Introduction
Subarachnoid hemorrhage (SAH) affects 30,000 people between the mid and late adulthood in the United States annually. It has a mortality rate of 30 to 45% and is more likely due to a congenital cranial base aneurysm rupture, followed by an aneurysm rupture of infectious or traumatic etiology and an arteriovenous malformation rupture.1 Approximately 15-20% of patients with SAH do not have a vascular lesion on initial four-vessel cerebral angiography, and the causes of this phenomenon are diverse and often not identified.2 Although SAH has a high mortality rate, it has been declining over time in western population; probably due to improvements in rate of smoking, hypertension, its clinical management, diagnostic accuracy and therapeutic measures.3
Apart from a clinical suspicion of a SAH based on a wide variety of signs including severe headache of abrupt onset, nausea, vomiting, nuchal rigidity and consciousness alteration, the electrocardiogram (ECG) can portray specific waveform morphology and rhythm alterations.1 It has been evidenced in previous studies that SAH is associated with a variety of cardiac alterations, which include ventricular tachycardia and fibrillation (mainly when a QTc is prolonged), torsade de pointes, sinus bradycardia, sinus tachycardia or atrial fibrillation, among others.1
Prevalence of ECG abnormalities in SAH patients
The prevalence of ECG abnormalities in SAH patients has been previously reported to range between 27 and 100%. This wide range of prevalence may be due to errors in study methods, number of patients or ECG readings. Because neurological patients do not routinely have ECG, underregistration is a common problem.1 Another observation is that even though some might consider ECG recordings prior to the SAH not important, these should also be included to have an objective comparison in order to better risk-stratify patients, especially in the first hours following the hemorrhage. Brouwers et al found that the most pronounced ECG changes occurred during the first 72 hours after SAH,4 while Di Pasquale et al found that 90% of patients had ECG abnormalities in the first 48 hours.5 These results outline the importance of starting early ECG monitoring in these patients.
Pathophysiology
Crompton and Smith, suggested that the hemorrhage causes localized arteriolar vasospasm resulting in ischemic damage to the hypothalamus and its periphery.6,7 This type of damage induces catecholamine release directly via the upper cervical sympathetic chain and indirectly via the pituitary-adrenal axis. Greenhoot et al, and Melville et al, demonstrated that direct electrical stimulation of the hypothalamus results in ECG abnormalities resembling those seen in SAH patients.8,9 Rogers et al, reproduced constant changes in T waves by stimulating the right and left side of the hypothalamus and stellate ganglia respectively, which suggest a neurogenic response that ultimately is responsible for any particular changes in rhythm and myocardial wall.10 Cruickshank et al, conducted a trial with 46 patients diagnosed with SAH by lumbar puncture recording a total of 197 ECGs and measured mean urinary catecholamines, mean urinary normetanephrine, mean urinary metanephrine and mean plasma cortisol. This trial demonstrated a statically significant correlation with high levels of plasma cortisol (100 μg/mL) with peaked P-waves (n = 40, plasma cortisol [PC] = 27 ± 10.8, p < 0.01), peaked T-waves (n = 21, PC = 27.3 ± 7.8, p < 0.05) and a PR interval lasting shorter than 0.13 msec (n = 11, PC = 27.5 ± 10, p < 0.01), as well as high mean urinary catecholamine values in the abnormal ECG group in a period of 2 weeks.11 Parekh et al found that patients with elevations in cardiac troponin I were more likely to have ECG abnormalities than patients without elevations in the biomarker.12
In general, factors that influence the development of ECG and ventricular function abnormalities include cerebral vasospasm (discussed above), hypoxia, electrolyte imbalance, and sudden increase in intracranial pressure triggering a sympathetic or vagal discharge due to compression or direct ischemic damage of brain structures,1 particularly, the hypothalamus.
Morphological wave changes
These are primarily alterations in repolarization encompassing the T wave, U wave, ST segment and QTc interval. ST-segment elevation or depression and T wave inversion are frequently confused with myocardial infarction (Figures 1 and 2).1,13 In the most extensive study ever done on the matter, Rudehill et al14 recruited 406 patients who were submitted to SAH preoperative ECG monitoring, and findings included high-amplitude R waves in 19% of subjects, ST depression in 15%, T-wave abnormalities in 32%, U waves greater than 1 mm in amplitude in 47%, and prolonged QTc interval (> 440 msec) in 23% of the patients. In a different study, Q wave alterations were found in 4 SAH patients after surgery and T wave alterations in 15 of them.15 A prolonged QTc interval and abnormally large T waves were also noted. These «cerebral» or «neurogenic» T waves are characterized by slurring, and having a broad base; although they can be notched or even flat.1
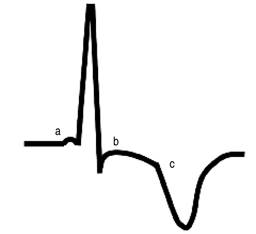
Figure 1: Schematic representation of the most common ECG findings on precordial leads (V3-V5): a. Short PR interval, b. ST segment depression, c. inverted and broad-based T wave.
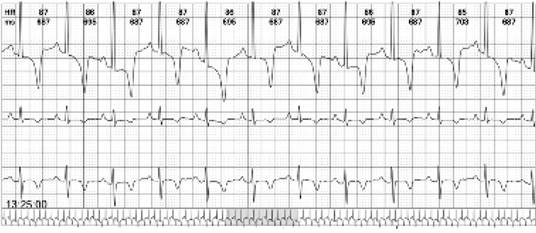
Figure 2: 24-hour Holter monitoring on a 64-year-old female with SAH. Recordings reveal characteristic inverted broad T waves and ST-segment depression. Taken with permission from CES Cardiology electrocardiographic database. Medellín, Colombia.
In a relatively large study, a total of 61 patients for 12 days or until surgery occurred with a 12-lead ECG and cardiac monitoring for arrhythmias; of these patients, 26 had daily 12-lead ECG readings, and 43 of them had readings three times per week. The most common findings were prominent U waves, ischemic T waves, ST-segment elevation or depression, indicators of left ventricular hypertrophy, flat T waves and short PR interval.4 Melin and Fogelholm found ECG changes in 86% of 14 subjects who died within seven days of SAH and in 73% of 62 subjects who survived more than 7 days.16 A total of 26 patients died within the first 6 hours of SAH instauration (even before an ECG could be obtained). Common findings among the obtained ECGs were abnormal Q waves, ST-segment deviation, neurogenic T waves and QTc longer than 430 msec. In the one study that included Holter monitoring, a 12-lead ECG and cardiac monitoring from 120 patients with SAH, were obtained on the day of admission. Changes included ST-segment deviations in 37% of patients, prominent U waves in 16%, and T-wave abnormalities in 12%. Also, 42% of patients had a prolonged QTc interval. Transitory ST-segment depression greater than 1.5 mm lasting 10 to 30 minutes was detected in 7 patients. ST-segment elevation lasting 20 minutes occurred in 1 patient during cerebral angiography along with bigeminal premature ventricular complexes. Abnormalities involving atrial depolarization including notably peaked P waves (> 2.5 mm in amplitude) and short PR intervals (< 100 msec), were also observed.5
Arrhythmias
Despite flaws in accurately monitoring for arrhythmias in previous studies, several alterations have been reported, such as sinus bradycardia, sinus tachycardia, wandering atrial pacemaker, atrial fibrillation, premature atrial, junctional, and ventricular complexes (Figure 3); atrioventricular block, torsade de pointes and ventricular tachycardia.1
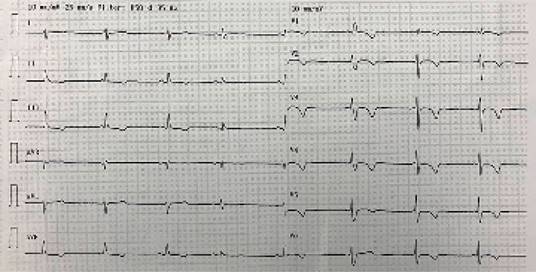
Figure 3: Twelve lead ECG in a 53-year-old man admitted to the intensive care unit due to SAH, exposing complete AV block with escape junctional rhythm, T wave inversion in precordial leads and 2 ventricular premature complexes. Taken with permission from CES Cardiology electrocardiographic database. Medellín, Colombia.
In his study, Di Pasquale et al, obtained a total of 107 correct Holter readings, and cardiac arrhythmias were detected in 96 (90%) of the 107 patients, premature ventricular complexes, including multiform premature ventricular complexes, couplets or triplets, and the R-on-T phenomenon, were detected in 49 patients (46%) and five of the patients with frequent premature ventricular complexes also had non-sustained ventricular tachycardia (defined as 3 or more consecutive premature ventricular complexes, torsade de pointes occurred in 4 patients and progressed to ventricular fibrillation and asystole in 1 of the 4.5
Holter monitoring was repeated 48 hours later in all patients who had malignant alterations, but no further arrhythmias occurred. Of the 107 patients, 39 (36%) had supraventricular arrhythmias, including premature supraventricular complexes, non-sustained supraventricular tachycardia, and atrial fibrillation.1 Thirty-two patients (30%) had sinus tachycardia (heart rate > 120/min), 32 (30%) sinus arrhythmia, 42 (39%) sinus bradycardia (heart rate < 50/min), and 23 (21%) sinoatrial blocks. Other rhythm disturbances, such as wandering pacemaker, sinus arrest greater than 3 seconds, 2:1 atrioventricular block, atrioventricular dissociation, and idioventricular rhythm were also found.1 Di Pasquale et al, found that both severity and frequency of arrhythmias were more significant in the 62 patients studied within 48 hours of SAH onset and that patients with malignant ventricular arrhythmias had a much more prolonged QTc interval (590 ± 52 msec) and serum levels of potassium less than 3.5 mmol/L.5
Wall motion abnormalities
Abnormalities in the myocardial wall movement can certainly confuse the attending physician when it comes to reading an ECG; especially given the fact that they can sometimes suggest other pathological entities such as myocardial ischemia or acute myocardial infarction.17 Although these changes have been reported to be benign in most cases, there is a significant prevalence of other very similar pathologies associated with SAH that can threaten the patient´s life and most always be taken in consideration and differentiated.18-20 For instance, the type of resemblance that these abnormalities have with myocardial infraction in ECG recordings can not only generate delay and confusion at the time of diagnosis, but also create possible incorrect therapeutic actions that can aggravate the patient’s condition such as antiplatelet, anticoagulant and fibrinolytic therapy.21
Taking into account that the alterations found in the ECG are not the only regarding cardiac perfusion per se, there has been evidence of dysfunction in the heart wall motion as well,22-24 specially Takotsubo cardiomyopathy (TCM).25-27 Van der Bilt et al conducted a prospective, multicenter, cohort study involving 277 patients with aneurysmal SAH; 58 of which (21%) developed wall motion abnormalities, and also reported that 25 of the 57 abnormalities (9%) consisted of hypercontractility of apical segments, compatible with TCM.25
TCM was first described in 1991 by Sato et al, as a clinical entity caused by a high level of psychosomatic stress.28 This syndrome consists of a global dysfunction of the left ventricle which in turn causes transient hypokinesis, akinesis or dyskinesis of the apex and midventricle with hyperkinesis of the basal left ventricle segments.29,30 The electrocardiographic presentation of TCM resembles an acute myocardial infarction, portraying elevation of the ST-segment, T-wave inversion and prolonged QTc interval.27,28 Multiple studies have shown the association between SAH and TCM, showing predominance female phenotype (76%), especially postmenopausal women, and the mean age upon presentation was 60 ± 8 years,30-32 with a prevalence of 4-15%,31,33-35 a mortality rate of < 2% and a recurrence rate between 5 and 11% in this population.32,36,37 Kadooka and collaborators presented a study with 450 patients with diagnosis of aneurysmal SAH. Ten of these patients developed TCM along with inverted of flattened T waves (100%), QTc prolongation (< 0.45 msg; 90%), ST-segment elevation (60%) and ST-segment depression (20%). In addition, the study found that the recovery of the wall motion varied between 1 to 2 weeks, as opposed to the improvement of abnormalities on ECG that had an average of 3-4 weeks in most of the patients.38 The importance of the period between the normalization of wall motion and the abnormalities in ECG lies on the risk of developing arrhythmias like non-sustained ventricular tachycardia, paroxysmal atrial fibrillation and ventricular fibrillation.1,38-40
In Table 1, all findings described by the authors mentioned in this review are included to give a better comprehensive view of all cardiac ECG and ventricular function alterations reported in patients with SAH.
Table 1: Most common wave electrocardiographical and ventricular function abnormalities found by each author.
| Summary of findings | ||
|---|---|---|
| Morphological wave changes | ||
| Brouwers et al(4) | PR interval | Short |
| Cropp and Manning et al(15) | Q wave | > 1 mm |
| Melin and Fogelholm et al(16) | T wave | Inverted |
| Brouwers et al(4) | Broad base | |
| Rudehill et al(14) | Notched | |
| Di Pasquale et al(5) | Flat | |
| Rudehill et al(14) | R wave | High amplitude |
| Brouwers et al(4) | U wave | Prominent (> 0.1 mv) |
| Rudehill et al(14) | ||
| Di Pasquale et al(5) | ||
| Melin and Fogelholm et al(16) | ST segment | Elevation |
| Brouwers et al(4) | Depression (more common) | |
| Rudehill et al(14) | ||
| Di Pasquale et al(5) | ||
| Melin and Fogelholm et al(16) | QTc interval | Prolonged |
| Rudehill et al(14) | ||
| Di Pasquale et al(5) | ||
| Arrythmias | ||
| Di Pasquale et al(5) | Supra-ventricular | Sinus bradycardia |
| Sinus tachycardia | ||
| Wandering atrial pacemaker | ||
| Atrial fibrillation | ||
| Ventricular | Ventricular tachycardia | |
| Torsade de pointes | ||
| Premature complexes | Atrial | |
| Junctional | ||
| Ventricular | ||
| Blocks | Sinoatrial | |
| Atrioventricular | ||
| Ventricular function abnormalities | ||
| Van der Bilt et al(25) | Takotsubo cardiomyopathy | ST elevation / depression |
| Kadooka et al(38) | T wave inversion | |
| QTc interval prolongation | ||
Conclusions
ECG abnormalities associated with aneurysmal SAH have been proved to affect discharge disposition and prolonged hospital stay,41 and because few studies have effectively involved Holter monitoring in these patients, this constitutes a matter of investigations with aims to standardize this extremely wide range of electrical cardiac complications in this central nervous system pathology.











 nueva página del texto (beta)
nueva página del texto (beta)


