | 2.0(1.3-3.1) | 0.001 | |
| LGR (%) | 1.58(0.99-1.99) | 1.02(8.85-1.32) | <.001 | 3.0(1.6-5.5) | 0.7 (0.2-2.5)) | 0.58 | |
| NLR (%) | 6.15(4.18-9.23) | 3.1(2.21-4.08) | <.001 | 4.0 (2.0-8.19) | 8.6(2.0-36.7) | 0.004 | |
|
Logistic regression description: Overall model fit: chi-square 81.123, df 5, p< 0.0001. Goodness-of-fit test: r2 = 0.483; Hosmer & Lemeshow test, chi-square, 12.47, df 8, p = 0.13. Model performance: AUC 0.938, 95% CI (0.89-0.98), p =<0.001. | |||||||
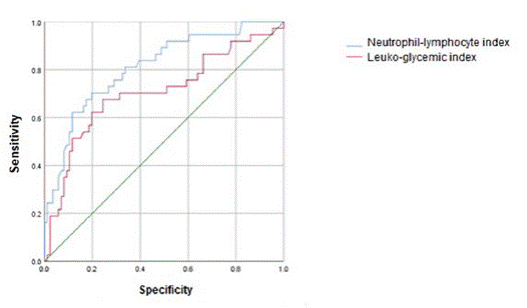
Both LGR and NLR demonstrated high performance for ventilatory support prediction, with 0.70 [95% CI (0.59-0.81), p<0.001] and 0.81 [95% CI (0.72-0.89), p<0.001], respectively. The best cut-off values, according to the Youden index, are 1.12 for LGR (sensitivity 0.70, specificity 0.40) and 3.59 for NLR (sensitivity 0.78, specificity 0.33). The operating characteristic curve analysis is shown in Figure 3.
Discussion
Guillain-Barre Syndrome (GBS) is an autoimmune disorder where antibodies are abnormally produced against peripheral nerve gangliosides. Cellular responses potentially play a role in the pathogenesis of GBS and complement-mediated nerve injury. Experimental allergic neuritis models have encountered several pro-inflammatory molecules related to acute nerve injury, such as IFN-γ, IL-1β, tumor necrosis factor (TNF), IL-6, and IL-10. All these inflammatory processes occur in the pre-symptomatic stages of patients with GBS and further portray worse clinical presentations with early ventilatory support requirements.15
Classically, old age has been considered a risk factor for ventilatory support and inability to walk independently at 1, 3, and 6 months.1 Despite decreased immunological responses in the elderly, as described previously by Hagen et al. this subset of patients has increased leukocyte, neutrophil, and lymphocyte counts when compared to younger patients with GBS, and they should be treated as high-risk patients for several complications and worse functional outcomes.15
The EGRIS scale, developed in European and North American populations, uses only clinical variables (MRC score, cranial/ bulbar weakness, and days between onset and hospital admission) to predict respiratory insufficiency and ventilatory support within the first week of admission.3 Early involvement of bulbar muscle strength can cause microaspiration, hence the large population of GBS patients with a high EGRIS score with pneumonia upon admission.16 In these cases, it is difficult to distinguish if the inflammatory process is due to sepsis associated with aspiration pneumonia or GBS inflammatory process. In our study, patients who presented some infectious process (pneumonia) on admission were excluded. We observed that high NLR is a risk factor, independent of the EGRIS scale score, for ventilation requirement; to our knowledge, this data had not been previously reported.
Consequently, we consider that the elevation of NLR in patients with GBS who require VMI is due to the systemic immune response of the disease, as in the case of other neurological autoimmune diseases (multiple sclerosis).9
We observed that the presence of dysautonomia is also a risk factor for the requirement of invasive mechanical ventilation, independent of the EGRIS scale score, which has not been previously reported. Cardiovascular dysautonomia is due to injury of the thoracic medullary roots exit that form the paraganglionic chains of the sympathetic autonomic system.
The diaphragmatic weakness that occurs in patients with SGB requiring VMI, in part, is attributable to damage of the phrenic nerves in their exit from the cervical roots. Therefore, we theorize that both the presence of dysautonomia and diaphragmatic weakness is due to severe root damage in GBS.3
The neutrophil-lymphocyte ratio is an indicator of systemic inflammation and has been described as a marker of severity and short overall survival in subjects with acute respiratory distress syndrome (ARDS). Wang et al. demonstrated an increase in in-hospital, 28-day, and 90-day mortality in patients with ARDS with a NLR of >14: 57.8% vs 43.2%, 58.9% vs 41.1,% and 58.0% vs 42.0%, respectively.17 Furthermore, inflammatory diseases have higher NLR, as recently described in a meta-analysis of 1550 patients with rheumatoid arthritis.6 Fewer evidence exists on its utility for neurological disorders. However, a higher NLR (classified by terciles and adjusted for clinical and laboratory values) was associated with unfavorable outcomes in terms of mRS score after acute ischemic stroke.18 Additionally, higher NLR values have been observed in patients with multiple sclerosis when compared to healthy controls.9
Scarce information is available on the usefulness of NLR as an indicator in autoimmune peripheral nerve disorders, such as GBS. One study reported a higher NLR in subjects with acute inflammatory demyelinating polyneuropathy (AIDP), but we did not find any association with electrophysiological subtypes.19
Other reports have associated this ratio with severe presentations of GBS, for example, Huang et al. reported a higher level of NLR in subjects with GDS ≥3 and a cut-off value of 2.295 for GBS occurrence and 3.05 for severity.20 Moreover, Pingping N et al. reported several ratios as risk factors for IMV in patients with GBS, particularly NLR, with an OR of 3.319 and a cut-off value of >3.5 for mechanical ventilation.21 We obtained a similar cut-off value of 3.59 with a sensibility of 78% and a specificity of 33%.
High serum glucose in a patient requiring ventilatory support due to any disease is considered a marker of severity.22 We observed higher serum glucose levels on admission in GBS patients with ventilatory support compared to those without, as demonstrated in other series.23 The increase in serum glucose levels might be directly related to the metabolic response to stress in acute inflammatory processes. On the other hand, an interesting fact is the high prevalence of autonomic dysfunction in severe GBS cases and in patients with mechanical support,23 where cortisol-induced glucose increment can be produced by adrenergic dysregulations.24
Although LGR has been extensively studied in acute myocardial infarction and relates to severity,25 few reports have demonstrated its correlation with severity in acute ischemic stroke.26 We observed a statistically significant OR in the univariable analysis for LGR but not in the multivariable analysis. To our knowledge, this is the first report of LGR in subjects with GBS. Further studies are needed to confirm the role of these indexes in other circumstances.











 nueva página del texto (beta)
nueva página del texto (beta)