Introduction
Seizure is a condition in which nerve cells make sudden and simultaneous discharges and is often accompanied by changes in the neural network and function. The term epilepsy is also defined as the presence of two or more seizures, and is one of the most common diseases in the world.1 Along with seizures, epilepsy is also associated with several other comorbidities, including cognitive deficits, that are very common in patients with epilepsy.2 At present, most cases of epilepsy are treated or controlled with anti-epileptic drugs (AEDS), which, as it has been reported, have limitations in performance, safety, and efficacy.3
Although there are many studies on how comorbidities with epilepsy develop, there is little information on how epileptic seizure causes memory impairment associated with learning.4 Oxidative stress and free radicals are among the most important mechanisms that can contribute to disorders such as epileptic seizures.5 Epileptic seizures have also been related to the increase of reactive oxygen species (ROS) levels and the production of superoxide anion (O2.-) in the brain.6 Oxidative stress is the consequence of a disproportion between the antioxidant resistance structure and the formation of ROS,7 it damages cell membranes and DNA, producing cell injury and death, as well as chronic illnesses such as Alzheimer's, cancer, epilepsy, Parkinson's, and heart diseases.8,9 The role of oxidative stress in epilepsy is well known,10 hence, maintaining low levels of ROS is crucial for normal cell function. Long-term increases in ROS pose an inherent risk of a higher neurological damage, as seen in epilepsy.11 Furthermore, nitric oxide (no) has been reported to be a potential neurotransmitter or retrograde messenger linked to synaptic plasticity, regulation of brain excitability, and triggering of seizure activity.
Nevertheless, no has been widely implicated in numerous neuroinflammatory and neurodegenerative conditions due to its prooxidant and antioxidant actions.12
Nowadays, antiepileptic drugs (AEDS) such as barbiturates and a variety of sedatives are adopted to impede or treat epilepsy. Although in recent years, many drugs have been introduced for dealing with epilepsy, they continue to show adverse and even deleterious effects that in most cases, even with a sufficient dosage, fail to properly stave off seizures. Therefore, they can pose a great negative impact on patients' quality of life both psychologically and behaviorally.13,14 One of the common approaches in the discovery and development of AEDS - which is the backbone for the cure of sick individuals with seizures and epilepsy - is the utilization of natural substances with medicinal properties.15 One of the herbs that has evinced beneficial medicinal properties and has been studied in traditional medicine is Buxus hyrcana (Buxaceae family), which grows in various part of the world.16,17 The recent phytochemical investigation on Buxus hyrcana Pojark has resulted in the isolation of triterpenoid alkaloids including hyrcanone, hyrcanol, hyrcatrienine, N(b)-dimethylcycloxobuxoviricine, and hyrcamine, through modern spectroscopic methods, while the known alkaloids, buxidin, buxandrine, buxabenzacinine, buxippine-K, and E-buxenone were identified and reported earlier.17 Previous studies have demonstrated many medicinal properties of the B. hyrcana, such as its antimalaria, anticancer, immunosuppressive - due to its active ingredients -, antifungal and antileishmanial activity; moreover, it has anti-inflammatory and antioxidant characteristics because of its triterpenoids and alkaloids compounds.16-21 Additionally, its anticonvulsive and neuroprotective properties have been recently documented in experimental animal models.22
Given the behavioral manifestations disorder associated with epilepsy and the effects of B. hyrcana on such disorders, it was hypothesized that the B. hyrcana extract (bhe) would have a positive effect on learning and memory in pentylenetetrazole (ptz)-kindled rats. The objective of this experiment was to assess the effects of bhe on behavioral disorders (i.e., learning and memory) caused by epilepsy and seizures in adult male rats.
Materials and methods
Animal ethics
This experiment was conducted at Shahid Beheshti University (Tehran, Iran) on adult Wistar rats (200 ± 20 g, 8 weeks old) obtained from the Shahid Beheshti University of Medical Sciences (Tehran, Iran). Animals were kept for one week in a room with constant conditions (12 h light/dark period with lighting starting at 7 a.m., 22 ± 2°C, 55 ± 5% relative humidity) in standard cages made from polycarbonate, in order for them to adapt to a new environment. The animals had free access to water and special feed for rats (Pars animal feed, Iran) throughout the experiment. Animals were randomly allocated in experimental groups (each one containing 6 rats) after one week of adaptation to the laboratory environment. Each animal was used only once during the trial, and efforts were made to reduce animal suffering while obtaining reliable scientific data. All experiments were done between 09:00 and 15:00. All conducted experiments pertaining animal rights and conservation in this study were performed in accordance with the standard ethical guidelines (European Communities Directive 2010/63/EU) and were approved by the Local Ethics Committee at the Shahid Beheshti University (ethical code: IR.SBU.1397.156).
Collection of plant material and preparation of extracts
The B. hyrcana herbs were obtained from the Institute of Medicinal Plants of Shahid Beheshti University. The extract was prepared according to cited references and previous work.22 Consequently, about 100 g of the aerial part of the plant was prepared, cleaned, dried in shade, and powdered by mechanical shredder. To prepare the hydroalcoholic extract, the plant powder was drenched in 1 L of ethanol 80% for 72 h, then the resulting mixture was filtered and concentrated in vacuum at 45°C using a rotary apparatus (EYEL A, Japan). The resultant extract was 37.8% dry and stored in the refrigerator at 4°C until the experiment. The dose required for the intraperitoneal (i.p.) injection was based on animal weight.
Medication and treatments
Pentylenetetrazole from Sigma Company (USA), phenobarbital sodium (PB) from Chemidarou Pharmaceutical Company (Iran), ketamine and xylazine from Alfasan Company (the Netherlands) were obtained. PTZ was prepared as a 1% v/w solution in saline. Phenobarbital sodium was used to establish a comparison with a conventional anticonvulsant drug. Phenobarbital sodium was dissolved in the physiological saline solution and administrated intraperitoneally in a dose of 30 mg kg-1 per body weight of animal. BHE was continued in normal saline solution via ip in doses of 300 and 600 mg kg-1 with PTZ during the experiment until complete animal kindling. Animals were randomly allocated into four groups of six: 1) control negative (CN) group receiving normal saline (0.3 mL per rat), 2) control positive (cp) group receiving PB (30 mg kg-1), 3 and 4) groups receiving bhe (300 and 600 mg kg-1, respectively).
Thirty minutes after vehicle injection, PB and BHE (300 and 600 mg kg-1) animals were challenged with the sub-threshold dose of PTZ (35 mg kg-1 per body weight). All injections were administered in the form of fresh solutions in a constant volume of 0.3 mL per rat throughout the study.22
Pentylenetetrazole-induced kindling test
The study was conducted in a double-blind manner (the experimenter was unaware of which animal belonged to which group). Pentylenetetrazole was injected in a sub-threshold dose of 35 mg kg-1 every 48-h interval for a period of one month. In order to record and measure seizure behavior, the animals were transferred individually to transparent plastic boxes and were observed immediately after the PTZ administration for one hour; they were recorded by a computer-connected camera. Seizure threshold was measured on a 6-step scale (Table 1). Animals were considered kindled if they showed stages 4 or 5 in two consecutive trials. Animals were given the PTZ challenge (70 mg kg-1) 7 days after the kindling development. Meanwhile, they underwent the passive avoidance learning test. The rate of memory retention was evaluated after the PTZ challenge.23
Table 1 Adapted Racine’s scale for pentylenetetrazole (PTZ) induced seizure in rats.
| Stage | Seizure intensity |
| 0 | No behavioral fluctuations |
| 1 | Ear and facial twitching |
| 2 | Nodding of the head, head clonus and myoclonic jerks |
| 3 | Unilateral forelimb clonus with lorditic posture |
| 4 | Bilateral forelimb clonus with rearing and falling |
| 5 | Generalized tonic-clonic seizure (GTCS) with loss of postural tone |
Shuttle box test
The shuttle box (Borj Sanat Azma, Iran) was used to test passive avoidance memory. The training was conducted in the shuttle box during the lighting phase, between 08:00 and 14:00. The shuttle box consists of two dark and light sections of identical dimensions (27 cm × 14.5 cm × 14 cm) that are opened or closed with a guillotine door. The floor of both compartments consists of steel rods at 1 cm apart from one another. The floor of the dark compartment can be electrified by a stimulator. The test process used in this study was described earlier.24 In summary, the animals in the acquisition phase were initially placed in the light chamber with their faces away from the door, and 10 s later the guillotine door was removed. Rats have an innate interest in dark environments. When the animal entered the dark chamber completely, the guillotine door was closed and after 30 s the animal was removed from the dark chamber and placed in its home cage. This habituation trial was repeated 30 min later. The animals were not given any shock during these stages. After the second iteration, the first acquisition trial began. Animal step-through latency during the acquisition phase (STLa) was measured as they entered their dark environments with all four legs. After the animal was fully enclosed in the dark chamber, the guillotine door was closed and a moderate shock (1.5 mA) was applied to the animal for 3 s. After 20 s, the animal was removed from the dark chamber and transferred to its cage.
After 5 min, the test was repeated. The rats were given a shock every time they entered the dark environment completely. Training was ended when the animal persisted in the light chamber for 120 consecutive sec. The shock number (SN) in acquisition trial was measured until acquisition. Memory retrieval was performed 24 h after the acquisition. The rats were placed in the light chamber and the guillotine door was removed after 30 s; the step-through latency to enter the dark environment during the retrieval phase (STLr) as well as the total time spent in the light compartment (TLC) were measured up to 300 s. If the rats did not enter the dark chamber within 300 s, the test would be concluded and a score of 300 would be documented for the rats.
Biochemical measurement
After the shuttle box test, the rats were quickly beheaded under deep anesthesia by ketamine and xylazine, and their brains were removed, washed with ice saline, and kept at -80°C for subsequent analyses.
Assessment of brain reduced glutathione (GSH) content
The levels of GSH was evaluated by Ellamn's reagent (5,50-dithio-bis-2-nitrobenzoic acid).25
Measurement of brain malondialdehyde (MDA) activity
Thiobarbituric acid reactive substances (TBARS) procedure was used to evaluate the brain MDA level.26
Measurement of brain nitric oxide (no) level
Indirect no (nitrites and nitrates) level in the brain tissues was calculated according to the Griess method. Summarizing, the supernatants were placed into a 96-well kit (Natrix™ Nitric Oxide (no) Assay Kit, Novin Navand Salamat, Iran). The absorbance of samples was read at 570 nm by an Elisa reader (Biotech, San Jose, ca, USA). The concentration of no was evaluated by a standard nitrite curve.27
Statistical Analysis
All statistical evaluations and graphs were performed with the GraphPad Prism software (version 8). All behavioral and biochemical tests were interpreted as mean ± standard error of the mean (SEM). The normality test carried out showed the data distribution as normal. One-way ANOVA was performed to compare the means of the statistics. The Tukey post hoc test was used when data were significant to compare the groups by pairs. The significance level was considered as P <0.05 for all the study groups. Effect sizes were estimated using the partial-eta squared (ηp2) statistic.
Results
The effect of BHE on seizures in PTZ-induced kindled rats BHE produced a significant rise in the mean of myoclonic jerk latency [F (3,20) = 15.65, P <0.001, ηp2 = 0.75] and a reduction in the mean of the number of myoclonic jerks [F (3,20) = 9.57, P <0.001, ηp2 = 0.64] in comparison with the CN group (Table 2). Post hoc evaluation showed a significant rise (P = 0.002) in the myoclonic jerk latency mean in the BHE treatment group in dose of 600 mg kg-1 compared to CN. However, BHE-treated groups in dose of 300 failed to show significant changes when compared to the CN group. A significant decline in the mean of the number of myoclonic jerks was observed in doses of 300 (P = 0.026) and 600 (P = 0.002) in the BHE-treated groups in comparison with CN (Table 2).
Table 2 Effect of BHE on seizure behavioral parameters in PTZ-induced kindling in rats.
| Treatments | Myoclonic Jerk Latency | Number of Myoclonic Jerks | GTCS latency | GTCS duration |
| CN | 36.80±3.29 | 14.40±0.51 | 336.8±5.32 | 22.82±0.81 |
| CP | 82.80±4.47c | 6.20±1.02b | 642.0±51.34c | 8.70±1.09c |
| BHE 300 | 51.00±8.28d | 9.00±2.05a | 413.4±23.46d | 14.53±0.66c,d |
| BHE 600 | 70.40±2.62b | 7.00±0.45b | 572.0±56.96b | 7.87±0.97c,e |
Data represent mean ± sem, a: P <0.05, b: P <0.01, c: P <0.001, as compared to control negative (cn) group; d: P <0.01, as compared to control positive (cp) group; e: P <0.001, as compared to bhe 300 group.
One-way ANOVA displayed significant changes in the mean of generalized tonic-clonic seizure (GTCS) latency [F (3,20) = 12.21, P <0.001, ηp2 = 0.70] as well as duration of GTCS [F (3,20) = 58.80, P <0.001, ηp2 = 0.92] between the experimental groups (Table 2). Further analysis with Tukey's test exposed that there was a significant (P = 0.004) increase in the mean of GTCS latency in the BHE group in a dose of 600 in comparison to the CN group. However, this increase in the mean of GTCS latency in a dose of 300 BHE was not significant compared to the CN group (Table 2). As for the GTCS duration parameter, the BHE receiving groups in doses of 300 and 600 also showed a significant difference in mean decrease in comparison to the CN group (P <0.001, Table 2).
Effect of bhe on passive avoidance memory in the shuttle box test
As shown in Figures 1 (a,b) , and confirmed by analysis of variance, there were no significant variances between the experimental groups concerning the STLa indexes [F (3,20) = 0.99, P = 0.423, ηp2 = 0.16] and the SN in acquisition trail [F (3, 20) = 1.60, P = 0.229, ηp2 = 0.23]. As for the STLr [F (3,20) = 17.63, P <0.001, ηp2 = 0.77] and TLC [F (3,20) = 5.31, P = 0.009, ηp2 = 0.50] indices, the memory retrieval stage one-way ANOVA displayed a significant change between the experimental groups (Figure 1c,d) (). The Tukey post hoc test indicated that the cp group produced a significant rise in the mean of STLr (P <0.001) and TLC (P = 0.018) indices in comparison to the CN group (Figure 1 (c,d)) . There was also a significant rise in the group receiving BHE in doses of 300 and 600 compared to the CN group in STLr (P = 0.005, and P <0.001, respectively) parameter. As for TLC parameter only the dose of 600 of BHE has showed a significance increase in comparison with CN group (P = 0.015).
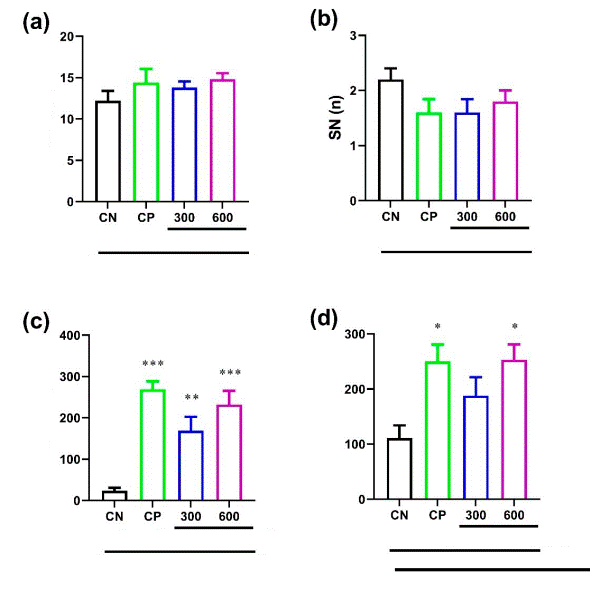
Figure 1 Effect of BHE on (a) step-through latency in the first acquisition trial (STLa), (b) shock number (SN) in acquisition trail, (c) step-through latency in retention trial (STLr), (d) time spent in the light compartment in retention trial (TLC) of passive avoidance learning (PAL) task in PTZ-induced kindling in rats. Each column and bar represent mean ± sem, * P <0.05, ** P <0.01, *** P <0.001, as compared to control negative (CN) group
Effect of BHE on oxidative stress factors
Effect of BHE on brain GSH levels
Significant differences were experienced in the mean of GSH levels in the brains of the rats between the different groups [F (3,20)=73.18, P <0.001, ηp2 = 0.93]. The post hoc examination suggested that the GSH level mean was significantly augmented in the BHE-treated groups in dose 600 compared to the CN group (P <0.001) (Figure 2a). In contrast, BHE-treated groups in dose of 300 failed to show significant changes when compared to the CN group (Figure 2a).
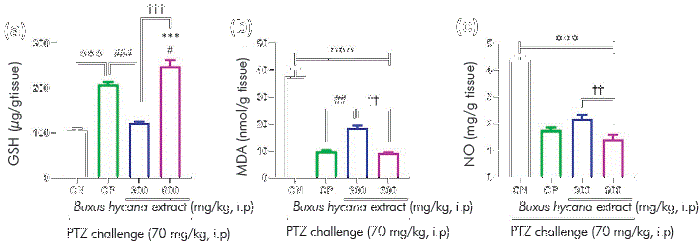
Figure 2 Effect of BHE on (a) GSH, (b) MDA, and (c) no levels in PTZ-induced kindling in rats. Data represents mean ± SEM, *** P<0.001, as compared to control negative (CN) group; # P <0.05, ## P <0.01, ### P <0.001, as compared to control positive (cp) group; †† P <0.01, ††† P <0.001, as compared to BHE 300 group.
The effect of BHE on brain MDA levels
Significant differences were observed in the mean of MDA level [F (3,20) = 82.65, P <0.001, ηp2 = 0.94] between the experimental groups. The post hoc investigation suggested that the MDA level was significantly decreased in the BHE-treated groups in doses of 300 and 600 compared to the CN group (P <0.001) (Figure 2b).
The effect of BHE on brain no levels
Analysis of variance displayed a significant change in the mean no level between the different groups [F (3,20) = 89.25, P<0.001, ηp2 = 0.94]. The no level in the BHE group in 300 and 600 doses (P<0.001) was significantly lower than that of the CN group (Figure 2c).
Discussion
This study identifies for the first time the positive effect of BHE on passive avoidance learning and memory in the experimental model of kindling epilepsy induced by PTZ. The data showed that pretreatment with BHE raises seizure threshold in PTZ-induced kindling. In addition, our findings revealed a significant decrease in epileptic demonstration in pre-treated BHE rats. BHE at doses of 300 and 600 mg kg-1 significantly elevated seizure threshold and decreased the duration of GTCS in kindled rats versus the control group. In line with this study, Azizi et al. (2018) observed that B. hyrcana exhibits neuroprotective and anticonvulsant characteristic in Wistar rats in the PTZ-induced seizure model. They have also documented that the dose of 600 mg kg-1 of the extract has the greatest effect, which is in consonance with our results.22
In the current work, the shuttle box test was adopted to evaluate memory retention and recall properties. The outcome of the study confirmed that doses of 300 and 600 mg kg-1 BHE increased the STLr and TLC compared to the control group. While no studies have been reported on the effect of BHE on memory impairment by convulsant agents so far, there is a report on the anticonvulsant and neuroprotective effects of B. hyrcana in rat model of PTZ-induced acute seizure. This study shows that B. hyrcana has protective activity against seizures which is in line with the result of this study.22
We examine the antioxidant capacity of BHE, therefor the total brain level of the GSH, MDA and no were evaluated. As the results show, BHE at the doses of 300 and 600 mg kg-1 significantly increase the GSH and decrease the MDA and no level, as compared to the PTZ group. Pentylenetetrazole also disrupts the blood-brain barrier (BBB) and disrupts brain function by creating free radicals.28 The brain is very sensitive to free radicals damage, because it contains copious amount of fatty acids and it has a high rate of oxidative metabolism.29 ROS are involved in the pathogenesis of various types of neurodegenerative ailments.30 Oxidative stress may play an important role in causing neurological damage due to seizures.31 Prolonged administration of PTZ induces free radicals and leads to seizure activity in animals. The oxidative stress induced by PTZ leads to tonic-clonic seizures and subsequent neurological death.32 The inhibition of brain neurons and production of free radicals by PTZ is one of the main reasons of epilepsy in experimental models of epilepsy. In epilepsy, seizure activity is always accompanied with amplified levels of ROS.33 Studies have focused on elucidating whether prolonged seizure activity in animals leads to increased ROS production and whether oxidative damage leads to seizure-induced brain injury or not. Accordingly, this study is consistent with the theory that oxidative stress in animals induced by PTZ is probably one of the parameters involved in the pathophysiology of epilepsy.34 no is considered to play a fundamental role in the production and the spread of epileptiform hyperactivity.12 Many studies propose that no plays a key role in the progress of kindling,12 and, recently, it has been reported that oxidative stress and nitrosative stress are the main pathological hallmarks of various neurodegenerative disorders including epilepsy.11,35 Recent research show that hyperactivation of N-methyl-D-aspartate (NMDA) receptors, which may be caused by no overproduction, results in oxidative stress due to ROS generation.36 The superoxide in the mitochondria sometimes combines with no to produce peroxynitrite, which further breaks down to release hydroxyl radicals, thus starting the development of lipid peroxidation and breakdown of formation of DNA leading to neural cell impairment.37
Flavonoids and iso-flavonoids with free radicals scavenging activity are able to fight against the oxidative stress in damaged cell or tissue.38 There is also ample evidence that flavonoids are involved in preventing the destruction of nerve cells caused by oxidative stress.39 It has been shown that in the epileptic condition the level of oxidative stress markers in the neural cells rise up and the levels of antioxidants like GSH will decrease.40 Based on studies on the active ingredients of the B. hyrcana it has been confirmed that it has characteristic to block the Glutathione-S-transferase enzyme (GST), an enzyme that conjugates the GSH with several complexes. As noted above, BHE has numerous flavonoids, which exert antioxidant properties;41 therefore, at least partially, the antiepileptic activity and ultimately the improvement in memory observed in this study may be due to the flavonoids present in BHE, as well as the control of GSH, MDA and no status in the brain.
Conclusion
Overall, the results of this study indicates that bhe has antiepileptic properties in ptz-kindled rats. Moreover, following the improvement of seizure symptoms, it was observed that the memory rate of the rats was enhanced. These effects can be attributed to the improvement of oxidative stress status, including an increased gsh content and decreased mda and no levels.











 nueva página del texto (beta)
nueva página del texto (beta)



